The Multiple Mechanisms of Cell Death Triggered by Resveratrol in Lymphoma and Leukemia
Abstract
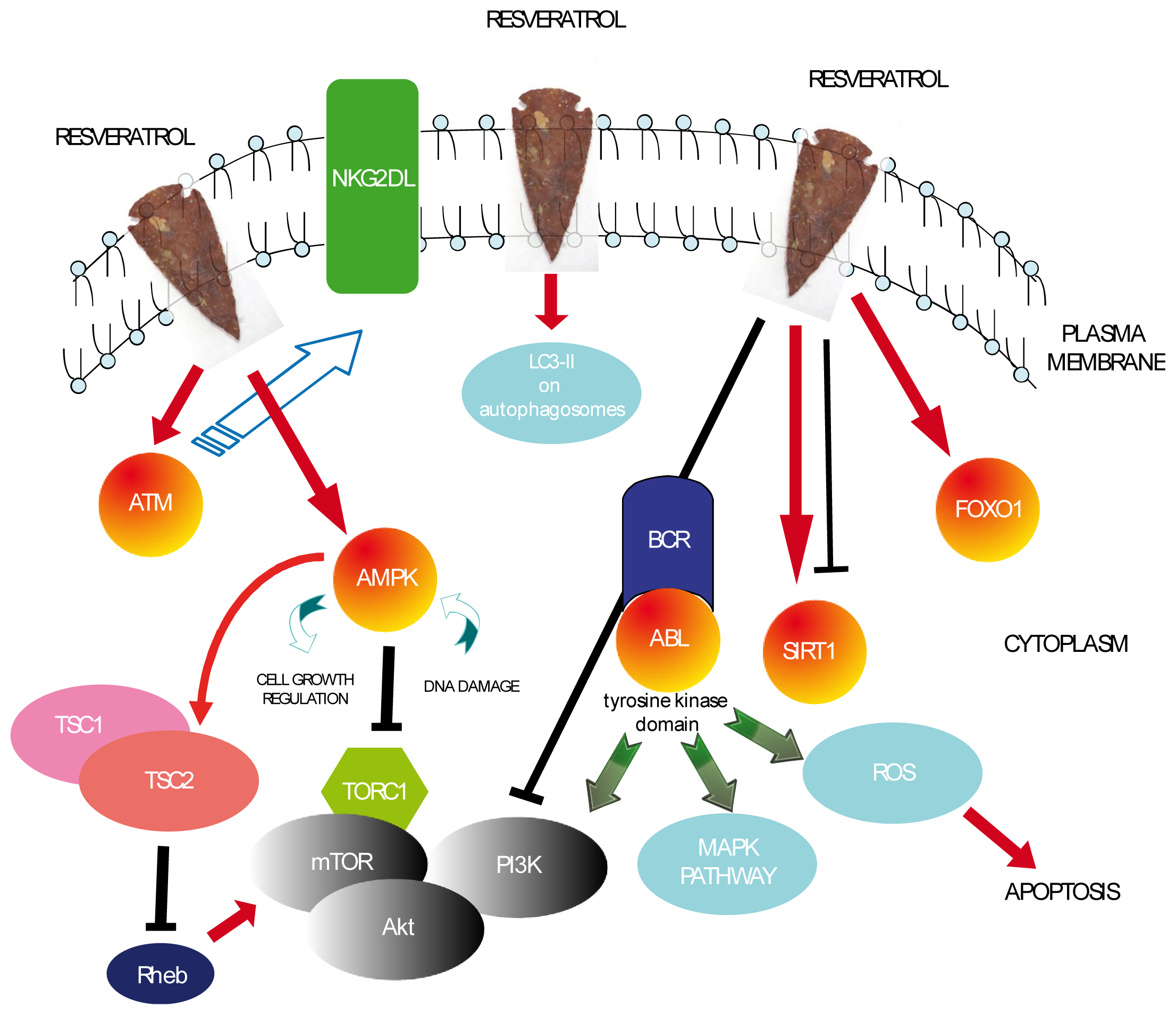
:1. Introduction
2. RSV in Non-Hodgkin Lymphoma and Leukemias
3. RSV in Hodgkin Lymphoma
4. In Vivo Tumor Experimental Models
5. RSV and Challenges in Humans
6. Conclusions and Perspectives
- -
- may exert its activity on human PBMCs in a bi-phasic/dose-dependent way;
- -
- is cytotoxic to lymphoma and leukemia cancer cells since it can trigger apoptosis, autophagy or senescence;
- -
- seems not to be toxic to human PBMCs, either resting or mitogenically stimulated;
- -
- is well tolerated by humans and its physiological metabolites may work as a circulating reservoir of the parent compound that can be re-generated within the cells of the intestinal tract;
- -
- is a chemically well-known molecule whose structure can be improved and modified by substitutions aimed at increasing the anticancer properties or the bioavailability of the active principle.
Acknowledgments
Conflicts of Interest
References
- Magrone, T.; Candore, G.; Caruso, C.; Jirillo, E.; Covelli, V. Polyphenols from red wine modulate immune responsiveness: Biological and clinical significance. Curr. Pharm. Des. 2008, 14, 2733–2748. [Google Scholar]
- Timmers, S.; Auwerx, J.; Schrauwen, P. The journey of resveratrol from yeast to human. Aging (Albany) 2012, 4, 146–158. [Google Scholar]
- Pezzuto, J.M. The phenomenon of resveratrol: Redefining the virtues of promiscuity. Ann. N. Y. Acad. Sci. 2011, 1215, 123–130. [Google Scholar]
- Agarwal, B.; Baur, J.A. Resveratrol and life extension. Ann. N. Y. Acad. Sci. 2011, 1215, 138–143. [Google Scholar]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar]
- Wood, J.G.; Rogina, B.; Lavu, S.; Howitz, K.; Helfand, S.L.; Tatar, M.; Sinclair, D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004, 430, 686–689. [Google Scholar]
- Lee, Y.J.; Im, J.H.; Won, S.Y.; Kim, Y.B.; Cho, M.K.; Nam, H.S.; Choi, Y.J.; Lee, S.H. Synergistic anti-cancer effects of resveratrol and chemotherapeutic agent clofarabine against human malignant mesothelioma MSTO-211H cells. Food Chem. Toxicol. 2012, 52, 61–68. [Google Scholar]
- Castillo-Pichardo, L.; Dharmawardhane, S.F. Grape polyphenols inhibit akt/mammalian target of rapamycin signaling and potentiate the effects of gefitinib in breast cancer. Nutr. Cancer 2012, 64, 1058–1069. [Google Scholar]
- Can, G.; Cakir, Z.; Kartal, M.; Gunduz, U.; Baran, Y. Apoptotic effects of resveratrol a grape polyphenol on imatinib-sensitive and resistant K562 chronic myeloid leukemia cells. Anticancer Res. 2012, 32, 2673–2678. [Google Scholar]
- Iguchi, K.; Toyama, T.; Ito, T.; Shakui, T.; Usui, S.; Oyama, M.; Iinuma, M.; Hirano, K. Anti-androgenic activity of resveratrol analogs in prostate cancer LNCaP cells. J. Androl. 2012, 33, 1208–1215. [Google Scholar]
- Juan, M.E.; Alfaras, I.; Planas, J.M. Colorectal cancer chemoprevention by trans-resveratrol. Pharmacol. Res. 2012, 65, 584–591. [Google Scholar]
- Hussain, A.R.; Uddin, S.; Bu, R.; Khan, O.S.; Ahmed, S.O.; Ahmed, M.; Al-Kuraya, K.S. Resveratrol suppresses constitutive activation of AKT via generation of ROS and induces apoptosis in diffuse large B cell lymphoma cell lines. PLoS One 2011, 6, e24703. [Google Scholar]
- Delmas, D.; Solary, E.; Latruffe, N. Resveratrol a phytochemical inducer of multiple cell death pathways: Apoptosis autophagy and mitotic catastrophe. Curr. Med. Chem. 2011, 18, 1100–1121. [Google Scholar]
- Wallace, D.C. Mitochondrial diseases in man and mouse. Science 1999, 283, 1482–1488. [Google Scholar]
- Mignotte, B.; Vayssiere, J.L. Mitochondria and apoptosis. Eur. J. Biochem. 1998, 252, 1–15. [Google Scholar]
- Sengottuvelan, M.; Deeptha, K.; Nalini, N. Resveratrol ameliorates DNA damage prooxidant and antioxidant imbalance in 12-dimethylhydrazine induced rat colon carcinogenesis. Chem.-Biol. Interact. 2009, 181, 193–201. [Google Scholar]
- Sengottuvelan, M.; Viswanathan, P.; Nalini, N. Chemopreventive effect of trans-resveratrol—A phytoalexin against colonic aberrant crypt foci and cell proliferation in 12-dimethylhydrazine induced colon carcinogenesis. Carcinogenesis 2006, 27, 1038–1046. [Google Scholar]
- Sener, G.; Topaloglu, N.; Sehirli, A.O.; Ercan, F.; Gedik, N. Resveratrol alleviates bleomycin-induced lung injury in rats. Pulm. Pharmacol. Ther. 2007, 20, 642–649. [Google Scholar]
- Ahmad, K.A.; Clement, M.V.; Hanif, I.M.; Pervaiz, S. Resveratrol inhibits drug-induced apoptosis in human leukemia cells by creating an intracellular milieu nonpermissive for death execution. Cancer Res. 2004, 64, 1452–1459. [Google Scholar]
- Ferrari, N.; Tosetti, F.; de Flora, S.; Donatelli, F.; Sogno, I.; Noonan, D.M.; Albini, A. Diet-derived phytochemicals: From cancer chemoprevention to cardio-oncological prevention. Curr. Drug Targets 2011, 12, 1909–1924. [Google Scholar]
- Bernhard, D.; Tinhofer, I.; Tonko, M.; Hubl, H.; Ausserlechner, M.J.; Greil, R.; Kofler, R.; Csordas, A. Resveratrol causes arrest in the S-phase prior to Fas-independent apoptosis in CEM-C7H2 acute leukemia cells. Cell Death Differ. 2000, 7, 834–842. [Google Scholar]
- Dorrie, J.; Gerauer, H.; Wachter, Y.; Zunino, S.J. Resveratrol induces extensive apoptosis by depolarizing mitochondrial membranes and activating caspase-9 in acute lymphoblastic leukemia cells. Cancer Res. 2001, 61, 4731–4739. [Google Scholar]
- Park, J.W.; Choi, Y.J.; Jang, M.A.; Lee, Y.S.; Jun, D.Y.; Suh, S.I.; Baek, W.K.; Suh, M.H.; Jin, I.N.; Kwon, T.K. Chemopreventive agent resveratrol a natural product derived from grapes reversibly inhibits progression through S and G2 phases of the cell cycle in U937 cells. Cancer Lett. 2001, 163, 43–49. [Google Scholar]
- Wieder, T.; Prokop, A.; Bagci, B.; Essmann, F.; Bernicke, D.; Schulze-Osthoff, K.; Dorken, B.; Schmalz, H.G.; Daniel, P.T.; Henze, G. Piceatannol a hydroxylated analog of the chemopreventive agent resveratrol is a potent inducer of apoptosis in the lymphoma cell line BJAB and in primary leukemic lymphoblasts. Leukemia 2001, 15, 1735–1742. [Google Scholar]
- Gogada, R.; Prabhu, V.; Amadori, M.; Scott, R.; Hashmi, S.; Chandra, D. Resveratrol induces p53-independent X-linked inhibitor of apoptosis protein (XIAP)-mediated Bax protein oligomerization on mitochondria to initiate cytochrome c release and caspase activation. J. Biol. Chem. 2011, 286, 28749–28760. [Google Scholar]
- Roman, V.; Billard, C.; Kern, C.; Ferry-Dumazet, H.; Izard, J.C.; Mohammad, R.; Mossalayi, D.M.; Kolb, J.P. Analysis of resveratrol-induced apoptosis in human B-cell chronic leukaemia. Br. J. Haematol. 2002, 117, 842–851. [Google Scholar]
- Surh, Y.J.; Hurh, Y.J.; Kang, J.Y.; Lee, E.; Kong, G.; Lee, S.J. Resveratrol an antioxidant present in red wine induces apoptosis in human promyelocytic leukemia (HL-60) cells. Cancer Lett. 1999, 140, 1–10. [Google Scholar]
- Jazirehi, A.R.; Bonavida, B. Resveratrol modifies the expression of apoptotic regulatory proteins and sensitizes non-Hodgkin’s lymphoma and multiple myeloma cell lines to paclitaxel-induced apoptosis. Mol. Cancer Ther. 2004, 3, 71–84. [Google Scholar]
- Luzi, C.; Brisdelli, F.; Cinque, B.; Cifone, G.; Bozzi, A. Differential sensitivity to resveratrol-induced apoptosis of human chronic myeloid (K562) and acute lymphoblastic (HSB-2) leukemia cells. Biochem. Pharmacol. 2004, 68, 2019–2030. [Google Scholar]
- Delmas, D.; Rebe, C.; Lacour, S.; Filomenko, R.; Athias, A.; Gambert, P.; Cherkaoui-Malki, M.; Jannin, B.; Dubrez-Daloz, L.; Latruffe, N.; et al. Resveratrol-induced apoptosis is associated with Fas redistribution in the rafts and the formation of a death-inducing signaling complex in colon cancer cells. J. Biol. Chem. 2003, 278, 41482–41490. [Google Scholar]
- Guha, P.; Dey, A.; Sen, R.; Chatterjee, M.; Chattopadhyay, S.; Bandyopadhyay, S.K. Intracellular GSH depletion triggered mitochondrial Bax translocation to accomplish resveratrol-induced apoptosis in the U937 cell line. J. Pharmacol. Exp. Ther. 2011, 336, 206–214. [Google Scholar]
- Suzuki, Y.; Ito, S.; Sasaki, R.; Asahi, M.; Ishida, Y. Resveratrol suppresses cell proliferation via inhibition of STAT3 phosphorylation and Mcl-1 and cIAP-2 expression in HTLV-1-infected T cells. Leuk. Res. 2013, 37, 1674–1679. [Google Scholar]
- Hayashibara, T.; Yamada, Y.; Nakayama, S.; Harasawa, H.; Tsuruda, K.; Sugahara, K.; Miyanishi, T.; Kamihira, S.; Tomonaga, M.; Maita, T. Resveratrol induces downregulation in survivin expression and apoptosis in HTLV-1-infected cell lines: A prospective agent for adult T cell leukemia chemotherapy. Nutr. Cancer 2002, 44, 193–201. [Google Scholar]
- Park, C.E.; Kim, M.J.; Lee, J.H.; Min, B.I.; Bae, H.; Choe, W.; Kim, S.S.; Ha, J. Resveratrol stimulates glucose transport in C2C12 myotubes by activating AMP-activated protein kinase. Exp. Mol. Med. 2007, 39, 222–229. [Google Scholar]
- Um, J.H.; Park, S.J.; Kang, H.; Yang, S.; Foretz, M.; McBurney, M.W.; Kim, M.K.; Viollet, B.; Chung, J.H. AMP-activated protein kinase-deficient mice are resistant to the metabolic effects of resveratrol. Diabetes 2010, 59, 554–563. [Google Scholar]
- Greer, E.L.; Brunet, A. Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans. Aging Cell 2009, 8, 113–127. [Google Scholar]
- Wang, A.; Liu, M.; Liu, X.; Dong, L.Q.; Glickman, R.D.; Slaga, T.J.; Zhou, Z.; Liu, F. Up-regulation of adiponectin by resveratrol: The essential roles of the Akt/FOXO1 and AMP-activated protein kinase signaling pathways and DsbA-L. J. Biol. Chem. 2011, 286, 60–66. [Google Scholar]
- Puissant, A.; Grosso, S.; Jacquel, A.; Belhacene, N.; Colosetti, P.; Cassuto, J.P.; Auberger, P. Imatinib mesylate-resistant human chronic myelogenous leukemia cell lines exhibit high sensitivity to the phytoalexin resveratrol. FASEB J. 2008, 22, 1894–1904. [Google Scholar]
- Puissant, A.; Robert, G.; Fenouille, N.; Luciano, F.; Cassuto, J.P.; Raynaud, S.; Auberger, P. Resveratrol promotes autophagic cell death in chronic myelogenous leukemia cells via JNK-mediated p62/SQSTM1 expression and AMPK activation. Cancer Res. 2010, 70, 1042–1052. [Google Scholar]
- Vakana, E.; Platanias, L.C. AMPK in BCR-ABL expressing leukemias Regulatory effects and therapeutic implications. Oncotarget 2011, 2, 1322–1328. [Google Scholar]
- Sabatini, D.M. mTOR and cancer: Insights into a complex relationship. Nat. Rev. Cancer 2006, 6, 729–734. [Google Scholar]
- Sarbassov, D.D.; Ali, S.M.; Sabatini, D.M. Growing roles for the mTOR pathway. Curr. Opin. Cell Biol. 2005, 17, 596–603. [Google Scholar]
- Puissant, A.; Auberger, P. AMPK- and p62/SQSTM1-dependent autophagy mediate Resveratrol-induced cell death in chronic myelogenous leukemia. Autophagy 2010, 6, 655–657. [Google Scholar]
- Hoffman, A.E.; Demanelis, K.; Fu, A.; Zhang, T.; Zhu, Y. Association of AMP-activated Protein Kinase with Risk and Progression of Non-Hodgkin Lymphoma. Cancer Epidemiol. Biomark. Prev. 2013, 22, 736–744. [Google Scholar]
- Radwan, F.F.; Zhang, L.; Hossain, A.; Doonan, B.P.; God, J.M.; Haque, A. Mechanisms regulating enhanced human leukocyte antigen class II-mediated CD4+ T cell recognition of human B-cell lymphoma by resveratrol. Leuk. Lymphoma 2012, 53, 305–314. [Google Scholar]
- Espinoza, J.L.; Takami, A.; Trung, L.Q.; Nakao, S. Ataxia-telangiectasia mutated kinase-mediated upregulation of NKG2D ligands on leukemia cells by resveratrol results in enhanced natural killer cell susceptibility. Cancer Sci. 2013, 104, 657–662. [Google Scholar]
- Eagle, R.A.; Trowsdale, J. Promiscuity and the single receptor: NKG2D. Nat. Rev. Immunol. 2007, 7, 737–744. [Google Scholar]
- Gasser, S.; Orsulic, S.; Brown, E.J.; Raulet, D.H. The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature 2005, 436, 1186–1190. [Google Scholar]
- Cavalieri, E.; Rogan, E. The molecular etiology and prevention of estrogen-initiated cancers: Ockham’s Razor: Pluralitas non est ponenda sine necessitate Plurality should not be posited without necessity. Mol. Asp. Med. 2013, 36C, 1–55. [Google Scholar]
- Gaikwad, N.W.; Yang, L.; Weisenburger, D.D.; Vose, J.; Beseler, C.; Rogan, E.G.; Cavalieri, E.L. Urinary biomarkers suggest that estrogen-DNA adducts may play a role in the aetiology of non-Hodgkin lymphoma. Biomarkers 2009, 14, 502–512. [Google Scholar]
- Frazzi, R.; Valli, R.; Tamagnini, I.; Casali, B.; Latruffe, N.; Merli, F. Resveratrol-mediated apoptosis of hodgkin lymphoma cells involves SIRT1 inhibition and FOXO3a hyperacetylation. Int. J. Cancer 2013, 132, 1013–1021. [Google Scholar]
- Gatto, D.; Brink, R. The germinal center reaction. J. Allergy Clin. Immunol. 2010, 126, 898–907. [Google Scholar]
- Liu, T.; Liu, P.Y.; Marshall, G.M. The critical role of the class III histone deacetylase SIRT1 in cancer. Cancer Res. 2009, 69, 1702–1705. [Google Scholar]
- Firestein, R.; Blander, G.; Michan, S.; Oberdoerffer, P.; Ogino, S.; Campbell, J.; Bhimavarapu, A.; Luikenhuis, S.; de Cabo, R.; Fuchs, C.; et al. The SIRT1 deacetylase suppresses intestinal tumorigenesis and colon cancer growth. PLoS One 2008, 3, e2020. [Google Scholar]
- Morris, B.J. Seven sirtuins for seven deadly diseases of aging. Free Radic. Biol. Med. 2013, 56, 133–171. [Google Scholar]
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; et al. Cancer chemopreventive activity of resveratrol a natural product derived from grapes. Science 1997, 275, 218–220. [Google Scholar]
- Yusuf, N.; Nasti, T.H.; Meleth, S.; Elmets, C.A. Resveratrol enhances cell-mediated immune response to DMBA through TLR4 and prevents DMBA induced cutaneous carcinogenesis. Mol. Carcinog. 2009, 48, 713–723. [Google Scholar]
- Roy, P.; Kalra, N.; Prasad, S.; George, J.; Shukla, Y. Chemopreventive potential of resveratrol in mouse skin tumors through regulation of mitochondrial and PI3K/AKT signaling pathways. Pharm. Res. 2009, 26, 211–217. [Google Scholar]
- Kowalczyk, M.C.; Kowalczyk, P.; Tolstykh, O.; Hanausek, M.; Walaszek, Z.; Slaga, T.J. Synergistic effects of combined phytochemicals and skin cancer prevention in SENCAR mice. Cancer Prev. Res. (Phila.) 2010, 3, 170–178. [Google Scholar]
- Banerjee, S.; Bueso-Ramos, C.; Aggarwal, B.B. Suppression of 712-dimethylbenz(a)anthraceneinduced mammary carcinogenesis in rats by resveratrol: Role of nuclear factor-kappaB cyclooxygenase 2 and matrix metalloprotease 9. Cancer Res. 2002, 62, 4945–4954. [Google Scholar]
- Provinciali, M.; Re, F.; Donnini, A.; Orlando, F.; Bartozzi, B.; di Stasio, G.; Smorlesi, A. Effect of resveratrol on the development of spontaneous mammary tumors in HER-2/neu transgenic mice. Int. J. Cancer 2005, 115, 36–45. [Google Scholar]
- Papoutsis, A.J.; Selmin, O.I.; Borg, J.L.; Romagnolo, D.F. Gestational exposure to the AhR agonist 2378-tetrachlorodibenzo-p-dioxin induces BRCA-1 promoter hypermethylation and reduces BRCA-1 expression in mammary tissue of rat offspring: Preventive effects of resveratrol. Mol. Carcinog. 2013. [Google Scholar] [CrossRef]
- Lee-Chang, C.; Bodogai, M.; Martin-Montalvo, A.; Wejksza, K.; Sanghvi, M.; Moaddel, R.; de Cabo, R.; Biragyn, A. Inhibition of breast cancer metastasis by resveratrol-mediated inactivation of tumor-evoked regulatory B cells. J. Immunol. 2013, 191, 4141–4151. [Google Scholar]
- Harper, C.E.; Cook, L.M.; Patel, B.B.; Wang, J.; Eltoum, I.A.; Arabshahi, A.; Shirai, T.; Lamartiniere, C.A. Genistein and resveratrol alone and in combination suppress prostate cancer in SV-40 tag rats. Prostate 2009, 69, 1668–1682. [Google Scholar]
- Yu, Y.H.; Chen, H.A.; Chen, P.S.; Cheng, Y.J.; Hsu, W.H.; Chang, Y.W.; Chen, Y.H.; Jan, Y.; Hsiao, M.; Chang, T.Y.; et al. MiR-520h-mediated FOXC2 regulation is critical for inhibition of lung cancer progression by resveratrol. Oncogene 2013, 32, 431–443. [Google Scholar]
- Cui, X.; Jin, Y.; Hofseth, A.B.; Pena, E.; Habiger, J.; Chumanevich, A.; Poudyal, D.; Nagarkatti, M.; Nagarkatti, P.S.; Singh, U.P.; et al. Resveratrol suppresses colitis and colon cancer associated with colitis. Cancer Prev. Res. (Phila.) 2010, 3, 549–559. [Google Scholar]
- Li, T.; Fan, G.X.; Wang, W.; Yuan, Y.K. Resveratrol induces apoptosis influences IL-6 and exerts immunomodulatory effect on mouse lymphocytic leukemia both in vitro and in vivo. Int. Immunopharmacol. 2007, 7, 1221–1231. [Google Scholar]
- Singh, N.P.; Singh, U.P.; Hegde, V.L.; Guan, H.; Hofseth, L.; Nagarkatti, M.; Nagarkatti, P.S. Resveratrol (trans-354′-trihydroxystilbene) suppresses EL4 tumor growth by induction of apoptosis involving reciprocal regulation of SIRT1 and NF-κB. Mol. Nutr. Food Res. 2011, 55, 1207–1218. [Google Scholar]
- Zunino, S.J.; Storms, D.H.; Newman, J.W.; Pedersen, T.L.; Keen, C.L.; Ducore, J.M. Dietary resveratrol does not delay engraftment sensitize to vincristine or inhibit growth of high-risk acute lymphoblastic leukemia cells in NOD/SCID mice. Int. J. Oncol. 2012, 41, 2207–2212. [Google Scholar]
- Zunino, S.J.; Storms, D.H.; Newman, J.W.; Pedersen, T.L.; Keen, C.L.; Ducore, J.M. Resveratrol given intraperitoneally does not inhibit the growth of high-risk t(4;11) acute lymphoblastic leukemia cells in a NOD/SCID mouse model. Int. J. Oncol. 2012, 40, 1277–1284. [Google Scholar]
- Li, T.; Wang, W.; Chen, H.; Ye, L. Evaluation of anti-leukemia effect of resveratrol by modulating STAT3 signaling. Int. Immunopharmacol. 2010, 10, 18–25. [Google Scholar]
- Zunino, S.J.; Storms, D.H. Resveratrol alters proliferative responses and apoptosis in human activated B lymphocytes in vitro. J. Nutr. 2009, 139, 1603–1608. [Google Scholar]
- Tomic, J.; McCaw, L.; Li, Y.; Hough, M.R.; Ben-David, Y.; Moffat, J.; Spaner, D.E. Resveratrol has anti-leukemic activity associated with decreased O-GlcNAcylated proteins. Exp. Hematol. 2013, 41, 675–686. [Google Scholar]
- Vang, O.; Ahmad, N.; Baile, C.A.; Baur, J.A.; Brown, K.; Csiszar, A.; Das, D.K.; Delmas, D.; Gottfried, C.; Lin, H.Y.; et al. What is new for an old molecule? Systematic review and recommendations on the use of resveratrol. PLoS One 2011, 6, e19881. [Google Scholar]
- Brown, V.A.; Patel, K.R.; Viskaduraki, M.; Crowell, J.A.; Perloff, M.; Booth, T.D.; Vasilinin, G.; Sen, A.; Schinas, A.M.; Piccirilli, G.; et al. Repeat dose study of the cancer chemopreventive agent resveratrol in healthy volunteers: Safety pharmacokinetics and effect on the insulin-like growth factor axis. Cancer Res. 2010, 70, 9003–9011. [Google Scholar]
- Patel, K.R.; Brown, V.A.; Jones, D.J.; Britton, R.G.; Hemingway, D.; Miller, A.S.; West, K.P.; Booth, T.D.; Perloff, M.; Crowell, J.A.; et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010, 70, 7392–7399. [Google Scholar]
- Sandhu, M.S.; Dunger, D.B.; Giovannucci, E.L. Insulin, insulin-like growth factor-I (IGF-I), IGF binding proteins, their biologic interactions, and colorectal cancer. J. Natl. Cancer Inst. 2002, 94, 972–980. [Google Scholar]
- Kari, F.W.; Dunn, S.E.; French, J.E.; Barrett, J.C. Roles for insulin-like growth factor-1 in mediating the anti-carcinogenic effects of caloric restriction. J. Nutr. Health Aging 1999, 3, 92–101. [Google Scholar]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar]
- Harper, C.E.; Patel, B.B.; Wang, J.; Arabshahi, A.; Eltoum, I.A.; Lamartiniere, C.A. Resveratrol suppresses prostate cancer progression in transgenic mice. Carcinogenesis 2007, 28, 1946–1953. [Google Scholar]
- Patel, K.R.; Andreadi, C.; Britton, R.G.; Horner-Glister, E.; Karmokar, A.; Sale, S.; Brown, V.A.; Brenner, D.E.; Singh, R.; Steward, W.P.; et al. Sulfate metabolites provide an intracellular pool for resveratrol generation and induce autophagy with senescence. Sci. Transl. Med. 2013, 5, 1–12. [Google Scholar]
- Shi, Y.; Tomic, J.; Wen, F.; Shaha, S.; Bahlo, A.; Harrison, R.; Dennis, J.W.; Williams, R.; Gross, B.J.; Walker, S.; et al. Aberrant O-GlcNAcylation characterizes chronic lymphocytic leukemia. Leukemia 2010, 24, 1588–1598. [Google Scholar]
- Delmas, D.; Aires, V.; Limagne, E.; Dutartre, P.; Mazue, F.; Ghiringhelli, F.; Latruffe, N. Transport stability and biological activity of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 48–59. [Google Scholar]
- Szekeres, T.; Saiko, P.; Fritzer-Szekeres, M.; Djavan, B.; Jager, W. Chemopreventive effects of resveratrol and resveratrol derivatives. Ann. N. Y. Acad. Sci. 2011, 1215, 89–95. [Google Scholar]
- Burkon, A.; Somoza, V. Quantification of free and protein-bound trans-resveratrol metabolites and identification of trans-resveratrol-C/O-conjugated diglucuronides—Two novel resveratrol metabolites in human plasma. Mol. Nutr. Food Res. 2008, 52, 549–557. [Google Scholar]
- Maier-Salamon, A.; Hagenauer, B.; Wirth, M.; Gabor, F.; Szekeres, T.; Jager, W. Increased transport of resveratrol across monolayers of the human intestinal Caco-2 cells is mediated by inhibition and saturation of metabolites. Pharm. Res. 2006, 23, 2107–2115. [Google Scholar]
- Maier-Salamon, A.; Hagenauer, B.; Reznicek, G.; Szekeres, T.; Thalhammer, T.; Jager, W. Metabolism and disposition of resveratrol in the isolated perfused rat liver: Role of Mrp2 in the biliary excretion of glucuronides. J. Pharm. Sci. 2008, 97, 1615–1628. [Google Scholar]
- Roupe, K.A.; Remsberg, C.M.; Yanez, J.A.; Davies, N.M. Pharmacometrics of stilbenes: Seguing towards the clinic. Curr. Clin. Pharmacol. 2006, 1, 81–101. [Google Scholar]
| Year | Tumor experimental model | Animal model | Reference |
|---|---|---|---|
| 1997 | Skin cancer | Female CD-1 mice | [56] |
| 2009 | Skin cancer | Female C3H/HeN mice | [57] |
| 2009 | Skin cancer | Female Swiss mice | [58] |
| 2010 | Skin cancer | SENCAR mice | [59] |
| 2002 | Breast cancer | Female Sprague-Dawley rats | [60] |
| 2005 | Breast cancer | FVB/N female mice | [61] |
| 2013 | Breast cancer | Female Sprague-Dawley rats | [62] |
| 2013 | Breast cancer | Female BALB/c mice | [63] |
| 2006 | Colorectal cancer | Male Wistar rats | [17] |
| 2010 | Colorectal cancer | C57 BL/6 mice | [66] |
| 2009 | Prostate cancer | Male Sprague-Dawley rats (SV-40 Tag) | [64] |
| 2013 | Lung cancer | Female SCID mice | [65] |
| Year | Tumor experimental model | Animal model | Reference |
|---|---|---|---|
| 2007 | L1210 mouse lymphocytic leukemia | Male BALB/c mice | [67] |
| 2011 | EL-4 mouse lypmhoma | NOD/SCID mice | [68] |
| 2012 | Human ALL | NOD/SCID mice | [69] |
| 2012 | Human ALL | NOD/SCID mice | [70] |
| 2010 | Human acute myeloblastic leukemia Kasumi-1 | Male BALB/c mice | [71] |
| 2009 | Human PBMCs | [72] | |
| 2013 | Human CLL | [73] |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Frazzi, R.; Tigano, M. The Multiple Mechanisms of Cell Death Triggered by Resveratrol in Lymphoma and Leukemia. Int. J. Mol. Sci. 2014, 15, 4977-4993. https://doi.org/10.3390/ijms15034977
Frazzi R, Tigano M. The Multiple Mechanisms of Cell Death Triggered by Resveratrol in Lymphoma and Leukemia. International Journal of Molecular Sciences. 2014; 15(3):4977-4993. https://doi.org/10.3390/ijms15034977
Chicago/Turabian StyleFrazzi, Raffaele, and Marco Tigano. 2014. "The Multiple Mechanisms of Cell Death Triggered by Resveratrol in Lymphoma and Leukemia" International Journal of Molecular Sciences 15, no. 3: 4977-4993. https://doi.org/10.3390/ijms15034977
APA StyleFrazzi, R., & Tigano, M. (2014). The Multiple Mechanisms of Cell Death Triggered by Resveratrol in Lymphoma and Leukemia. International Journal of Molecular Sciences, 15(3), 4977-4993. https://doi.org/10.3390/ijms15034977





