Circulating YKL-40 Level, but not CHI3L1 Gene Variants, Is Associated with Atherosclerosis-Related Quantitative Traits and the Risk of Peripheral Artery Disease
Abstract
:1. Introduction
2. Results
2.1. Clinical and Biochemical Characteristics
| Baseline Characteristics | Total | Men | Women | p Value |
|---|---|---|---|---|
| Number | 612 | 323 | 289 | |
| Age (years) | 46.2 ± 10.0 | 45.6 ± 10.0 | 47.0 ± 10.0 | 0.082 |
| Systolic BP (mm Hg) | 113.1 ± 16.1 | 114.0 ± 14.3 | 112.1 ± 17.8 | 0.169 |
| Diastolic BP (mm Hg) | 75.0 ± 10.0 | 76.8 ± 9.7 | 73.1 ± 10.0 | <0.001 |
| Mean BP (mm Hg) | 87.7 ± 11.2 | 89.2 ± 10.4 | 86.1±11.8 | 0.001 |
| Total cholesterol (mg/dL) | 198.8 ± 36.4 | 200.7 ± 36.9 | 196.6 ± 35.9 | 0.164 |
| HDL-cholesterol (mg/dL) | 55.3 ± 14.3 | 49.8 ± 11.9 | 61.3 ± 14.3 | <0.001 |
| LDL-cholesterol (mg/dL) | 116.1 ± 32.9 | 118.4 ± 33.8 | 113.5 ± 31.6 | 0.064 |
| Triglyceride (mg/dL) | 142.4 ± 118.5 | 171.8 ± 147.2 | 109.9 ± 60.6 | <0.001 |
| Body mass index (kg/m2) | 24.3 ± 3.5 | 24.9 ± 3.1 | 23.6 ± 3.7 | <0.001 |
| Diabetes mellitus (%) | 5.1 | 5.9 | 4.2 | 0.330 |
| Current smokers (%) | 19.3 | 33.1 | 3.8 | <0.001 |
| Fasting plasma glucose (mg/dL) | 96.2 ± 22.5 | 98.9 ± 25.7 | 93.2 ± 17.7 | 0.002 |
| Fasting serum insulin (μU/mL) | 9.2 ± 4.9 | 9.8 ± 5.5 | 8.6 ± 3.9 | 0.003 |
| HOMA-IR index | 2.2 ± 1.4 | 2.4 ± 1.6 | 2.0 ± 1.1 | <0.001 |
| QUICKI | 0.35 ± 0.25 | 0.34 ± 0.25 | 0.35 ± 0.24 | <0.001 |
| Adiponectin (mg/L) | 7.2 ± 5.2 | 5.4 ± 4.0 | 9.1 ± 5.6 | <0.001 |
| Resistin (ng/mL) | 18.6 ± 14.3 | 17.9 ± 12.2 | 19.2 ± 16.4 | 0.234 |
| Lipocalin 2 (ng/mL) | 80.4 ± 52.0 | 83.9 ± 59.8 | 76.5 ± 41.7 | 0.078 |
| CRP (mg/L) | 1.08 ± 1.39 | 1.10 ± 1.38 | 1.07 ± 1.40 | 0.186 |
| Fibrinogen (mg/dL) | 264.9 ± 70.3 | 262.8 ± 72.4 | 267.4 ± 68.0 | 0.424 |
| sE-selectin (ng/mL) | 53.3 ± 25.1 | 60.0 ± 25.8 | 45.7 ± 21.8 | <0.001 |
| sP-selectin (ng/mL) | 139.9 ± 117.0 | 154.5 ± 131.9 | 123.5 ± 95.5 | 0.001 |
| SAA (mg/L) | 6.1 ± 15.4 | 7.0 ± 19.6 | 5.1 ± 8.9 | 0.131 |
| sICAM1 (ng/mL) | 241.3 ± 110.6 | 243.9 ± 109.2 | 238.4 ± 112.3 | 0.583 |
| sVCAM1 (ng/mL) | 491.0 ± 131.9 | 494.2 ± 148.7 | 487.4 ± 110.3 | 0.652 |
| MMP1 (pg/mL) | 467.2 ± 1151.8 | 338.9 ± 545.4 | 609.7 ± 1560.8 | 0.673 |
| MMP2 (ng/mL) | 126.5 ± 40.9 | 123.2 ± 41.2 | 130.1 ± 40.4 | 0.038 |
| MMP-9 (ng/mL) | 143.5 ± 111.9 | 155.1 ± 115.8 | 130.6 ± 105.9 | 0.007 |
| MCP1 (pg/mL) | 73.0 ± 58.3 | 78.0 ± 66.1 | 67.4 ± 47.6 | 0.010 |
| sTNFRII (pg/mL) | 3270.9 ± 947.5 | 3328.1 ± 981.5 | 3207.4 ± 905.7 | 0.102 |
| Creatinine (mg/dL) | 0.99 ± 0.46 | 1.12 ± 0.44 | 0.84 ± 0.44 | <0.001 |
| eGFR (mL/min/1.73 m2) | 81.03 ± 14.94 | 79.38 ± 13.55 | 82.93 ± 16.21 | 0.007 |
| YKL-40 (ng/mL) | 92.37 ± 90.32 | 95.08 ± 101.50 | 89.14 ± 74.94 | 0.283 |
| 8-OHdG/creatinine (ng/mg) | 38.12 ± 24.44 | 35.65 ± 24.97 | 40.9 ± 23.56 | 0.008 |
| Demographics | Total (n = 86) | Survival ( n = 76) | Mortality (n = 10) | p Value | |
|---|---|---|---|---|---|
| Age | 71.65 ± 10.90 | 71.89 ± 10.05 | 71.67 ± 16.00 | 0.951 | |
| Sex (male/female) | 50/36 | 44/32 | 6/4 | 1.000 | |
| Body mass index | 23.94 ± 3.82 | 24.11 ± 3.69 | 23.46 ± 4.50 | 0.321 | |
| Smoking | 33.3% | 33.8% | 30.0% | 1.000 | |
| Diabetes mellitus | 73.3% | 75.0% | 60.0% | 0.447 | |
| Dyslipidemia | 42.2% | 43.8% | 30.0% | 0.507 | |
| Hypertension | 84.9% | 86.8% | 70.0% | 0.172 | |
| Congestive heart failure | 17.4% | 17.1% | 20.0% | 1.000 | |
| Stroke | 15.1% | 14.5% | 20.0% | 0.644 | |
| End stage renal disease | 41.2% | 38.7% | 60.0% | 0.305 | |
| Coronary artery disease | 55.8% | 56.6% | 50.0% | 0.744 | |
| Rutheford grade | 3: severe claudication | 22.6% | 24.3% | 10% | 0.443 |
| ≥4: critical limb ischemia | 77.4% | 75.7% | 90% | ||
| YKL-40 (ng/mL) | 406.74 ± 286.66 | 405.19 ± 299.00 | 418.47 ± 175.90 | 0.408 | |
| rs4950928 G-allele carriers | 29.1% | 30.3% | 20% | 0.502 | |
2.2. Associations between YKL-40 Levels and Clinical and Biochemical Correlates
| Clinical Biochemical Parameters | Unadjusted | Adjusted for Age and Sex | ||||
|---|---|---|---|---|---|---|
| r | p Value | r | p Value | Adjusted p | ||
| Anthropology | Age | 0.346 | 4.85 × 10−18 | – | – | – |
| Body mass index | 0.022 | 0.596 | −0.008 | 0.853 | NS | |
| Waist circumference | 0.088 | 0.032 | 0.029 | 0.481 | NS | |
| Blood pressure | Systolic BP | 0.122 | 0.005 | −0.015 | 0.734 | NS |
| Diastolic BP | 0.067 | 0.125 | −0.004 | 0.934 | NS | |
| Glucose metabolism | Fasting plasma glucose | 0.082 | 0.049 | 0.069 | 0.099 | NS |
| Fasting serum insulin | 0.064 | 0.127 | 0.074 | 0.077 | NS | |
| HOMA-IR index | 0.085 | 0.041 | 0.094 | 0.024 | NS | |
| QUICKI | −0.062 | 0.136 | −0.067 | 0.109 | NS | |
| Lipid profiles | Total cholesterol | 0.008 | 0.843 | −0.050 | 0.234 | NS |
| LDL-cholesterol | −0.083 | 0.046 | −0.146 | 4.34 × 10−4 | 0.013 | |
| HDL-cholesterol | −0.062 | 0.136 | −0.065 | 0.121 | NS | |
| Triglyceride | 0.226 | 3.95 × 10−8 | 0.220 | 9.4 × 10−8 | 2.82 × 10−6 | |
| Renal function | Microalbumin/creatinine | 0.123 | 0.003 | 0.096 | 0.021 | NS |
| eGFR | −0.186 | 3.25 × 10−5 | 1.9 × 10−4 | 0.997 | NS | |
| Inflammation marker | CRP | 0.172 | 3.25 × 10−5 | 0.142 | 0.001 | 0.003 |
| Fibrinogen | 0.127 | 0.002 | 0.058 | 0.158 | NS | |
| sE-selectin | 0.147 | 3.6 × 10−4 | 0.144 | 0.001 | 0.003 | |
| sP-selectin | −0.023 | 0.584 | −0.047 | 0.259 | NS | |
| sVCAM1 | 0.211 | 2.69 × 10−7 | 0.155 | 1.79 × 10−4 | 0.005 | |
| sICAM1 | 0.107 | 0.010 | 0.094 | 0.023 | NS | |
| TNFRII | 0.196 | 1.81 × 10−6 | 0.150 | 2.82 × 10−4 | 0.008 | |
| MCP1 | 0.037 | 0.372 | 0.022 | 0.600 | NS | |
| MMP1 | −0.032 | 0.443 | −0.023 | 0.572 | NS | |
| MMP2 | 0.052 | 0.211 | 0.008 | 0.854 | NS | |
| MMP9 | 0.014 | 0.735 | 0.038 | 0.366 | NS | |
| SAA | −0.005 | 0.910 | 0.007 | 0.870 | NS | |
| Adipokines | Leptin | 6.57 × 10−5 | 0.999 | 0.023 | 0.571 | NS |
| Resistin | 0.116 | 0.006 | 0.115 | 0.006 | NS | |
| Lipocalin2 | 0.149 | 3.82 × 10−4 | 0.162 | 1.09 × 10−4 | 0.003 | |
| Adiponectin | 0.069 | 0.092 | 0.056 | 0.173 | NS | |
| Oxidative stress | Homocysteine | 0.108 | 0.008 | 0.100 | 0.016 | NS |
| 8-OHdG/creatinine | 0.077 | 0.062 | 0.046 | 0.262 | NS | |
| Atherosclerotic Risk Factors | YKL40 Levels (Means ± SD) (N) | p Value a | p Value b | |
|---|---|---|---|---|
| Current smoker | Noncurrent | 88.64 ± 74.09 (472) | 0.118 | 0.010 |
| Current | 107.42 ± 137.05 (177) | |||
| Hypertension | Without | 88.02 ± 90.44 (472) | 0.001 | 0.499 |
| With | 109.90 ± 88.09 (117) | |||
| Diabetes mellitus | Nil | 90.59 ± 87.87 (558) | 0.102 | 0.557 |
| Yes | 124.37 ± 124.16 (31) | |||
| Obesity | Nonobese | 93.29 ± 103.38 (353) | 0.281 | 0.878 |
| Obese | 91.00 ± 66.33 (236) | |||
| Metabolic syndrome | Nil | 90.89 ± 93.09 (478) | 0.099 | 0.674 |
| Yes | 98.74 ± 77.32 (111) | |||
| Insulin resistance | Nil | 87.82 ± 75.68 (441) | 0.063 | 0.127 |
| Yes | 105.92 ± 123.46 (148) | |||
2.3. Associations of the CHI3L1 Genotypes/Haplotypes with Circulation Levels of YKL40 in Subjects from Health Examination and in PAD Patients
| CHI3L1 Genotypes | Health Examination Subjects | PAD | |||||
|---|---|---|---|---|---|---|---|
| YKL-40 Level Means ± SD (N) | p Value | Adjusted p | YKL-40 Level Means ± SD (N) | * p Value | Adjusted p | ||
| rs10399931 | CC | 104.77 ± 110.27 (232) | 3.12 × 10−6 | 1.87 × 10−5 | 548.74 ± 356.70 (30) | 0.011 | 0.066 |
| CT | 82.79 ± 65.46 (267) | 346.76 ± 197.28 (40) | |||||
| TT | 74.98 ± 88.40 (79) | 290.43 ± 232.65 (16) | |||||
| CC | 104.77 ± 110.27 (232) | 6 × 10−10 | 3.6 × 10−9 | 548.74 ± 356.70 (30) | 0.001 | 0.006 | |
| CT + TT | 81.01 ± 71.29 (346) | 330.67 ± 207.41 (56) | |||||
| rs10399805 | AA | 102.67 ± 114.85 (42) | 0.328 | 1.00 | 568.36 ± 417.22 (10) | 0.072 | 0.432 |
| AG | 87.79 ± 71.23 (230) | 452.92 ± 282.13 (32) | |||||
| GG | 93.14 ± 98.01 (308) | 336.42 ± 236.43 (44) | |||||
| AA + AG | 90.09 ± 79.44 (272) | 0.362 | 1.00 | 480.40 ± 317.60 (42) | 0.074 | 0.444 | |
| GG | 93.14 ± 98.01 (308) | 336.42 ± 236.43 (44) | |||||
| rs4950928 | CC | 100.99 ± 99.59 (404) | 8.36 × 10−7 | 5.02 × 10−6 | 467.63 ± 302.62 (61) | 1.94 × 10−4 | 1.16 × 10−3 |
| CG | 71.64 ± 56.42 (154) | 283.39 ± 174.75 (21) | |||||
| GG | 59.65 ± 53.66 (21) | 125.73 ± 77.11 (4) | |||||
| CC | 100.99 ± 99.59 (404) | 2.29 × 10−10 | 1.37 × 10−9 | 467.63 ± 302.62 (61) | 6.61 × 10−6 | 3.97 × 10−5 | |
| CG + GG | 70.20 ± 56.09 (175) | 258.17 ± 172.25 (25) | |||||
2.4. Associations Analysis of YKL-40 with Clinical Parameters and Various Biomarker Levels
2.5. Comparison of Circulating YKL-40 Level and CHI3L1 Gene Variants between Subjects from Health Examination and PAD Patients
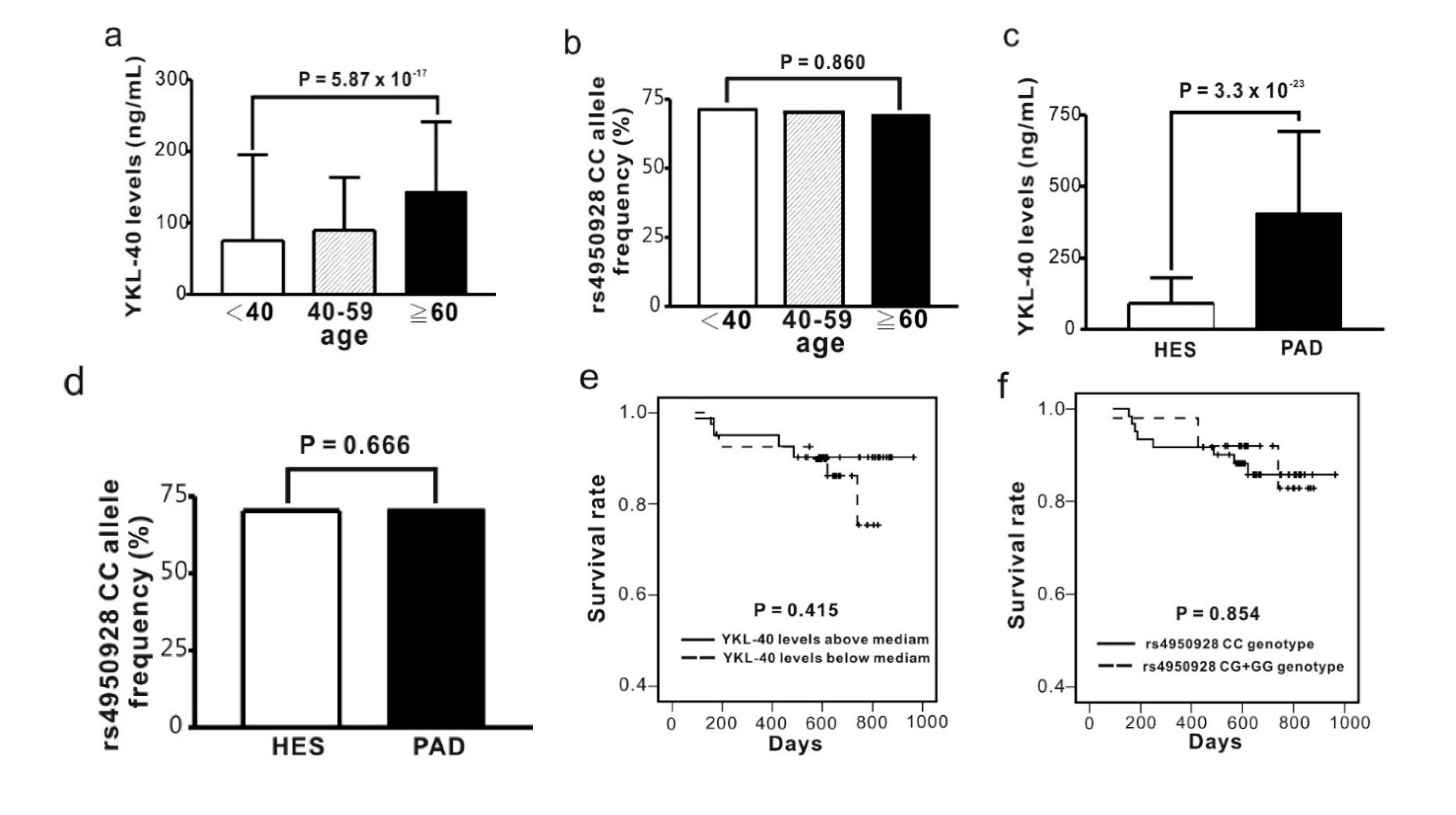
| Haplotypes | Health Examination Subjects | PAD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SNP1 | SNP2 | SNP3 | Frequency | Estimate | p Value | Adjusted p | Frequency | Estimate | * p Value | Adjusted p | |
| H1 | C | G | C | 35.22% | 0.172 | 1.78 × 10−8 | 7.12 × 10−8 | 27.91% | 0.248 | 0.037 | 0.148 |
| H2 | C | A | C | 26.93% | 0.049 | 0.136 | 0.544 | 30.23% | 0.179 | 0.086 | 0.344 |
| H3 | T | G | C | 21.04% | −0.067 | 0.066 | 0.264 | 25.00% | 0.037 | 0.343 | 1.000 |
| H4 | T | G | G | 15.30% | −0.252 | 1.72 × 10−10 | 6.88 × 10−8 | 16.86% | −0.560 | 1.06 × 10−6 | 4.24 × 10−6 |
3. Discussion
3.1. Association between YKL-40 Levels and CHI3L1 Gene Promoter Variants and the Risk of Atherosclerotic Cardiovascular Disease
3.2. Association of Clinical Parameters with YKL-40 Level
3.3. Roles of Association between YKL-40 Level and Biomarker Levels
4. Material and Methods
4.1. Study Population
4.2. Genomic DNA Extraction and Genotyping
4.3. Laboratory Examination
4.4. Assays
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Johansen, J.S. Studies on serum YKL-40 as a biomarker in diseases with inflammation, tissue remodelling, fibroses and cancer. Dan. Med. Bull. 2006, 53, 172–209. [Google Scholar]
- Rathcke, C.N.; Vestergaard, H. YKL-40, a new inflammatory marker with relation to insulin resistance and with a role in endothelial dysfunction and atherosclerosis. Inflamm. Res. 2006, 55, 221–227. [Google Scholar]
- Rathcke, C.N.; Vestergaard, H. YKL-40—An emerging biomarker in cardiovascular disease and diabetes. Cardiovasc. Diabetol. 2009, 8, 61. [Google Scholar]
- Lee, C.G.; da Silva, C.A.; Cruz, C.S.D.; Ahangari, F.; Ma, B.; Kang, M.J. Role of chitin and chitinase/chitinase-like proteins in inflammation, tissue remodeling, and injury. Annu. Rev. Physiol. 2011, 73, 479–501. [Google Scholar]
- Boot, R.G.; van Achterberg, T.A.; van Aken, B.E. Strong induction of members of the chitinase family of proteins in atherosclerosis: Chitotriosidase and human cartilage gp-39 expressed in lesion macrophages. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 687–694. [Google Scholar]
- Gong, Z.; Xing, S.; Zheng, F.; Xing, Q. Increased expression of chitinase 3-like 1 in aorta of patients with atherosclerosis and suppression of atherosclerosis in apolipoprotein E-knockout mice by chitinase 3-like 1 gene silencing. Mediat. Inflamm. 2014, 2014, 905463. [Google Scholar]
- Rathcke, C.N.; Johansen, J.S.; Vestergaard, H. YKL-40, a biomarker of inflammation, is elevated in patients with type 2 diabetes and is related to insulin resistance. Inflamm. Res. 2006, 55, 53–59. [Google Scholar]
- Rathcke, C.N.; Persson, F.; Tarnow, L.; Vestergaard, H. YKL-40, a marker of inflammation and endothelial dysfunction, is elevated in patients with type 1 diabetes and increases with levels of albuminuria. Diabetes Care 2009, 32, 323–328. [Google Scholar]
- Zheng, J.L.; Lu, L.; Hu, J.; Zhang, R.Y.; Zhang, Q.; Chen, Q.J.; Shen, W.F. Genetic polymorphisms in chitinase 3-like 1 (CHI3L1) are associated with circulating YKL-40 levels, but not with angiographic coronary artery disease in a Chinese population. Cytokine 2011, 54, 51–55. [Google Scholar]
- Xie, F.; Qian, Q.; Chen, Z. Chitinase 3-like 1 gene-329G/A polymorphism, plasma concentration and risk of coronary heart disease in a Chinese population. Gene 2012, 10, 499,135–138. [Google Scholar]
- Batinic, K.; Höbaus, C.; Grujicic, M.; Steffan, A.; Jelic, F.; Lorant, D.; Hörtenhuber, T.; Hoellerl, F. YKL-40 is elevated in patients with peripheral arterial disease and diabetes or pre-diabetes. Atherosclerosis 2012, 222, 557–563. [Google Scholar]
- Rathcke, C.N.; Thomsen, S.B.; Linneberg, A. Variations of CHI3L1, levels of the encoded glycoprotein YKL-40 and prediction of fatal and non-fatal ischemic stroke. PLoS One 2012, 7, e43498. [Google Scholar]
- Johansen, J.S.; Pedersen, A.N.; Schroll, M. High serum YKL-40 level in a cohort of octogenarians is associated with increased risk of all-cause mortality. Clin. Exp. Immunol. 2008, 151, 260–266. [Google Scholar]
- Johansen, J.S.; Bojesen, S.E.; Tybjaerg-Hansen, A.; Mylin, A.K.; Price, P.A.; Nordestgaard, B.G. Plasma YKL-40 and total and disease-specific mortality in the general population. Clin. Chem. 2010, 56, 1580–1591. [Google Scholar]
- Kastrup, J.; Johansen, J.S.; Winkel, P.; Hansen, J.F.; Hildebrandt, P.; Jensen, G.B.; Jespersen, C.M.; Kjøller, E.; Kolmos, H.J.; Lind, I.; et al. High serum YKL-40 concentration is associated with cardiovascular and all-cause mortality in patients with stable coronary artery disease. Eur. Heart J. 2009, 30, 1066–1072. [Google Scholar]
- Rathcke, C.N.; Raymond, I.; Kistorp, C.; Hildebrandt, P.; Faber, J.; Vestergaard, H. Low grade inflammation as measured by levels of YKL-40: Association with an increased overall and cardiovascular mortality rate in an elderly population. Int. J. Cardiol. 2010, 143, 35–42. [Google Scholar]
- Kruit, A.; Grutters, J.C.; Ruven, H.J.; van Moorsel, C.C.; van den Bosch, J.M. A CHI3L1 gene polymorphism is associated with serum levels of YKL-40, a novel sarcoidosis marker. Respir. Med. 2007, 101, 1563–1571. [Google Scholar]
- Zhao, X.; Tang, R.; Gao, B.; Shi, Y.; Zhou, J.; Guo, S.; Zhang, J.; Wang, Y.; Tang, W.; Meng, J.; et al. Functional variants in the promoter region of Chitinase 3-like 1 (CHI3L1) and susceptibility to schizophrenia. Am. J. Hum. Genet. 2007, 80, 12–18. [Google Scholar]
- Ober, C.; Tan, Z.; Sun, Y.; Possick, J.D.; Pan, L.; Nicolae, R.; Radford, S.; Parry, R.R.; Heinzmann, A.; Deichmann, K.A.; et al. Effect of variation in CHI3L1 on serum YKL-40 level, risk of asthma, and lung function. N. Engl. J. Med. 2008, 358, 1682–1691. [Google Scholar]
- Yamamori, H.; Hashimoto, R.; Ohi, K.; Yasuda, Y.; Fukumoto, M.; Kasahara, E.; Sekiyama, A.; Umeda-Yano, S.; Okada, T.; Iwase, M.; et al. A promoter variant in the chitinase 3-like 1 gene is associated with serum YKL-40 level and personality trait. Neurosci. Lett. 2012, 513, 204–208. [Google Scholar]
- Henningsen, K.M.; Therkelsen, S.K.; Johansen, J.S.; Bruunsgaard, H.; Svendsen, J.H. Plasma YKL-40, a new biomarker for atrial fibrillation? Europace 2009, 11, 1032–1036. [Google Scholar]
- Thomsen, S.B.; Rathcke, C.N.; Skaaby, T.; Linneberg, A.; Vestergaard, H. The association between genetic variations of CHI3L1, levels of the encoded glycoprotein YKL-40 and the lipid profile in a Danish population. PLoS One 2012, 7, e47094. [Google Scholar]
- Rosén, C.; Andersson, C.H.; Andreasson, U.; Molinuevo, J.L.; Bjerke, M.; Rami, L.; Lladó, A.; Blennow, K.; Zetterberg, H. Increased level of Chitotriosidase and YKL-40 in cerebrospinal fluid from patients with Alzheimer’s disease. Dement. Geriatr. Cogn. Disord. EXTRA 2014, 4, 297–304. [Google Scholar]
- Antonell, A.; Mansilla, A.; Rami, L.; Lladó, A.; Iranzo, A.; Olives, J.; Balasa, M.; Sánchez-Valle, R.; Molinuevo, J.L. Cerebrospinal fluid level of YKL-40 protein in preclinical and prodromal Alzheimer’s disease. J. Alzheimer’s Dis. 2014, 42, 901–908. [Google Scholar]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Ramírez, B.; Rotellar, F.; Valentí, V.; Silva, C.; Gil, M.J.; Salvador, J.; Frühbeck, G.; et al. Increased circulating and visceral adipose tissue expression levels of YKL-40 in obesity-associated type 2 diabetes are related to inflammation: Impact of conventional weight loss and gastric bypass. J. Clin. Endocrinol. Metab. 2011, 96, 200–209. [Google Scholar]
- Nishikawa, K.C.; Millis, A.J. gp38k (CHI3L1) is a novel adhesion and migration factor for vascular cells. Exp. Cell Res. 2003, 287, 79–87. [Google Scholar]
- Conrozier, T.; Carlier, M.C.; Mathieu, P.; Colson, F.; Debard, A.L.; Richard, S.; Favret, H.; Bienvenu, J.; Vignon, E. Serum levels of YKL-40 and C reactive protein in patients with hip osteoarthritis and healthy subjects: A cross sectional study. Ann. Rheum. Dis. 2000, 59, 828–831. [Google Scholar]
- Matsumoto, T.; Tsurumoto, T. Serum YKL-40 levels in rheumatoid arthritis: Correlations between clinical and laborarory parameters. Clin. Exp. Rheumatol. 2001, 19, 655–660. [Google Scholar]
- Wang, Y.; Ripa, R.S.; Johansen, J.S.; Gabrielsen, A.; Steinbruchel, D.A.; Friis, T.; Bindslev, L.; Haack-Sørensen, M.; Jørgensen, E.; Kastrup, J. YKL-40 a new biomarker in patients with acute coronary syndrome or stable coronary artery disease. Scand. Cardiovasc. J. 2008, 42, 295–302. [Google Scholar]
- Nielsen, A.R.; Erikstrup, C.; Johansen, J.S.; Fischer, C.P.; Plomgaard, P.; Krogh-Madsen, R.; Taudorf, S.; Lindegaard, B.; Pedersen, B.K. Plasma YKL-40: A BMI-independent marker of type 2 diabetes. Diabetes 2008, 57, 3078–3082. [Google Scholar]
- Rathcke, C.N.; Holmkvist, J.; Jørgensen, T.; Borch-Johnsen, K.; Hansen, T.; Pedersen, O.B.; Vestergaard, H. Variation in CHI3LI in relation to type 2 diabetes and related quantitative traits. PLoS One 2009, 4, e5469. [Google Scholar]
- Iwata, T.; Kuwajima, M.; Sukeno, A.; Ishimaru, N.; Hayashi, Y.; Wabitsch, M.; Mizusawa, N.; Itakura, M.; Yoshimoto, K. YKL-40 secreted from adipose tissue inhibits degradation of type I collagen. Biochem. Biophys. Res. Commun. 2009, 388, 511–516. [Google Scholar]
- Briguori, C.; Quintavalle, C.; Donnarumma, E.; Condorelli, G. Novel biomarkers for contrast-induced acute kidney injury. Biomed. Res. Int. 2014, 2014, 568738. [Google Scholar]
- Hsu, L.A.; Chang, C.J.; Wu, S.; Teng, M.S.; Chou, H.H.; Chang, H.H.; Chang, P.Y.; Ko, Y.L. Association between functional variants of the ICAM1 and CRP genes and metabolic syndrome in Taiwanese subjects. Metabolism 2010, 59, 1710–1716. [Google Scholar]
- Ko, Y.L.; Hsu, L.A.; Hsu, K.H.; Ko, Y.H.; Lee, Y.S. The interactive effects of hepatic lipase gene promoter polymor-phisms with sex and obesity on high-density-lipoprotein cholesterol levels in Taiwanese–Chinese. Atherosclerosis 2004, 172, 135–142. [Google Scholar]
- Teng, M.S.; Hsu, L.A.; Wu, S.; Chou, H.H.; Chang, C.J.; Sun, Y.Z.; Juan, S.H.; Ko, Y.L. Mediation analysis reveals a sex-dependent association between ABO gene variants and TG/HDL-C ratio that is suppressed by sE-selectin level. Atherosclerosis 2013, 228, 406–412. [Google Scholar]
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative insulin sensitivity check index: A simple, accurate method for assessing insulin sensitivity in humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410. [Google Scholar]
- Matsuo, Y.; Kurita, M.; Park, J.K.; Tanaka, K.; Nakagawa, T.; Kawamukai, M.; Matsuda, H. Purification, characterization and gene analysis of N-acetylglucosaminidase from Enterobacter sp. G-1. Biosci. Biotechnol. Biochem. 1999, 63, 1261–1268. [Google Scholar]
- Wu, T.L.; Tsao, K.C.; Chang, C.P.; Li, C.N.; Sun, C.F.; Wu, J.T. Development of ELISA on microplate for serum C-reactive protein and establishment of age-dependent normal reference range. Clin. Chim. Acta 2002, 322, 163–168. [Google Scholar]
- Chan, E.C.; Chang, C.P.; Wu, T.L.; Wu, J.T. Enzymatic assay of homocysteine on microtiter plate or a TECAN analyzer using crude lysate containing recombinant methionine γ-lyase. Ann. Clin. Lab. Sci. 2005, 35, 155–160. [Google Scholar]
- Wu, T.L.; Tsai, I.C.; Chang, P.Y.; Tsao, K.C.; Sun, C.F.; Wu, L.L.; Wu, J.T. Establishment of an in-house ELISA and the reference range for serum amyloid A (SAA): Complementarity between SAA and C-reactive protein as markers of inflammation. Clin. Chim. Acta 2007, 376, 72–76. [Google Scholar]
- Chiou, C.C.; Chang, P.Y.; Chan, E.C.; Wu, T.L.; Tsao, K.C.; Wu, J.T. Urinary 8-hydroxydeoxyguanosine and its analogs as DNA marker of oxidative stress: Development of an ELISA and measurement in both bladder and prostate cancers. Clin. Chim. Acta 2003, 334, 87–94. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.; Hsu, L.-A.; Cheng, S.-T.; Teng, M.-S.; Yeh, C.-H.; Sun, Y.-C.; Huang, H.-L.; Ko, Y.-L. Circulating YKL-40 Level, but not CHI3L1 Gene Variants, Is Associated with Atherosclerosis-Related Quantitative Traits and the Risk of Peripheral Artery Disease. Int. J. Mol. Sci. 2014, 15, 22421-22437. https://doi.org/10.3390/ijms151222421
Wu S, Hsu L-A, Cheng S-T, Teng M-S, Yeh C-H, Sun Y-C, Huang H-L, Ko Y-L. Circulating YKL-40 Level, but not CHI3L1 Gene Variants, Is Associated with Atherosclerosis-Related Quantitative Traits and the Risk of Peripheral Artery Disease. International Journal of Molecular Sciences. 2014; 15(12):22421-22437. https://doi.org/10.3390/ijms151222421
Chicago/Turabian StyleWu, Semon, Lung-An Hsu, Shih-Tsung Cheng, Ming-Sheng Teng, Ching-Hua Yeh, Yu-Chen Sun, Hsuan-Li Huang, and Yu-Lin Ko. 2014. "Circulating YKL-40 Level, but not CHI3L1 Gene Variants, Is Associated with Atherosclerosis-Related Quantitative Traits and the Risk of Peripheral Artery Disease" International Journal of Molecular Sciences 15, no. 12: 22421-22437. https://doi.org/10.3390/ijms151222421
APA StyleWu, S., Hsu, L.-A., Cheng, S.-T., Teng, M.-S., Yeh, C.-H., Sun, Y.-C., Huang, H.-L., & Ko, Y.-L. (2014). Circulating YKL-40 Level, but not CHI3L1 Gene Variants, Is Associated with Atherosclerosis-Related Quantitative Traits and the Risk of Peripheral Artery Disease. International Journal of Molecular Sciences, 15(12), 22421-22437. https://doi.org/10.3390/ijms151222421





