Prenatal Exposure to Arsenic and Cadmium Impacts Infectious Disease-Related Genes within the Glucocorticoid Receptor Signal Transduction Pathway
Abstract
:1. Introduction
2. Results
2.1. The Comparative Genomic Enrichment Method (CGEM)
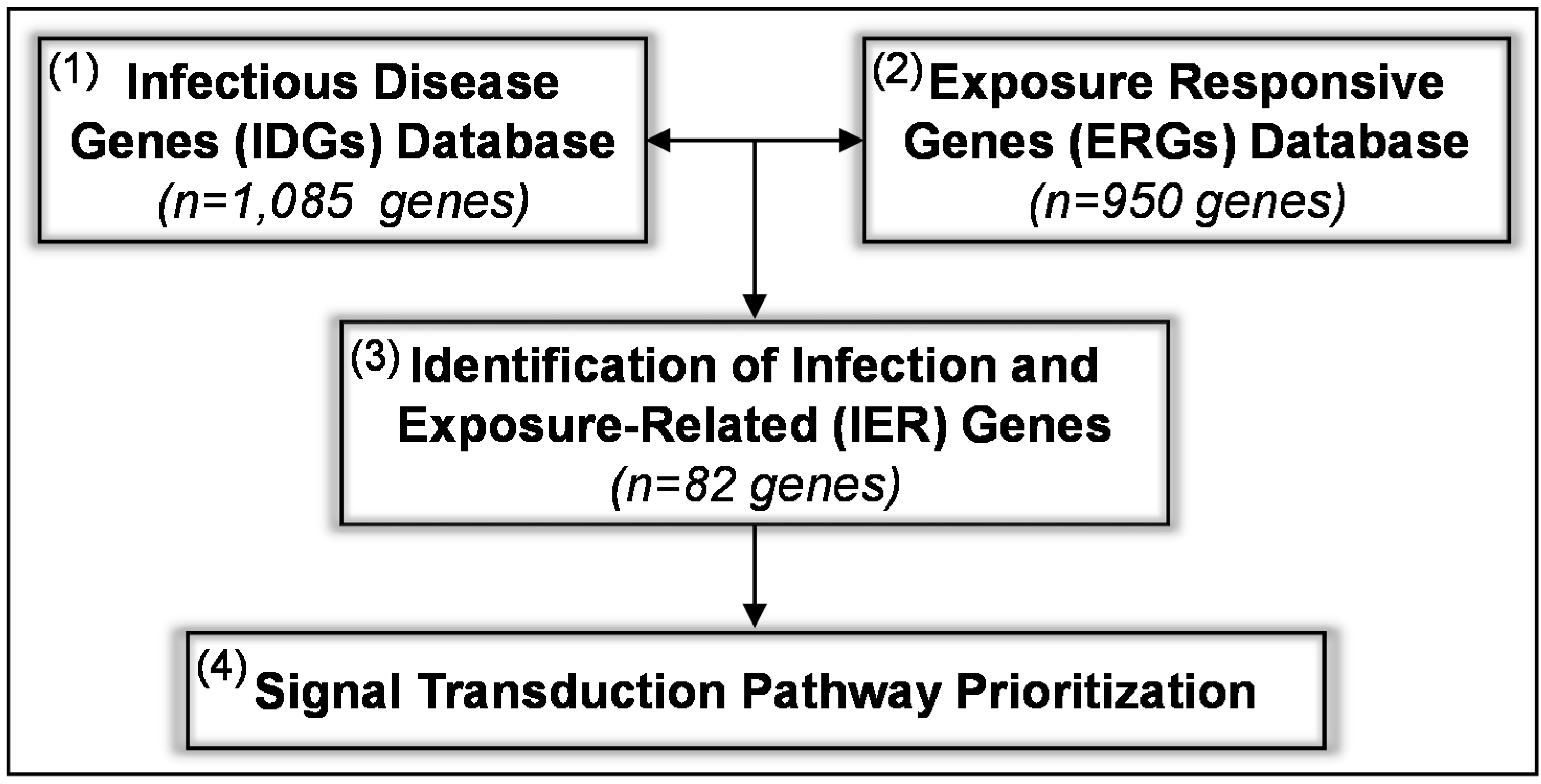
2.2. Application of the CGEM Identifies Infection and Exposure-Related (IER) Genes
| Study | Metalloid/Metal | Biological Media | Measurements in Biological Media a | Measurements in Drinking Water a |
|---|---|---|---|---|
| [39] | iAs | Urine b | GW8: 136 µg/L (26–341) c,d | Not Reported |
| [16] | iAs | Urine b | 64.5 µg/L (6.2–319.7) e | 51.7 µg/L (ND–326) |
| [40] | iAs | Urine b | GW5–14: Median: 66 µg/L (3–740) d | Not Reported |
| [41] | iAs | Urine f | GW24–28: Median: 4.4 μg/L (1.8–11.9) e | 0.36 µg/L (0.02–3.55) g |
| [42] | iAs | Toenail Clippings | 4.8 µg/g (0.1–68.63) | Not Reported |
| [43] | iAs | Cord blood | 5.79 µg/g (1.31–10.37) d,e | 8.38 µg/L (0.17–61.63) |
| [44] | iAs | Urine b | GW ≤ 28: 12.35 µg/L (0.05–260.3) | 14.8 µg/L (1–230) |
| [34] | iAs | Urine b | GW24–28: Median: 4.1 µg/L (0.45–300) | 1.2 µg/L (0.03–100) |
| [36] | iAs | Urine b | 64.5 µg/L (6.2–319.7) e | 51.7 µg/L (ND–326) |
| [35] | iAs | Urine b | 64.5 µg/L (6.2–319.7) e | 51.7 µg/L (ND–326) |
| [33] | Cd | Peripheral Blood | 0.44 µg/L (ND–1.05) e | Not Reported |
| [45] | Cd | Urine | Median Urine GW8: 0.77 µg/L (0.25–2.4) h | Not Reported |
| Peripheral Blood | Median Blood GW14: 1.3 (0.54–3.1) d,h µg/kg |
| Study | Metalloid | Biological Media | Measurements in Biological Media a | Measurements in Drinking Water a | Health Endpoint | Duration of Health Endpoint Observation |
|---|---|---|---|---|---|---|
| [25] | iAs | Urine b | GW8: 152 µg/L (1–1211) | Not Reported | LRTI, Severe | Mean: 75 Days |
| GW30: 166 µg/L (2–1440) | LRTI, Diarrhea | |||||
| [26] | iAs | Urine b | GW6–10: 152 µg/L (1–2020) | Not Reported | ARI c | 12 Months |
| GW30: 146 µg/L (4–1126) | ||||||
| [15] | iAs | Urine d | GW24–28: 6 µg/L (0.45–58.3) | 5.2 µg/L (0.01–67.5) | Infection e, LRTI, URTI | 4 Months |
2.3. The IER Genes Are Enriched for Involvement in the Glucocorticoid Receptor (GR) Signal Transduction Pathway
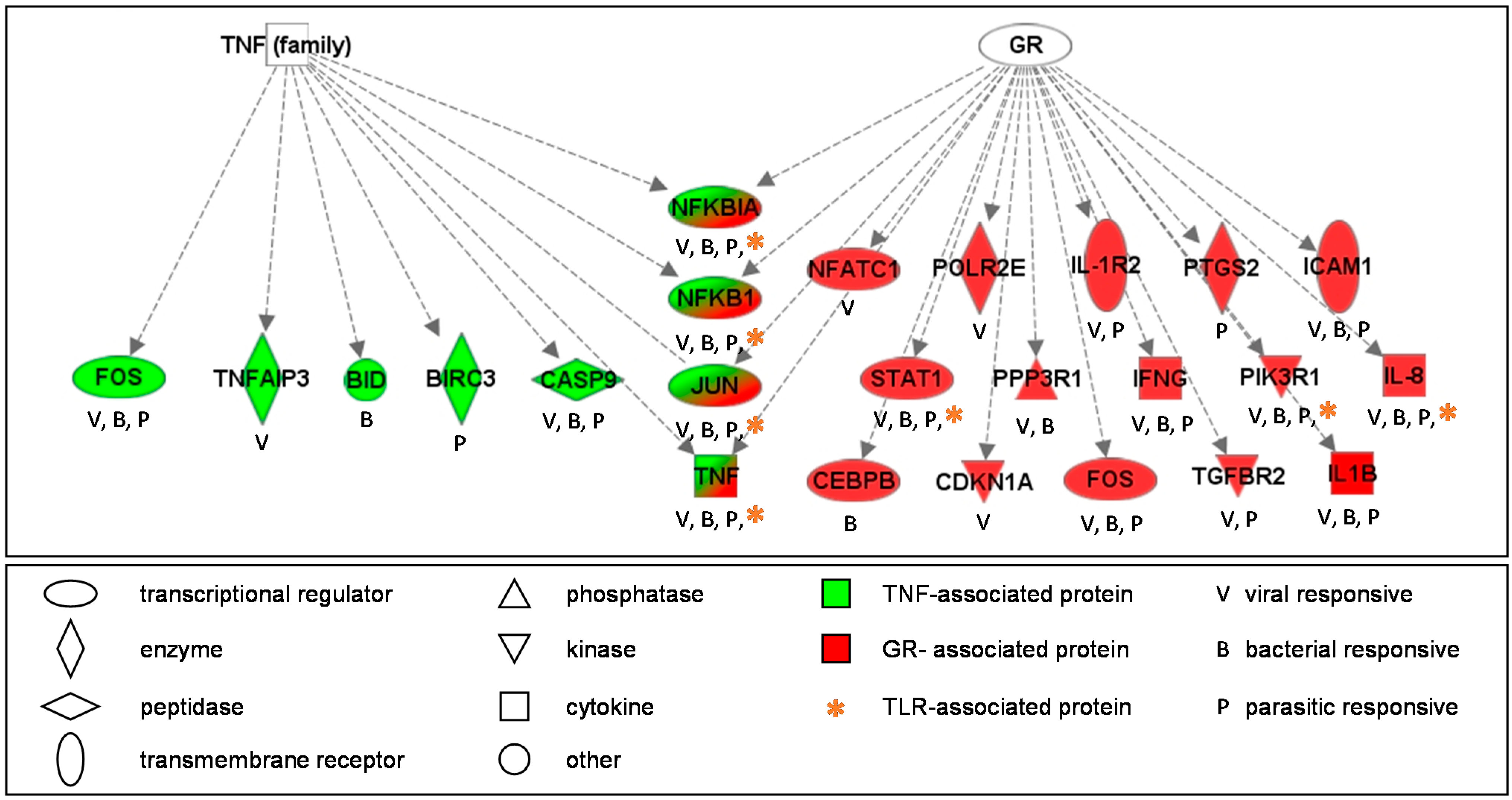
2.4. Validation of ERG-Association with iAs and Cd Using the Comparative Toxicogenomics Database (CTD)
3. Discussion
4. Experimental Section
4.1. Identifying Infectious Disease Genes (IDGs)
4.2. Identifying Exposure Responsive Genes (ERGs)
4.3. Pathway and Upstream Regulator Analysis
4.4. Validating Gene-Environment Interactions with an Alternative Genomics Database
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO World Health Organization (WHO). Burden of Disease: Dalys. Available online: http://www.who.int/healthinfo/global_burden_disease/GBD_report_2004update_part4.pdf (accessed on 26 August 2014).
- Smith, A.H.; Lingas, E.O.; Rahman, M. Contamination of drinking-water by arsenic in Bangladesh: A public health emergency. Bull. World Health Organ. 2000, 78, 1093–1103. [Google Scholar] [PubMed]
- WHO World Health Organization (WHO). The Top 10 Causes of Death. Available online: http://www.who.int/mediacentre/factsheets/fs310/en/ (accessed on 26 August 2014).
- Scholthof, K.B. The disease triangle: Pathogens, the environment and society. Nat. Rev. Microbiol. 2007, 5, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Collinge, S.K.; Ray, C. Disease Ecology: Community Structure and Pathogen Dynamics; Oxford University Press: Oxford, UK; New York, NY, USA, 2006; Volume xii, p. 227. [Google Scholar]
- Arcavi, L.; Benowitz, N.L. Cigarette smoking and infection. Arch. Intern. Med. 2004, 164, 2206–2216. [Google Scholar] [CrossRef] [PubMed]
- Tchounwou, P.B.; Ayensu, W.K.; Ninashvili, N.; Sutton, D. Environmental exposure to mercury and its toxicopathologic implications for public health. Environ. Toxicol. 2003, 18, 149–175. [Google Scholar] [CrossRef] [PubMed]
- Kamanyire, R.; Karalliedde, L. Organophosphate toxicity and occupational exposure. Occup. Med. 2004, 54, 69–75. [Google Scholar] [CrossRef]
- ATSDR Agency for Toxic Substances and Disease Registry (ATSDR). 2013. Hazardous Substance Priority List. 2013. Available online: http://www.atsdr.cdc.gov/spl/ (accessed on 26 August 2014). [Google Scholar]
- Uddin, R.; Huda, N.H. Arsenic poisoning in Bangladesh. Oman. Med. J. 2011, 26, 207. [Google Scholar] [CrossRef] [PubMed]
- Naujokas, M.F.; Anderson, B.; Ahsan, H.; Aposhian, H.V.; Graziano, J.H.; Thompson, C.; Suk, W.A. The broad scope of health effects from chronic arsenic exposure: Update on a worldwide public health problem. Environ. Health Perspect. 2013, 121, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Järup, L.; Akesson, A. Current status of cadmium as an environmental health problem. Toxicol. Appl. Pharmacol. 2009, 238, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Steinmaus, C.; Ferreccio, C.; Acevedo, J.; Yuan, Y.; Liaw, J.; Duran, V.; Cuevas, S.; Garcia, J.; Meza, R.; Valdes, R.; et al. Increased lung and bladder cancer incidence in adults after in utero and early-life arsenic exposure. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1529–1538. [Google Scholar] [CrossRef]
- Smith, A.H.; Yunus, M.; Khan, A.F.; Ercumen, A.; Yuan, Y.; Smith, M.H.; Liaw, J.; Balmes, J.; von Ehrenstein, O.; Raqib, R.; et al. Chronic respiratory symptoms in children following in utero and early life exposure to arsenic in drinking water in Bangladesh. Int. J. Epidemiol. 2013, 42, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Farzan, S.F.; Karagas, M.R.; Chen, Y. In utero and early life arsenic exposure in relation to long-term health and disease. Toxicol. Appl. Pharmacol. 2013, 272, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Bailey, K.; Fry, R.C. Long-term health consequences of prenatal arsenic exposure: Links to the genome and the epigenome. Rev. Environ. Health 2014, 29, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Dauphine, D.C.; Ferreccio, C.; Guntur, S.; Yuan, Y.; Hammond, S.K.; Balmes, J.; Smith, A.H.; Steinmaus, C. Lung function in adults following in utero and childhood exposure to arsenic in drinking water: Preliminary findings. Int. Arch. Occup. Environ. Health 2011, 84, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Steinmaus, C.M.; Ferreccio, C.; Romo, J.A.; Yuan, Y.; Cortes, S.; Marshall, G.; Moore, L.E.; Balmes, J.R.; Liaw, J.; Golden, T.; et al. Drinking water arsenic in Northern Chile: High cancer risks 40 years after exposure cessation. Cancer Epidemiol. Biomark. Prev. 2013, 22, 623–630. [Google Scholar] [CrossRef]
- Smith, A.H.; Marshall, G.; Liaw, J.; Yuan, Y.; Ferreccio, C.; Steinmaus, C. Mortality in young adults following in utero and childhood exposure to arsenic in drinking water. Environ. Health Perspect. 2012, 120, 1527–1531. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.H.; Marshall, G.; Yuan, Y.; Ferreccio, C.; Liaw, J.; von Ehrenstein, O.; Steinmaus, C.; Bates, M.N.; Selvin, S. Increased mortality from lung cancer and bronchiectasis in young adults after exposure to arsenic in utero and in early childhood. Environ. Health Perspect. 2006, 114, 1293–1296. [Google Scholar] [CrossRef] [PubMed]
- Salpietro, C.D.; Gangemi, S.; Minciullo, P.L.; Briuglia, S.; Merlino, M.V.; Stelitano, A.; Cristani, M.; Trombetta, D.; Saija, A. Cadmium concentration in maternal and cord blood and infant birth weight: A study on healthy non-smoking women. J. Perinat. Med. 2002, 30, 395–399. [Google Scholar] [PubMed]
- Shirai, S.; Suzuki, Y.; Yoshinaga, J.; Mizumoto, Y. Maternal exposure to low-level heavy metals during pregnancy and birth size. J. Environ. Sci. Health A 2010, 45, 1468–1474. [Google Scholar] [CrossRef]
- Hopenhayn, C.; Ferreccio, C.; Browning, S.R.; Huang, B.; Peralta, C.; Gibb, H.; Hertz-Picciotto, I. Arsenic exposure from drinking water and birth weight. Epidemiology 2003, 14, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Hack, M.; Klein, N.K.; Taylor, H.G. Long-term developmental outcomes of low birth weight infants. Future Child. 1995, 5, 176–196. [Google Scholar] [PubMed]
- Rahman, A.; Vahter, M.; Ekstrom, E.C.; Persson, L.A. Arsenic exposure in pregnancy increases the risk of lower respiratory tract infection and diarrhea during infancy in Bangladesh. Environ. Health Perspect. 2011, 119, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Raqib, R.; Ahmed, S.; Sultana, R.; Wagatsuma, Y.; Mondal, D.; Hoque, A.M.; Nermell, B.; Yunus, M.; Roy, S.; Persson, L.A.; et al. Effects of in utero arsenic exposure on child immunity and morbidity in rural Bangladesh. Toxicol. Lett. 2009, 185, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Farzan, S.F.; Korrick, S.; Li, Z.; Enelow, R.; Gandolfi, A.J.; Madan, J.; Nadeau, K.; Karagas, M.R. In utero arsenic exposure and infant infection in a United States cohort: A prospective study. Environ. Res. 2013, 126, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Ahsan, K.B.; Kippler, M.; Mily, A.; Wagatsuma, Y.; Hoque, A.M.; Ngom, P.T.; El Arifeen, S.; Raqib, R.; Vahter, M. In utero arsenic exposure is associated with impaired thymic function in newborns possibly via oxidative stress and apoptosis. Toxicol. Sci. 2012, 129, 305–314. [Google Scholar] [CrossRef]
- Holaskova, I.; Elliott, M.; Hanson, M.L.; Schafer, R.; Barnett, J.B. Prenatal cadmium exposure produces persistent changes to thymus and spleen cell phenotypic repertoire as well as the acquired immune response. Toxicol. Appl. Pharmacol. 2012, 265, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.L.; Brundage, K.M.; Schafer, R.; Tou, J.C.; Barnett, J.B. Prenatal cadmium exposure dysregulates sonic hedgehog and Wnt/β-catenin signaling in the thymus resulting in altered thymocyte development. Toxicol. Appl. Pharmacol. 2010, 242, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.V. The genomics and genetics of human infectious disease susceptibility. Annu. Rev. Genomics Hum. Genet. 2001, 2, 373–400. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.H.; Sarkar, S.N.; Ram, G.C.; Tripathi, H.C. Immunosuppressive effect of subchronic exposure to a mixture of eight heavy metals, found as groundwater contaminants in different areas of India, through drinking water in male rats. Arch. Environ. Contam. Toxicol. 2007, 53, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.P.; Smeester, L.; Rojas, D.; DeBussycher, T.; Wu, M.C.; Wright, F.A.; Zhou, Y.H.; Laine, J.E.; Rager, J.E.; Swamy, G.K.; et al. Cadmium exposure and the epigenome: Exposure-associated patterns of DNA methylation in leukocytes from mother-baby pairs. Epigenetics 2014, 9, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Koestler, D.C.; Avissar-Whiting, M.; Houseman, E.A.; Karagas, M.R.; Marsit, C.J. Differential DNA methylation in umbilical cord blood of infants exposed to low levels of arsenic in utero. Environ. Health Perspect. 2013, 121, 971–977. [Google Scholar] [PubMed]
- Rojas, D.; Rager, J.E.; Smeester, L.; Bailey, K.A.; Drobná, Z.; Rubio-Andrade, M.; Stýblo, M.; García-Vargas, G.; Fry, R.C. Prenatal arsenic exposure and the epigenome: Identifying sites of 5-methyl cytosine alterations that predict functional changes in gene expression in newborn cord blood and subsequent birth outcomes. Toxicol. Sci. 2014. [Google Scholar] [CrossRef]
- Rager, J.E.; Bailey, K.A.; Smeester, L.; Miller, S.K.; Parker, J.S.; Laine, J.E.; Drobná, Z.; Currier, J.; Douillet, C.; Olshan, A.F.; et al. Prenatal arsenic exposure and the epigenome: Altered microRNAs associated with innate and adaptive immune signaling in newborn cord blood. Environ. Mol. Mutagen. 2014, 55, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Concha, G.; Vogler, G.; Lezcano, D.; Nermell, B.; Vahter, M. Exposure to inorganic arsenic metabolites during early human development. Toxicol. Sci. 1998, 44, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Kippler, M.; Hoque, A.M.; Raqib, R.; Ohrvik, H.; Ekström, E.C.; Vahter, M. Accumulation of cadmium in human placenta interacts with the transport of micronutrients to the fetus. Toxicol. Lett. 2010, 192, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Mahabbat-e Khoda, S.; Rekha, R.S.; Gardner, R.M.; Ameer, S.S.; Moore, S.; Ekstrom, E.C.; Vahter, M.; Raqib, R. Arsenic-associated oxidative stress, inflammation, and immune disruption in human placenta and cord blood. Environ. Health Perspect. 2011, 119, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Broberg, K.; Ahmed, S.; Engström, K.; Hossain, M.B.; Jurkovic Mlakar, S.; Bottai, M.; Grandér, M.; Raqib, R.; Vahter, M. Arsenic exposure in early pregnancy alters genome-wide DNA methylation in cord blood, particularly in boys. J. Dev. Orig. Health Dis. 2014, 5, 288–298. [Google Scholar] [CrossRef]
- Fei, D.L.; Koestler, D.C.; Li, Z.; Giambelli, C.; Sanchez-Mejias, A.; Gosse, J.A.; Marsit, C.J.; Karagas, M.R.; Robbins, D.J. Association between in utero arsenic exposure, placental gene expression, and infant birth weight: A US birth cohort study. Environ. Health 2013, 12, 58. [Google Scholar] [CrossRef] [PubMed]
- Fry, R.C.; Navasumrit, P.; Valiathan, C.; Svensson, J.P.; Hogan, B.J.; Luo, M.; Bhattacharya, S.; Kandjanapa, K.; Soontararuks, S.; Nookabkaew, S.; et al. Activation of inflammation/NF-κB signaling in infants born to arsenic-exposed mothers. PLoS Genet. 2007, 3, e207. [Google Scholar] [CrossRef] [PubMed]
- Intarasunanont, P.; Navasumrit, P.; Waraprasit, S.; Chaisatra, K.; Suk, W.A.; Mahidol, C.; Ruchirawat, M. Effects of arsenic exposure on DNA methylation in cord blood samples from newborn babies and in a human lymphoblast cell line. Environ. Health 2012, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Kile, M.L.; Baccarelli, A.; Hoffman, E.; Tarantini, L.; Quamruzzaman, Q.; Rahman, M.; Mahiuddin, G.; Mostofa, G.; Hsueh, Y.M.; Wright, R.O.; et al. Prenatal arsenic exposure and DNA methylation in maternal and umbilical cord blood leukocytes. Environ. Health Perspect. 2012, 120, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Kippler, M.; Engstrom, K.; Mlakar, S.J.; Bottai, M.; Ahmed, S.; Hossain, M.B.; Raqib, R.; Vahter, M.; Broberg, K. Sex-specific effects of early life cadmium exposure on DNA methylation and implications for birth weight. Epigenetics 2013, 8, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Murphy, C.G.; Johnson, R.; Lay, J.M.; Lennon-Hopkins, K.; Saraceni-Richards, C.; Sciaky, D.; King, B.L.; Rosenstein, M.C.; Wiegers, T.C.; et al. The comparative toxicogenomics database: Update 2013. Nucleic Acids Res. 2013, 41, D1104–D1114. [Google Scholar] [CrossRef] [PubMed]
- Chilakapati, J.; Wallace, K.; Ren, H.; Fricke, M.; Bailey, K.; Ward, W.; Creed, J.; Kitchin, K. Genome-wide analysis of BEAS-2B cells exposed to trivalent arsenicals and dimethylthioarsinic acid. Toxicology 2010, 268, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.I.; Sternberg, E.M. Role of the hypothalamic-pituitary-adrenal axis, glucocorticoids and glucocorticoid receptors in toxic sequelae of exposure to bacterial and viral products. J. Endocrinol. 2004, 181, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Kadmiel, M.; Cidlowski, J.A. Glucocorticoid receptor signaling in health and disease. Trends Pharmacol. Sci. 2013, 34, 518–530. [Google Scholar] [CrossRef] [PubMed]
- Simons, S.S., Jr.; Chakraborti, P.K.; Cavanaugh, A.H. Arsenite and cadmium(II) as probes of glucocorticoid receptor structure and function. J. Biol. Chem. 1990, 265, 1938–1945. [Google Scholar] [PubMed]
- Ahir, B.K.; Sanders, A.P.; Rager, J.E.; Fry, R.C. Systems biology and birth defects prevention: Blockade of the glucocorticoid receptor prevents arsenic-induced birth defects. Environ. Health Perspect. 2013, 121, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Bodwell, J.E.; Kingsley, L.A.; Hamilton, J.W. Arsenic at very low concentrations alters glucocorticoid receptor (GR)-mediated gene activation but not GR-mediated gene repression: Complex dose-response effects are closely correlated with levels of activated GR and require a functional GR DNA binding domain. Chem. Res. Toxicol. 2004, 17, 1064–1076. [Google Scholar] [CrossRef] [PubMed]
- Kaltreider, R.C.; Davis, A.M.; Lariviere, J.P.; Hamilton, J.W. Arsenic alters the function of the glucocorticoid receptor as a transcription factor. Environ. Health Perspect. 2001, 109, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Curtin, N.M.; Boyle, N.T.; Mills, K.H.; Connor, T.J. Psychological stress suppresses innate IFN-γ production via glucocorticoid receptor activation: Reversal by the anxiolytic chlordiazepoxide. Brain Behav. Immun. 2009, 23, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Makino, Y.; Miura, T.; Hirano, F.; Okamoto, K.; Komura, K.; Sato, Y.; Makino, I. Ligand-independent activation of the glucocorticoid receptor by ursodeoxycholic acid. Repression of IFN-γ-induced MHC class II gene expression via a glucocorticoid receptor-dependent pathway. J. Immunol. 1996, 156, 1601–1608. [Google Scholar] [PubMed]
- Young, H.A.; Hardy, K.J. Role of interferon-γ in immune cell regulation. J. Leukoc. Biol. 1995, 58, 373–381. [Google Scholar] [PubMed]
- Rossouw, M.; Nel, H.J.; Cooke, G.S.; van Helden, P.D.; Hoal, E.G. Association between tuberculosis and a polymorphic NF-κB binding site in the interferon gamma gene. Lancet 2003, 361, 1871–1872. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, M.M.; Riley, E.M. Innate immunity to malaria. Nat. Rev. Immunol. 2004, 4, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Torres, O.A.; Calzada, J.E.; Beraún, Y.; Morillo, C.A.; González, A.; González, C.I.; Martín, J. Role of the IFNG +874T/A polymorphism in Chagas disease in a Colombian population. Infect. Genet. Evol. 2010, 10, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Song, X.; Berezov, A.; Zhang, G.; Li, Y.; Zhang, H.; Murali, R.; Li, B.; Greene, M.I. Human glucocorticoid-induced TNF receptor ligand regulates its signaling activity through multiple oligomerization states. Proc. Natl. Acad. Sci. USA 2008, 105, 5465–5470. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A. When signaling pathways collide: Positive and negative regulation of toll-like receptor signal transduction. Immunity 2008, 29, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Goeddel, D.V. TNF-R1 signaling: A beautiful pathway. Science 2002, 296, 1634–1635. [Google Scholar] [CrossRef] [PubMed]
- Van der Bruggen, T.; Nijenhuis, S.; van Raaij, E.; Verhoef, J.; van Asbeck, B.S. Lipopolysaccharide-induced tumor necrosis factor α production by human monocytes involves the Raf-1/MEK1-MEK2/ERK1-ERK2 pathway. Infect. Immun. 1999, 67, 3824–3829. [Google Scholar] [PubMed]
- Cabrera, M.; Shaw, M.A.; Sharples, C.; Williams, H.; Castes, M.; Convit, J.; Blackwell, J.M. Polymorphism in tumor necrosis factor genes associated with mucocutaneous leishmaniasis. J. Exp. Med. 1995, 182, 1259–1264. [Google Scholar] [CrossRef] [PubMed]
- Conway, D.J.; Holland, M.J.; Bailey, R.L.; Campbell, A.E.; Mahdi, O.S.; Jennings, R.; Mbena, E.; Mabey, D.C. Scarring trachoma is associated with polymorphism in the tumor necrosis factor α (TNF-α) gene promoter and with elevated TNF-α levels in tear fluid. Infect. Immun. 1997, 65, 1003–1006. [Google Scholar] [PubMed]
- McGuire, W.; Hill, A.V.; Allsopp, C.E.; Greenwood, B.M.; Kwiatkowski, D. Variation in the TNF-α promoter region associated with susceptibility to cerebral malaria. Nature 1994, 371, 508–510. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; McGuire, W.; Mascie-Taylor, C.G.; Saha, B.; Hazra, S.K.; Hill, A.V.; Kwiatkowski, D. Tumor necrosis factor promoter polymorphism and susceptibility to lepromatous leprosy. J. Infect. Dis. 1997, 176, 530–532. [Google Scholar] [CrossRef] [PubMed]
- Falvo, J.V.; Tsytsykova, A.V.; Goldfeld, A.E. Transcriptional control of the TNF gene. Curr. Dir. Autoimmune 2010, 11, 27–60. [Google Scholar]
- Banerjee, N.; Nandy, S.; Kearns, J.K.; Bandyopadhyay, A.K.; Das, J.K.; Majumder, P.; Basu, S.; Banerjee, S.; Sau, T.J.; States, J.C.; et al. Polymorphisms in the TNF-α and IL10 gene promoters and risk of arsenic-induced skin lesions and other nondermatological health effects. Toxicol. Sci. 2011, 121, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Kayama, F.; Yoshida, T.; Elwell, M.R.; Luster, M.I. Role of tumor necrosis factor-α in cadmium-induced hepatotoxicity. Toxicol. Appl. Pharmacol. 1995, 131, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Beane, J.; Sebastiani, P.; Liu, G.; Brody, J.S.; Lenburg, M.E.; Spira, A. Reversible and permanent effects of tobacco smoke exposure on airway epithelial gene expression. Genome Biol. 2007, 8, R201. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xie, Y.; Ward, J.M.; Diwan, B.A.; Waalkes, M.P. Toxicogenomic analysis of aberrant gene expression in liver tumors and nontumorous livers of adult mice exposed in utero to inorganic arsenic. Toxicol. Sci. 2004, 77, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Jirtle, R.L.; Skinner, M.K. Environmental epigenomics and disease susceptibility. Nat. Rev. Genet. 2007, 8, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Marketon, J.I.; Sternberg, E.M. The glucocorticoid receptor: A revisited target for toxins. Toxins 2010, 2, 1357–1380. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Araki, M.; Goto, S.; Hattori, M.; Hirakawa, M.; Itoh, M.; Katayama, T.; Kawashima, S.; Okuda, S.; Tokimatsu, T.; et al. KEGG for linking genomes to life and the environment. Nucleic Acids Res. 2008, 36, D480–D484. [Google Scholar] [CrossRef] [PubMed]
- Bailey, K.A.; Laine, J.; Rager, J.E.; Sebastian, E.; Olshan, A.; Smeester, L.; Drobna, Z.; Styblo, M.; Rubio-Andrade, M.; Garcia-Vargas, G.; et al. Prenatal arsenic exposure and shifts in the newborn proteome: Interindividual differences in Tumor Necrosis Factor (TNF)-responsive signaling. Toxicol. Sci. 2014, 139, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Genomatix; Software; Inc. Genomatic Software Suite. 2014. Available online: http://www.genomatix.de/ (accessed on 27 June 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rager, J.E.; Yosim, A.; Fry, R.C. Prenatal Exposure to Arsenic and Cadmium Impacts Infectious Disease-Related Genes within the Glucocorticoid Receptor Signal Transduction Pathway. Int. J. Mol. Sci. 2014, 15, 22374-22391. https://doi.org/10.3390/ijms151222374
Rager JE, Yosim A, Fry RC. Prenatal Exposure to Arsenic and Cadmium Impacts Infectious Disease-Related Genes within the Glucocorticoid Receptor Signal Transduction Pathway. International Journal of Molecular Sciences. 2014; 15(12):22374-22391. https://doi.org/10.3390/ijms151222374
Chicago/Turabian StyleRager, Julia E., Andrew Yosim, and Rebecca C. Fry. 2014. "Prenatal Exposure to Arsenic and Cadmium Impacts Infectious Disease-Related Genes within the Glucocorticoid Receptor Signal Transduction Pathway" International Journal of Molecular Sciences 15, no. 12: 22374-22391. https://doi.org/10.3390/ijms151222374
APA StyleRager, J. E., Yosim, A., & Fry, R. C. (2014). Prenatal Exposure to Arsenic and Cadmium Impacts Infectious Disease-Related Genes within the Glucocorticoid Receptor Signal Transduction Pathway. International Journal of Molecular Sciences, 15(12), 22374-22391. https://doi.org/10.3390/ijms151222374




