Molecular Characteristics and Biochemical Functions of VpPR10s from Vitis pseudoreticulata Associated with Biotic and Abiotic Stresses
Abstract
:1. Introduction
2. Results
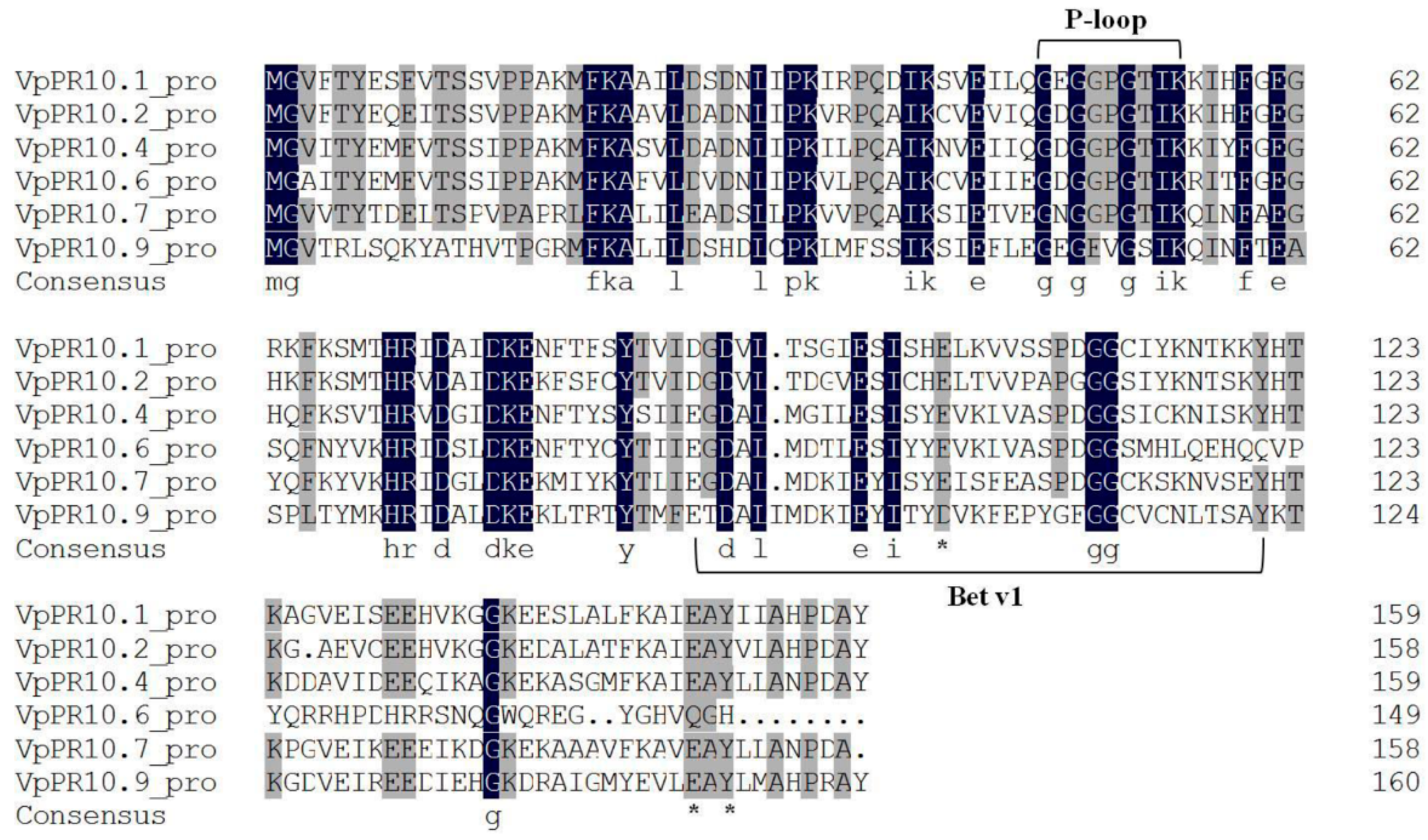
2.1. Sequence Alignment of VpPR10s
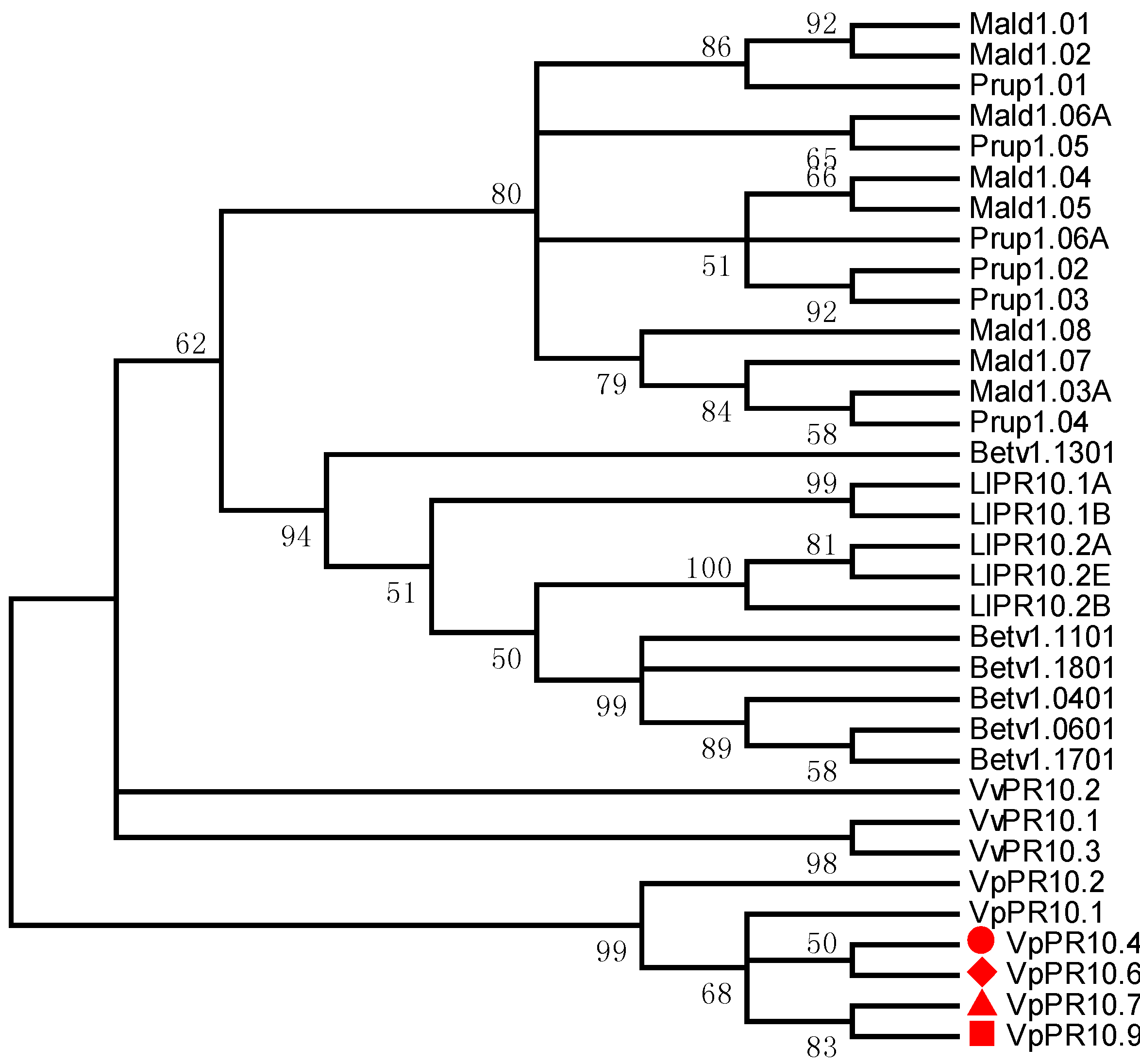
2.2. Phylogenetic Analyses of VpPR10s
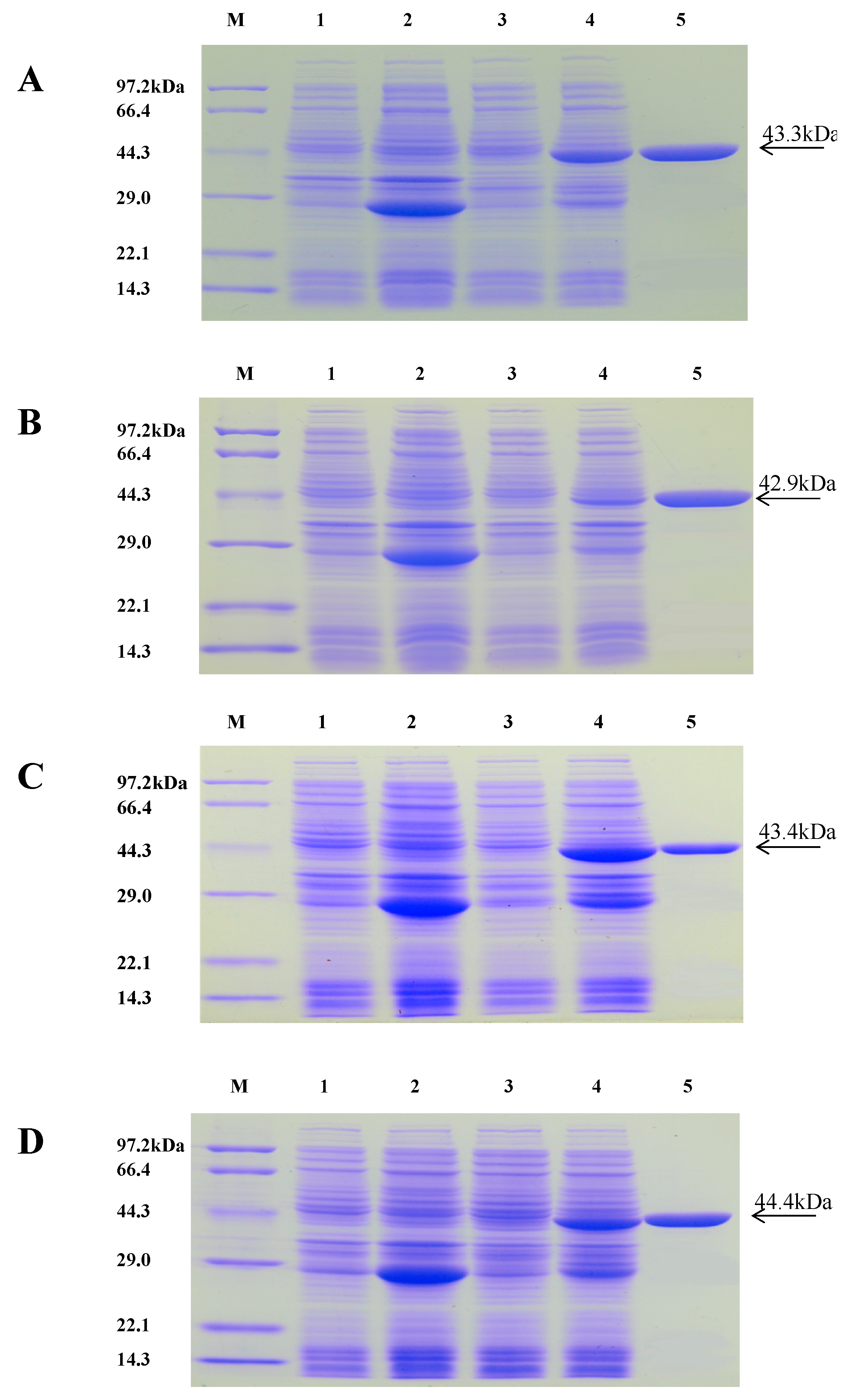
2.3. Purification of Recombinant Proteins
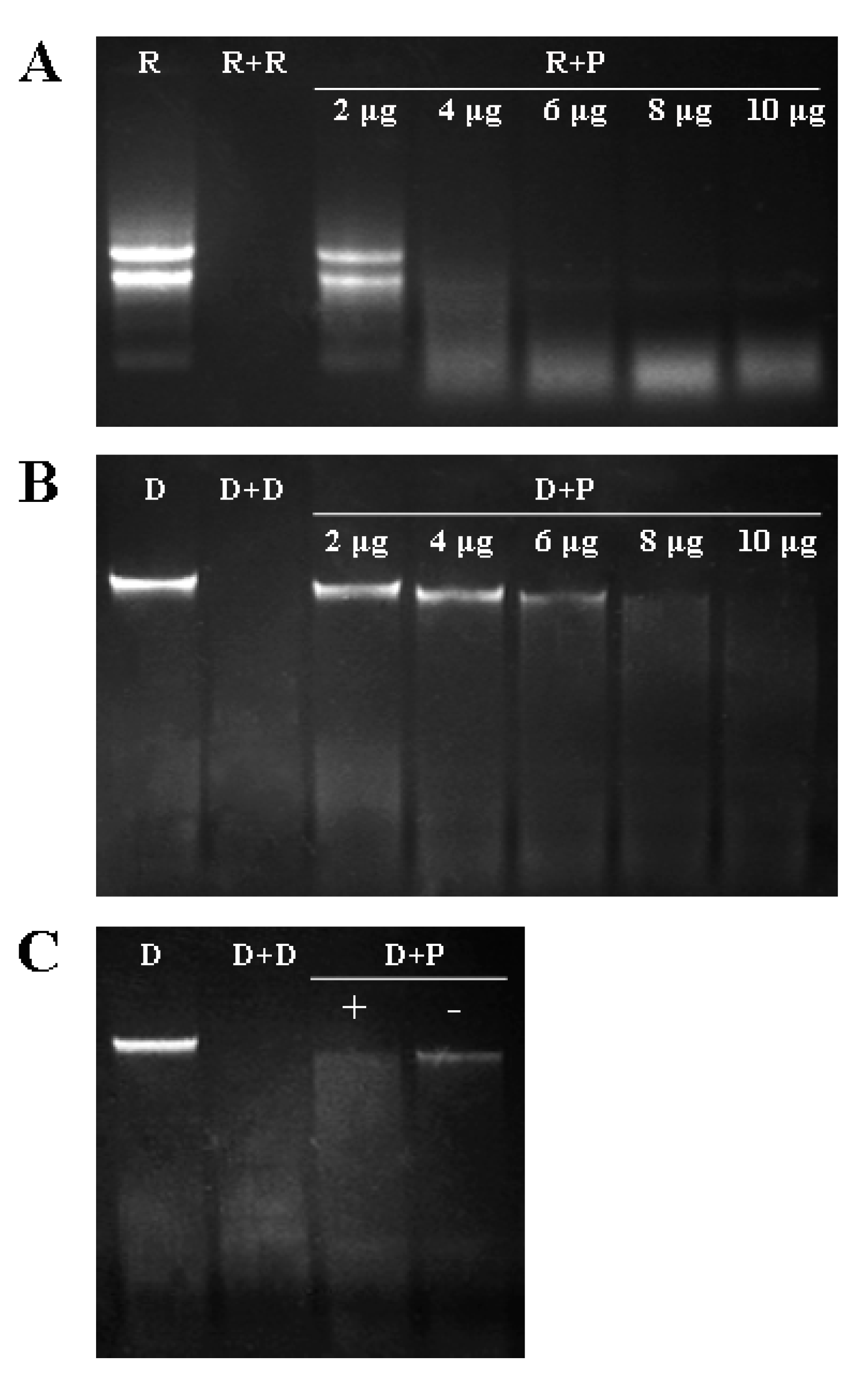
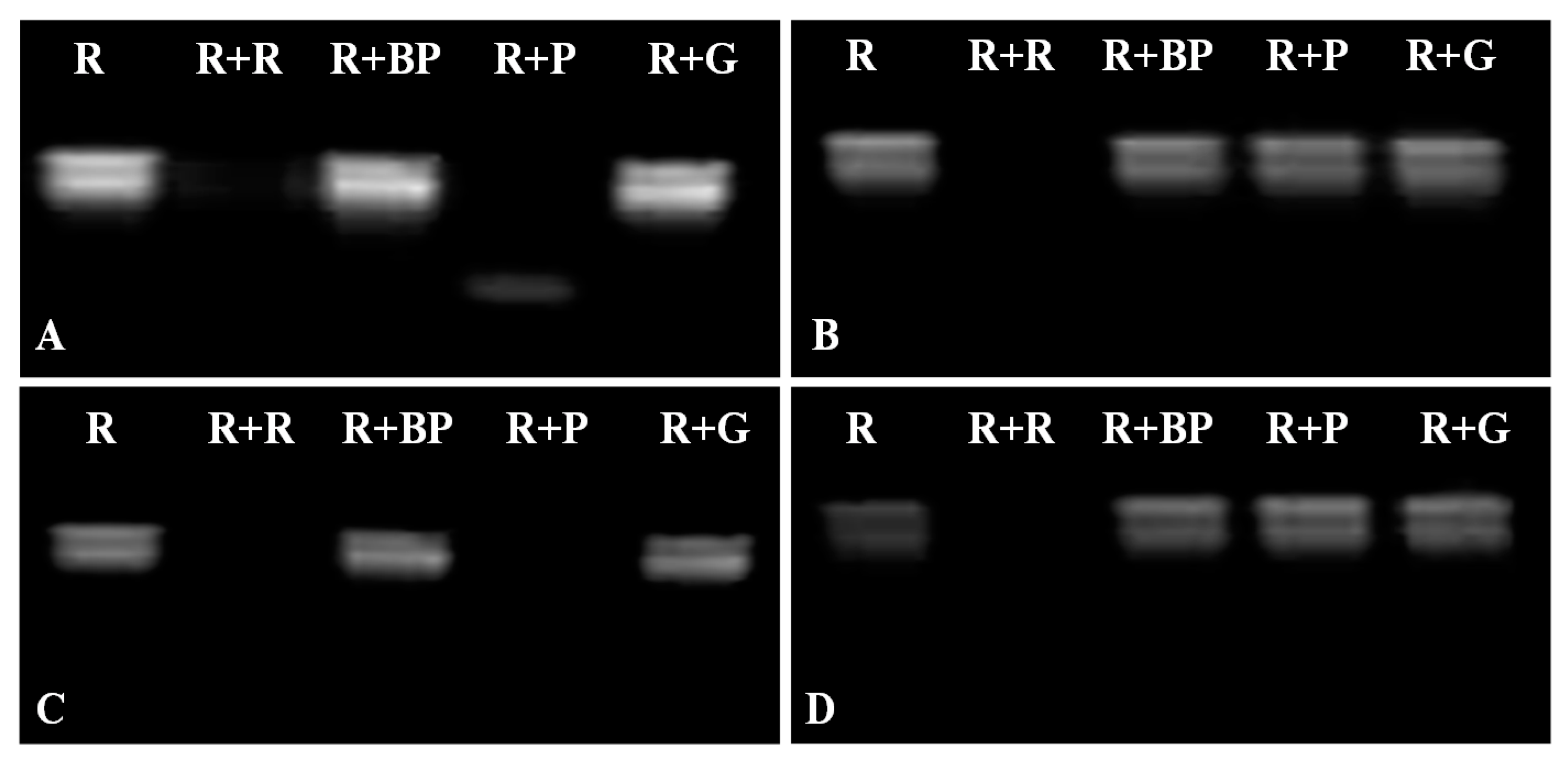
2.4. DNase and RNase Activities of VpPR10s
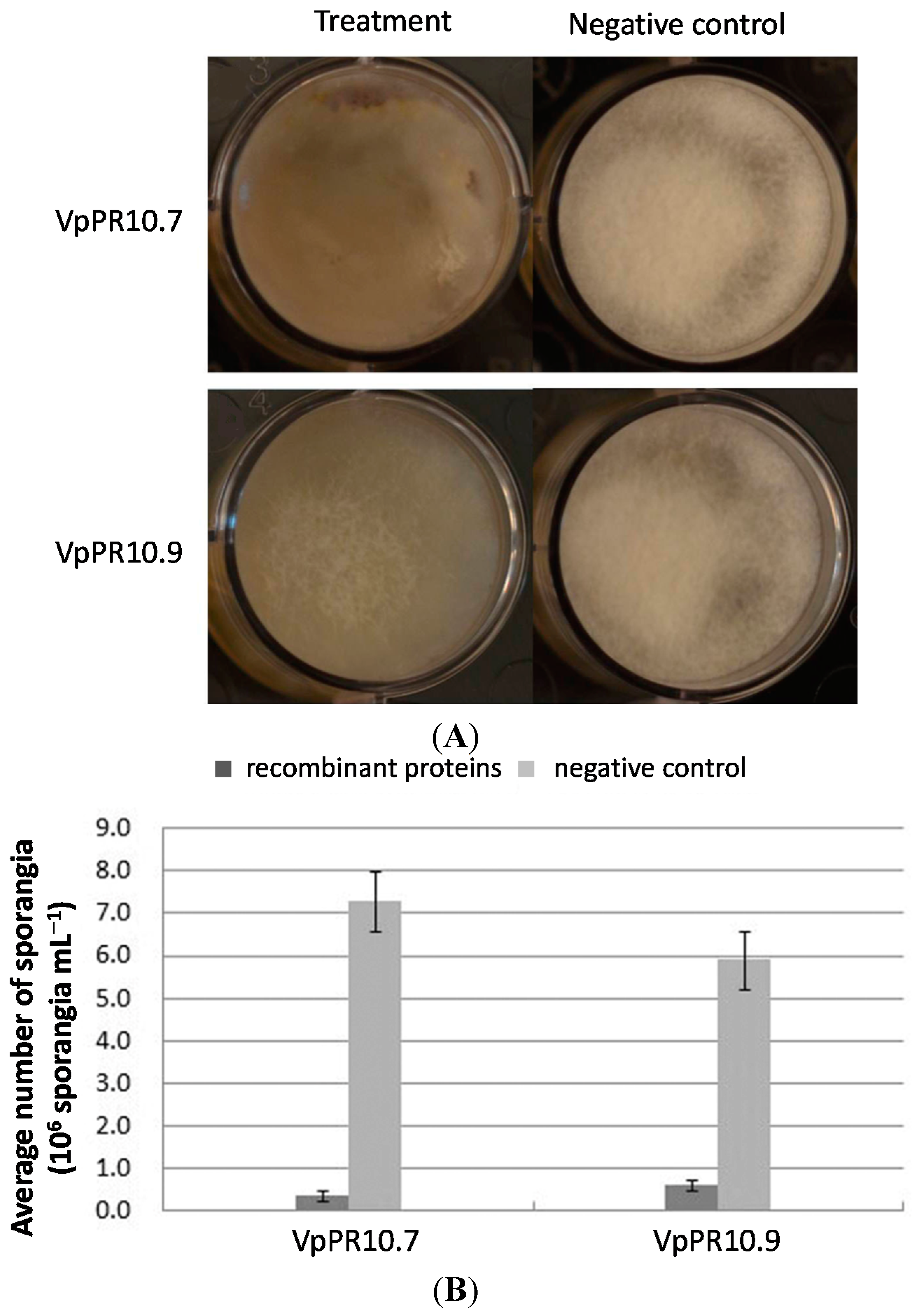
2.5. In Vitro Anti-Fungal Activities of Recombinant VpPR10s Proteins
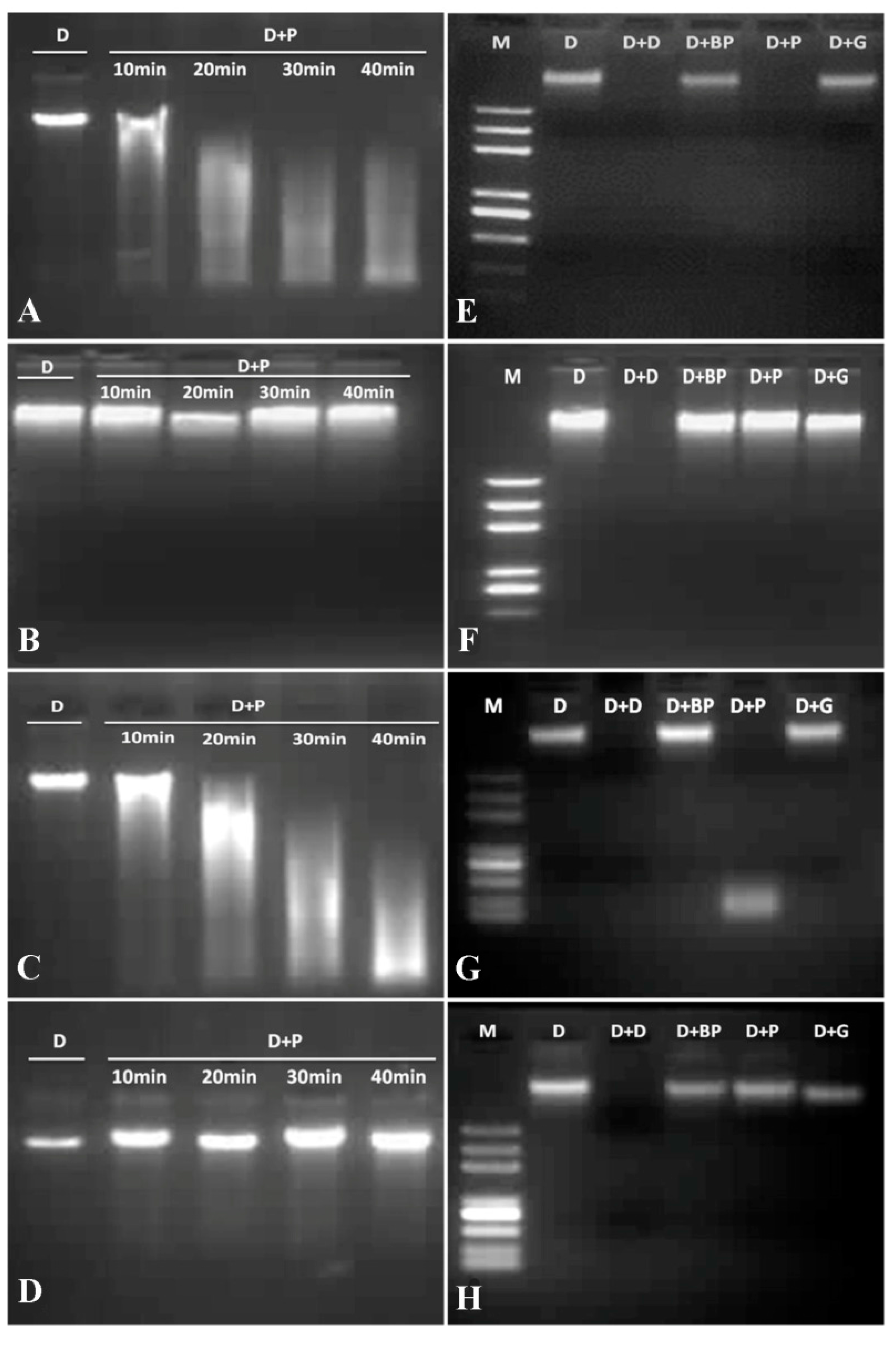
2.6. Influences of VpPR10s on E. coli under Abiotic Stresses

3. Discussion
4. Experimental Section
4.1. Plant Material
4.2. Full-Length Cloning of VpPR10s
| Primer Name | Sequence (5' to 3') |
|---|---|
| VpPR10.4F1 | 5'-ATGGGTGTTATTACTTATGAGATG-3' |
| VpPR10.4R1 | 5'-TTAATAAGCATCAGGATTTGCCAAG-3' |
| VpPR10.4F2 | 5'-CTACTTTGGTGAAGGTCACCAATTCAAGAGCGTGACACA-3' |
| VpPR10.4R2 | 5'-CACGCTCTTGAATTGGTGACCTTCACCAAAGTAGATC-3' |
| VpPR10.6F1 | 5'-ATGGGTGCTATCACTTATGAAATGGA-3' |
| VpPR10.6R1 | 5'-TTAATAGGCATCAGGATTGGCCAAG-3' |
| VpPR10.6F2 | 5'-GATTACTTTCGGTGAAGGCAGCCAATTCAACTACGTG-3' |
| VpPR10.6R2 | 5'-GTTGAATTGGCTGCCTTCACCGAAAGTAATCTTCTTG-3' |
| VpPR10.7F1 | 5'-ATGGGTGTTGTCACTTACACTG-3' |
| VpPR10.7R1 | 5'-TCAGGCATCAGGATTAGCTAAGAG-3' |
| VpPR10.7F2 | 5'-GAACTTTGCTGAAGGCTACCAATTCAAATATGTGAAGC-3' |
| VpPR10.7R2 | 5'-GCTTCACATATTTGAATTGGTAGCCTTCAGCAAAGTTCA-3' |
| VpPR10.9F1 | 5'-ATGGGTGTCACAAGACTCAGT-3' |
| VpPR10.9R1 | 5'-TCAAGTATAGGCGCGAGGGTGT-3' |
| VpPR10.9F2 | 5'-CAGATCAACTTCACTGAAGCTAGTCCTTTAACATACATG-3' |
| VpPR10.9R2 | 5'-GTATGTTAAAGGACTAGCTTCAGTGAAGTTGATCTGTTTG-3' |
4.3. Sequence Alignment of VpPR10s
4.4. Prokaryotic Expression of VpPR10s and Purification of Recombinant Proteins
4.5. DNase and RNase Activities Assays of Recombinant Proteins
4.6. In Vitro Anti-Fungal Activity Assay
4.7. Functional Analysis of VpPR10s Proteins under Different Abiotic Stresses
5. Conclusions
Acknowledgments
Author Contributions
Abbreviations
| ABA | abscisic acid |
| CTAB | cetyltrimethyl ammonium bromide |
| IPTG | isopropyl-β-d-thiogalactoside |
| JA | jasmonic acid |
| PDA | potato dextrose agar |
| PR | pathogenesis-related |
| SA | salicylic acid |
| SDS-PAGE | Tsodium dodecyl sulfate polyacrylamide gel electrophoresis |
| Vp | Vitis pseudoreticulata |
Conflicts of Interest
References
- Brewer, M.T.; Milgroom, M.G. Phylogeography and population structure of the grape powdery mildew fungus, Erysiphe necator, from diverse Vitis species. BMC Evolut. Biol. 2010, 10, 268. [Google Scholar]
- Cadle-Davidson, L.; Chicoine, D.R.; Consolie, N.H. Variation within and among Vitis spp for foliar resistance to the powdery mildew pathogen Erysiphe necator. Plant Dis. 2011, 95, 202–211. [Google Scholar]
- Caffi, T.; Rossi, V.; Legler, S.E.; Bugiani, R. A mechanistic model simulating ascosporic infections by Erysiphe necator, the powdery mildew fungus of grapevine. Plant Pathol. 2011, 60, 522–531. [Google Scholar]
- Donald, T.M.; Pellerone, F.; Adam Blondon, A.-F.; Bouquet, A.; Thomas, M.R.; Dry, I.B. Identification of resistance gene analogs linked to a powdery mildew resistance locus in grapevine. Theor. Appl. Genet. 2002, 104, 610–618. [Google Scholar]
- Bisson, L.F.; Waterhouse, A.L.; Ebeler, S.E.; Andrew Walker, M.; Lapsley, J.T. The present and future of the international wine industry. Nature 2002, 418, 696–699. [Google Scholar]
- Wang, Y.; Liu, Y.; He, P.; Chen, J.; Lamikanra, O.; Lu, J. Evaluation of foliar resistance to Uncinula necator in Chinese wild Vitis species. Vitis 1995, 34, 159–164. [Google Scholar]
- Xu, W.; Yu, Y.; Zhou, Q.; Ding, J.H.; Dai, L.M.; Xie, X.Q.; Xu, Y.; Zhang, C.H.; Wang, Y.J. Expression pattern, genomic structure, and promoter analysis of the gene encoding stilbene synthase from Chinese wild Vitis pseudoreticulata. J. Exp. Bot. 2011, 62, 2745–2761. [Google Scholar]
- Van Loon, L.C.; Pierpoint, W.S.; Boller, T.; Conejero, V. Recommendations for naming plant pathogenesis-related proteins. Plant Mol. Biol. Rep. 1994, 12, 245–264. [Google Scholar]
- Van Loon, L.C.; van Strien, E.A. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol. Mol. Plant Pathol. 1999, 55, 85–97. [Google Scholar]
- Christensen, A.B.; Cho, B.H.; Næsby, M.; Gregersen, P.L.; Brandt, J.; Madriz-Ordeñana, K.; Collinge, D.B.; Thordal-Christensen, H. The molecular characterization of two barley proteins establishes the novel PR-17 family of pathogenesis-related proteins. Mol. Plant Pathol. 2002, 3, 135–144. [Google Scholar]
- Somssich, I.E.; Schmelzer, E.; Kawalleck, P.; Hahlbrock, K. Gene structure and in situ transcript localization of pathogenesis-related protein 1 in parsley. Mol. Gen. Genet. MGG 1988, 213, 93–98. [Google Scholar]
- Walter, M.H.; Liu, J.W.; Grand, C.; Lamb, C.J.; Hess, D. Bean pathogenesis-related (PR) proteins deduced from elicitor-induced transcripts are members of a ubiquitous new class of conserved PR proteins including pollen allergens. Mol. Gen. Genet. MGG 1990, 222, 353–360. [Google Scholar]
- Warner, S.A.; Scott, R.; Draper, J. Characterisation of a wound induced transcript from the monocot asparagus that shares similarity with a class of intracellular pathogenesis-related (PR) proteins. Plant Mol. Biol. 1992, 19, 555–561. [Google Scholar]
- Huang, J.C.; Chang, F.C.; Wang, C.S. Characterization of a lily tapetal transcript that shares sequence similarity with a class of intracellular pathogenesis-related (IPR) proteins. Plant Mol. Biol. 1997, 34, 681–686. [Google Scholar]
- Breiteneder, H.; Pettenburger, K.; Bito, A.; Valenta, R.; Kraft, D.; Rumpold, H.; Scheiner, O.; Breitenbach, M. The gene coding for the major birch pollen allergen Bet v 1, is highly homologous to a pea disease resistance response gene. EMBO J. 1989, 8, 1935–1938. [Google Scholar]
- Swoboda, I.; Jilek, A.; Ferreira, F.; Engel, E.; Hoffmann-Sommergruber, K.; Scheiner, O.; Kraft, D.; Breiteneder, H.; Pittenauer, E.; Schmid, E.; et al. Isoforms of Bet v 1, the major birch pollen allergen, analyzed by liquid chromatography, mass spectrometry, and cDNA cloning. J. Biol. Chem. 1995, 270, 2607–2613. [Google Scholar]
- Breda, C.; Sallaud, C.; EL Turk, J.; Buffard, D.; de Kozak, I.; Esnault, R.; Kondorosi, A. Defense reaction in Medicago sativa: A gene encoding a class 10 PR protein is expressed in vascular bundles. Mol. Plant-Microbe Interact. 1996, 9, 713–719. [Google Scholar]
- Esnault, R.; Buffard, D.; Breda, C.; Sallaud, C.; el Turk, J.; Kondorosi, A. Pathological and molecular characterizations of alfalfa interactions with compatible and incompatible bacteria, Xanthomonas campestris pv. alfalfae and Pseudomonas syringae pv. pisi. Mol. Plant-Microbe Interact. 1993, 6, 655–664. [Google Scholar]
- Liu, J.J.; Ekramoddoullah, A.K. Root-specific expression of a western white pine PR10 gene is mediated by different promoter regions in transgenic tobacco. Plant Mol. Biol. 2003, 52, 103–120. [Google Scholar]
- Ukaji, N.; Kuwabara, C.; Takezawa, D.; Arakawa, K.; Fujikawa, S. Accumulation of pathogenesis-related (PR) 10/Bet v 1 protein homologues in mulberry (Morus bombycis Koidz.) tree during winter. Plant Cell Environ. 2004, 27, 1112–1121. [Google Scholar]
- Apold, J.; Florvaag, E.; Elsayed, S. Comparative studies on tree-pollen allergens. Int. Arch. Allergy Immunol. 1981, 64, 439–447. [Google Scholar]
- Constabel, C.P.; Brisson, N. Stigma- and vascular-specific expression of the PR-10a gene of potato: A novel pattern of expression of a pathogenesis-related gene. Mol. Plant-Microbe Interact. 1995, 8, 104–113. [Google Scholar]
- Swoboda, I.; Hoffmann Sommergruber, K.; O’Ríordáin, G.; Scheiner, O.; Heberle-Bors, E.; Vicente, O. Bet v 1 proteins, the major birch pollen allergens and members of a family of conserved pathogenesis-related proteins, show ribonuclease activity in vitro. Physiol. Plant 1996, 96, 433–438. [Google Scholar]
- Warner, S.A.; Scott, R.; Draper, J. Isolation of an asparagus intracellular PR gene (AoPR1) wound-responsive promoter by the inverse polymerase chain reaction and its characterization in transgenic tobacco. Plant J. 1993, 3, 191–201. [Google Scholar]
- Warner, S.A.; Gill, A.; Draper, J. The developmental expression of the asparagus intracellular PR protein (AoPR1) gene correlates with sites of phenylpropanoid biosynthesis. Plant J. 1994, 6, 31–43. [Google Scholar]
- Atkinson, R.G.; Perry, J.; Matsui, T.; Ross, G.S.; MacRae, E.A. A stress-, pathogenesis-, and allergen-related cDNA in apple fruit is also ripening-related. NZ J. Crop Hort. Sci. 1996, 24, 103–107. [Google Scholar]
- Pühringer, H.; Moll, D.; Hoffmann Sommergruber, K.; Watillon, B.; Katinger, H.; da Câmara Machado, M.L. The promoter of an apple Ypr10 gene, encoding the major allergen Mal d 1, is stress-and pathogen-inducible. Plant Sci. 2000, 152, 35–50. [Google Scholar]
- Wu, F.; Yan, M.; Li, Y.; Chang, S.; Song, X.; Zhou, Z.; Gong, W. cDNA cloning, expression, and mutagenesis of a PR-10 protein SPE-16 from the seeds of Pachyrrhizus erosus. Biochem. Biophys. Res. Commun. 2003, 312, 761–766. [Google Scholar]
- Mylona, P.; Moerman, M.; Yang, W.C.; Gloudemans, T.; van de Kerckhove, J.; van Kamme, A.; Bisseling, T.; Franssen, H.J. The root epidermis-specific pea gene RH2 is homologous to a pathogenesis-related gene. Plant Mol. Biol. 1994, 26, 39–50. [Google Scholar]
- Sikorski, M.M.; Biesiadka, J.; Kasperska, A.E.; Kopcińska, J.; Łotocka, B.; Golinowski, W.; Legocki, A.B. Expression of genes encoding PR10 class pathogenesis-related proteins is inhibited in yellow lupine root nodules. Plant Sci. 1999, 149, 125–137. [Google Scholar]
- Walter, M.H.; Liu, J.W.; Wünn, J.; Hess, D. Bean ribonuclease-like pathogenesis-related protein genes (Ypr10) display complex patterns of developmental, dark-induced and exogenous-stimulus-dependent expression. Eur. J. Biochem. 1996, 239, 281–293. [Google Scholar]
- Yamamoto, M.; Torikai, S.; Oeda, K. A major root protein of carrots with high homology to intracellular pathogenesis-related (PR) proteins and pollen allergens. Plant Cell Physiol. 1997, 38, 1080–1086. [Google Scholar]
- Liu, J.J.; Ekramoddoullah, A.K.; Piggott, N.; Zamani, A. Molecular cloning of a pathogen/wound-inducible PR10 promoter from Pinus monticola and characterization in transgenic Arabidopsis plants. Planta 2005, 221, 159–169. [Google Scholar]
- Liu, J.J.; Ekramoddoullah, A.K. Characterization, expression and evolution of two novel subfamilies of Pinus monticola cDNAs encoding pathogenesis-related (PR)-10 proteins. Tree Physiol. 2004, 24, 1377–1385. [Google Scholar]
- Hoffmann Sommergruber, K.; Vanek Krebitz, M.; Radauer, C.; Wen, J.; Ferreira, F.; Scheiner, O.; Breiteneder, H. Genomic characterization of members of the Bet v 1 family: Genes coding for allergens and pathogenesis-related proteins share intron positions. Gene 1997, 197, 91–100. [Google Scholar]
- Zhou, X.J.; Lu, S.; Xu, Y.H.; Wang, J.W.; Chen, X.Y. A cotton cDNA (GaPR-10) encoding a pathogenesis-related 10 protein with in vitro ribonuclease activity. Plant Sci. 2002, 162, 629–636. [Google Scholar]
- Chadha, P.; Das, R.H. A pathogenesis related protein, AhPR10 from peanut: An insight of its mode of antifungal activity. Planta 2006, 225, 213–222. [Google Scholar]
- Lebel, S.; Schellenbaum, P.; Walter, B.; Maillot, P. Characterisation of the Vitis vinifera PR10 multigene family. BMC Plant Biol. 2010, 10, 184. [Google Scholar]
- Bufe, A.; Spangfort, M.D.; Kahlert, H.; Schlaak, M.; Becker, W.-M. The major birch pollen allergen, Bet v 1, shows ribonuclease activity. Planta 1996, 199, 413–415. [Google Scholar]
- Bantignies, B.; Séguin, J.; Muzac, I.; Dédaldéchamp, F.; Gulick, P.; Ibrahim, R. Direct evidence for ribonucleolytic activity of a PR-10-like protein from white lupin roots. Plant Mol. Biol. 2000, 42, 871–881. [Google Scholar]
- Park, C.J.; Kim, K.J.; Shin, R.; Park, J.M.; Shin, Y.C.; Paek, K.H. Pathogenesis-prelated protein 10 isolated from hot pepper functions as a ribonuclease in an antiviral pathway. Plant J. 2004, 37, 186–198. [Google Scholar]
- Soh, H.C.; Park, A.R.; Park, S.; Back, K.; Yoon, J.B.; Park, H.G.; Kim, Y.S. Comparative analysis of pathogenesis-related protein 10 (PR10) genes between fungal resistant and susceptible peppers. Eur. J. Plant Pathol. 2012, 132, 37–48. [Google Scholar]
- Kim, S.G.; Kim, S.T.; Wang, Y.M.; Yu, S.; Choi, I.S.; Kim, Y.C.; Kim, W.T.; Agrawal, G.K.; Rakwal, R.; Kang, K.Y. The RNase activity of rice probenazole-induced protein 1 (PBZ1) plays a key role in cell death in plants. Mol. Cells 2011, 31, 25–31. [Google Scholar]
- Srivastava, S.; Emery, R.N.; Kurepin, L.V.; Reid, D.M.; Fristensky, B.; Kav, N.N.V. Pea PR 10.1 is a ribonuclease and its transgenic expression elevates cytokinin levels. Plant Growth Regul. 2006, 49, 17–25. [Google Scholar]
- He, M.Y.; Xu, Y.; Cao, J.J.; Zhu, Z.G.; Jiao, Y.T.; Wang, Y.J.; Guan, X.; Yang, Y.Z.; Xu, W.R.; Fu, Z.F. Subcellular localization and functional analyses of a PR10 protein gene from Vitis pseudoreticulata in response to Plasmopara viticola infection. Protoplasma 2013, 250, 129–140. [Google Scholar]
- Kim, S.T.; Yu, S.; Kang, Y.H.; Kim, S.G.; Kim, J.Y.; Kim, S.H.; Kang, K.Y. The rice pathogen-related protein 10 (JIOsPR10) is induced by abiotic and biotic stresses and exhibits ribonuclease activity. Plant Cell Rep. 2008, 27, 593–603. [Google Scholar]
- Jwa, N.S.; Kumar Agrawal, G.; Rakwal, R.; Park, C.H.; Prasad Agrawal, V. Molecular cloning and characterization of a novel jasmonate inducible pathogenesis-related class 10 protein gene, JIOsPR10, from rice (Oryza sativa L.) seedling leaves. Biochem. Biophys. Res. Commun. 2001, 286, 973–983. [Google Scholar]
- Hashimoto, M.; Kisseleva, L.; Sawa, S.; Furukawa, T.; Komatsu, S.; Koshiba, T. A novel rice PR10 protein, RSOsPR10, specifically induced in roots by biotic and abiotic stresses, possibly via the jasmonic acid signaling pathway. Plant Cell Physiol. 2004, 45, 550–559. [Google Scholar]
- Xie, Y.R.; Chen, Z.Y.; Brown, R.L.; Bhatnagar, D. Expression and functional characterization of two pathogenesis-related protein 10 genes from Zea mays. J. Plant Physiol. 2010, 167, 121–130. [Google Scholar]
- Jellouli, N.; Ben Jouira, H.; Skouri, H.; Ghorbel, A.; Gourgouri, A.; Mliki, A. Proteomic analysis of Tunisian grapevine cultivar Razegui under salt stress. J. Physiol. 2008, 165, 471–481. [Google Scholar]
- Krishnaswamy, S.; Baral, P.K.; James, M.N.; Kav, N.N.V. Site-directed mutagenesis of histidine 69 and glutamic acid 148 alters the ribonuclease activity of pea ABR17 (PR10.4). Plant Physiol. Biochem. 2011, 49, 958–962. [Google Scholar]
- Yan, Q.J.; Qi, X.W.; Jiang, Z.Q.; Yang, S.Q.; Han, L.J. Characterization of a pathogenesis-related class 10 protein (PR-10) from Astragalus mongholicus with ribonuclease activity. Plant Physiol. Biochem. 2008, 46, 93–99. [Google Scholar]
- Guevara Morato, M.Á.; de Lacoba, M.G.; García Luque, I.; Serra, M.T. Characterization of a pathogenesis-related protein 4 (PR-4) induced in Capsicum chinense L3 plants with dual RNase and DNase activities. J. Exp. Bot. 2010, 61, 3259–3271. [Google Scholar]
- Liu, J.J.; Ekramoddoullah, A.K. The family 10 of plant pathogenesis-related proteins: Their structure, regulation, and function in response to biotic and abiotic stresses. Physiol. Mol. Plant Pathol. 2006, 68, 3–13. [Google Scholar]
- Gao, Z.S.; van de Weg, W.E.; Schaart, J.G.; Schouten, H.J.; Tran, D.H.; Kodde, L.P.; van der Meer, I.M.; van der Geest, A.H.M.; Kodde, J.; Breiteneder, H.; et al. Genomic cloning and linkage mapping of the Mal d 1 (PR-10) gene family in apple (Malus domestica). Theor. Appl. Genet. 2005, 111, 171–183. [Google Scholar]
- Agarwal, P.; Bhatt, V.; Singh, R.; Das, M.; Sopory, S.K.; Chikara, J. Pathogenesis-related gene, JcPR-10a from Jatropha curcas exhibit RNase and antifungal activity. Mol. Biotechnol. 2013, 54, 1–14. [Google Scholar]
- A pathogenesis related protein, VpPR-10.1, from Vitis pseudoreticulata: An insight of its mode of antifungal activity. PLoS One 2014, 9, e95102.
- Constabel, C.P.; Bertrand, C.; Brisson, N. Transgenic potato plants overexpressing the pathogenesis-related STH-2 gene show unaltered susceptibility to Phytophthora infestans and potato virus X. Plant Mol. Biol. 1993, 22, 775–782. [Google Scholar]
- Lodhi, M.A.; Ye, G.N.; Weeden, N.F.; Reisch, B.I. A simple and efficient method for DNA extraction from grapevine cultivars and Vitis species. Plant Mol. Biol. Rep. 1994, 12, 6–13. [Google Scholar]
- Basic Local Alignment Search Tool. Available online: http://www.ncbi.nlm.nih.gov/BLAST (accessed on 30 September 2013).
- Xu, Y.; Yu, H.; He, M.Y.; Yang, Y.Z.; Wang, Y.J. Isolation and expression analysis of a novel pathogenesis-related protein 10 gene from Chinese wild Vitis pseudoreticulata induced by Uncinula necator. Biologia 2010, 65, 653–659. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Wei, J.; Zou, Y.; Xu, K.; Wang, Y.; Cui, L.; Xu, Y. Molecular Characteristics and Biochemical Functions of VpPR10s from Vitis pseudoreticulata Associated with Biotic and Abiotic Stresses. Int. J. Mol. Sci. 2014, 15, 19162-19182. https://doi.org/10.3390/ijms151019162
Wang L, Wei J, Zou Y, Xu K, Wang Y, Cui L, Xu Y. Molecular Characteristics and Biochemical Functions of VpPR10s from Vitis pseudoreticulata Associated with Biotic and Abiotic Stresses. International Journal of Molecular Sciences. 2014; 15(10):19162-19182. https://doi.org/10.3390/ijms151019162
Chicago/Turabian StyleWang, Lan, Jinyu Wei, Ying Zou, Keyao Xu, Yuejin Wang, Lu Cui, and Yan Xu. 2014. "Molecular Characteristics and Biochemical Functions of VpPR10s from Vitis pseudoreticulata Associated with Biotic and Abiotic Stresses" International Journal of Molecular Sciences 15, no. 10: 19162-19182. https://doi.org/10.3390/ijms151019162
APA StyleWang, L., Wei, J., Zou, Y., Xu, K., Wang, Y., Cui, L., & Xu, Y. (2014). Molecular Characteristics and Biochemical Functions of VpPR10s from Vitis pseudoreticulata Associated with Biotic and Abiotic Stresses. International Journal of Molecular Sciences, 15(10), 19162-19182. https://doi.org/10.3390/ijms151019162





