Serum Starvation-Induced Voltage-Gated Potassium Channel Kv7.5 Expression and Its Regulation by Sp1 in Canine Osteosarcoma Cells
Abstract
:1. Introduction
2. Results
2.1. Identification of Kv7.5 in the CCL-183 Canine Osteosarcoma Cell Line
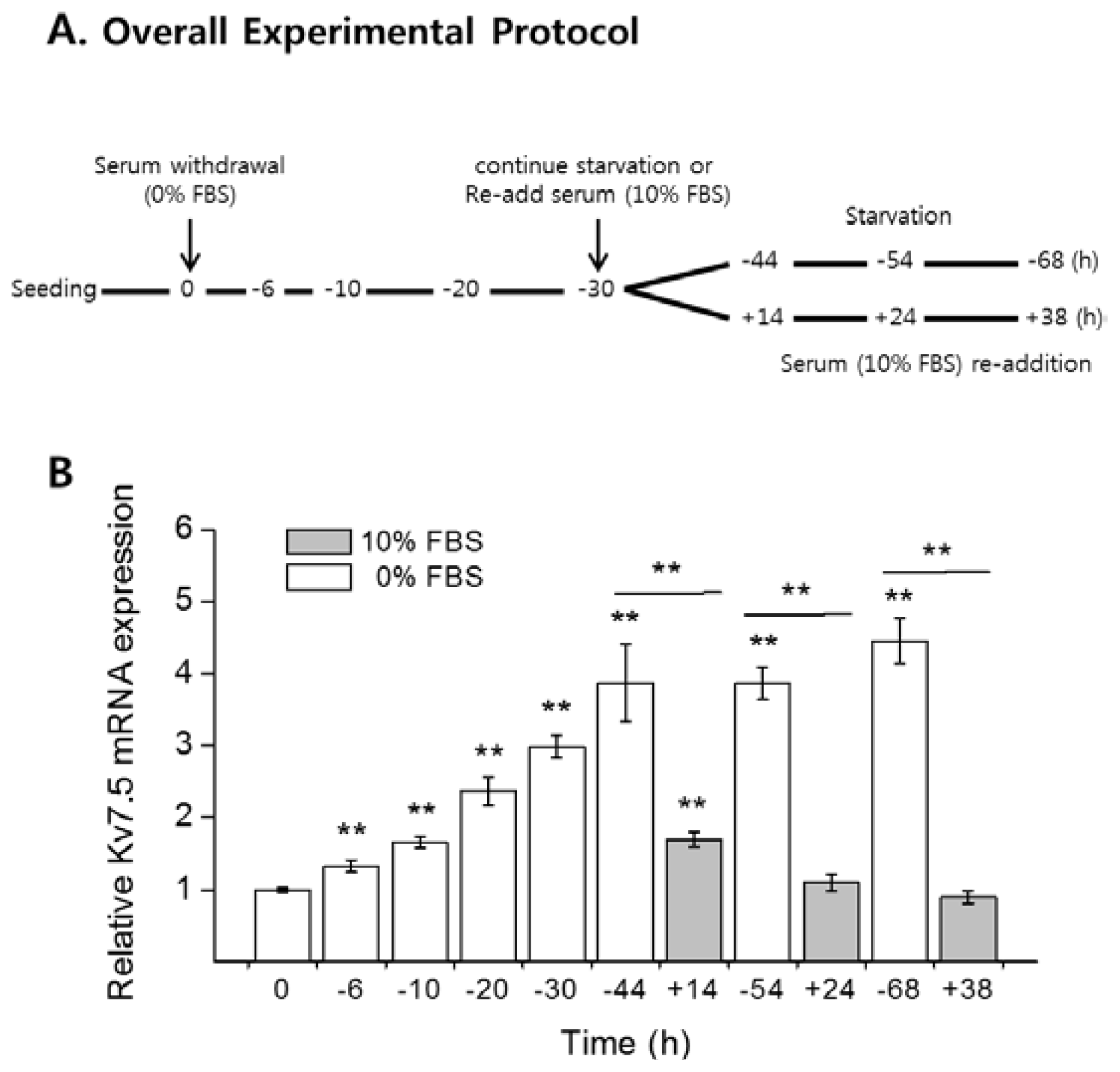
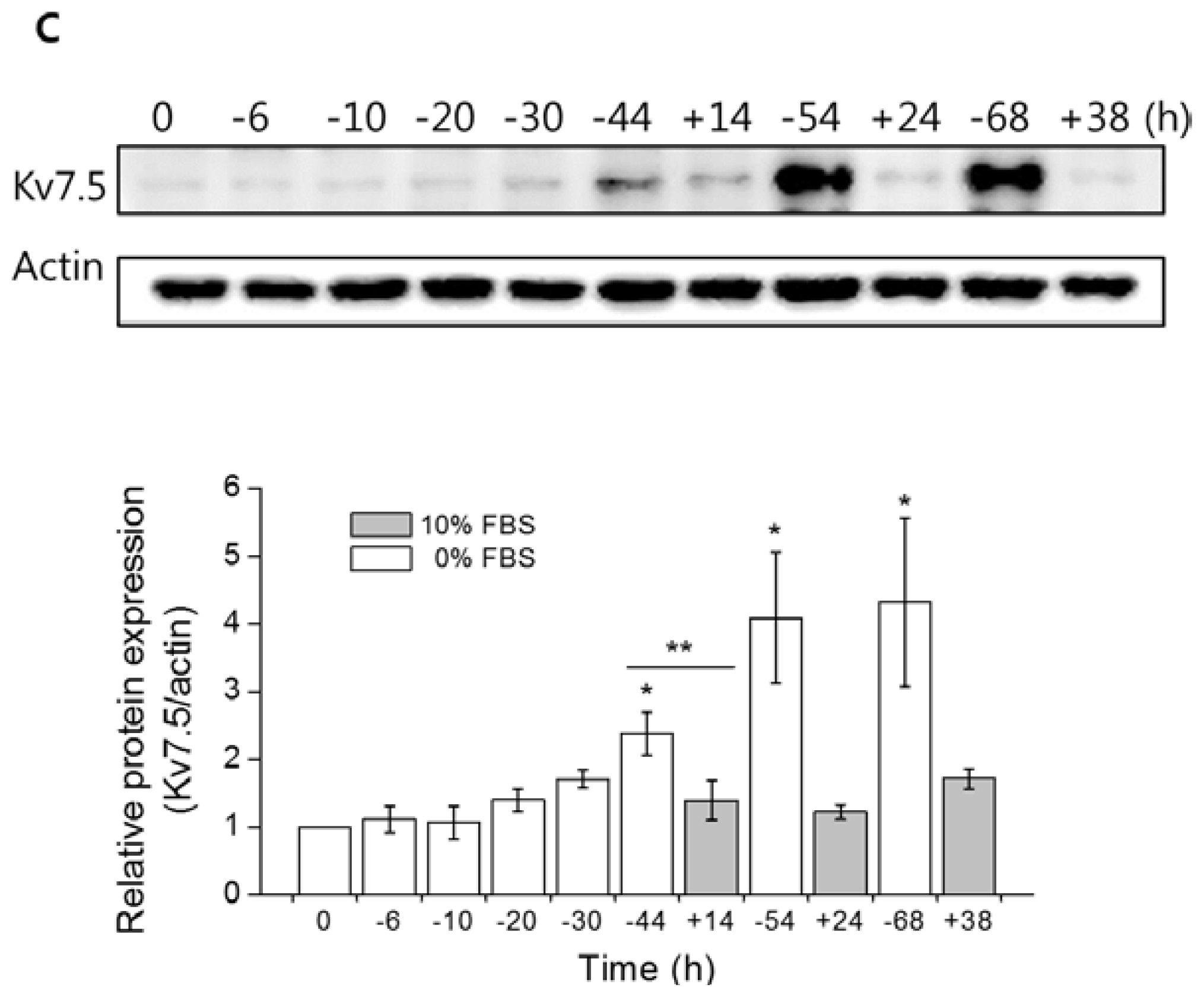
2.2. Serum Starvation Upregulates Kv7.5 Transcripts and Protein in a Time-Dependent Manner
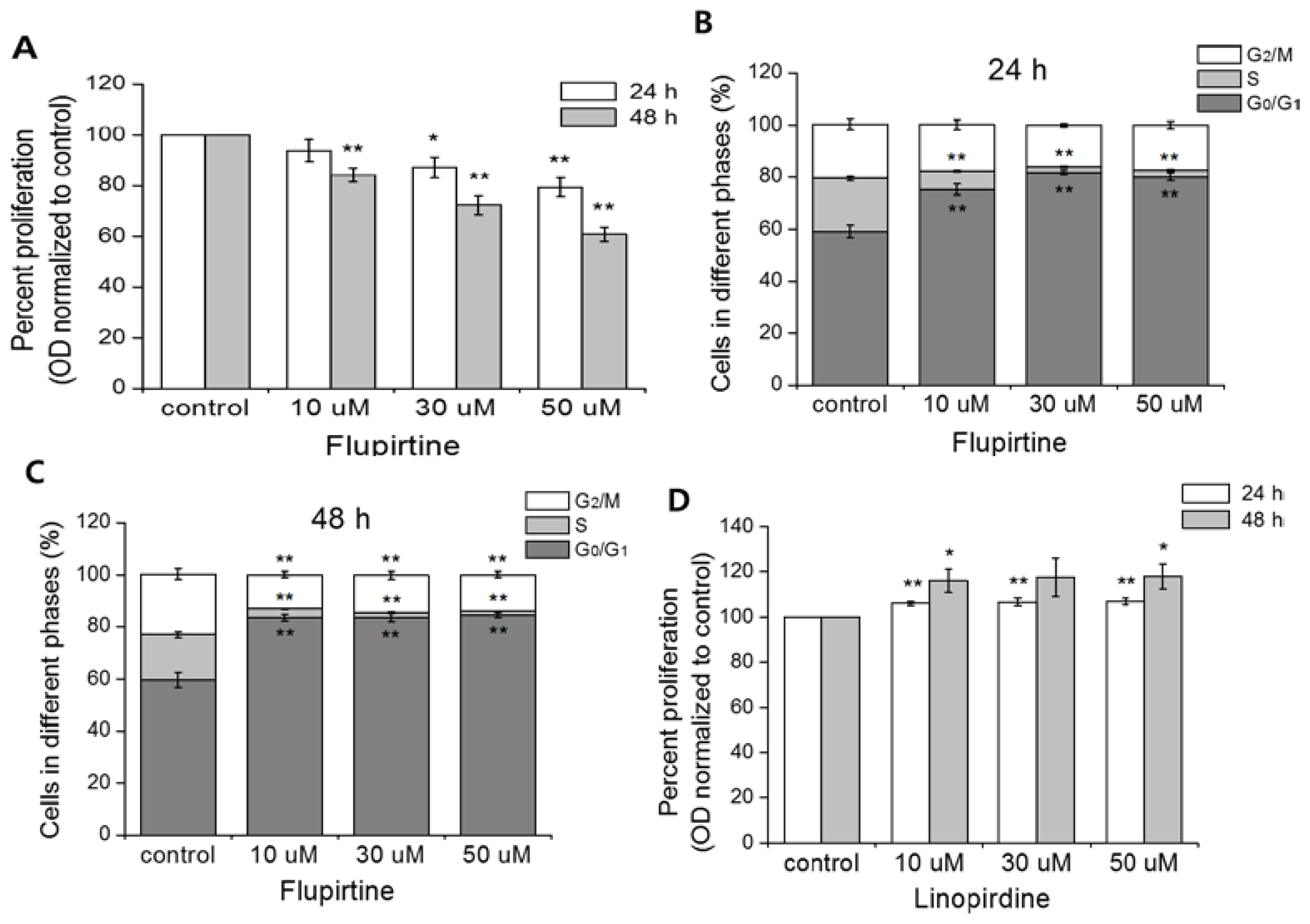
2.3. Involvement of Kv7.5 in CCL-183 Cell Proliferation
2.4. Flupirtine, a Kv7 Opener, Arrests Cells in the G0/G1 Phase
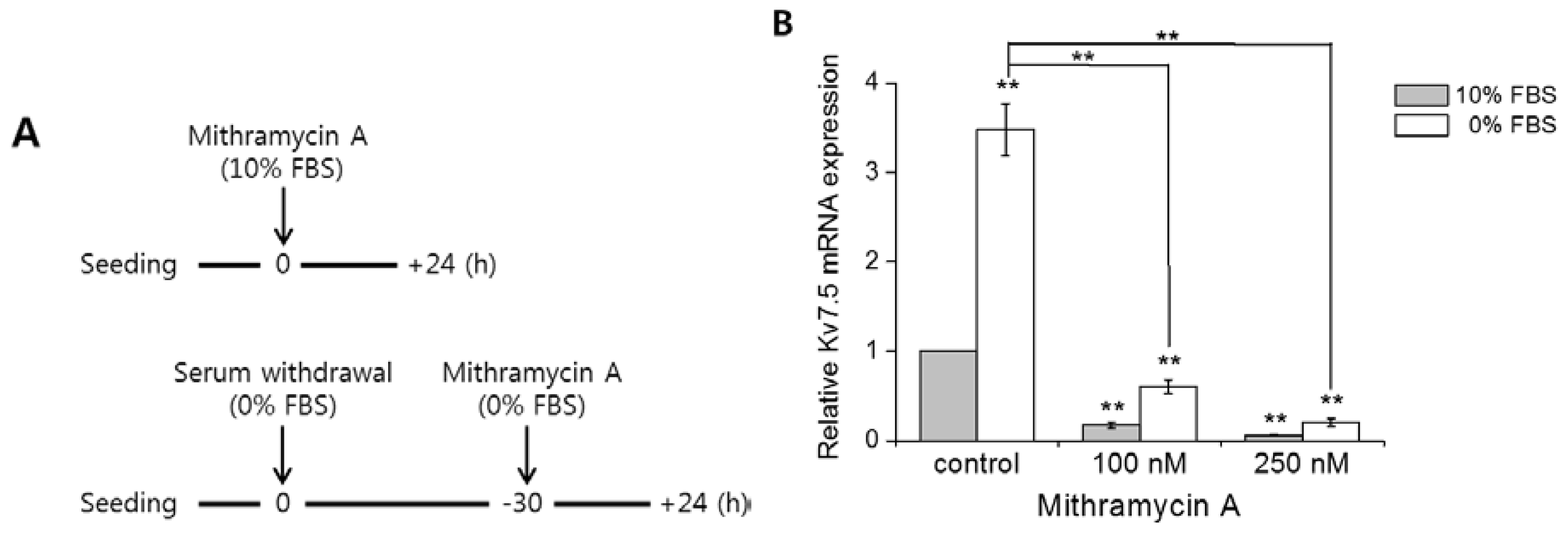
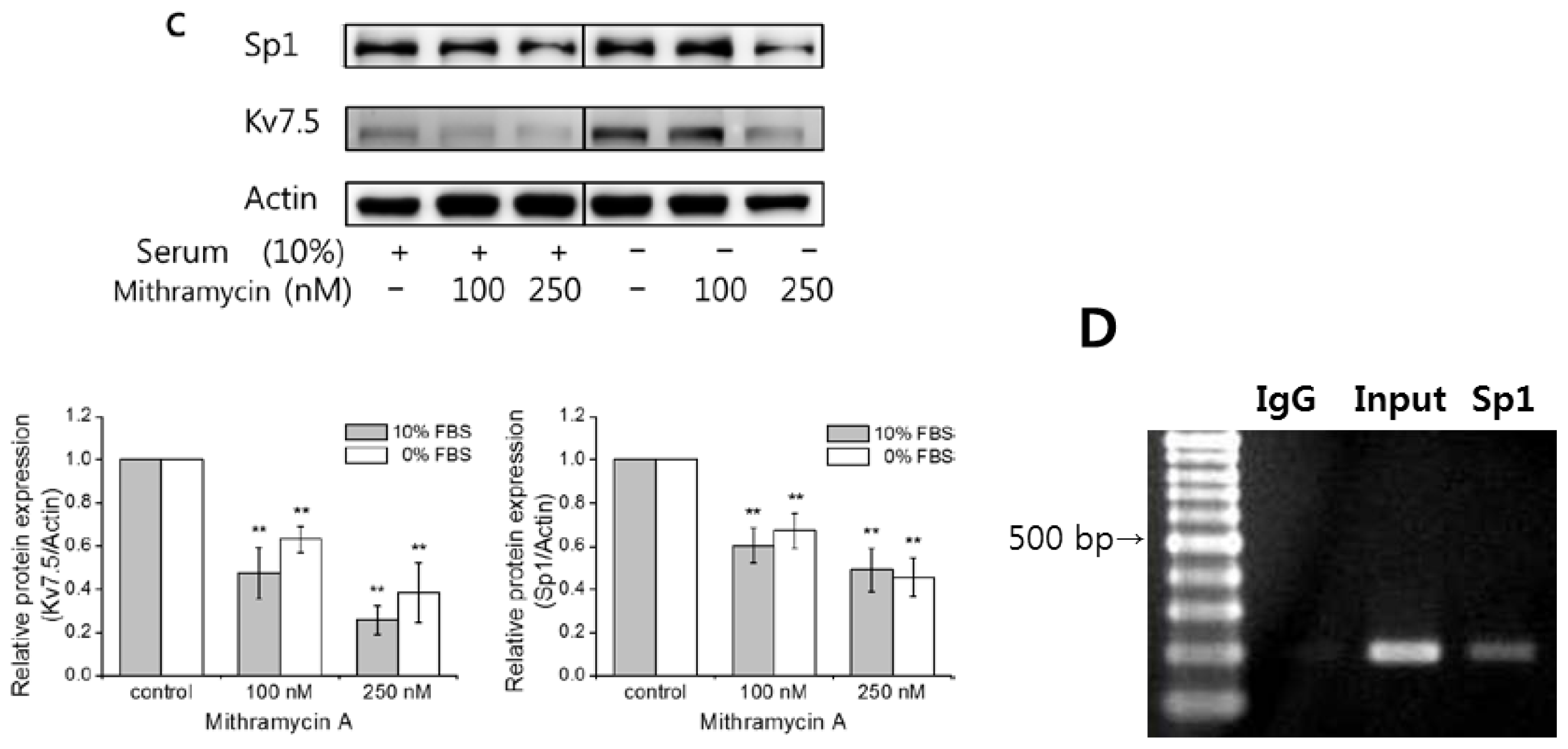
2.5. Mithramycin a Blocks Kv7.5 Transcription by Inhibiting Sp1 Binding to the Kv7.5 Promoter
2.6. Serum Dependent Cell Cycle Progression
3. Discussion
4. Experimental Section
4.1. Cell Culture
4.2. RNA Isolation: Reverse Transcription PCR (RT-PCR)
4.3. Quantitative Real-Time PCR (qPCR)
4.4. Small Interfering RNA (siRNA) Transfection
4.5. Drugs and Antibodies
4.6. MTT Cell Proliferation Assay
4.7. Cell Cycle Analysis by Flow Cytometry
4.8. Western Blot Analysis
4.9. Chromatin Immunoprecipitation (ChIP) Assay
4.10. Statistical Analysis
5. Conclusions
Supplementary Information
ijms-15-00977-s001.pdfAcknowledgments
Conflicts of Interest
References
- Lee, S.Y.; Maniak, P.J.; Ingbar, D.H.; O’Grady, S.M. Adult alveolar epithelial cells express multiple subtypes of voltage-gated K+ channels that are located in apical membrane. Am. J. Physiol. Cell Physiol 2003, 284, C1614–C1624. [Google Scholar]
- Vallejo-Gracia, A.; Bielanska, J.; Hernandez-Losa, J.; Castellvi, J.; Ruiz-Marcellan, M.C.; Ramony Cajal, S.; Condom, E.; Manils, J.; Soler, C.; Comes, N.; et al. Emerging role for the voltage-dependent K+ channel Kv1.5 in B-lymphocyte physiology: Expression associated with human lymphoma malignancy. J. Leukoc. Biol 2013, 94, 779–789. [Google Scholar]
- Villalonga, N.; David, M.; Bielanska, J.; Vicente, R.; Comes, N.; Valenzuela, C.; Felipe, A. Immunomodulation of voltage-dependent K+ channels in macrophages: Molecular and biophysical consequences. J. Gen. Physiol 2010, 135, 135–147. [Google Scholar]
- Kim, H.J.; Jang, S.H.; Jeong, Y.A.; Ryu, P.D.; Kim, D.Y.; Lee, S.Y. Involvement of Kv4.1 K+ channels in gastric cancer cell proliferation. Biol. Pharm. Bull 2010, 33, 1754–1757. [Google Scholar]
- Suzuki, T.; Takimoto, K. Selective expression of HERG and Kv2 channels influences proliferation of uterine cancer cells. Int. J. Oncol 2004, 25, 153–159. [Google Scholar]
- Abdul, M.; Santo, A.; Hoosein, N. Activity of potassium channel-blockers in breast cancer. Anticancer Res 2003, 23, 3347–3351. [Google Scholar]
- Cherubini, A.; Taddei, G.L.; Crociani, O.; Paglierani, M.; Buccoliero, A.M.; Fontana, L.; Noci, I.; Borri, P.; Borrani, E.; Giachi, M.; et al. HERG potassium channels are more frequently expressed in human endometrial cancer as compared to non-cancerous endometrium. Br. J. Cancer 2000, 83, 1722–1729. [Google Scholar]
- Jang, S.H.; Choi, C.; Hong, S.G.; Yarishkin, O.V.; Bae, Y.M.; Kim, J.G.; O’Grady, S.M.; Yoon, K.A.; Kang, K.S.; Ryu, P.D.; et al. Silencing of Kv4.1 potassium channels inhibits cell proliferation of tumorigenic human mammary epithelial cells. Biochem. Biophys. Res. Commun 2009, 384, 180–186. [Google Scholar]
- Jang, S.H.; Kang, K.S.; Ryu, P.D.; Lee, S.Y. Kv1.3 voltage-gated K+ channel subunit as a potential diagnostic marker and therapeutic target for breast cancer. BMB Rep 2009, 42, 535–539. [Google Scholar]
- Crociani, O.; Guasti, L.; Balzi, M.; Becchetti, A.; Wanke, E.; Olivotto, M.; Wymore, R.S.; Arcangeli, A. Cell cycle-dependent expression of HERG1 and HERG1B isoforms in tumor cells. J. Biol. Chem 2003, 278, 2947–2955. [Google Scholar]
- Czarnecki, A.; Dufy-Barbe, L.; Huet, S.; Odessa, M.F.; Bresson-Bepoldin, L. Potassium channel expression level is dependent on the proliferation state in the GH3 pituitary cell line. Am. J. Physiol. Cell Physiol 2003, 284, C1054–C1064. [Google Scholar]
- Bielanska, J.; Hernandez-Losa, J.; Moline, T.; Somoza, R.; Cajal, S.R.; Condom, E.; Ferreres, J.C.; Felipe, A. Increased voltage-dependent K+ channel Kv1.3 and Kv1.5 expression correlates with leiomyosarcoma aggressiveness. Oncol. Lett 2012, 4, 227–230. [Google Scholar]
- Brown, D.A.; Adams, P.R. Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone. Nature 1980, 283, 673–676. [Google Scholar]
- Biervert, C.; Schroeder, B.C.; Kubisch, C.; Berkovic, S.F.; Propping, P.; Jentsch, T.J.; Steinlein, O.K. A potassium channel mutation in neonatal human epilepsy. Science 1998, 279, 403–406. [Google Scholar]
- Maljevic, S.; Wuttke, T.V.; Seebohm, G.; Lerche, H. KV7 channelopathies. Pflugers Arch 2010, 460, 277–288. [Google Scholar]
- Soldovieri, M.V.; Miceli, F.; Taglialatela, M. Driving with no brakes: Molecular pathophysiology of Kv7 potassium channels. Physiology 2011, 26, 365–376. [Google Scholar]
- Roura-Ferrer, M.; Sole, L.; Martinez-Marmol, R.; Villalonga, N.; Felipe, A. Skeletal muscle Kv7 (KCNQ) channels in myoblast differentiation and proliferation. Biochem. Biophys. Res. Commun 2008, 369, 1094–1097. [Google Scholar]
- Ipavec, V.; Martire, M.; Barrese, V.; Taglialatela, M.; Curro, D. Kv7 channels regulate muscle tone and nonadrenergic noncholinergic relaxation of the rat gastric fundus. Pharmacol. Res 2011, 64, 397–409. [Google Scholar]
- Brueggemann, L.I.; Kakad, P.P.; Love, R.B.; Solway, J.; Dowell, M.L.; Cribbs, L.L.; Byron, K.L. Kv7 potassium channels in airway smooth muscle cells: Signal transduction intermediates and pharmacological targets for bronchodilator therapy. Am. J. Physiol. Lung Cell. Mol. Physiol 2012, 302, L120–L132. [Google Scholar]
- Svalo, J.; Bille, M.; Parameswaran Theepakaran, N.; Sheykhzade, M.; Nordling, J.; Bouchelouche, P. Bladder contractility is modulated by Kv7 channels in pig detrusor. Eur. J. Pharmacol 2013, 715, 312–320. [Google Scholar]
- Mani, B.K.; O’Dowd, J.; Kumar, L.; Brueggemann, L.I.; Ross, M.; Byron, K.L.; Vascular, K.C.N.Q. (Kv7) potassium channels as common signaling intermediates and therapeutic targets in cerebral vasospasm. J. Cardiovasc. Pharmacol 2013, 61, 51–62. [Google Scholar]
- Iannotti, F.A.; Panza, E.; Barrese, V.; Viggiano, D.; Soldovieri, M.V.; Taglialatela, M. Expression, localization, and pharmacological role of Kv7 potassium channels in skeletal muscle proliferation, differentiation, and survival after myotoxic insults. J. Pharmacol. Exp. Ther 2010, 332, 811–820. [Google Scholar]
- Dynan, W.S.; Tjian, R. The promoter-specific transcription factor Sp1 binds to upstream sequences in the SV40 early promoter. Cell 1983, 35, 79–87. [Google Scholar]
- Kadonaga, J.T.; Courey, A.J.; Ladika, J.; Tjian, R. Distinct regions of Sp1 modulate DNA binding and transcriptional activation. Science 1988, 242, 1566–1570. [Google Scholar]
- Chang, W.C.; Hung, J.J. Functional role of post-translational modifications of Sp1 in tumorigenesis. J. Biomed. Sci 2012, 19, 94. [Google Scholar]
- Jensen, H.S.; Callo, K.; Jespersen, T.; Jensen, B.S.; Olesen, S.P. The KCNQ5 potassium channel from mouse: A broadly expressed M-current like potassium channel modulated by zinc, pH, and volume changes. Brain Res. Mol. Brain Res 2005, 139, 52–62. [Google Scholar]
- Blume, S.W.; Snyder, R.C.; Ray, R.; Thomas, S.; Koller, C.A.; Miller, D.M. Mithramycin inhibits SP1 binding and selectively inhibits transcriptional activity of the dihydrofolate reductase gene in vitro and in vivo. J. Clin. Investig. 1991, 88, 1613–1621. [Google Scholar]
- Yuan, P.; Wang, L.; Wei, D.; Zhang, J.; Jia, Z.; Li, Q.; Le, X.; Wang, H.; Yao, J.; Xie, K. Therapeutic inhibition of Sp1 expression in growing tumors by mithramycin a correlates directly with potent antiangiogenic effects on human pancreatic cancer. Cancer 2007, 110, 2682–2690. [Google Scholar]
- Goodchild, C.S.; Nelson, J.; Cooke, I.; Ashby, M.; Jackson, K. Combination therapy with flupirtine and opioid: Open-label case series in the treatment of neuropathic pain associated with cancer. Pain Med 2008, 9, 939–949. [Google Scholar]
- Friedel, H.A.; Fitton, A. Flupirtine. A review of its pharmacological properties, and therapeutic efficacy in pain states. Drugs 1993, 45, 548–569. [Google Scholar]
- Burgmaier, G.; Schonrock, L.M.; Kuhlmann, T.; Richter-Landsberg, C.; Bruck, W. Association of increased bcl-2 expression with rescue from tumor necrosis factor-alpha-induced cell death in the oligodendrocyte cell line OLN-93. J. Neurochem 2000, 75, 2270–2276. [Google Scholar]
- Wood, J.P.; Pergande, G.; Osborne, N.N. Prevention of glutathione depletion-induced apoptosis in cultured human RPE cells by flupirtine. Restor. Neurol. Neurosci 1998, 12, 119–125. [Google Scholar]
- Davis, P.K.; Ho, A.; Dowdy, S.F. Biological methods for cell-cycle synchronization of mammalian cells. BioTechniques 2001, 30, 1322–1331. [Google Scholar]
- Wang, Z. Roles of K+ channels in regulating tumour cell proliferation and apoptosis. Pflugers Arch 2004, 448, 274–286. [Google Scholar]
- Spitzner, M.; Ousingsawat, J.; Scheidt, K.; Kunzelmann, K.; Schreiber, R. Voltage-gated K+ channels support proliferation of colonic carcinoma cells. FASEB J 2007, 21, 35–44. [Google Scholar]
- Jang, S.H.; Choi, S.Y.; Ryu, P.D.; Lee, S.Y. Anti-proliferative effect of Kv1.3 blockers in A549 human lung adenocarcinoma in vitro and in vivo. Eur. J. Pharmacol. 2011, 651, 26–32. [Google Scholar]
- Jehle, J.; Schweizer, P.A.; Katus, H.A.; Thomas, D. Novel roles for hERG K+ channels in cell proliferation and apoptosis. Cell Death Dis 2011, 2. [Google Scholar] [CrossRef]
- Glassmeier, G.; Hempel, K.; Wulfsen, I.; Bauer, C.K.; Schumacher, U.; Schwarz, J.R. Inhibition of HERG1 K+ channel protein expression decreases cell proliferation of human small cell lung cancer cells. Pflugers Arch 2012, 463, 365–376. [Google Scholar]
- Asher, V.; Warren, A.; Shaw, R.; Sowter, H.; Bali, A.; Khan, R. The role of Eag and HERG channels in cell proliferation and apoptotic cell death in SK-OV-3 ovarian cancer cell line. Cancer Cell Int 2011, 11. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.S.; Sayeed, M.M.; Wurster, R.D. In vitro antitumor activity of cromakalim in human brain tumor cells. Pharmacology 1994, 49, 69–74. [Google Scholar]
- Iannotti, F.A.; Barrese, V.; Formisano, L.; Miceli, F.; Taglialatela, M. Specification of skeletal muscle differentiation by repressor element-1 silencing transcription factor (REST)-regulated Kv7.4 potassium channels. Mol. Biol. Cell 2013, 24, 274–284. [Google Scholar]
- Mucha, M.; Ooi, L.; Linley, J.E.; Mordaka, P.; Dalle, C.; Robertson, B.; Gamper, N.; Wood, I.C. Transcriptional control of KCNQ channel genes and the regulation of neuronal excitability. J. Neurosci 2010, 30, 13235–13245. [Google Scholar]
- Yang, G.; Pei, Y.; Teng, H.; Cao, Q.; Wang, R. Specificity protein-1 as a critical regulator of human cystathionine gamma-lyase in smooth muscle cells. J. Biol. Chem 2011, 286, 26450–26460. [Google Scholar]
- Ming, L.; Sakaida, T.; Yue, W.; Jha, A.; Zhang, L.; Yu, J. Sp1 and p73 activate PUMA following serum starvation. Carcinogenesis 2008, 29, 1878–1884. [Google Scholar]
- Vivar, O.I.; Lin, C.L.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-Diindolylmethane induces a G1 arrest in human prostate cancer cells irrespective of androgen receptor and p53 status. Biochem. Pharmacol 2009, 78, 469–476. [Google Scholar]
- Firestone, G.L.; Bjeldanes, L.F. Indole-3-carbinol and 3-3′-diindolylmethane antiproliferative signaling pathways control cell-cycle gene transcription in human breast cancer cells by regulating promoter-Sp1 transcription factor interactions. J. Nutr 2003, 133, S2448–S2455. [Google Scholar]
- Beardsley, A.; Fang, K.; Mertz, H.; Castranova, V.; Friend, S.; Liu, J. Loss of caveolin-1 polarity impedes endothelial cell polarization and directional movement. J. Biol. Chem 2005, 280, 3541–3547. [Google Scholar]


| Gene | Sequences | Product size | Accession number |
|---|---|---|---|
| Kv7.2 | F 5′-CCATTGGTTATGGGGACAAG-3′ R 5′-ATAGAACCTCCAGGCCGACT-3′ | 212 | JN546558.1 |
| Kv7.3 | F 5′-GCTTCAGCATCTCCCAAGAC-3′ R 5′-GGGAGGGGTCCATACTGAAT-3′ | 188 | XM_532334.3 |
| Kv7.4 | F 5′-TGGCCAAAAGGAAATTCAAG-3′ R 5′-CCCCTTGTCTCCCTTCTCTC-3′ | 179 | XM_539568.3 |
| Kv7.5 | F 5′-CGCTTTCGTTTTTCTCCTTG-3′ R 5′-GCAGACCAGATCCGAATGAT-3′ | 156 | XM_003431766.1 |
| Sp1 | F 5′-TGCAGCAGAATTGAGTCACC-3′ R 5′-CACAACATACTGCCCACCAG-3′ | 246 | XM_543633.3 |
| GAPDH | F 5′-AAGGTCATCCCTGAGCTGAA-3′ R 5′-GACCACCTGGTCCTCAGTGT-3′ | 192 | NM_001003142.1 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, B.H.; Ryu, P.D.; Lee, S.Y. Serum Starvation-Induced Voltage-Gated Potassium Channel Kv7.5 Expression and Its Regulation by Sp1 in Canine Osteosarcoma Cells. Int. J. Mol. Sci. 2014, 15, 977-993. https://doi.org/10.3390/ijms15010977
Lee BH, Ryu PD, Lee SY. Serum Starvation-Induced Voltage-Gated Potassium Channel Kv7.5 Expression and Its Regulation by Sp1 in Canine Osteosarcoma Cells. International Journal of Molecular Sciences. 2014; 15(1):977-993. https://doi.org/10.3390/ijms15010977
Chicago/Turabian StyleLee, Bo Hyung, Pan Dong Ryu, and So Yeong Lee. 2014. "Serum Starvation-Induced Voltage-Gated Potassium Channel Kv7.5 Expression and Its Regulation by Sp1 in Canine Osteosarcoma Cells" International Journal of Molecular Sciences 15, no. 1: 977-993. https://doi.org/10.3390/ijms15010977
APA StyleLee, B. H., Ryu, P. D., & Lee, S. Y. (2014). Serum Starvation-Induced Voltage-Gated Potassium Channel Kv7.5 Expression and Its Regulation by Sp1 in Canine Osteosarcoma Cells. International Journal of Molecular Sciences, 15(1), 977-993. https://doi.org/10.3390/ijms15010977




