Structural Characterization of the Degradation Products of a Minor Natural Sweet Diterpene Glycoside Rebaudioside M under Acidic Conditions
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Reagents and Chemicals
3.2. Plant Material
3.3. HPLC Conditions
3.4. General Instrumentation
3.5. Degradation of Rebaudioside M (1)
3.5.1. Isolation of Degradation Compounds 2–4
3.5.2. Physical and Spectroscopic Data of Compounds 2–4
3.5.3. General Procedure for Acid Hydrolysis and Determination of Sugar Configuration in 2–4
3.5.4. Enzymatic Hydrolysis of 4
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Mosettig, E.; Nes, W.R. Stevioside. II. The structure of the aglucon. J. Org. Chem 1955, 20, 884–899. [Google Scholar]
- Mosettig, E.; Beglinger, U.; Dolder, F.; Lichiti, H.; Quitt, P.; Waters, J.A. The absolute configuration of steviol and isosteviol. J. Am. Chem. Soc 1963, 85, 2305–2309. [Google Scholar]
- Brandle, J.E.; Starrratt, A.N.; Gijen, M. Stevia rebaudiana: Its agricultural, biological and chemical properties. Can. J. Plant Sci 1998, 78, 527–536. [Google Scholar]
- Wayne, E.S.; Lin, L. NMR studies of the conformation of the natural sweetener rebaudioside A. Carbohydr. Res 2009, 344, 2533–2538. [Google Scholar]
- Prakash, I.; Chaturvedula, V.S.P.; Markosyan, A. Isolation, characterization and sensory evaluation of a hexa β-d-glucopyranosyl diterpene from Stevia rebaudiana. Nat. Prod. Commun 2013, 8, 1523–1526. [Google Scholar]
- Prakash, I.; Chaturvedula, V.S.P. Additional minor diterpene glycosides from Stevia rebaudiana bertoni. Molecules 2013, 18, 13510–13519. [Google Scholar]
- Chaturvedula, V.S.P.; Mani, U.; Prakash, I. Diterpene glycosides from Stevia rebaudiana. Molecules 2011, 16, 3552–3562. [Google Scholar]
- Chaturvedula, V.S.P.; Prakash, I. A new diterpenoid glycoside from Stevia rebaudiana. Molecules 2011, 16, 2937–2943. [Google Scholar]
- Chaturvedula, V.S.P.; Prakash, I. Structures of the novel diterpene glycosides from Stevia rebaudiana. Carbohydr. Res 2011, 346, 1057–1060. [Google Scholar]
- Chaturvedula, V.S.P.; Prakash, I. Hydrogenation of the exocyclic olefinic bond at C-16/C-17 position of ent-kaurane diterpene glycosides of Stevia rebaudiana using various catalysts. Int. J. Mol. Sci 2013, 14, 15669–15680. [Google Scholar]
- Prakash, I.; Clos, J.F.; Chaturvedula, V.S.P. Stability of rebaudioside A under acidic conditions and its degradation products. Food Res. Int 2012, 48, 65–75. [Google Scholar]
- Prakash, I.; Campbell, M.; Miguel, R.I.S.; Chaturvedula, V.S.P. Synthesis and sensory evaluation of ent-kaurane diterpene glycosides. Molecules 2012, 17, 8908–8916. [Google Scholar]
- Prakash, I.; Campbell, M.; Chaturvedula, V.S.P. Catalytic hydrogenation of the sweet principles of Stevia rebaudiana, rebaudioside B, rebaudioside C, and rebaudioside D and sensory evaluation of their reduced derivatives. Int. J. Mol. Sci 2012, 13, 15126–15136. [Google Scholar]
- Chaturvedula, V.S.P.; Klucik, J.; Mani, U.; Prakash, I. Synthesis of ent-kaurane diterpene glycosides. Molecules 2011, 16, 8402–8409. [Google Scholar]
- Chaturvedula, V.S.P.; Clos, J.F.; Prakash, I. Stability study of steviol glycosides in mock beverages using fluorescent light exposure under ICH guidelines. Int. J. Pharm. Pharm. Sci 2011, 3, 316–323. [Google Scholar]
- Chaturvedula, V.S.P.; Clos, J.F.; Prakash, I. Stability of steviol glycosides in mock beverages under acidic conditions. Int. J. Pharm. Pharm. Sci 2011, 3, 421–425. [Google Scholar]
- Clos, J.F.; DuBois, G.E.; Prakash, I. Photostability of rebaudioside A and stevioside in beverages. J. Agric. Food Chem 2008, 56, 8507–8513. [Google Scholar]
- Tanaka, T.; Nakashima, T.; Ueda, T.; Tomii, K.; Kouno, I. Facile discrimination of aldose enantiomers by reversed-phase HPLC. Chem. Pharm. Bull 2007, 55, 899–901. [Google Scholar]
- Kohda, H.; Kasai, R.; Yamsaki, K.; Murakami, K.; Tanaka, O. New sweet diterpene glucosides from Stevia rebaudiana. Phytochemistry 1976, 15, 981–983. [Google Scholar]
- Avent, A.G.; Hanson, J.R.; DeOliviera, B.H. Hydrolysis of the diterpenoid glycoside, stevioside. Phytochemistry 1990, 29, 2712–2715. [Google Scholar]
- Ohtani, K.; Aikawa, Y.; Kasai, R.; Chou, W.; Yamasaki, K.; Tanaka, O. Minor diterpene glycosides from sweet leaves of Rubus suavissimus. Phytochemistry 1992, 31, 1553–1559. [Google Scholar]

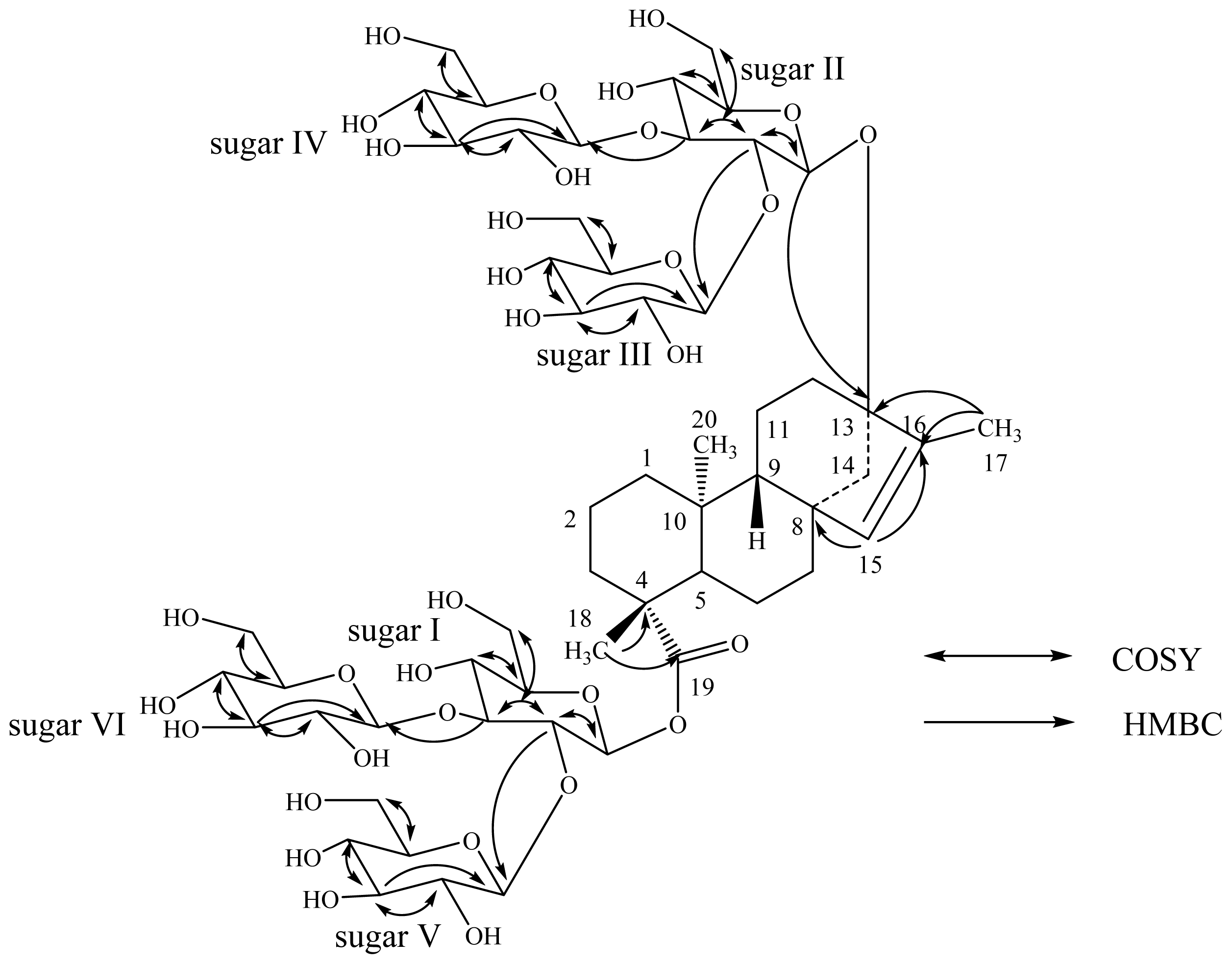
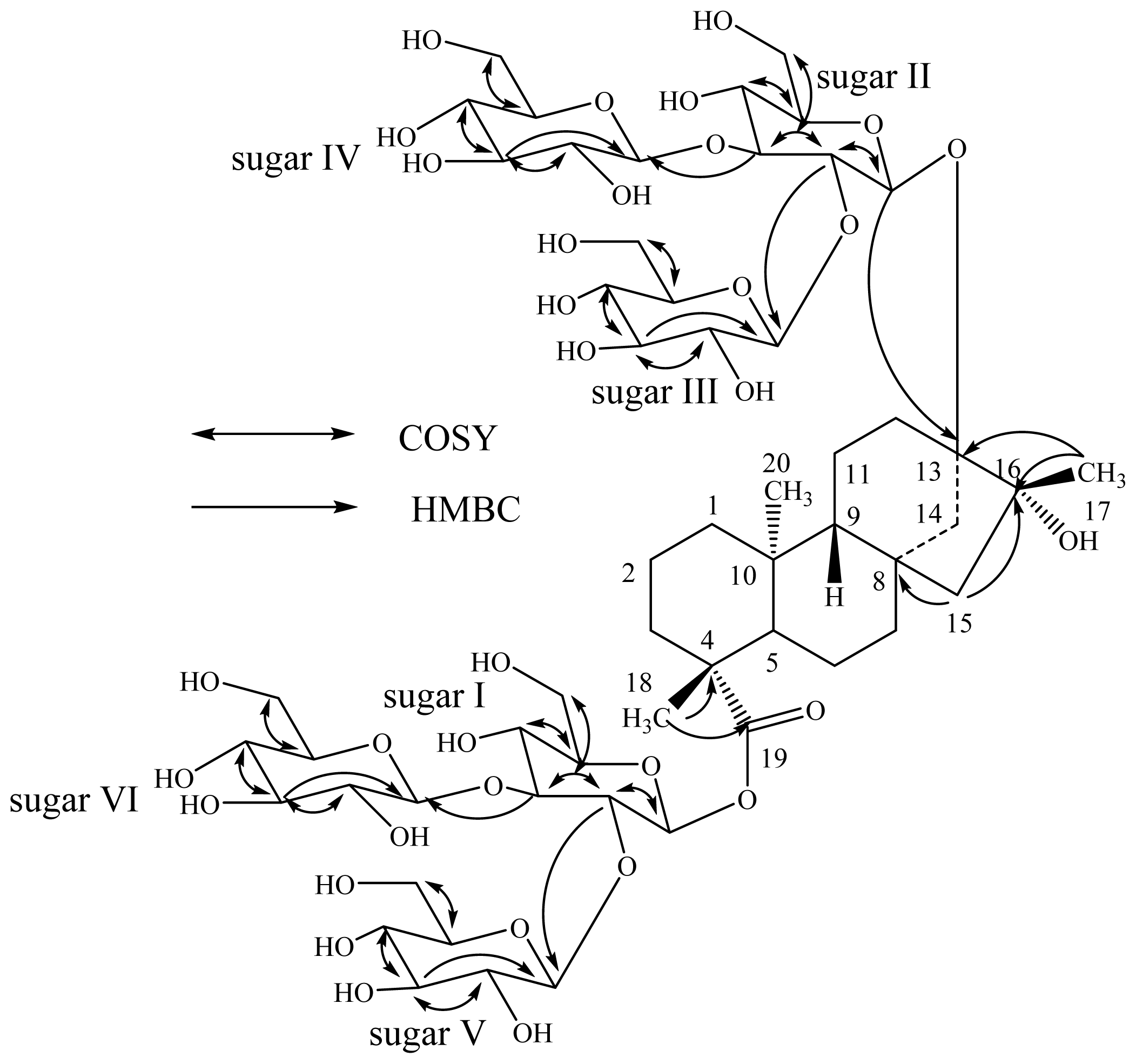
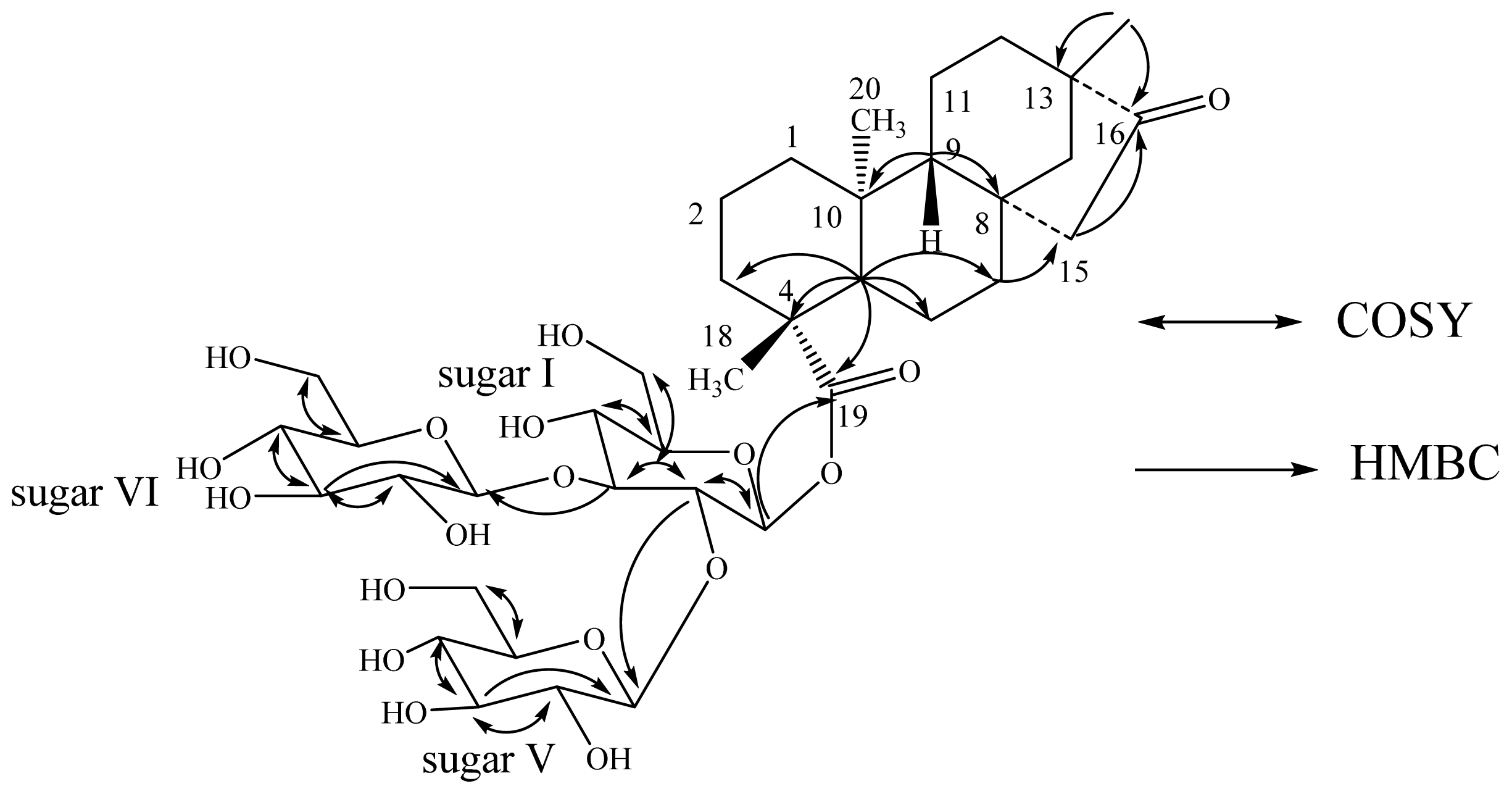
| Position | 2 | 3 | 4 | |||
|---|---|---|---|---|---|---|
| 1H | 13C | 1H | 13C | 1H | 13C | |
| 1 | 0.77 t (12.8), 1.76 d (12.8) | 40.2 | 0.78 t (13.2), 1.75 d (13.0) | 40.3 | 0.77 td (3.8, 13.2), 1.47 m | 39.4 |
| 2 | 1.36 m, 2.25 m | 19.3 | 1.34 m, 2.23 m | 19.3 | 2.02 m | 21.6 |
| 3 | 1.03 m, 2.33 m | 38.0 | 1.00 td (3.9, 12.8), 2.32 d (12.3) | 38.4 | 1.07 m, 2.77 d (13.1) | 37.3 |
| 4 | - | 44.0 | - | 43.8 | - | 44.1 |
| 5 | 1.05 d (12.7) | 56.9 | 1.04 d (12.7) | 57.1 | 1.06 m | 57.1 |
| 6 | 2.21 m, 2.31 m | 21.8 | 2.11 d (13.5), 2.43 m | 23.1 | 1.38 m, 2.04 m | 19.4 |
| 7 | 1.49 m, 1.89 m | 40.0 | 1.37 m, 1.88 m | 42.8 | 1.34 m, 1.61 m | 41.3 |
| 8 | - | - | - | |||
| 9 | 0.83 d (8.4) | 47.0 | 0.84 d (8.1) | 54.8 | 1.04 m | 54.4 |
| 10 | - | 39.6 | - | - | ||
| 11 | 1.56 m, 1.67 m | 20.9 | 1.52 m, 1.71 m | 19.8 | 1.04 m, 1.46 m | 20.1 |
| 12 | 1.84 t (12.2), 2.36 m | 29.9 | 1.85 m, 2.67 m | 31.6 | 1.24 m, 1.51 m | 37.1 |
| 13 | - | 89.6 | - | 87.6 | - | 54.2 |
| 14 | 2.17 d (9.8), 2.69 d (9.8) | 47.1 | 2.44 m, 2.58 d (9.9) | 40.3 | 1.83 d (18.5), 2.62 dd (3.1, 18.5) | 48.4 |
| 15 | 5.03 s | 134.3 | 1.41 d (14.3), 1.83 m | 54.3 | 1.27 m, 1.36 m | 53.8 |
| 16 | - | 144.4 | - | 77.1 | - | 220.7 |
| 17 | 1.89 s | 12.7 | 1.32 s | 22.2 | 0.98 s | 19.9 |
| 18 | 1.35 s | 28.0 | 1.28 s | 27.7 | 1.42 s | 28.8 |
| 19 | - | 176.7 | - | 176.9 | - | 175.8 |
| 20 | 1.33 s | 16.4 | 1.31 s | 16.0 | 0.81s | 13.8 |
| 1′ | 6.33 d (8.4) | 94.5 | 6.35 d (8.3) | 94.5 | 6.19 d (8.1) | 93.0 |
| 2′ | 4.51 t (8.8) | 76.6 | 4.53 t (8.7) | 76.4 | 4.51 t (8.9) | 76.8 |
| 3′ | 4.99 m | 88.3 | 4.98 m | 88.5 | 4.27 t m | 88.4 |
| 4′ | 4.19 m | 69.6 | 4.21 m | 69.7 | 4.17 m | 69.1 |
| 5′ | 4.10 m | 78.1 | 4.14 m | 78.3 | 3.98 m | 78.1 |
| 6′ | 4.21 m, 4.33 m | 61.5 | 4.22 m, 4.30 m | 61.5 | 4.24 m, 4.52 m | 62.0 |
| 1″ | 5.47 d (8.0) | 95.7 | 5.79 d (6.7) | 96.0 | ||
| 2″ | 4.13 m | 81.1 | 4.14 m | 80.4 | ||
| 3″ | 4.89 t (8.9) | 87.6 | 5.09 m | 87.9 | ||
| 4″ | 4.04 t (8.9) | 70.0 | 4.00 m | 69.9 | ||
| 5″ | 3.90 m | 77.4 | 4.17 m | 77.2 | ||
| 6″ | 4.17 m, 4.31 m | 62.4 | 4.13 m, 4.34 m | 62.0 | ||
| 1″′ | 5.45 d (7.0) | 104.5 | 5.63 d (7.3) | 103.8 | ||
| 2″′ | 4.15 m | 75.6 | 4.13 m | 75.6 | ||
| 3″′ | 4.14 m | 78.1 | 4.09 m | 78.0 | ||
| 4″′ | 4.02 m | 72.8 | 4.08 m | 72.2 | ||
| 5″′ | 3.77 m | 77.3 | 3.70 m | 77.5 | ||
| 6″′ | 4.31 m, 4.53 d (10.9) | 63.5 | 4.32 m, 4.49 m | 63.2 | ||
| 1″″ | 5.44 d (7.5) | 103.9 | 5.53 d (7.9) | 103.8 | ||
| 2″″ | 3.98 m | 75.3 | 4.02 m | 75.3 | ||
| 3″″ | 4.46 t (9.0) | 77.6 | 4.55 t (9.0) | 77.6 | ||
| 4″″ | 4.14 m | 71.0 | 4.22 m | 71.0 | ||
| 5″″ | 3.99 m | 77.8 | 4.09 m | 78.0 | ||
| 6″″ | 4.12 m, 4.33 m | 61.8 | 4.13 m, 4.34 m | 62.0 | ||
| 1″″′ | 5.81 d (6.9) | 103.8 | 5.76 d (7.8) | 104.0 | 5.79 d (7.9) | 103.4 |
| 2″″′ | 4.20 m | 75.2 | 4.28 m | 74.8 | 3.99 m | 75.7 |
| 3″″′ | 4.20 m | 78.2 | 4.20 m | 78.1 | 4.32 m | 78.3 |
| 4″″′ | 4.10 m | 73.1 | 4.13 m | 73.5 | 4.13 m | 72.4 |
| 5″″′ | 3.89 m | 77.8 | 3.92 | 77.6 | 4.02 m | 78.4 |
| 6″″′ | 4.31 m, 4.62 d (11.1) | 63.6 | 4.29 m, 4.64 dd (2.6, 11.6) | 63.8 | 4.36 m, 4.56 m | 63.2 |
| 1″″′ | 5.33 d (7.7) | 103.9 | 5.22 d (7.8) | 104.0 | 5.35 d (7.9) | 104.5 |
| 2″″′ | 3.97 m | 75.1 | 3.95 m | 75.1 | 4.04 t (8.2) | 75.2 |
| 3″″′ | 4.35 t (8.3) | 77.7 | 4.28 m | 77.7 | 4.21 m | 78.3 |
| 4″″′ | 4.11 m | 70.9 | 4.08 m | 70.8 | 4.14 m | 71.2 |
| 5″″′ | 3.85 m | 77.7 | 3.76 m | 77.8 | 3.85 m | 78.7 |
| 6″″′ | 4.12 m, 4.33 m | 61.8 | 4.13 m, 4.34 m | 62.0 | 4.24 m, 4.33 m | 61.6 |
| Time (min) | % of Mobile Phase A | % of Mobile Phase B |
|---|---|---|
| 0.0 | 75 | 25 |
| 20.0 | 69 | 31 |
| 20.5 | 50 | 50 |
| 25.0 | 40 | 60 |
| 25.1 | 75 | 25 |
| 30.0 | 75 | 25 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Prakash, I.; Chaturvedula, V.S.P.; Markosyan, A. Structural Characterization of the Degradation Products of a Minor Natural Sweet Diterpene Glycoside Rebaudioside M under Acidic Conditions. Int. J. Mol. Sci. 2014, 15, 1014-1025. https://doi.org/10.3390/ijms15011014
Prakash I, Chaturvedula VSP, Markosyan A. Structural Characterization of the Degradation Products of a Minor Natural Sweet Diterpene Glycoside Rebaudioside M under Acidic Conditions. International Journal of Molecular Sciences. 2014; 15(1):1014-1025. https://doi.org/10.3390/ijms15011014
Chicago/Turabian StylePrakash, Indra, Venkata Sai Prakash Chaturvedula, and Avetik Markosyan. 2014. "Structural Characterization of the Degradation Products of a Minor Natural Sweet Diterpene Glycoside Rebaudioside M under Acidic Conditions" International Journal of Molecular Sciences 15, no. 1: 1014-1025. https://doi.org/10.3390/ijms15011014




