Exploring the Role of Genetic Variability and Lifestyle in Oxidative Stress Response for Healthy Aging and Longevity
Abstract
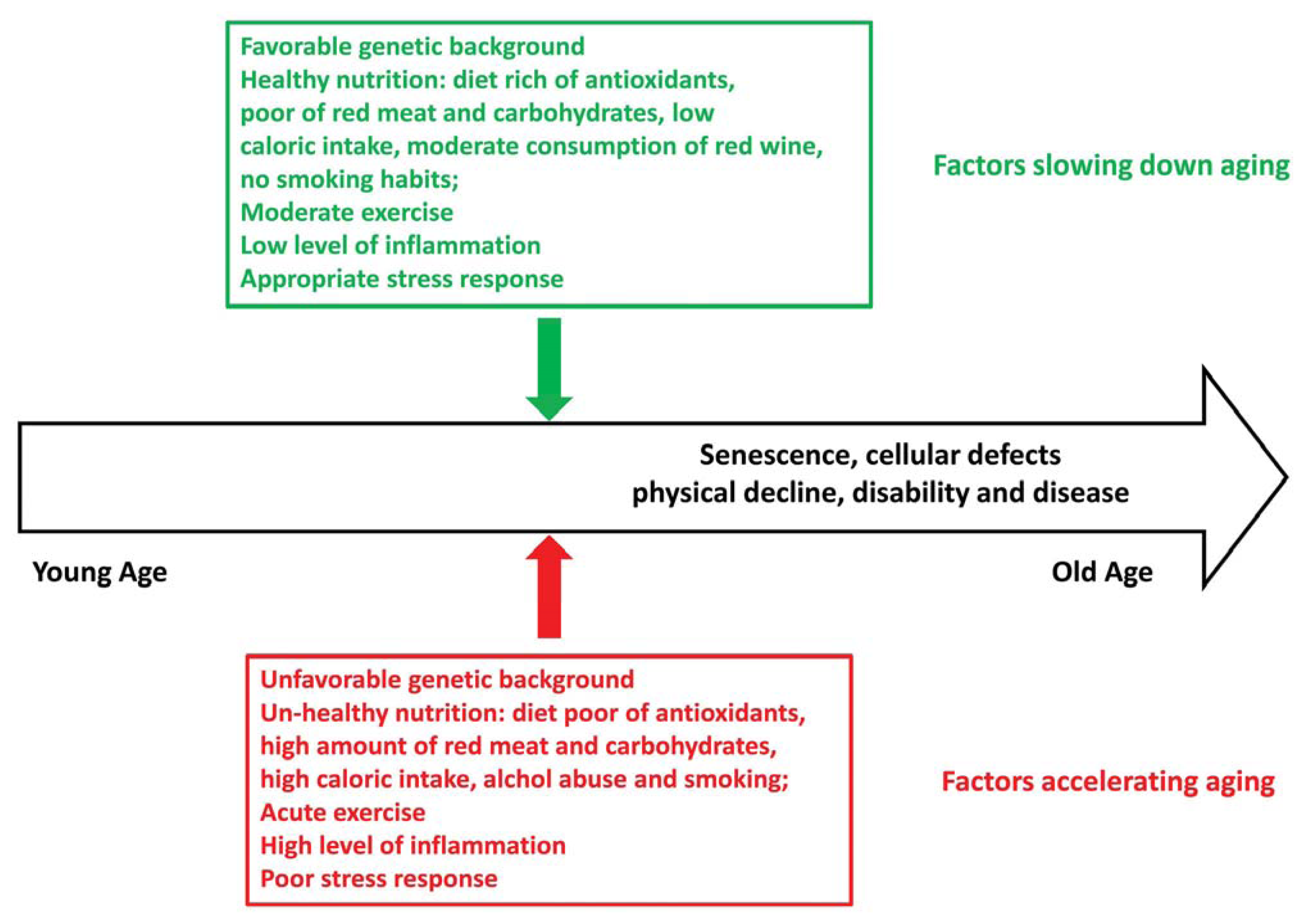
:1. Introduction
1.1. Oxidative Stress at the Cellular Level
1.2. Oxidative Stress and Aging
2. Role of Genetic Variability in Oxidative Stress Response for Healthy Aging and Longevity
Mitochondrial DNA Variability, Oxidative Stress and Aging: A Complex Interaction
3. Role of Lifestyle in the Oxidative Stress Response for Healthy Aging and Longevity
3.1. Physical Activity
3.2. Dietary Intake of Antioxidants
3.3. Antioxidant Supplementation to Diet
4. Conclusions
Acknowledgments
Conflict of interest
References
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar]
- Pamplona, R. Mitochondrial DNA damage and animal longevity: Insights from comparative studies. J. Aging Res. 2011, 807108:1–807108:9. [Google Scholar]
- Kregel, K.C.; Zhang, H.J. An integrated view of oxidative stress in aging: Basic mechanisms, functional effects, and pathological considerations. Am. J. Physiol. Regul. Integr. Comp. Physiol 2007, 292, R18–R36. [Google Scholar]
- Pérez, V.I.; Bokov, A.; van Remmen, H.; Mele, J.; Ran, Q.; Ikeno, Y.; Richardson, A. Is the oxidative stress theory of aging dead? Biochim. Biophys. Acta 2009, 1790, 1005–1014. [Google Scholar]
- Anson, R.M.; Bohr, V.A. Mitochondria, oxidative DNA damage, and aging. J. Am. Aging Assoc 2000, 23, 199–218. [Google Scholar]
- Li, J.; Holbrook, N.J. Common mechanisms for declines in oxidative stress tolerance and proliferation with aging. Exp. Gerontol 2003, 35, 292–299. [Google Scholar]
- Ikeyama, S.; Kokkonen, G.; Shack, S.; Wang, X.T.; Holbrook, N.J. Loss in oxidative stress tolerance with aging linked to reduced extracellular signal-regulated kinase and Akt kinase activities. FASEB J 2002, 16, 114–116. [Google Scholar]
- Murphy, C.T. Using whole-genome transcriptional analyses to identify molecular mechanisms of Aging. Drug Discov. Today 2006, 3, 41–46. [Google Scholar]
- Butterfield, D.A.; Perluigi, M.; Sultana, R. Oxidative stress in Alzheimer’s disease brain: New insights from redox proteomics. Eur. J. Pharmacol 2006, 545, 39–50. [Google Scholar]
- Wood-Kaczmar, A.S.; Gandhi, S.; Wood, W.N. Understanding the molecular causes of Parkinsons’s disease. Trends Mol. Med 2006, 12, 521–528. [Google Scholar]
- Aviram, M. Review of human studies on oxidative damage and antioxidant protection related to cardiovascular diseases. Free Radic. Res 2000, 33, S85–S97. [Google Scholar]
- Parthasarathy, S.; Litvinov, D.; Selvarajan, K.; Garelnabi, M. Lipid peroxidation and decomposition-conflicting roles in plaque vulnerability and stability. Biochim. Biophys. Acta 2008, 1781, 221–231. [Google Scholar]
- Hitchon, C.A.; El-Gabalawy, H.S. Oxidation in rheumatoid arthritis. Arthritis Res. Ther 2004, 6, 265–278. [Google Scholar]
- Halliwell, B. Oxidative stress and cancer: Have we moved forward? Biochem. J 2007, 401, 1–11. [Google Scholar]
- Willcox, J.K.; Ash, S.L.; Catignani, G.L. Antioxidants and prevention of chronic disease. Crit. Rev. Food Sci. Nutr 2004, 44, 275–295. [Google Scholar]
- Toyokuni, S.; Okamoto, K.; Yodoi, J.; Hiai, H. Persistent oxidative stress in cancer. FEBS Lett 1995, 358, 1–3. [Google Scholar]
- Nunomura, A.; Perry, G.; Pappolla, M.A.; Wade, R.; Hirai, K.; Chiba, S.; Smith, M.A. RNA oxidation is a prominent feature of vulnerable neurons in Alzheimer’s disease. J. Neurosci 1999, 19, 1959–1964. [Google Scholar]
- Nunomura, A.; Chiba, S.; Kosaka, K.; Takeda, A.; Castellani, R.J.; Smith, M.A.; Perry, G. Neuronal RNA oxidation is a prominent feature of dementia with Lewy bodies. Neuroreport 2002, 13, 2035–2039. [Google Scholar]
- Kikuchi, A.; Takeda, A.; Onodera, H.; Kimpara, T.; Hisanaga, K.; Sato, N.; Nunomura, A.; Castellani, R.J.; Perry, G.; Smith, M.A.; et al. Systemic increase of oxidative nucleic acid damage in Parkinson’s disease and multiple system atrophy. Neurobiol. Dis 2002, 9, 244–248. [Google Scholar]
- Martinet, W.; de Meyer, G.R.; Herman, A.G.; Kockx, M.M. Reactive oxygen species induce RNA damage in human atherosclerosis. Eur. J. Clin. Invest 2004, 34, 323–327. [Google Scholar]
- Tateyama, M.; Takeda, A.; Onodera, Y.; Matsuzaki, M.; Hasegawa, T.; Nunomura, A.; Hirai, K.; Perry, G.; Smith, M.A.; Itoyama, Y. Oxidative stress and predominant Aβ42(43) deposition in myopathies with rimmed vacuoles. Acta Neuropathol 2003, 105, 581–585. [Google Scholar]
- Beckman, J.S.; Koppenol, W.H. Nitric oxide, superoxide, and peroxynitrite: The good, the bad, and ugly. Am. J. Physiol 1996, 271, C1424–C1437. [Google Scholar]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev 2007, 87, 315–424. [Google Scholar]
- Calcerrada, P.; Peluffo, G.; Radi, R. Nitric oxide-derived oxidants with a focus on peroxynitrite: Molecular targets, cellular responses and therapeutic implications. Curr. Pharm. Des 2011, 17, 3905–3932. [Google Scholar]
- Harman, D. Prolongation of the normal life span by radiation protection chemicals. J. Gerontol 1957, 12, 257–263. [Google Scholar]
- Harman, D. A biologic clock: The mitochondria? J. Am. Geriatr. Soc 1972, 20, 145–147. [Google Scholar]
- Fusco, D.; Colloca, G.; Lo Monaco, M.R.; Cesari, M. Effects of antioxidant supplementation on the aging process. Clin. Interv. Aging 2007, 2, 377–387. [Google Scholar]
- Sastre, J.; Pallardó, F.V.; Viña, J. Mitochondrial oxidative stress plays a key role in aging and apoptosis. IUBMB Life 2000, 49, 427–435. [Google Scholar]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci 2001, 56, M146–M156. [Google Scholar]
- Swindell, W.R. Dietary restriction in rats and mice: A meta-analysis and review of the evidence for genotype-dependent effects on lifespan. Ageing Res. Rev 2012, 11, 254–270. [Google Scholar]
- Houthoofd, K.; Braeckman, B.P.; Lenaerts, I.; Brys, K.; de Vreese, A.; van Eygen, S.; Vanfleteren, J.R. No reduction of metabolic rate in food restricted Caenorhabditis elegans. Exp. Gerontol 2002, 37, 1359–1369. [Google Scholar]
- Puca, A.A.; Carrizzo, A.; Villa, F.; Ferrario, A.; Casaburo, M.; Maciag, A.; Vecchione, C. Vascular aging: The role of oxidative stress. Int. J. Biochem. Cell Biol 2013, 45, 556–559. [Google Scholar]
- Ristow, M.; Zarse, K. How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis). Exp. Gerontol 2010, 45, 410–418. [Google Scholar]
- Burtner, C.R.; Kennedy, B.K. Progeria syndromes and ageing: What is the connection? Nat. Rev. Mol. Cell. Biol 2010, 11, 567–578. [Google Scholar]
- Cluett, C.; Melzer, D. Human genetic variations: Beacons on the pathways to successful ageing. Mech. Ageing Dev 2009, 130, 553–563. [Google Scholar]
- Kenyon, C. A pathway that links reproductive status to lifespan in Caenorhabditis elegans. Ann. N. Y. Acad. Sci 2010, 1204, 156–162. [Google Scholar]
- Kuningas, M.; Mooijaart, S.P.; van Heemst, D.; Zwaan, B.J.; Slagboom, P.E.; Westendorp, R.G. Genes encoding longevity: From model organisms to humans. Aging Cell 2008, 7, 270–280. [Google Scholar]
- Vijg, J.; Suh, Y. Genetics of longevity and aging. Annu. Rev. Med 2005, 56, 193–212. [Google Scholar]
- Bartke, A. Mutations prolong life in flies; implications for aging in mammals. Trends Endocrinol. Metab 2001, 12, 233–234. [Google Scholar]
- Herskind, A.M.; McGue, M.; Holm, N.V.; Sørensen, T.I.; Harvald, B.; Vaupel, J.W. The heritability of human longevity: A population-based study of 2872 Danish twin pairs born 1870–1900. Hum. Genet 1996, 97, 319–323. [Google Scholar]
- Hjelmborg, J.; Iachine, I.; Skytthe, A.; Vaupel, J.W.; McGue, M.; Koskenvuo, M.; Kaprio, J.; Pedersen, N.L.; Christensen, K. Genetic influence on human lifespan and longevity. Hum. Genet 2006, 119, 312–321. [Google Scholar]
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; de Luca, M.; Ottaviani, E.; de Benedictis, G. Inflamm-aging: An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci 2000, 908, 244–254. [Google Scholar]
- Sansoni, P.; Cossarizza, A.; Brianti, V.; Fagnoni, F.; Snelli, G.; Monti, D.; Marcato, A.; Passeri, G.; Ortolani, C.; Forti, E.; et al. Lymphocyte subsets and natural killer cell activity in healthy old people and centenarians. Blood 1993, 82, 2767–2773. [Google Scholar]
- Mezzetti, A.; Lapenna, D.; Romano, F.; Costantini, F.; Pierdomenico, S.D.; de Cesare, D.; Cuccurullo, F.; Riario-Sforza, G.; Zuliani, G.; Fellin, R. Systemic oxidative stress and its relationship with age and illness. Associazione Medica “Sabin”. J. Am. Geriatr. Soc 1996, 44, 823–827. [Google Scholar]
- Mariotti, S.; Barbesino, G.; Caturegli, P.; Bartalena, L.; Sansoni, P.; Fagnoni, F.; Monti, D.; Fagiolo, U.; Franceschi, C.; Pinchera, A. Complex alteration of thyroid function in healthy centenarians. J. Clin. Endocrinol. Metab 1993, 77, 1130–1134. [Google Scholar]
- Paolisso, G.; Gambardella, A.; Ammendola, S.; D’Amore, A.; Balbi, V.; Varricchio, M.; D’Onofrio, F. Glucose tolerance and insulin action in healty centenarians. Am. J. Physiol 1996, 270, E890–E894. [Google Scholar]
- Passarino, G.; Montesanto, A.; Dato, S.; Giordano, S.; Domma, F.; Mari, V.; Feraco, E.; de Benedictis, G. Sex and age specificity of susceptibility genes modulating survival at old age. Hum. Hered 2006, 62, 213–220. [Google Scholar]
- Suzuki, M.; Willcox, D.C.; Rosenbaum, M.W.; Willcox, B.J. Oxidative stress and longevity in okinawa: An investigation of blood lipid peroxidation and tocopherol in okinawan centenarians. Curr. Gerontol. Geriatr. Res 2010, 380460:1–380460:10. [Google Scholar]
- Paolisso, G.; Tagliamonte, M.R.; Rizzo, M.R.; Manzella, D.; Gambardella, A.; Varricchio, M. Oxidative stress and advancing age: Results in healthy centenarians. J. Am. Geriatr. Soc 1998, 46, 833–838. [Google Scholar]
- Di Pietro, F.; Dato, S.; Carpi, F.M.; Corneveaux, J.J.; Serfaustini, S.; Maoloni, S.; Mignini, F.; Huentelman, M.J.; Passarino, G.; Napolioni, V. TP53*P72 allele influences negatively female life expectancy in a population of central Italy: Cross-sectional study and genetic-demographic approach analysis. J. Gerontol. A Biol. Sci. Med. Sci 2013, 68, 539–545. [Google Scholar]
- Van Heemst, D.; Mooijaart, S.P.; Beekman, M.; Schreuder, J.; de Craen, A.J.; Brandt, B.W.; Slagboom, P.E.; Westendorp, R.G. Variation in the human TP53 gene affects old age survival and cancer mortality. Exp. Gerontol 2005, 40, 11–15. [Google Scholar]
- Altilia, S.; Santoro, A.; Malagoli, D.; Lanzarini, C.; Ballesteros Álvarez, J.A.; Galazzo, G.; Porter, D.C.; Crocco, P.; Rose, G.; Passarino, G.; et al. TP53 codon 72 polymorphism affects accumulation of mtDNA damage in human cells. Aging (Albany N. Y.) 2012, 4, 28–39. [Google Scholar]
- Soerensen, M.; Christensen, K.; Stevnsner, T.; Christiansen, L. The Mn-superoxide dismutase single nucleotide polymorphism rs4880 and the glutathione peroxidase 1 single nucleotide polymorphism rs1050450 are associated with aging and longevity in the oldest old. Mech. Ageing Dev 2009, 130, 308–314. [Google Scholar]
- Lunetta, K.L.; D’Agostino, R.B., Sr.; Karasik, D.; Benjamin, E.J.; Guo, C.Y.; Govindaraju, R.; Kiel, D.P.; Kelly-Hayes, M.; Massaro, J.M.; Pencina, M.J.; et al. Genetic correlates of longevity and selected age-related phenotypes: A genome-wide association study in the Framingham Study. BMC Med. Genet. 2007, 8 Suppl 1. [Google Scholar] [CrossRef]
- Montesanto, A.; Crocco, P.; Tallaro, F.; Pisani, F.; Mazzei, B.; Mari, V.; Corsonello, A.; Lattanzio, F.; Passarino, G.; Rose, G. Common polymorphisms in nitric oxide synthase (NOS) genes influence quality of aging and longevity in humans. Biogerontology 2013, 14, 177–186. [Google Scholar]
- Altomare, K.; Greco, V.; Bellizzi, D.; Berardelli, M.; Dato, S.; DeRango, F.; Garasto, S.; Rose, G.; Feraco, E.; Mari, V.; et al. The allele (A)−110 in the promoter region of the HSP70-1 gene is unfavorable to longevity in women. Biogerontology 2003, 4, 215–220. [Google Scholar]
- Singh, R.; Kølvraa, S.; Bross, P.; Christensen, K.; Gregersen, N.; Tan, Q.; Jensen, U.B.; Eiberg, H.; Rattan, S.I. Heat-shock protein 70 genes and human longevity: A view from Denmark. Ann. N. Y. Acad. Sci 2006, 1067, 301–308. [Google Scholar]
- Ross, O.A.; Curran, M.D.; Crum, K.A.; Rea, I.M.; Barnett, Y.A.; Middleton, D. Increased frequency of the 2437T allele of the heat shock protein 70-Hom gene in an aged Irish population. Exp. Gerontol 2003, 38, 561–565. [Google Scholar]
- Kim, S.; Bi, X.; Welsh, M.D.A.; Myers, L.; Welsch, M.A.; Cherry, K.E.; Arnold, J.; Poon, L.W.; Jazwinski, M.S. Telomere maintenance genes SIRT1 and XRCC6 impact age-related decline in telomere length but only SIRT1 is associated with human longevity. Biogerontology 2012, 13, 119–131. [Google Scholar]
- Kuningas, M.; Putters, M.; Westendorp, R.G.; Slagboom, P.E.; van Heemst, D. SIRT1 gene, age-related diseases, and mortality: The Leiden 85-plus study. J. Gerontol. A Biol. Sci. Med. Sci 2007, 62, 960–965. [Google Scholar]
- Dato, S.; Rose, G.; Altomare, K.; Bellizzi, D.; Garasto, S.; Greco, V.; Passarino, G.; Feraco, E.; Mari, V.; Barbi, C.; et al. Variability of the SIRT3 gene, human silent information regulator Sir2 homologue, and survivorship in the elderly. Exp. Gerontol 2003, 38, 1065–1070. [Google Scholar]
- Lescai, F.; Blanché, H.; Nebel, A.; Beekman, M.; Sahbatou, M.; Flachsbart, F.; Slagboom, E.; Schreiber, S.; Sorbi, S.; Passarino, G.; et al. Human longevity and 11p15.5: A study in 1321 centenarians. Eur. J. Hum. Genet 2009, 17, 1515–1519. [Google Scholar]
- Bellizzi, D.; Rose, G.; Cavalcante, P.; Covello, G.; Dato, S.; de Rango, F.; Greco, V.; Maggiolini, M.; Feraco, E.; Mari, V.; et al. A novel VNTR enhancer within the SIRT3 gene, a human homologue of SIR2, is associated with survival at oldest ages. Genomics 2005, 85, 258–263. [Google Scholar]
- Rose, G.; Crocco, P.; D’Aquila, P.; Montesanto, A.; Bellizzi, D.; Passarino, G. Two variants located in the upstream enhancer region of human UCP1 gene affect gene expression and are correlated with human longevity. Exp. Gerontol 2011, 46, 897–904. [Google Scholar]
- Rose, G.; Crocco, P.; de Rango, F.; Montesanto, A.; Passarino, G. Further support to the uncoupling-to-survive theory: The genetic variation of human UCP genes is associated with longevity. PLoS One 2011, 6, e29650. [Google Scholar]
- Crocco, P.; Montesanto, A.; Passarino, G.; Rose, G. A common polymorphism in the UCP3 promoter influences hand grip strength in elderly people. Biogerontology 2011, 1, 265–271. [Google Scholar]
- Dato, S.; Soerensen, M.; Montesanto, A.; Lagani, V.; Passarino, G.; Christensen, K.; Christiansen, L. UCP3 polymorphisms, hand grip performance and survival at old age: Association analysis in two Danish middle aged and elderly cohorts. Mech. Ageing Dev 2012, 133, 530–537. [Google Scholar]
- Soerensen, M.; Dato, S.; Tan, Q.; Thinggaard, M.; Kleindorp, R.; Beekman, M.; Jacobsen, R.; Suchiman, H.E.; de Craen, A.J.; Westendorp, R.G.; et al. Human longevity and variation in GH/IGF-1/insulin signaling, DNA damage signaling and repair and pro/antioxidant pathway genes: Cross sectional and longitudinal studies. Exp. Gerontol 2012, 47, 379–387. [Google Scholar]
- Di Cianni, F.; Campa, D.; Tallaro, F.; Rizzato, C.; de Rango, F.; Barale, R.; Passarino, G.; Canzian, F.; Gemignani, F.; Montesanto, A.; et al. MAP3K7 and GSTZ1 are associated with human longevity: A two-stage case-control study using a multilocus genotyping. Age 2013, 35, 1357–1366. [Google Scholar]
- Bonafè, M.; Marchegiani, F.; Cardelli, M.; Olivieri, F.; Cavallone, L.; Giovagnetti, S.; Pieri, C.; Marra, M.; Antonicelli, R.; Troiano, L.; et al. Genetic analysis of paraoxonase (PON1) locus reveals an increased frequency of Arg192 allele in centenarians. Eur. J. Hum. Genet 2002, 10, 292–296. [Google Scholar]
- Marchegiani, F.; Marra, M.; Spazzafumo, L.; James, R.W.; Boemi, M.; Olivieri, F.; Cardelli, M.; Cavallone, L.; Bonfigli, A.R.; Franceschi, C. Paraoxonase activity and genotype predispose to a successful aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci 2006, 61, 541–546. [Google Scholar]
- Rea, I.M.; McKeown, P.P.; McMaster, D.; Young, I.S.; Patterson, C.; Savage, M.J.; Belton, C.; Marchegiani, F.; Olivieri, F.; Bonafe, M.; et al. Paraoxonase polymorphisms PON1192 and 55 and longevity in Italian centenarians and Irish nonagenarians. A pooled analysis. Exp. Gerontol 2004, 39, 629–635. [Google Scholar]
- Caliebe, A.; Kleindorp, R.; Blanché, H.; Christiansen, L.; Puca, A.A.; Rea, I.M.; Slagboom, E.; Flachsbart, F.; Christensen, K.; Rimbach, G.; et al. No or only population-specific effect of PON1 on human longevity: A comprehensive meta-analysis. Ageing Res. Rev 2010, 9, 238–244. [Google Scholar]
- Campo, S.; Sardo, M.A.; Trimarchi, G.; Bonaiuto, M.; Fontana, L.; Castaldo, M.; Bonaiuto, A.; Saitta, C.; Bitto, A.; Manduca, B.; et al. Association between serum paraoxonase (PON1) gene promoter T(-107)C polymorphism, PON1 activity and HDL levels in healthy Sicilian octogenarians. Exp. Gerontol 2004, 39, 1089–1094. [Google Scholar]
- Li, Y.; Wang, W.J.; Cao, H.; Lu, J.; Wu, C.; Hu, F.Y.; Guo, J.; Zhao, L.; Yang, F.; Zhang, Y.X.; et al. Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum. Mol. Genet 2009, 18, 4897–4904. [Google Scholar]
- Willcox, B.J.; Donlon, T.A.; He, Q.; Chen, R.; Grove, J.S.; Yano, K.; Masaki, K.H.; Willcox, D.C.; Rodriguez, B.; Curb, J.D. FOXO3A genotype is strongly associated with human longevity. Proc. Natl. Acad. Sci. USA 2008, 105, 13987–13992. [Google Scholar]
- Anselmi, C.V.; Malovini, A.; Roncarati, R.; Novelli, V.; Villa, F.; Condorelli, G.; Bellazzi, R.; Puca, A.A. Association of the FOXO3A locus with extreme longevity in a southern Italian centenarian study. Rejuvenation Res 2009, 12, 95–104. [Google Scholar]
- Flachsbart, F.; Caliebe, A.; Kleindorp, R.; Blanché, H.; von Eller-Eberstein, H.; Nikolaus, S.; Schreiber, S.; Nebel, A. Association of FOXO3A variation with human longevity confirmed in German centenarians. Proc. Natl. Acad. Sci. USA 2009, 106, 2700–2705. [Google Scholar]
- Pawlikowska, L.; Hu, D.; Huntsman, S.; Sung, A.; Chu, C.; Chen, J.; Joyner, A.H.; Schork, N.J.; Hsueh, W.C.; Reiner, A.P.; et al. Association of common genetic variation in the insulin/IGF1 signaling pathway with human longevity. Aging Cell 2009, 8, 460–472. [Google Scholar]
- Soerensen, M.; Dato, S.; Christensen, K.; McGue, M.; Stevnsner, T.; Bohr, V.A.; Christiansen, L. Replication of an association of variation in the FOXO3A gene with human longevity using both case-control and longitudinal data. Aging Cell 2010, 9, 1010–1017. [Google Scholar]
- Soerensen, M.; Dato, S.; Tan, Q.; Thinggaard, M.; Kleindorp, R.; Beekman, M.; Suchiman, H.E.; Jacobsen, R.; McGue, M.; Stevnsner, T.; et al. Evidence from case-control and longitudinal studies supports associations of genetic variation in APOE, CETP, and IL6 with human longevity. Age 2013, 35, 487–500. [Google Scholar]
- Kojima, T.; Kamei, H.; Aizu, T.; Arai, Y.; Takayama, M.; Nakazawa, S.; Ebihara, Y.; Inagaki, H.; Masui, Y.; Gondo, Y.; et al. Association analysis between longevity in the Japanese population and polymorphic variants of genes involved in insulin and insulin-like growth factor 1 signaling pathways. Exp. Gerontol 2004, 39, 1595–1598. [Google Scholar]
- Van Heemst, D.; Beekman, M.; Mooijaart, S.P.; Heijmans, B.T.; Brandt, B.W.; Zwaan, B.J.; Slagboom, P.E.; Westendorp, R.G. Reduced insulin/IGF-1 signalling and human longevity. Aging Cell 2005, 4, 79–85. [Google Scholar]
- Bonafè, M.; Barbieri, M.; Marchegiani, F.; Olivieri, F.; Ragno, E.; Giampieri, C.; Mugianesi, E.; Centurelli, M.; Franceschi, C.; Paolisso, G. Polymorphic variants of insulin-like growth factor I (IGF-I) receptor and phosphoinositide 3-kinase genes affect IGF-I plasma levels and human longevity: Cues for an evolutionarily conserved mechanism of life span control. J. Clin. Endocrinol. Metab 2003, 88, 3299–3304. [Google Scholar]
- Stessman, J.; Maaravi, Y.; Hammerman-Rozenberg, R.; Cohen, A.; Nemanov, L.; Gritsenko, I.; Gruberman, N.; Ebstein, R.P. Candidate genes associated with ageing and life expectancy in the Jerusalem longitudinal study. Mech. Ageing Dev 2005, 126, 333–339. [Google Scholar]
- Soerensen, M.; Thinggaard, M.; Nygaard, M.; Dato, S.; Tan, Q.; Hjelmborg, J.; Andersen-Ranberg, K.; Stevnsner, T.; Bohr, V.A.; Kimura, M.; et al. Genetic variation in TERT and TERC and human leukocyte telomere length and longevity: A cross-sectional and longitudinal analysis. Aging Cell 2012, 11, 223–227. [Google Scholar]
- Atzmon, G.; Cho, M.; Cawthon, R.M.; Budagov, T.; Katz, M.; Yang, X.; Siegel, G.; Bergman, A.; Huffman, D.M.; Schechter, C.B.; et al. Evolution in health and medicine Sackler colloquium: Genetic variation in human telomerase is associated with telomere length in Ashkenazi centenarians. Proc. Natl. Acad. Sci. USA 2010, 107 Suppl 1, 1710–1717. [Google Scholar]
- Concetti, F.; Lucarini, N.; Carpi, F.M.; di Pietro, F.; Dato, S.; Capitani, M.; Nabissi, M.; Santoni, G.; Mignini, F.; Passarino, G.; et al. The functional VNTR MNS16A of the TERT gene is associated with human longevity in a population of Central Italy. Exp. Gerontol 2013, 48, 587–592. [Google Scholar]
- Rose, G.; Romeo, G.; Dato, S.; Crocco, P.; Bruni, A.C.; Hervonen, A.; Majamaa, K.; Sevini, F.; Franceschi, C.; Passarino, G. the GEHA Project Consortium. Somatic point mutations in mtDNA control region are influenced by genetic background and associated with healthy aging: A GEHA study. PLoS One 2010, 5, e13395. [Google Scholar]
- Rose, G.; Passarino, G.; Scornaienchi, V.; Romeo, G.; Dato, S.; Bellizzi, D.; Mari, V.; Feraco, E.; Maletta, R.; Bruni, A.; et al. The mitochondrial DNA control region shows genetically correlated levels of heteroplasmy in leukocytes of centenarians and their offspring. BMC Genomics 2007, 8. [Google Scholar] [CrossRef]
- Tanaka, M.; Gong, J.S.; Zhang, J.; Yoneda, M.; Yagi, K. Mitochondrial genotype associated with longevity. Lancet 1998, 351, 185–186. [Google Scholar]
- De Benedictis, G.; Rose, G.; Carrieri, G.; de Luca, M.; Falcone, E.; Passarino, G.; Bonafè, M.; Monti, D.; Baggio, G.; Bertolini, S.; et al. Mitochondrial DNA inherited variants are associated with successful aging and longevity in humans. FASEB J 1999, 13, 1532–1536. [Google Scholar]
- Ross, O.A.; McCormack, R.; Curran, M.D.; Duguid, R.A.; Barnett, Y.A.; Rea, I.M.; Middleton, D. Mitochondrial DNA polymorphism: Its role in longevity of the Irish population. Exp. Gerontol 2001, 36, 1161–1178. [Google Scholar]
- Niemi, A.K.; Hervonen, A.; Hurme, M.; Karhunen, P.J.; Jylhä, M.; Majamaa, K. Mitochondrial DNA polymorphisms associated with longevity in a Finnish population. Hum. Genet 2003, 112, 29–33. [Google Scholar]
- Bilal, E.; Rabadan, R.; Alexe, G.; Fuku, N.; Ueno, H.; Nishigaki, Y.; Fujita, Y.; Ito, M.; Arai, Y.; Hirose, N.; et al. Mitochondrial DNA haplogroup D4a is a marker for extreme longevity in Japan. PLoS One 2008, 3, e2421. [Google Scholar]
- Cai, X.Y.; Wang, X.F.; Li, S.L.; Qian, J.; Qian, D.G.; Chen, F.; Yang, Y.J.; Yuan, Z.Y.; Xu, J.; Bai, Y.; et al. Association of mitochondrial DNA haplogroups with exceptional longevity in a Chinese population. PLoS One 2009, 4, e6423. [Google Scholar]
- Feng, J.; Zhang, J.; Liu, M.; Wan, G.; Qi, K.; Zheng, C.; Lv, Z.; Hu, C.; Zeng, Y.; Gregory, S.G.; et al. Association of mtDNA haplogroup F with healthy longevity in the female Chuang population, China. Exp. Gerontol 2011, 46, 987–993. [Google Scholar]
- Crawford, A.; Fassett, R.G.; Geraghty, D.P.; Kunde, D.A.; Ball, M.J.; Robertson, I.K.; Coombes, J.S. Relationship between single nucleotide polymorphisms of antioxidant enzymes and disease. Gene 2012, 501, 89–103. [Google Scholar]
- Vera, E.; de Jesus, B.B.; Foronda, M.; Flores, J.M.; Blasco, M.A. Telomerase reverse transcriptase synergizes with calorie restriction to increase health span and extend mouse longevity. PLoS One 2013, 8, e53760. [Google Scholar]
- Vina, J.; Gambini, J.; Lopez-Grueso, R.; Abdelaziz, K.M.; Jove, M.; Borras, C. Females live longer than males: Role of oxidative stress. Curr. Pharm. Des 2011, 17, 3959–3965. [Google Scholar]
- Spazzafumo, L.; Olivieri, F.; Abbatecola, A.M.; Castellani, G.; Monti, D.; Lisa, R.; Galeazzi, R.; Sirolla, C.; Testa, R.; Ostan, R.; et al. Remodelling of biological parameters during human ageing: Evidence for complex regulation in longevity and in type 2 diabetes. Age 2013, 35, 419–429. [Google Scholar]
- Beekman, M.; Blanché, H.; Perola, M.; Hervonen, A.; Bezrukov, V.; Sikora, E.; Flachsbart, F.; Christiansen, L.; de Craen, A.J.; Kirkwood, T.B.; et al. Genome-wide linkage analysis for human longevity: Genetics of healthy aging study. Aging Cell 2013, 12, 184–193. [Google Scholar]
- Tranah, G.J. Mitochondrial-nuclear epistasis: Implications for human aging and longevity. Ageing Res. Rev 2011, 10, 238–252. [Google Scholar]
- De Benedictis, G.; Carrieri, G.; Garasto, S.; Rose, G.; Varcasia, O.; Bonafè, M.; Franceschi, C.; Jazwinski, S.M. Does a retrograde response in human aging and longevity exist? Exp. Gerontol 2000, 35, 795–801. [Google Scholar]
- Santoro, A.; Balbi, V.; Balducci, E.; Pirazzini, C.; Rosini, F.; Tavano, F.; Achilli, A.; Siviero, P.; Minicuci, N.; Bellavista, E.; et al. Evidence for sub-haplogroup h5 of mitochondrial DNA as a risk factor for lateonset Alzheimer’s disease. PLoS One 2010, 5, e12037. [Google Scholar]
- Carrieri, G.; Bonafè, M.; de Luca, M.; Rose, G.; Varcasia, O.; Bruni, A.; Maletta, R.; Nacmias, B.; Sorbi, S.; Corsonello, F.; et al. Mitochondrial DNA haplogroups and APOE4 allele are non-independent variables in sporadic Alzheimer’s disease. Hum. Genet 2001, 108, 194–198. [Google Scholar]
- Garesse, R.; Vallejo, C.G. Animal mitochondrial biogenesis and function: A regulatory cross-talk between two genomes. Gene 2001, 263, 1–16. [Google Scholar]
- Piantadosi, C.A.; Suliman, H.B. Redox regulation of mitochondrial biogenesis. Free Radic. Biol. Med 2012, 53, 2043–2053. [Google Scholar]
- Yoboue, E.D.; Devin, A. Reactive oxygen species-mediated control of mitochondrial biogenesis. Int. J. Cell Biol. 2012. [Google Scholar] [CrossRef]
- Scarpulla, R.C. Transcriptional activators and coactivators in the nuclear control of mitochondrial function in mammalian cells. Gene 2002, 286, 81–89. [Google Scholar]
- Scarpulla, R.C. Nucleus-encoded regulators of mitochondrial function: Integration of respiratory chain expression, nutrient sensing and metabolic stress. Biochim. Biophys. Acta 2012, 1819, 1088–1097. [Google Scholar]
- Jazwinski, S.M. Yeast longevity and aging—The mitochondrial connection. Mech. Ageing Dev 2005, 126, 243–248. [Google Scholar]
- Gogvadze, V.; Orrenius, S. Mitochondrial regulation of apoptotic cell death. Chem. Biol. Interact 2006, 163, 4–14. [Google Scholar]
- Tait, S.W.; Green, D.R. Mitochondria and cell signalling. J. Cell Sci 2012, 125, 807–815. [Google Scholar]
- Butow, R.A.; Avadhani, N.G. Mitochondrial signaling: The retrograde response. Mol. Cell 2004, 14, 1–15. [Google Scholar]
- King, M.P.; Attardi, G. Human cells lacking mtDNA: Repopulation with exogenous mitochondria by complementation. Science 1989, 246, 500–503. [Google Scholar]
- Vives-Bauza, C.; Gonzalo, R.; Manfredi, G.; Garcia-Arumi, E.; Andreu, A.L. Enhanced ROS production and antioxidant defenses in cybrids harbouring mutations in mtDNA. Neurosci. Lett 2006, 391, 136–141. [Google Scholar]
- Bellizzi, D.; Cavalcante, P.; Taverna, D.; Rose, G.; Passarino, G.; Salvioli, S.; Franceschi, C.; de Benedictis, G. Gene expression of cytokines and cytokine receptors is modulated by the common variability of the mitochondrial DNA in cybrid cell lines. Genes Cells 2006, 11, 883–891. [Google Scholar]
- Bellizzi, D.; Taverna, D.; D’Aquila, P.; de Blasi, S.; de Benedictis, G. Mitochondrial DNA variability modulates mRNA and intra-mitochondrial protein levels of HSP60 and HSP75: Experimental evidence from cybrid lines. Cell Stress Chaperones 2009, 14, 265–271. [Google Scholar]
- D’Aquila, P.; Rose, G.; Panno, M.L.; Passarino, G.; Bellizzi, D. SIRT3 gene expression: A link between inherited mitochondrial DNA variants and oxidative stress. Gene 2012, 497, 323–329. [Google Scholar]
- Herndon, L.A.; Schmeissner, P.J.; Dudaronek, J.M.; Brown, P.A.; Listner, K.M.; Sakano, Y.; Paupard, M.C.; Hall, D.H.; Driscoll, M. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 2002, 419, 808–814. [Google Scholar]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T. for the Lancet Physical Activity Series Working Group. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar]
- Fiatarone, M.A.; O’Neill, E.F.; Ryan, N.D.; Clements, K.M.; Solares, G.R.; Nelson, M.E.; Roberts, S.B.; Kehayias, J.J.; Lipsitz, L.A.; Evans, W.J. Exercise training and nutritional supplementation for physical frailty in very elderly people. N. Engl. J. Med 1994, 330, 1769–1775. [Google Scholar]
- Polidori, M.C.; Mecocci, P.; Cherubini, A.; Senin, U. Physical activity and oxidative stress during aging. Int. J. Sports Med 2000, 21, 154–157. [Google Scholar]
- Singh, S.; Melkani, G.C.; Rani, C.; Gaur, S.P.; Agrawal, V.; Agrawal, C.G. Oxidative stress and metabolic control in non-insulin dependent diabetes mellitus. Indian J. Biochem. Biophys 1997, 34, 512–517. [Google Scholar]
- Olinski, R.; Gackowski, D.; Rozalski, R.; Foksinski, M.; Bialkowski, K. Oxidative DNA damage in cancer patients: A cause or a consequence of the disease development? Mutat. Res 2003, 531, 177–190. [Google Scholar]
- Dengel, D.R.; Hagberg, J.M.; Pratley, R.E.; Rogus, E.M.; Goldberg, A.P. Improvements in blood pressure, glucose metabolism, and lipoprotein lipids after aerobic exercise plus weight loss in obese, hypertensive middle-aged men. Metabolism 1998, 47, 1075–1082. [Google Scholar]
- Evans, W.J. Exercise training guidelines for the elderly. Med. Sci. Sports Exerc 1999, 31, 12–17. [Google Scholar]
- Wojtaszewski, J.F.; Nielsen, P.; Hansen, B.F.; Richter, E.A.; Kiens, B. Isoform-specific and exercise intensity-dependent activation of 5′-AMP-activated protein kinase in human skeletal muscle. J. Physiol 2000, 528, 221–226. [Google Scholar]
- Arroll, B.; Beaglehole, R. Does physical activity lower blood pressure: A critical review of the clinical trials. J. Clin. Epidemiol 1992, 45, 439–447. [Google Scholar]
- Thompson, P.D.; Buchner, D.; Piña, I.L.; Balady, G.J.; Williams, M.A.; Marcus, B.H.; Berra, K.; Blair, S.N.; Costa, F.; Franklin, B.; et al. Exercise and physical activity in the prevention and treatment of atherosclerotic cardiovascular disease. Circulation 2003, 107, 3109–3116. [Google Scholar]
- Corbi, G.; Conti, V.; Scapagnini, G.; Filippelli, A.; Ferrara, N. Role of sirtuins, calorie restriction and physical activity in aging. Front. Biosci 2012, 4, 768–778. [Google Scholar]
- Paffenbarger, R.S., Jr; Hyde, R.T.; Wing, A.L.; Lee, I.M.; Jung, D.L.; Kampert, J.B. The association of changes in physical-activity level and other lifestyle characteristics with mortality among men. N. Engl. J. Med. 1993, 328, 538–545. [Google Scholar]
- Fulle, S.; Protasi, F.; di Tano, G.; Pietrangelo, T.; Beltramin, A.; Boncompagni, S.; Vecchiet, L.; Fanò, G. The contribution of reactive oxygen species to sarcopenia and muscle ageing. Exp. Gerontol 2004, 39, 17–24. [Google Scholar]
- Semba, R.D.; Blaum, C.; Guralnik, J.M.; Moncrief, D.T.; Ricks, M.O.; Fried, LP. Carotenoid and vitamin E status are associated with indicators of sarcopenia among older women living in the community. Aging Clin. Exp. Res. 2003, 15, 482–487. [Google Scholar]
- Hu, F.B.; Willett, W.C.; Li, T.; Stampfer, M.J.; Colditz, G.A.; Manson, J.E. Adiposity as compared with physical activity in predicting mortality among women. N. Engl. J. Med 2004, 351, 2694–2703. [Google Scholar]
- Cesari, M.; Pahor, M.; Bartali, B.; Cherubini, A.; Penninx, B.W.; Williams, G.R.; Atkinson, H.; Martin, A.; Guralnik, J.M.; Ferrucci, L. Antioxidants and physical performance in elderly persons: The Invecchiare in Chianti (InCHIANTI) study. Am. J. Clin. Nutr 2004, 79, 289–294. [Google Scholar]
- Muller, F.L.; Lustgarten, M.S.; Jang, Y.; Richardson, A.; van Remmen, H. Trends in oxidative aging theories. Free Radic. Biol. Med 2007, 43, 477–503. [Google Scholar]
- Cotman, C.; Engesser-Cesar, C. Exercise enhances and protects brain function. Exerc. Sports Sci. Rev 2002, 30, 75–79. [Google Scholar]
- Vaynman, S.; Ying, Z.; Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci 2004, 20, 2580–2590. [Google Scholar]
- Van Praag, H.; Shubert, T.; Zhao, C.; Gage, F.H. Exercise enhances learning and hippocampal neurogenesis in aged mice. J. Neurosci 2005, 25, 8680–8685. [Google Scholar]
- Dik, M.G.; Deeg, D.J.H.; Visser, M.; Jonker, C. Early life physical activity and cognition at old age. J. Clin. Exp. Neuropsychol 2003, 25, 643–653. [Google Scholar]
- INSERM Collective Expertise Centre, Physical Activity: Contexts and Effects on Health. In INSERM Collective Expert Reports; Institut national de la santé et de la recherche médicale: Paris, France, 2008.
- Cardoso, A.M.; Bagatini, M.D.; Roth, M.A.; Martins, C.C.; Rezer, J.F.; Mello, F.F.; Lopes, L.F.; Morsch, V.M.; Schetinger, M.R. Acute effects of resistance exercise and intermittent intense aerobic exercise on blood cell count and oxidative stress in trained middle-aged women. Braz. J. Med. Biol. Res 2012, 45, 1172–1182. [Google Scholar]
- Ji, L.L.; Leeuwenburgh, C.; Leichtweis, S.; Gore, M.; Fiebig, R.; Hollander, J.; Bejma, J. Oxidative stress and aging. Role of exercise and its influences on antioxidant systems. Ann. N. Y. Acad. Sci 1998, 854, 102–117. [Google Scholar]
- Zerba, E.; Komorowski, T.E.; Faulkner, J.A. Free radical injury to skeletal muscles of young, adult, and old mice. Am. J. Physiol 1990, 258, C429–C435. [Google Scholar]
- Watson, T.A.; MacDonald-Wicks, L.K.; Garg, M.L. Oxidative stress and antioxidants in athletes undertaking regular exercise training. Int. J. Sport Nutr. Exerc. Metab 2005, 15, 131–146. [Google Scholar]
- Pedersen, B.K.; Ostrowski, K.; Rohde, T.; Bruunsgaard, H. The cytokine response to strenuous exercise. Can. J. Physiol. Pharmacol 1998, 76, 505–511. [Google Scholar]
- Urquiaga, I.; Strobel, P.; Perez, D.; Martinez, C.; Cuevas, A.; Castillo, O.; Marshall, G.; Rozowski, J.; Leighton, F. Mediterranean diet and red wine protect against oxidative damage in young volunteers. Atherosclerosis 2010, 211, 694–699. [Google Scholar]
- Riccioni, G.; Bucciarelli, T.; Mancini, B.; di Ilio, C.; Capra, V.; D’Orazio, N. The role of the antioxidant vitamin supplementation in the prevention of cardiovascular diseases. Expert Opin. Investig. Drugs 2007, 16, 25–32. [Google Scholar]
- Vasto, S.; Rizzo, C.; Caruso, C. Centenarians and diet: What they eat in the Western part of Sicily. Immun. Ageing 2012, 9, 10, :1–10:6.. [Google Scholar]
- Covas, M.I.; Gambert, P.; Fitó, M.; de la Torre, R. Wine and oxidative stress: Up-to-date evidence of the effects of moderate wine consumption on oxidative damage in humans. Atherosclerosis 2010, 208, 297–304. [Google Scholar]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Suzuki, M. The Okinawan diet: Health implications of a low-calorie, nutrient-dense, antioxidant-rich dietary pattern low in glycemic load. J. Am. Coll. Nutr 2009, 28, 500S–516S. [Google Scholar]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med 2002, 113, 71S–88S. [Google Scholar]
- Huang, H.Y.; Appel, L.J.; Croft, K.D.; Miller, E.R., III; Mori, T.A.; Puddey, I.B. Effects of vitamin C and vitamin E on in vivo lipid peroxidation: Results of a randomized controlled trial. Am. J. Clin. Nutr. 2002, 76, 549–555. [Google Scholar]
- Maxwell, S.R. Prospects for the use of antioxidant therapies. Drugs 1995, 49, 345–361. [Google Scholar]
- Steinberg, D. A critical look at the evidence for the oxidation of LDL in atherogenesis. Atherosclerosis 1997, 131, S5–S7. [Google Scholar]
- Witztum, J.L. The oxidation hypothesis of atherosclerosis. Lancet 1994, 344, 793–795. [Google Scholar]
- Di Mascio, P.; Kaiser, S.P.; Devasagayam, T.P.; Sies, H. Biological significance of active oxygen species: In vitro studies on singlet oxygen-induced DNA damage and on the singlet oxygen quenching ability of carotenoids, tocopherols and thiols. Adv. Exp. Med. Biol 1991, 283, 71–77. [Google Scholar]
- Upritchard, J.E.; Schuurman, C.R.; Wiersma, A.; Tijburg, L.B.; Coolen, S.A.; Rijken, P.J.; Wiseman, S.A. Spread supplemented with moderate doses of vitamin E and carotenoids reduces lipid peroxidation in healthy, nonsmoking adults. Am. J. Clin. Nutr 2003, 78, 985–992. [Google Scholar]
- Prince, M.R.; LaMuraglia, G.M.; MacNichol, E.F., Jr. Increased preferential absorption in human atherosclerotic plaque with oral beta carotene. Implications for laser endarterectomy. Circulation 1988, 78, 338–344. [Google Scholar]
- Gaziano, J.M.; Manson, J.E.; Branch, L.G.; Colditz, G.A.; Willett, W.C.; Buring, J.E. A prospective study of consumption of carotenoids in fruits and vegetables and decreased cardiovascular mortality in the elderly. Ann. Epidemiol 1995, 5, 255–260. [Google Scholar]
- Niki, E.; Noguchi, N.; Tsuchihashi, H.; Gotoh, N. Interaction among vitamin C, vitamin E, and beta-carotene. Am. J. Clin. Nutr 1995, 62, 1322S–1326S. [Google Scholar]
- Aydogan, S.; Yerer, M.B.; Goktas, A. Melatonin and nitric oxide. J. Endocrinol. Invest 2006, 29, 281–287. [Google Scholar]
- Nishida, S. Metabolic effects of melatonin on oxidative stress and diabetes mellitus. Endocrine 2005, 27, 131–136. [Google Scholar]
- Pieri, C.; Marra, M.; Moroni, F.; Marcheselli, F.; Benatti, C. The modulation of intracellular glutathione level modulates the mitochondrial response in proliferating rat splenocytes. Arch. Gerontol. Geriatr 1995, 21, 115–125. [Google Scholar]
- Gaetke, L.M.; Chow, C.K. Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 2003, 189, 147–163. [Google Scholar]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Curb, J.D.; Suzuki, M. Caloric restriction and human longevity: What can we learn from the Okinawans? Biogerontology 2006, 7, 173–177. [Google Scholar]
- Rajpathak, S.N.; Liu, Y.; Ben-David, O.; Reddy, S.; Atzmon, G.; Crandall, J.; Barzilai, N. Lifestyle factors of people with exceptional longevity. J. Am. Geriatr. Soc 2011, 59, 1509–1512. [Google Scholar]
- Witte, A.V.; Fobker, M.; Gellner, R.; Knecht, S.; Flöel, A. Caloric restriction improves memory in elderly humans. Proc. Natl. Acad. Sci. USA 2009, 106, 1255–1260. [Google Scholar]
- Ingram, D.K.; Weindruch, R.; Spangler, E.L.; Freeman, J.R.; Walford, R.L. Dietary restriction benefits learning and motor performance of aged mice. J. Gerontol 1987, 42, 78–81. [Google Scholar]
- Park, S.K.; Kim, K.; Page, G.P.; Allison, D.B.; Weindruch, R.; Prolla, T.A. Gene expression profiling of aging in multiple mouse strains: Identification of aging biomarkers and impact of dietary antioxidants. Aging Cell 2009, 8, 484–495. [Google Scholar]
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009, 325, 201–204. [Google Scholar]
- Montiel, T.; Quiroz-Baez, R.; Massieu, L.; Arias, C. Role of oxidative stress on β-amyloid neurotoxicity elicited during impairment of energy metabolism in the hippocampus: Protection by antioxidants. Exp. Neurol 2006, 200, 496–508. [Google Scholar]
- Pavlik, V.N.; Doody, R.S.; Rountree, S.D.; Darby, E.J. Vitamin E use is associated with improved survival in an Alzheimer’s disease cohort. Dement. Geriat. Cogn. Disord 2009, 28, 536–540. [Google Scholar]
- Shargorodsky, M.; Ortal, D.; Matas, Z.; Zimlichma, N. Effect of long-term treatment with antioxidants (vitamin C, vitamin E, coenzyme Q10 and selenium) on arterial compliance, humoral factors and inflammatory markers in patients with multiple cardiovascular risk factors. Nutr. Metab 2010, 7, 55, :1–55:8.. [Google Scholar]
- Block, K.I.; Koch, A.C.; Mead, M.N.; Tothy, P.K.; Newmanc, R.A.; Gyllenhaal, C. Impact of antioxidant supplementation on chemotherapeutic efficacy: A systematic review of the evidence from randomized controlled trials. Cancer Treat. Rev 2007, 33, 407–418. [Google Scholar]
- Mocchegiani, E.; Santarelli, L.; Tibaldi, A.; Muzzioli, M.; Bulian, D.; Cipriano, K.; Olivieri, F.; Fabris, N. Presence of links between zinc and melatonin during the circadian cycle in old mice: Effects on thymic endocrine activity and on the survival. J. Neuroimmunol 1998, 86, 111–122. [Google Scholar]
- Porquet, D.; Casadesús, G.; Bayod, S.; Vicente, A.; Canudas, A.M.; Vilaplana, J.; Pelegrí, C.; Sanfeliu, C.; Camins, A.; Pallàs, M.; et al. Dietary resveratrol prevents Alzheimer’s markers and increases life span in SAMP8. Age 2012. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.S.; Lee, B.S.; Semnani, S.; Avanesian, A.; Um, C.Y.; Jeon, H.J.; Seong, K.M.; Yu, K.; Min, K.J.; Jafari, M. Curcumin extends life span, improves health span, and modulates the expression of age-associated aging genes in Drosophila melanogaster. Rejuvenation Res 2010, 13, 561–570. [Google Scholar]
- Pearson, K.J.; Baur, J.A.; Lewis, K.N.; Peshkin, L.; Price, N.; Labinskyy, N.; Swindell, W.R.; Kamara, D.; Minor, R.K.; Perez, E.; et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab 2008, 8, 157–168. [Google Scholar]
- Lönn, M.E.; Dennis, J.M.; Stocker, R. Actions of “antioxidants” in the protection against atherosclerosis. Free Radic. Biol. Med 2012, 53, 863–884. [Google Scholar]
- Zureik, M.; Galan, P.; Bertrais, S.; Mennen, L.; Czernichow, S.; Blacher, J.; Ducimetiere, P.; Hercberg, S. Effects of long-term daily low-dose supplementation with antioxidant vitamins and minerals on structure and function of large arteries. Arterioscler. Thromb. Vasc. Biol 2004, 24, 1485–1491. [Google Scholar]
- Song, Y.; Cook, N.R.; Albert, C.M.; van Denburgh, M.; Manson, J.E. Effects of vitamins C and E and beta-carotene on the risk of type 2 diabetes in women at high risk of cardiovascular disease: A randomized controlled trial. Am. J. Clin. Nutr 2009, 90, 429–437. [Google Scholar]
- Hercberg, S.; Ezzedine, K.; Guinot, C.; Preziosi, P.; Galan, P.; Bertrais, S.; Estaquio, C.; Briançon, S.; Favier, A.; Latreille, J.; et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J. Nutr 2007, 137, 2098–2105. [Google Scholar]
- Lawenda, B.D.; Kelly, K.M.; Ladas, E.J.; Sagar, S.M.; Vickers, A.; Blumberg, J.B. Should supplemental antioxidant administration be avoided during chemotherapy and radiation therapy? J. Natl. Cancer Inst 2008, 100, 773–783. [Google Scholar]
- Ward, N.C.; Wu, J.H.; Clarke, M.W.; Puddey, I.B.; Burke, V.; Croft, K.D.; Hodgson, J.M. The effect of vitamin E on blood pressure in individuals with type 2 diabetes: A randomized; double-blind; placebo-controlled trial. J. Hypertens 2007, 25, 227–234. [Google Scholar]
- Yang, L.; Pascal, M.; Wu, X.H. Review of selenium and prostate cancer prevention. Asian Pac. J. Cancer Prev 2013, 14, 2181–2184. [Google Scholar]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr 2005, 81, 341–354. [Google Scholar]
- Goldwyn, S.; Lazinsky, A.; Wei, H. Promotion of health by soy isoflavones: Efficacy, benefit and safety concerns. Drug Metabol. Drug Interact 2000, 17, 261–289. [Google Scholar]
- Terada, A.; Yoshida, M.; Seko, Y.; Kobayashi, T.; Yoshida, K.; Nakada, M.; Nakada, K.; Echizen, H.; Ogata, H.; Rikihisa, T. Active oxygen species generation and cellular damage by additives of parenteral preparations: Selenium and sulfhydryl compounds. Nutrition 1999, 15, 651–655. [Google Scholar]
- Berger, M.M. Can oxidative damage be treated nutritionally? Clin. Nutr 2005, 24, 172–183. [Google Scholar]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of Vitamin A. Am. J. Clin. Nutr 2006, 83, 191–201. [Google Scholar]
- Lee, K.W.; Lee, H.J.; Surh, Y.; Lee, C.Y. Vitamin C and cancer chemoprevention: A reappraisal. Am. J. Clin. Nutr 2003, 78, 1074–1078. [Google Scholar]
- Calabrese, V.; Cornelius, C.; Dinkova-Kostova, A.T.; Iavicoli, I.; di Paola, R.; Koverech, A.; Cuzzocrea, S.; Rizzarelli, E.; Calabrese, E.J. Cellular stress responses, hormetic phytochemicals and vitagenes in aging and longevity. Biochim. Biophys. Acta 2012, 1822, 753–783. [Google Scholar]
- Warner, H.R.; Ingram, D.; Miller, R.A.; Nadon, N.L.; Richardson, A.G. Program for testing biological interventions to promote healthy aging. Mech. Ageing Dev 2000, 115, 199–207. [Google Scholar]
- Nadon, N.L.; Strong, R.; Miller, R.A.; Nelson, J.; Javors, M.; Sharp, Z.D.; Peralba, J.M.; Harrison, D.E. Design of aging intervention studies: The NIA interventions testing program. Age 2008, 30, 187–199. [Google Scholar]
| Gene * | Biological function | Polymorphism | Position of the variation | Direction of association ** | References |
|---|---|---|---|---|---|
| Tp53 | Tumour suppressor p53 | rs1042522 | 534 C/G (aa33) | G POSITIVE (Italian; the Netherlands) | [50–52] |
| GPX1 | Glutathione peroxidase 1 | rs1050450 | 958 C/T | T POSITIVE (Danish) | [53] |
| SOD2 | Manganese superoxide dismutase | rs4880 rs911847 | 201 T/C (aa16) C/T variation near-gene | C POSITIVE (Danish) POSITIVE (U.S. Caucasian European) | [53] [54] |
| NOS1 | Nitric Oxide Synthase1 | rs1879417 | 2087 C/T (aa608) | C NEGATIVE (Italian) | [55] |
| NOS2 | Nitric Oxide Synthase 2 | rs2297518 | −34640 G/A | A NEGATIVE (Italian) | [55] |
| HSPA1A | Heat shock protein | rs1043618 | −110 A/T | Allele A NEGATIVE (Italian) AA Genotype NEGATIVE (Danish) | [56] [57] |
| HSPA1B | Heat shock protein | rs1061581 | 1059 G/A | POSITIVE (Danish) | [57] |
| HSPA1L | Heat shock protein | rs2227956 | 1661 C/T (aa493) | NEGATIVE (Irish) | [58] |
| SIRT1 | Sirtuin protein deacetylase 1 | rs7896005 rs3758391 | A/G Intron 2 −1138 T/C | G POSITIVE (U.S. Caucasian European) T POSITIVE (The Netherlands) | [59] [60] |
| SIRT3 | Sirtuin protein deacetylase 3 | rs11555236 rs939915 VNTR (72bp) | 477 G/T (aa159) −1409 A/T Intron 5 | POSITIVE (Italian) POSITIVE (Italian, German) Allele 2 NEGATIVE (Italian) | [61] [62] [63] |
| UCP1 | Uncoupling protein | rs1800592; rs7687015 | −3826 A/G; −3737 C/A | A POSITIVE; C POSITIVE (Italian) | [64] |
| UCP2 | Uncoupling protein | rs660339 | 544 C/T (aa55) | POSITIVE (Italian) | [65] |
| UCP3 | Uncoupling protein | rs1800849; rs15763 rs11235972 | −55 C/T; 118 C/T A/G Intron 3 | T POSITIVE (Italian) A NEGATIVE(Danish) | [66] [67] |
| SLC25A27 (UCP4) | Uncoupling protein | rs9472817 | C/G Intron 8 | G NEGATIVE(Italian) | [65] |
| TXNRD1 | Thioredoxin reductase 1 | rs10047589 | 2189 C/T | T POSITIVE (Danish) | [68] |
| XDH | Xanthine dehydrogenase | rs207444 | C/T Intron 3 | T POSITIVE (Danish) | [68] |
| MAP3K7 | Mitogen-activated protein kinase kinase kinase 7 | rs282070 | C/G Intron 1 | POSITIVE (Italian) | [69] |
| GSTZ1 | Glutathione S-transferase zeta 1 | rs2111699 | A/G Intron 1 | POSITIVE (Italian) | [69] |
| PON1 | Paraoxonase | rs662 rs705379 rs2374983 | 575 A/G −107 T/C A/G near PON1 | POSITIVE (Italian; Irish; German) CC POSITIVE (Italian) POSITIVE (U.S. Caucasian European) | [70–73] [74] [54] |
| FOXO1A | Forkhead box protein O1 A | rs2755209; rs2755213 rs4943794; rs10507486 | A/C Intron 1; C/T Intron 1 C/G Intron 1; C/T Intron 1 | POSITIVE (Chinese) POSITIVE (U.S. Caucasian European ) | [75] [54] |
| FOXO3A | Forkhead box protein O3 A | rs4946936 rs2802292 rs2802288 rs3800231 rs13220810 rs12206094; rs7762395 rs9486902 rs479744; rs9400239 rs2764264; rs13217795 | 2326 T/C G/T Intron 2 A/G Intron 2 A/G Intron 3 C/T Intron 2 C/T Intron 2; A/G Intron 2 C/T Upstream G/T Downstream; C/T Intron 2 C/T Intron 2; C/T Intron 2 | POSITIVE (Chinese) G POSITIVE (Chinese; Japanese; American; Italian) POSITIVE (Italian) POSITIVE (German; Ashkenazi Jewish POSITIVE (German; Danish) POSITIVE (Danish) POSITIVE (Danish) POSITIVE (German; Danish) POSITIVE (Chinese, Danish) | [75] [75–77] [77] [78,79] [78,80] [80] [80] [78,80] [75,80] |
| APOE | Apolipoprotein E | rs429358 (ɛ4) rs7412 (ɛ2) | 388 T/C (aa130) 526 C/T (aa176) | NEGATIVE (Italian, Danish, Finnish, French, Japanese) POSITIVE (Italian, Danish, Finnish, French, Japanese) | [81] and references therein |
| INS | Insulin | rs3842755 | +286 G/T | POSITIVE (Danish) | [68] |
| INSR | Insulin receptor | rs3745548 | A/G Intron 10 | POSITIVE (Japanese) | [82] |
| IGF1 | Insulin Growth Factor 1 | CA repeat (promoter) | Intron 1 | POSITIVE (The Netherlands) | [83] |
| IGF1R | Insulin Growth Factor 1 receptor | rs2229765 | 3179 G/A | A allele POSITIVE (Italian) | [84] |
| IGF2 | Insulin Growth Factor 2 | rs112276039 | 490 T/C | A allele POSITIVE (Ashkenazi Jewish) | [85] |
| IGF2R | Insulin Growth Factor 2 receptor | rs9456497 | A/G Intron 4 | POSITIVE (Danish) | [68] |
| IRS1 | Insulin Receptor Substrate 1 | rs1801278 | 2963 G/A | POSITIVE (The Netherlands) | [83] |
| GH1 | Growth Hormone 1 | rs2665802 | +1169 A/T (Intron 4) | T POSITIVE (The Netherlands) | [83] |
| GHSR | Growth Hormone Secretagogue Receptor Type 1 | rs572169 | 520 A/G | POSITIVE (Danish) | [68] |
| AKT | V-Akt Murine Thymoma Viral Oncogene Homolog 1 | rs3803304 | +19835 G/C | POSITIVE (U.S. Caucasian European; Ashkenazi Jewish) | [79] |
| TERC | Telomerase RNA component | rs3772190 rs12696304 | (169500487) C/T 6578 G/C | Allele A POSITIVE (Danish) POSITIVE (Ashkenazi Jewish) | [86] [87] |
| TERT | Telomerase reverse transcriptase | rs2853669; rs2736098, rs33954691; rs2853691 MNS16A VNTR | −245 T/C; 915 G/A; 3039 C/T; +893 A/G Downstream exon 16 | POSITIVE (Ashkenazi Jewish) L allele NEGATIVE (Italian) | [87] [88] |
| mtDNA | Mitochondrial DNA | Heteroplasmy Haplogroup J Haplogroup D4 Haplogroup F | High level mtDNA Heteroplasmy POSITIVE (Italian) POSITIVE (Italian, Irish, Finnish) POSITIVE (Japanese) POSITIVE (China) | [88,89] [90–93] [94] [95,96] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dato, S.; Crocco, P.; D'Aquila, P.; De Rango, F.; Bellizzi, D.; Rose, G.; Passarino, G. Exploring the Role of Genetic Variability and Lifestyle in Oxidative Stress Response for Healthy Aging and Longevity. Int. J. Mol. Sci. 2013, 14, 16443-16472. https://doi.org/10.3390/ijms140816443
Dato S, Crocco P, D'Aquila P, De Rango F, Bellizzi D, Rose G, Passarino G. Exploring the Role of Genetic Variability and Lifestyle in Oxidative Stress Response for Healthy Aging and Longevity. International Journal of Molecular Sciences. 2013; 14(8):16443-16472. https://doi.org/10.3390/ijms140816443
Chicago/Turabian StyleDato, Serena, Paolina Crocco, Patrizia D'Aquila, Francesco De Rango, Dina Bellizzi, Giuseppina Rose, and Giuseppe Passarino. 2013. "Exploring the Role of Genetic Variability and Lifestyle in Oxidative Stress Response for Healthy Aging and Longevity" International Journal of Molecular Sciences 14, no. 8: 16443-16472. https://doi.org/10.3390/ijms140816443
APA StyleDato, S., Crocco, P., D'Aquila, P., De Rango, F., Bellizzi, D., Rose, G., & Passarino, G. (2013). Exploring the Role of Genetic Variability and Lifestyle in Oxidative Stress Response for Healthy Aging and Longevity. International Journal of Molecular Sciences, 14(8), 16443-16472. https://doi.org/10.3390/ijms140816443






