Exercise Therapy Augments the Ischemia-Induced Proangiogenic State and Results in Sustained Improvement after Stroke
Abstract
:1. Introduction
2. Results and Discussion
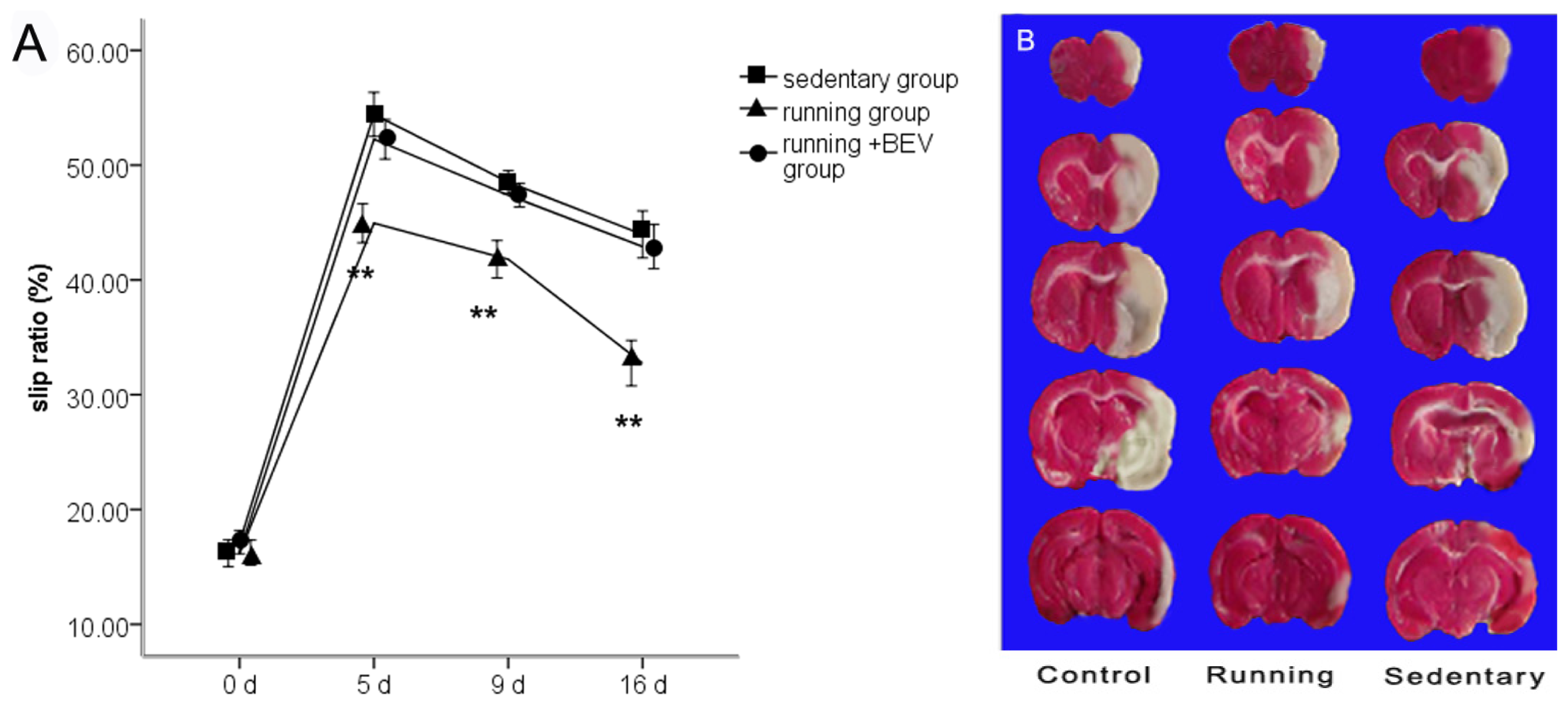
2.1. Neurobehavioral Scores
2.2. Infarct Volume
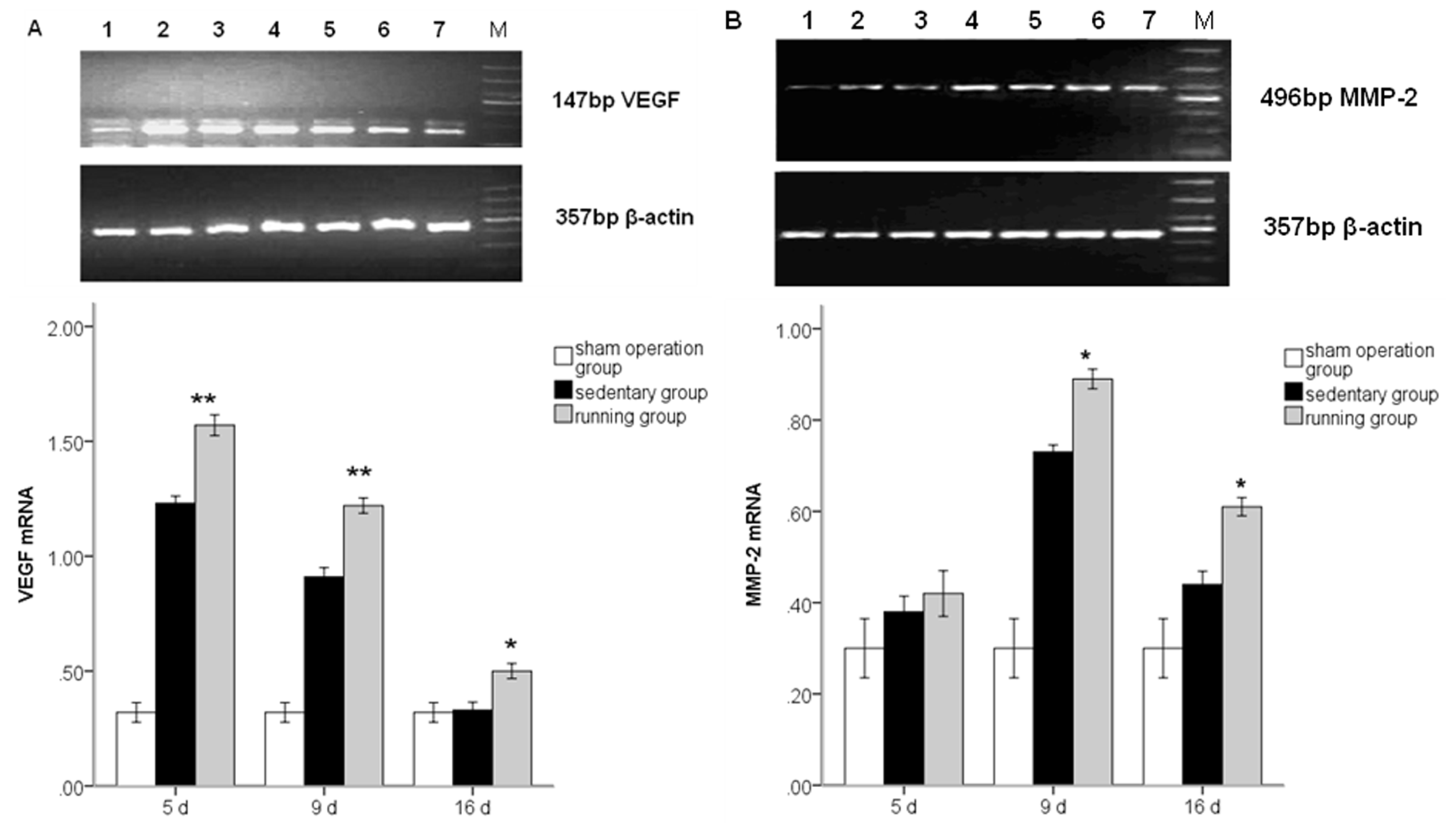
2.3. Running Enhances MMP2 and VEGF mRNA Levels
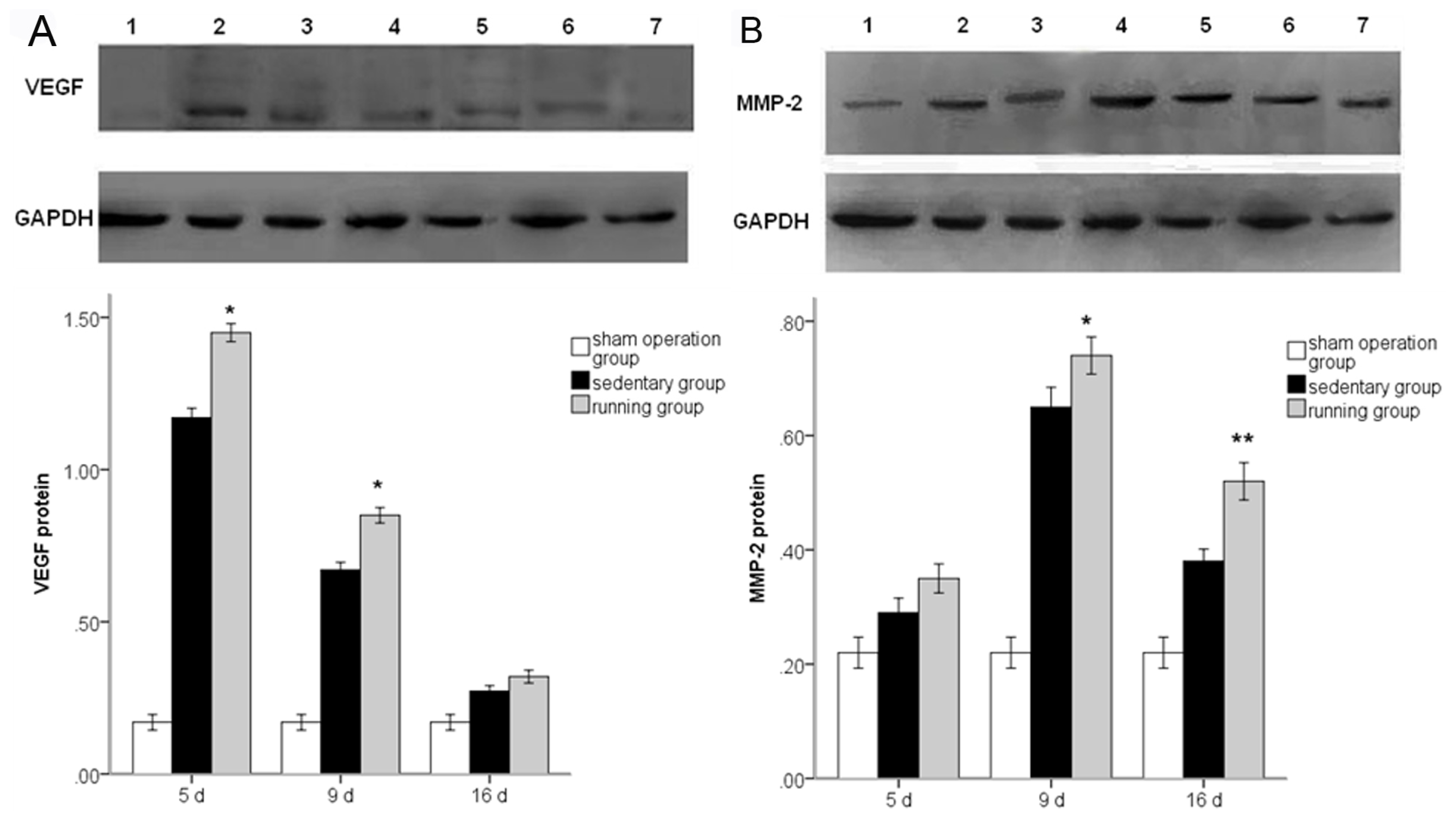
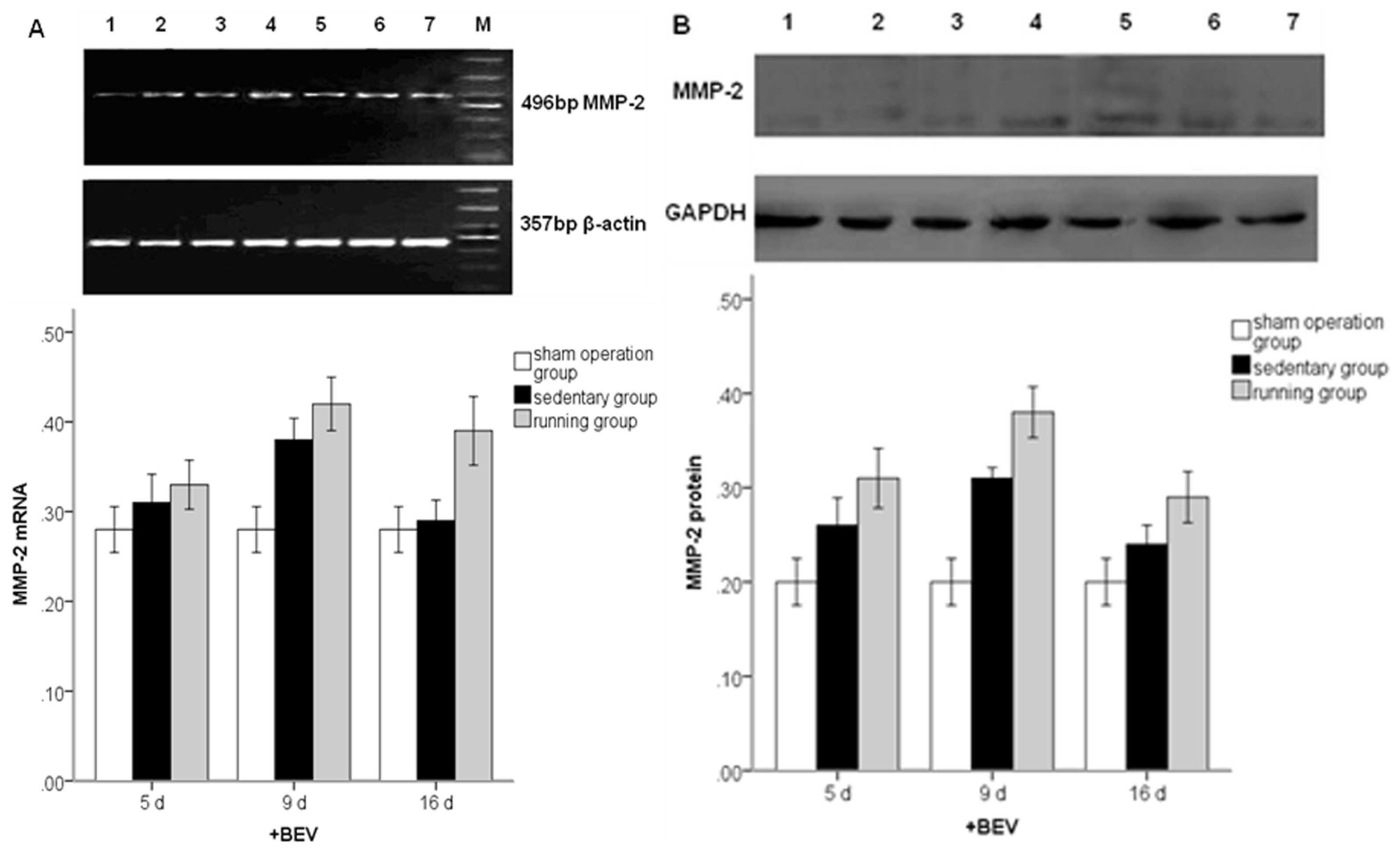
2.4. Effect of Running on MMP2 and VEGF Protein Expressions and the Effect of BEV
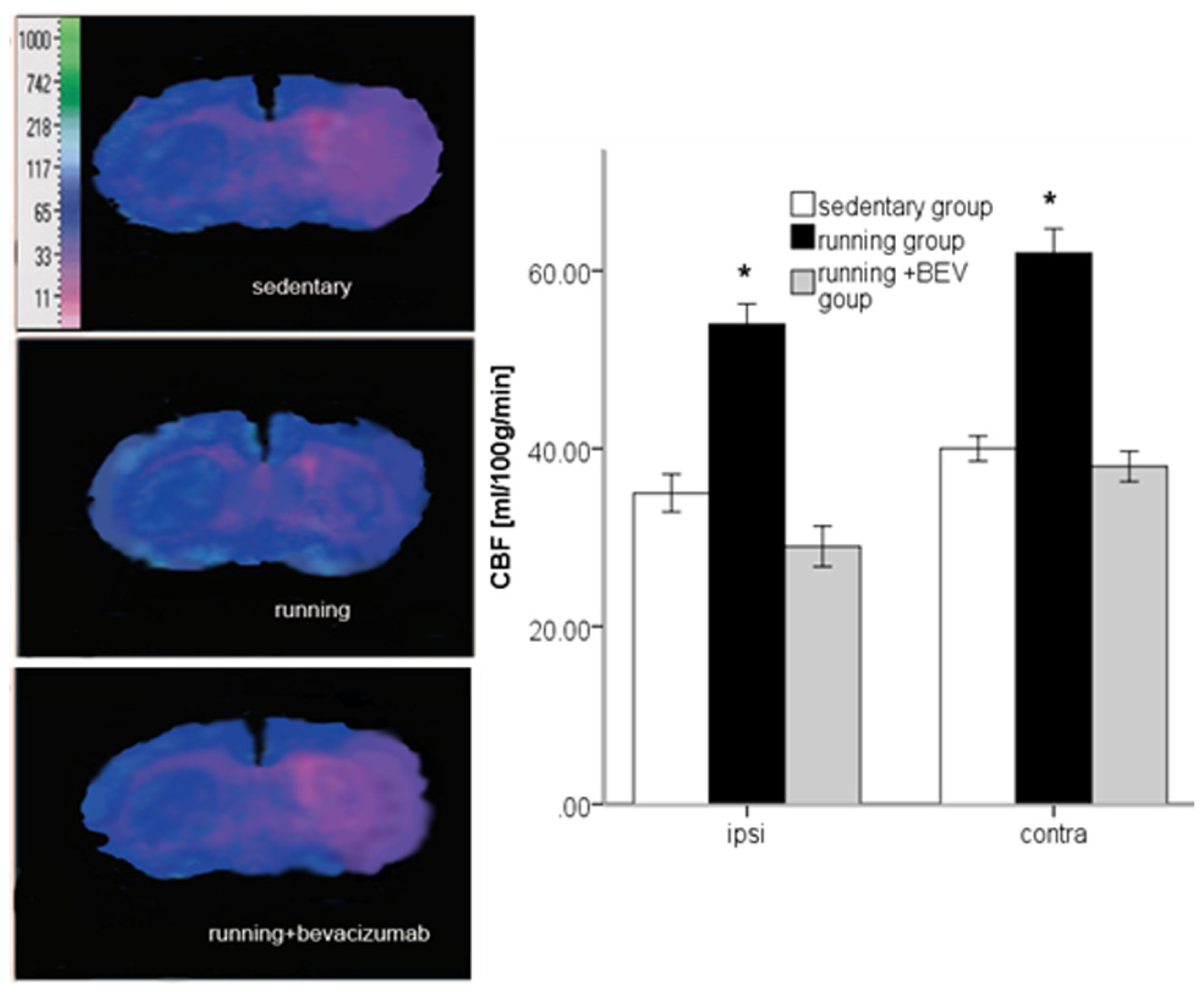
2.5. Running Increases rCBF
2.6. Discussion
3. Experimental Section
3.1. Animals and Treatment
3.2. Cerebral Ischemia and Measurement of Physiological Parameters
3.3. Neurobehavioral Tests
3.4. Measurement of Cerebral Infarction Volume
3.5. Reverse-Transcription Polymerase Chain Reaction
3.6. Western Blot Analysis
3.7. CBF Measurements
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Moseley, A.M.; Stark, A.; Cameron, I.D.; Pollock, A. Treadmill training and body weight support for walking after stroke. Stroke 2003, 34, 3006. [Google Scholar]
- Ploughman, M.; Attwood, Z.; White, N.; Doré, J.J.; Corbett, D. Endurance exercise facilitates relearning of forelimb motor skill after focal ischemia. Eur. J. Neurosci 2007, 25, 3453–3460. [Google Scholar]
- Langhammer, B.; Lindmark, B. Functional exercise and physical fitness post stroke: The importance of exercise maintenance for motor control and physical fitness after stroke. Stroke Res. Treat 2012, 2012, 864835. [Google Scholar]
- Thored, P.; Wood, J.; Arvidsson, A.; Cammenga, J.; Kokaia, Z.; Lindvall, O. Long-term neuroblast migration along blood vessels in an area with transient angiogenesis and increased vascularization after stroke. Stroke 2007, 38, 3032–3039. [Google Scholar]
- Ohab, J.J.; Fleming, S.; Blesch, A.; Carmichael, S.T. A neurovascular niche for neurogenesis after stroke. J. Neurosci 2006, 26, 13007–13016. [Google Scholar]
- Taguchi, A.; Soma, T.; Tanaka, H.; Kanda, T.; Nishimura, H.; Yoshikawa, H.; Tsukamoto, Y.; Iso, H.; Fujimori, Y.; Stern, D.M.; et al. Administration of CD34+ cells after stroke enhances neurogenesis via angiogenesis in a mouse model. J. Clin. Invest 2004, 114, 330–338. [Google Scholar]
- Navaratna, D.; Guo, S.; Arai, K.; Lo, E.H. Mechanisms and targets for angiogenic therapy after stroke. Cell Adh. Migr 2009, 3, 216–223. [Google Scholar]
- Xiong, Y.; Mahmood, A.; Chopp, M. Angiogenesis, neurogenesis and brain recovery of function following injury. Curr. Opin. Investig. Drugs 2010, 11, 298–308. [Google Scholar]
- Ding, Y.H.; Li, J.; Yao, W.X.; Rafols, J.A.; Clark, J.C.; Ding, Y. Exercise preconditioning upregulates cerebral integrins and enhances cerebrovascular integrity in ischemic rats. Acta Neuropathol 2006, 112, 74–84. [Google Scholar]
- Gertz, K.; Priller, J.; Kronenberg, G.; Fink, K.B.; Winter, B.; Schröck, H.; Ji, S.; Milosevic, M.; Harms, C.; Böhm, M.; et al. Physical activity improves long-term stroke outcome via Endothelial nitric oxide synthase—Dependent augmentation of neovascularization and cerebral blood flow. Circ. Res 2006, 99, 1132–1140. [Google Scholar]
- Cobbs, C.S.; Chen, J.; Greenberg, D.A.; Graham, S.H. Vascular endothelial growth factor expression in transient focal cerebral ischemia in the rat. Neurosci. Lett 1998, 249, 79–82. [Google Scholar]
- Kuo, N.T.; Benhayon, D.; Przybylski, R.J.; Martin, R.J.; LaManna, J.C. Prolonged hypoxia increases vascular endothelial growth factor mRNA and protein in adult mouse brain. J. Appl. Physiol 1999, 86, 260–264. [Google Scholar]
- Lee, M.Y.; Ju, W.K.; Cha, J.H.; Son, B.C.; Chun, M.H.; Kang, J.K.; Park, C.K. Expression of vascular endothelial growth factor mRNA following transient forebrain ischemia in rats. Neurosci. Lett 1999, 265, 107–110. [Google Scholar]
- Pichiule, P.; Chavez, J.C.; Xu, K.; LaManna, J.C. Vascular endothelial growth factor upregulation in transient global ischemia by cardiac arrest and resuscitation in rat brain. Mol. Brain Res. 1999, 74, 83–90. [Google Scholar]
- Marti, H.J.; Bernaudin, M.; Bellail, A.; Schoch, H.; Euler, M.; Pettic, E.; Risau, W. Hypoxia-induced vascular endothelial growth factor expression precedes neovascularization after cerebral ischemia. Am. J. Pathol 2000, 156, 965–976. [Google Scholar]
- Lennmyr, F.; Ata, K.A.; Funa, K.; Olsson, Y.; Terent, A. Expression of vascular endothelial growth factor (VEGF) and its receptors (Flt-1 and Flk-1) following permanent and transient occlusion of the middle cerebral artery in the rat. J. Neuropathol. Exp. Neurol 1998, 57, 874–882. [Google Scholar]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nature Med 1995, 1, 27–31. [Google Scholar]
- Louissaint, A.J.; Rao, S.; Leventhal, C.; Goldman, S.A. Coordinated interaction of neurogenesis and angiogenesis in the adult songbird brain. Neuron 2002, 34, 945–960. [Google Scholar]
- Matrisian, L.M. The matrix-degrading metalloproteinases. BioEssays 1992, 14, 455–463. [Google Scholar]
- Rivera, S.; Khrestchatisky, M.; Kaczmarek, L.; Rosenberg, G.A.; Jaworski, D.M. Metzincin proteases and their inhibitors: Foes or friends in nervous system physiology? J. Neurosci 2010, 30, 15337–15357. [Google Scholar]
- Huntley, G.W. Synaptic circuit remodelling by matrix metalloproteinases in health and disease. Nat. Rev. Neurosci 2012, 13, 743–757. [Google Scholar]
- Mignatti, P.; Rifkin, D.B. Plasminogen activators and matrix metalloproteinases in angiogenesis. Enzyme Protein 1996, 49, 117–137. [Google Scholar]
- Milward, E.; Kim, K.J.; Szklarczyk, A.; Nguyen, T.; Melli, G.; Nayak, M.; Deshpande, D.; Fitzsimmons, C.; Hoke, A.; Kerr, D.; et al. Cleavage of myelin associated glycoprotein by matrix metalloproteinases. J. Neuroimmunol 2008, 193, 140–148. [Google Scholar]
- Zuo, J.; Neubauer, D.; Dyess, K.; Ferguson, T.A.; Muir, D. Degradation of chondroitin sulfate proteoglycan enhances the neurite-promoting potential of spinal cord tissue. Exp. Neurol 1998, 154, 654–662. [Google Scholar]
- Adya, R.; Tan, B.K.; Punn, A.; Chen, J.; Randeva, H.S. Visfatin induces human endothelial VEGF and MMP-2/9 production via MAPK and PI3K/Akt signalling pathways: Novel insights into visfatin-induced angiogenesis. Cardiovasc. Res 2008, 78, 356–365. [Google Scholar]
- Carmeli, E.; Moas, M.; Lennon, S.; Powers, S.K. High intensity exercise increases expression of matrix metalloproteinases in fast skeletal muscle fibres. Exp. Physio 2005, 90, 613–619. [Google Scholar]
- Rullman, E.; Norrbom, J.; Strömberg, A.; Wågsäter, D.; Rundqvist, H.; Haas, T.; Gustafsson, T. Endurance exercise activates matrix metalloproteinases in human skeletal muscle. J. Appl. Physol 2009, 106, 804–812. [Google Scholar]
- Samal, B.; Sun, Y.; Stearns, G.; Xie, C.; Suggs, S.; McNiece, I. Cloning and characterization of the cDNA encoding a novel human pre-B-cell colony-enhancing factor. Mol. Cell Biol 1994, 14, 1431–1437. [Google Scholar]
- Goldlust, S.A.; Cavaliere, R.; Newton, H.B.; Hsu, M.; Deangelis, L.M.; Batchelor, T.T.; Gilbert, M.R.; Lassman, A.B. Bevacizumab for glioblastoma refractory to vascular endothelial growth factor receptor inhibitors. J. Neurooncol 2012, 107, 407–411. [Google Scholar]
- Ghazi, N.G. Bevacizumab for neovascular age-related macular degeneration (ABC trial): Multicenter randomized double-masked study. Expert Rev. Clin. Pharmacol 2010, 3, 747–752. [Google Scholar]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–57. [Google Scholar]
- Johansson, B.B. Environmental influence on recovery after brain lesions-experimental and clinical data. J. Rehabil. Med 2003, 41, 11–16. [Google Scholar]
- Lee, S.R.; Kim, H.Y.; Rogowska, J.; Zhao, B.Q.; Bhide, P.; Parent, J.M.; Lo, E.H. Involvement of matrix metalloproteinase in neuroblast cell migration from the subven-tricular zone after stroke. J. Neurosci 2006, 26, 3491–3495. [Google Scholar]
- Wang, L.; Zhang, Z.G.; Zhang, R.L.; Gregg, S.R.; Hozeska-Solgot, A.; LeTourneau, Y.; Wang, Y.; Chopp, M. Matrix metalloprotein-ase 2 (MMP2) and MMP9 secreted by erythropoietin-activated endothelial cells promote neural progenitor cell migration. J. Neurosci 2006, 26, 5996–6003. [Google Scholar]
- Reeves, T.M.; Prins, M.L.; Zhu, J.; Povlishock, J.T.; Phillips, L.L. Matrix metalloproteinase inhibition alter functional and structural correlates of deafferentation-induced sprouting in the dentate gyrus. J. Neurosci 2003, 23, 10182–10189. [Google Scholar]
- Yong, V.W. Metalloproteinases: Mediators of pathology and regeneration in the CNS. Nat. Rev. Neurosci 2005, 6, 931–944. [Google Scholar]
- Zhang, H.; Adwanikar, H.; Werb, Z.; Noble-Haeusslein, L.J. Matrix metalloproteinases and neurotrauma: Evolving roles in injury and reparative processes. Neuroscientist 2010, 16, 156–170. [Google Scholar]
- Rosenberg, G.A.; Cunningham, L.A.; Wallace, J.; Alexander, S.; Estrada, E.Y.; Grossetete, M.; Razhagi, A.; Miller, K.; Gearing, A. Immuno-histochemistry of matrix metalloproteinases in reperfusion injury to rat brain: Activation of MMP-9 linked to stromelysin-1 and microglia in cell cultures. Brain Res 2001, 893, 104–112. [Google Scholar]
- Romanic, A.M.; White, R.F.; Arleth, A.J.; Ohlstein, E.H.; Barone, F.C. Matrix metalloproteinase expression increases after cerebral focal ischemia in rats: Inhibition of matrix metalloproteinase-9 reduces infarct size. Stroke 1998, 29, 1020–1030. [Google Scholar]
- Ihara, M.; Tomimoto, H.; Kinoshita, M.; Oh, J.; Noda, M.; Wakita, H.; Akiguchi, I.; Shibasaki, H. Chronic cerebral hypoperfusion induces MMP-2 but not MMP-9 expression in the microglia and vascular endothelium of white matter. J. Cereb. Blood Flow Metab. 2001, 21, 828–834. [Google Scholar]
- Olson, M.W.; Gervasi, D.C.; Mobashery, S.; Fridman, R. Kinetic analysis of the binding of human matrix metalloproteinase-2 and -9 to tissue inhibitor of metalloproteinase TIMP-1 and TIMP-2. J. Biol. Chem 1997, 272, 29975–29983. [Google Scholar]
- Gibbons, G.H.; Dzau, V.J. The emerging concept of vascular remodeling. N. Engl. J. Med 1994, 330, 1431–1438. [Google Scholar]
- Bendeck, M.P.; Zempo, N.; Clowes, A.W.; Galardy, R.E.; Reidy, M.A. Smooth muscle cell migration and matrix metalloproteinase expression after arterial injury in the rat. Circ. Res 1994, 75, 539–545. [Google Scholar]
- Godin, D.; Ivan, E.; Johnson, C.; Magid, R.; Galis, Z.S. Remodeling of carotid artery is associated with increased expression of matrix metalloproteinases in mouse blood flow cessation model. Circulation 2000, 102, 2861–2866. [Google Scholar]
- Galis, Z.S.; Muszynski, M.; Sukhova, G.K.; Simon-Morrissey, E.; Unemori, E.N.; Lark, M.W.; Amento, E.; Libby, P. Cytokine-stimulated human vascular smooth muscle cells synthesize a complement of enzymes required for extracellular matrix digestion. Circ. Res 1994, 75, 181–189. [Google Scholar]
- Thomas, K.A. Vascular endothelial growth factor, a potent and selective angiogenic agent. J. Biol. Chem 1996, 271, 603–606. [Google Scholar]
- Sun, Y.; Jin, K.; Xie, L.; Childs, J.; Mao, X.O.; Logvinova, A.; Greenberg, D.A. VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. J. Clin. Invest 2003, 111, 1843–1851. [Google Scholar]
- Van Bruggen, N.; Thibodeaux, H.; Palmer, J.T.; Lee, W.P.; Fu, L.; Cairns, B.; Tumas, D.; Gerlai, R.; Williams, S.P.; Campagne, M.V.L.; et al. VEGF antagonism reduces edema formation and tissue damage after ischemia/reperfusion injury in the mouse brain. J. Clin. Invest 1999, 104, 1613–1620. [Google Scholar]
- Jin, K.; Minami, M.; Lan, J.Q.; Mao, X.O.; Batteur, S.; Simon, R.P.; Greenberg, D.A. Neurogenesis in dentate subgranular zone and rostral subventricular zone after focal cerebral ischemia in the rat. Proc. Natl. Acad. Sci. USA 2001, 98, 4710–475. [Google Scholar]
- Jin, K.; Zhu, Y.; Sun, Y.; Mao, X.O.; Xie, L.; Greenberg, D.A. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 11946–11950. [Google Scholar]
- Lamoreaux, W.J.; Fitzgerald, M.E.; Reiner, A.; Hasty, K.A.; Charles, S.T. Vascular endothelial growth factor increases release of gelatinase A and decreases release of tissue inhibitor of metalloproteinases by microvascular endothelial cells in vitro. Microvasc. Res 1998, 55, 29–42. [Google Scholar]
- Jackson, D.W.; McGuire, P.G.; Eriqat, C.; Das, A. Both vascular endothelial growth factor (VEGF) and hypoxia increase gelatinase A (MMP-2) activity in vascular endothelial cells in vitro. Invest. Ophthalmol. Vis. Sci 1997, 38, S500. [Google Scholar]
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar]
- Endres, M.; Gertz, K.; Lindauer, U.; Katchanov, J.; Schultze, J.; Schröck, H.; Nickenig, G.; Kuschinsky, W.; Dirnagl, U.; Laufs, U. Mechanisms of stroke protection by physical activity. Ann. Neurol 2003, 54, 582–590. [Google Scholar]
- Zhao, C.S.; Wang, J.; Zhao, S.S.; Nie, Y.X. Constraint-induced movement therapy enhanced neurogenesis and behavioral recovery after stroke in adult rats. Tohoku J. Exp. Med 2009, 218, 301–308. [Google Scholar]
- Lee, R.; Kermani, P.; Teng, K.K.; Hempstead, B.L. Regulation of cell survival by secreted proneurotrophins. Science 2001, 294, 1945–1948. [Google Scholar]
- Hwang, J.J.; Park, M.H.; Choi, S.Y.; Koh, J.Y. Activation of the Trk signaling pathway by extracellular zinc. Role of metalloproteinases. J. Biol. Chem 2005, 280, 11995–12001. [Google Scholar]
| Group | Number | MCAo/R | BEV Treatment | Time for Running (hours post MCAO) | Duration of Running (days) | Timing for Sacrifice after MCAo (days) |
|---|---|---|---|---|---|---|
| 1 | 5 | - | - | - | - | 0 |
| 2 | 5 | + | - | - | - | 1 |
| 3 | 5 | + | - | 48 | 3 | 5 |
| 4 | 5 | + | - | 48 | 7 | 9 |
| 5 | 15 | + | - | 48 | 14 | 16 |
| 6 | 5 | + | - | - | - | 5 |
| 7 | 5 | + | - | - | - | 9 |
| 8 | 15 | + | - | - | - | 16 |
| 9 | 10 | + | + | 48 | 14 | 16 |
| 10 | 10 | + | + | - | - | 16 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, Y.; Qiang, L.; He, M. Exercise Therapy Augments the Ischemia-Induced Proangiogenic State and Results in Sustained Improvement after Stroke. Int. J. Mol. Sci. 2013, 14, 8570-8584. https://doi.org/10.3390/ijms14048570
Ma Y, Qiang L, He M. Exercise Therapy Augments the Ischemia-Induced Proangiogenic State and Results in Sustained Improvement after Stroke. International Journal of Molecular Sciences. 2013; 14(4):8570-8584. https://doi.org/10.3390/ijms14048570
Chicago/Turabian StyleMa, Yuewen, Lin Qiang, and Man He. 2013. "Exercise Therapy Augments the Ischemia-Induced Proangiogenic State and Results in Sustained Improvement after Stroke" International Journal of Molecular Sciences 14, no. 4: 8570-8584. https://doi.org/10.3390/ijms14048570
APA StyleMa, Y., Qiang, L., & He, M. (2013). Exercise Therapy Augments the Ischemia-Induced Proangiogenic State and Results in Sustained Improvement after Stroke. International Journal of Molecular Sciences, 14(4), 8570-8584. https://doi.org/10.3390/ijms14048570




