Pesticide Removal from Aqueous Solutions by Adding Salting Out Agents
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Potassium Inorganic Salts on the ABS
2.2. Effect of IL Family and Cation Size and on the ABS
2.3. PCP Extraction
3. Experimental Section
3.1. Materials
3.2. Solubility Curves Determination
3.3. PCP Extraction
3.4. PCP Analysis
3.5. pH Determination
4. Conclusions
Supplementary Information
ijms-14-20954-s001.pdf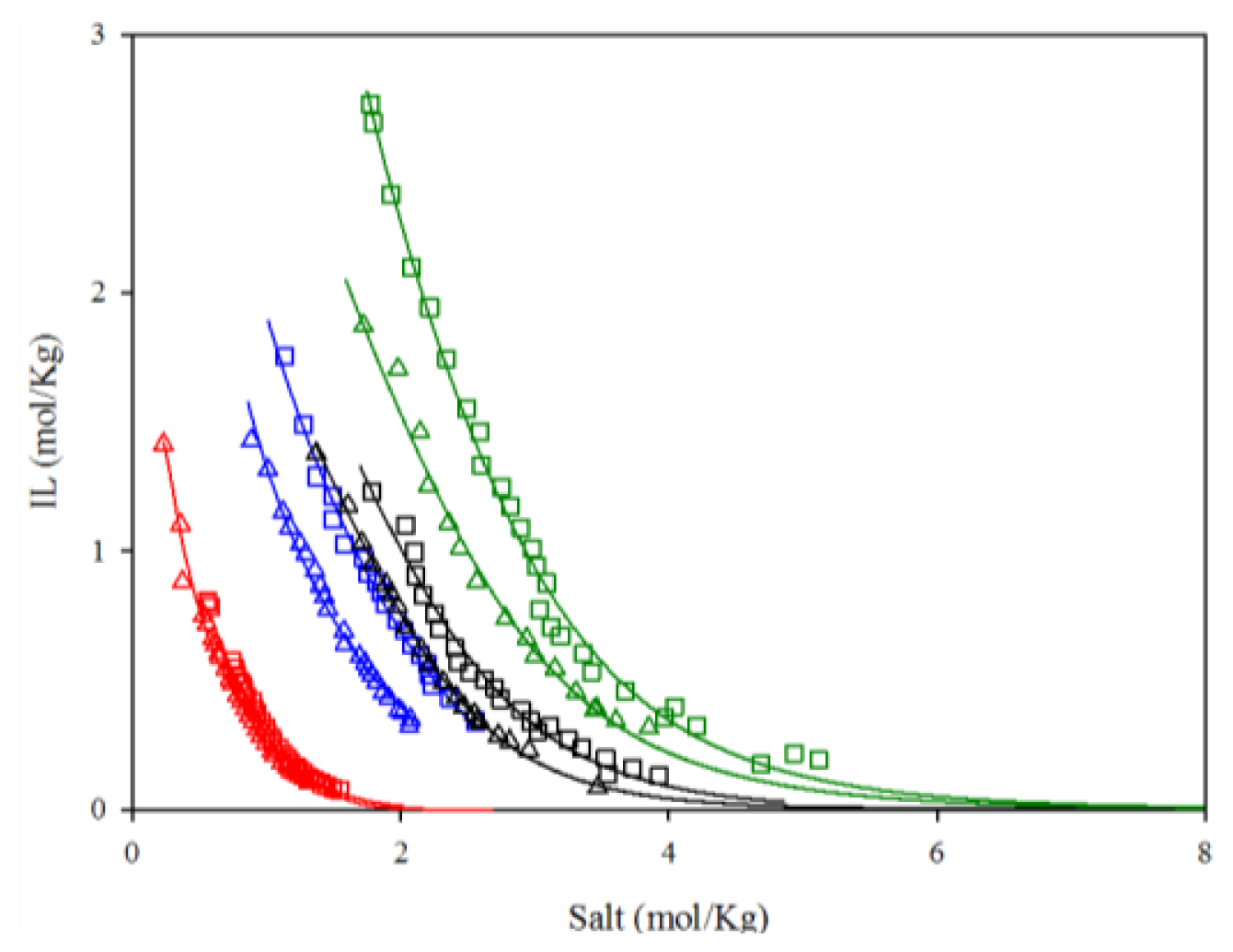
| Equation 1 | a | b | c | n | m | σ |
|---|---|---|---|---|---|---|
| C2C1im C1SO4 + K2CO3 + H2O | 112.84 | −0.3184 | 3.0 × 10−5 | 0.499 | ||
| 111.63 | −0.4100 | 3.8 × 10−4 | 0.4 | 2.4 | 0.484 | |
| C2C1im C1SO4 + K2HPO4 + H2O | 80.00 | −0.2495 | 2.1 × 10−5 | 0.315 | ||
| 80.01 | −0.2648 | 7.4 × 10−5 | 0.5 | 2.7 | 0.361 | |
| C4C1im C1SO4 + K2CO3 + H2O | 80.00 | −0.2969 | 5.0 × 10−5 | 0.464 | ||
| 80.00 | −0.3782 | 1.2 × 10−4 | 0.4 | 2.8 | 0.463 | |
| C4C1im C1SO4 + K2HPO4 + H2O | 60.00 | −0.2156 | 5.3 × 10−5 | 0.152 | ||
| 30.61 | 0.0085 | 1.2 × 10−3 | 1.0 | 2.2 | 0.146 | |
| P4441 C1SO4 + K2CO3 + H2O | 70.01 | −0.3191 | 2.7 × 10−4 | 0.616 | ||
| 71.28 | −0.1445 | 1.0 × 10−3 | 0.9 | 2.4 | 0.243 | |
| P4441 C1SO4 + K2HPO4 + H2O | 80.00 | −0.3584 | 1.4 × 10−4 | 0.335 | ||
| 33.54 | 0.1417 | 2.4 × 10−2 | 0.5 | 1.6 | 0.105 | |
| C1Py C1SO4 + K2CO3 + H2O | 100.01 | −0.2465 | 2.9 × 10−5 | 0.999 | ||
| 100.07 | −0.494 | 1.9 × 10−2 | 0.7 | 1.3 | 0.717 | |
| C1Py C1SO4 + K2HPO4 + H2O | 70.00 | −0.1979 | 2.5 × 10−5 | 0.456 | ||
| 79.99 | −0.3226 | 1.5 × 10−3 | 0.3 | 1.9 | 0.327 | |
| Equation 2 | d | e | f | σ |
|---|---|---|---|---|
| C2C1im C1SO4 + K2CO3 + H2O | −28.38 | 105.10 | −0.1494 | 0.988 |
| C2C1im C1SO4 + K2HPO4 + H2O | −22.17 | 81.12 | −4.9460 | 0.468 |
| C4C1im C1SO4 + K2CO3 + H2O | −13.60 | 44.82 | −10.0118 | 0.510 |
| C4C1im C1SO4 + K2HPO4 + H2O | −21.04 | 74.61 | −3.1855 | 0.400 |
| P4441 C1SO4 + K2CO3 + H2O | −16.08 | 46.51 | −4.4159 | 0.556 |
| P4441 C1SO4 + K2HPO4 + H2O | −17.72 | 54.93 | −3.8697 | 0.309 |
| C1Py C1SO4 + K2CO3 + H2O | −21.82 | 78.61 | −8.7861 | 0.617 |
| C1Py C1SO4 + K2HPO4 + H2O | −31.56 | 122.26 | 2.8666 | 0.393 |
| V*123/(g/mol) | f213 | σ | |
|---|---|---|---|
| C2C1im C1SO4 + K2CO3 + H2O | 4.8 | 0.987 | 0.021 |
| C2C1im C1SO4 + K2HPO4 + H2O | 7.3 | 0.984 | 0.014 |
| C4C1im C1SO4 + K2CO3 + H2O | 8.0 | 0.983 | 0.046 |
| C4C1im C1SO4 + K2HPO4 + H2O | 12.0 | 0.978 | 0.027 |
| P4441 C1SO4 + K2CO3 + H2O | 20.0 | 0.973 | 0.028 |
| P4441 C1SO4 + K2HPO4 + H2O | 30.0 | 0.962 | 0.018 |
| C1Py C1SO4 + K2CO3 + H2O | 12.0 | 0.967 | 0.015 |
| C1Py C1SO4 + K2HPO4 + H2O | 20.0 | 0.954 | 0.011 |
| System | K | E (%) |
|---|---|---|
| C2C1im C1SO4 + K2HPO4 + H2O | 105 | 92 |
| C4C1im C1SO4 + K2HPO4 + H2O | 138 | 94 |
| P4441 C1SO4 + K2HPO4 + H2O | 1140 | 99 |
| C1Py C1SO4 + K2HPO4 + H2O | 183 | 96 |
| Chemical name | Supplier | Mass fraction purity | Method of analysis |
|---|---|---|---|
| C4C1im C1SO4 | Synthesized | 0.99 | NMR and positive FAMBS |
| C2C1im C1SO4 | Merck | 0.99 | None |
| C1Py C1SO4 | Synthesized | 0.99 | NMR and positive FAMBS |
| P4441 C1SO4 | Cytec | 0.99 | None |
| K2CO3 | Sigma-Aldrich | 0.98 | None |
| K2HPO4 | Sigma-Aldrich | 0.98 | None |
Acknowledgments
Conflicts of Interest
References
- Registration Eligibility Decision for Pentachlorophenol. Available online: http://www.epa.gov/oppsrrd1/REDs/pentachlorophenol_red.pdf (accessed on 12 October 2013).
- Basic Information about Pentachlorophenol in Drinking Water. Available online: http://water.epa.gov/drink/contaminants/basicinformation/pentachlorophenol.cfm (accessed on 12 October 2013).
- Pilli, S.R.; Banerjee, T.; Mohanty, K. Extraction of pentachlorophenol and dichlorodiphenyl trichloroethane from aqueous solutions using ionic liquids. J. Ind. Eng. Chem 2012, 18, 1983–1996. [Google Scholar]
- Albertsson, P.A. Partitioning of Cell Particles and Macromolecules; John Wiley and Sons: New York, NY, USA; p. 1986.
- Deive, F.J.; Rodríguez, A.; Pereiro, A.B.; Araújo, J.M.M.; Longo, M.A.; Coelho, M.A.Z.; Canongia Lopes, J.N.; Esperança, J.M.S.S.; Rebelo, L.P.N.; Marrucho, I.M. Ionic liquid-based aqueous biphasic system for lipase extraction. Green Chem 2011, 13, 390–396. [Google Scholar]
- Ulloa, G.; Coutens, C.; Sánchez, M.; Sineiro, J.; Fábregas, J.; Deive, F.J.; Rodríguez, A.; Núñez, M.J. On the double role of surfactants as microalga cell lysis agents and antioxidants extractants. Green Chem 2012, 14, 1044–1051. [Google Scholar]
- Luechau, F.; Ling, T.C.; Lyddiatt, A. Partition of plasmid DNA in polymer-salt aqueous two-phase systems. Sep. Purif. Technol 2009, 66, 397–404. [Google Scholar]
- Bulgariu, L.; Bulgariu, D. Extraction of metal ions in aqueous polyethylene glycol-inorganic salt two-phase systems in the presence of inorganic extractants: Correlation between extraction behaviour and stability constants of extracted species. J. Chromatogr. A 2008, 1196, 117–124. [Google Scholar]
- Mokhtarani, B.; Karimzadeh, R.; Amini, M.H.; Manesh, S.D. Partitioning of Ciprofloxacin in aqueous two-phase system of poly(ethylene glycol) and sodium sulphate. Biochem. Eng. J 2008, 38, 241–247. [Google Scholar]
- Martins, J.P.; dos Reis Coimbra, J.S.; Oliveira, F.C.; Sanaiotti, G.; da Silva, C.A.S.; Mendes da Silva, L.H.; da Silva, M.C.H. Liquid-liquid equilibrium of aqueous two-phase system composed of poly(ethylene glycol) 400 and sulfate salts. J. Chem. Eng. Data 2010, 55, 1247–1251. [Google Scholar]
- Zafarani-Moattar, M.T.; Hamzehzadeh, S. Partitioning of amino acids in the aqueous biphasic system containing the water-miscible ionic liquid 1-butyl-3-methylimidazolium bromide and the water-structuring salt potassium citrate. Biotechnol. Prog 2011, 27, 986–997. [Google Scholar]
- Zafarani-Moattar, M.T.; Hamzehzadeh, S. Salting-out effect, preferential exclusion, and phase separation in aqueous solutions of chaotropic water-miscible ionic liquids and kosmotropic salts: Effects of temperature, anions, and cations. J. Chem. Eng. Data 2010, 55, 1598–1610. [Google Scholar]
- Gutowski, K.E.; Broker, G.A.; Willauer, H.D.; Huddleston, J.G.; Swatloski, R.P.; Holbrey, J.D.; Rogers, R.D. Controlling the aqueous miscibility of ionic liquids: Aqueous biphasic systems of water-miscible ionic liquids and water-structuring salts for recycle, metathesis, and separations. J. Am. Chem. Soc 2003, 125, 6632–6633. [Google Scholar]
- Freire, M.G.; Carvalho, P.J.; Silva, A.M.S.; Santos, L.M.N.B.F.; Rebelo, L.P.N.; Marrucho, I.M.; Coutinho, J.A.P. Ion specific effects on the mutual solubilities of water and hydrophobic ionic liquids. J. Phys. Chem. B 2009, 113, 202–211. [Google Scholar]
- Han, J.A.; Wang, Y.; Yu, C.L.; Li, C.X.; Yan, Y.S.; Liu, Y.; Wang, L.A. Separation, concentration and determination of chloramphenicol in environment and food using an ionic liquid/salt aqueous two-phase flotation system coupled with high-performance liquid chromatography. Anal. Chim. Acta 2011, 685, 138–145. [Google Scholar]
- Freire, M.G.; Neves, C.M.S.S.; Silva, A.M.S.; Santos, L.M.N.B.F.; Marrucho, I.M.; Rebelo, L.P.N.; Shah, J.K.; Maginn, E.J.; Coutinho, J.A.P. 1H NMR and molecular dynamics evidence for an unexpected interaction on the origin of salting-in/salting-out phenomena. J. Phys. Chem. B 2010, 114, 2004–2014. [Google Scholar]
- Bösmann, A.; Datsevich, L.; Jess, A.; Lauter, A.; Schmitz, C.; Wasserscheid, P. Deep desulfurization of diesel fuel by extraction with ionic liquids. Chem. Commun 2001. [Google Scholar] [CrossRef]
- Plechkova, N.V.; Seddon, K.R. Applications of ionic liquids in the chemical industry. Chem. Soc. Rev 2008, 37, 123–150. [Google Scholar]
- Earle, M.J.; Esperança, J.M.S.S.; Gilea, M.A.; Canongia Lopes, J.N.; Rebelo, L.P.N.; Magee, J.W.; Seddon, K.R.; Widegren, J.A. The distillation and volatility of ionic liquids. Nature 2006, 439, 831–834. [Google Scholar]
- Smiglak, M.; Reichert, W.M.; Holbrey, J.D.; Wilkes, J.S.; Sun, L.T.; Thrasher, J.S.; Kirichenko, K.; Singh, S.; Katritzky, A.R.; Rogers, R.D. Combustible ionic liquids by design: Is laboratory safety another ionic liquid myth? Chem. Commun 2006, 24, 2554–2556. [Google Scholar]
- Pereiro, A.B.; Rodriguez, A. Azeotrope-breaking using [BMIM] [MeSO4] ionic liquid in an extraction column. Sep. Purif. Technol 2008, 62, 733–738. [Google Scholar]
- Petkovic, M.; Seddon, K.R.; Rebelo, L.P.N.; Pereira, C.S. Ionic liquids: A pathway to environmental acceptability. Chem. Soc. Rev 2011, 40, 1383–1403. [Google Scholar]
- Deive, F.J.; Rodríguez, A.; Varela, A.; Rodrígues, C.; Leitão, M.C.; Houbraken, J.A.M.P.; Pereiro, A.B.; Longo, M.A.; Sanromán, M.A.; Samson, R.A.; et al. Impact of ionic liquids on extreme microbial biotypes from soil. Green Chem 2011, 13, 687–696. [Google Scholar]
- Bekou, E.; Dionysiou, D.D.; Qian, R.-Y.; Botsaris, G.D. Extraction of chlorophenols from water using room temperature ionic liquids. ACS Symp. Ser 2003, 856, 544–560. [Google Scholar]
- Khodadoust, A.P.; Chandrasekaran, S.; Dionysiou, D.D. Preliminary assessment of imidazolium-based room-temperature ionic liquids for extraction of organic contaminants. Environ. Sci. Technol 2006, 40, 2339–2345. [Google Scholar]
- Fan, J.; Fan, Y.; Pei, Y.; Wu, K.; Wang, J.; Fan, M. Solvent extraction of selected endocrine-disrupting phenols using ionic liquids. Sep. Purif. Technol 2008, 61, 324–331. [Google Scholar]
- Katsuta, S.; Nakamura, K.; Kudo, Y.; Takeda, Y.; Kato, H. Partition behavior of chlorophenols and nitrophenols between hydrophobic ionic liquids and water. J. Chem. Eng. Data 2011, 56, 4083–4089. [Google Scholar]
- Gomez, E.; Calvar, N.; Dominguez, A.; Macedo, E.A. Synthesis and temperature dependence of physical properties of four pyridinium-based ionic liquids: Influence of the size of the cation. J. Chem. Thermodyn 2010, 42, 1324–1329. [Google Scholar]
- Ventura, S.P.M.; Sousa, S.G.; Serafim, L.S.; Lima, A.S.; Freire, M.F.; Coutinho, J.A.P. Ionic-liquid-based aqueous biphasic systems with controlled pH: The ionic liquid anion effect. J. Chem. Eng. Data 2012, 57, 507–512. [Google Scholar]
- Wang, Y.; Han, J.; Liua, J.; Hua, Y.; Shenga, C.; Wua, Y. Liquid–liquid equilibrium phase behavior of iminazolium-based ionic liquid aqueous two-phase systems composed of 1-alkyl-3-methyl imidazolium tetrafluoroborate and different electrolytes ZnSO4, MgSO4 and Li2SO4 at 298.15 K: Experimental and correlation. Thermochim. Acta 2013, 557, 68–76. [Google Scholar]
- Deive, F.J.; Rivas, M.A.; Rodríguez, A. Sodium carbonate as phase promoter in aqueous solutions of imidazolium and pyridinium ionic liquids. J. Chem. Thermodyn 2011, 43, 1153–1158. [Google Scholar]
- Shahriari, S.; Neves, C.M.S.S.; Freire, M.G.; Coutinho, J.A.P. Role of the hofmeister series in the formation of ionic-liquid-based aqueous biphasic systems. J. Phys. Chem. B 2012, 116, 7252–7258. [Google Scholar]
- Freire, M.G.; Cláudio, A.F.M.; Araújo, J.M.M.; Coutinho, J.A.P.; Marrucho, I.M.; Canongia Lopes, J.N.; Rebelo, L.P.N. Aqueous biphasic systems: A boost brought about by using ionic liquids. Chem. Soc. Rev 2012, 41, 4966–4995. [Google Scholar]
- Guan, Y.; Lilley, T.H.; Treffy, T.E. A new excluded volume theory and its application to the coexistence curves of aqueous polymer two-phase systems. Macromolecules 1993, 26, 3971–3979. [Google Scholar]
- The Free Chemical Database. Available online: http://www.chemspider.com (accessed on 12 October 2013).
- Pereiro, A.B.; Santamarta, F.; Tojo, E.; Rodríguez, A.; Canosa, J.; Tojo, J. Temperature dependence of physical properties of ionic liquid 1,3-dimethylimidazolium methyl sulphate. J. Chem. Eng. Data 2006, 51, 952–954. [Google Scholar]
- Pereiro, A.B.; Verdía, P.; Tojo, E.; Rodríguez, A. Physical properties of 1-butyl-3-methylimidazolium methyl sulfate as a function of temperature. J. Chem. Eng. Data 2007, 52, 377–380. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moscoso, F.; Deive, F.J.; Esperança, J.M.S.S.; Rodríguez, A. Pesticide Removal from Aqueous Solutions by Adding Salting Out Agents. Int. J. Mol. Sci. 2013, 14, 20954-20965. https://doi.org/10.3390/ijms141020954
Moscoso F, Deive FJ, Esperança JMSS, Rodríguez A. Pesticide Removal from Aqueous Solutions by Adding Salting Out Agents. International Journal of Molecular Sciences. 2013; 14(10):20954-20965. https://doi.org/10.3390/ijms141020954
Chicago/Turabian StyleMoscoso, Fátima, Francisco J. Deive, José M. S. S. Esperança, and Ana Rodríguez. 2013. "Pesticide Removal from Aqueous Solutions by Adding Salting Out Agents" International Journal of Molecular Sciences 14, no. 10: 20954-20965. https://doi.org/10.3390/ijms141020954
APA StyleMoscoso, F., Deive, F. J., Esperança, J. M. S. S., & Rodríguez, A. (2013). Pesticide Removal from Aqueous Solutions by Adding Salting Out Agents. International Journal of Molecular Sciences, 14(10), 20954-20965. https://doi.org/10.3390/ijms141020954





