Synthesis and Activity of Novel Acylthiourea with Hydantoin
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Synthesis
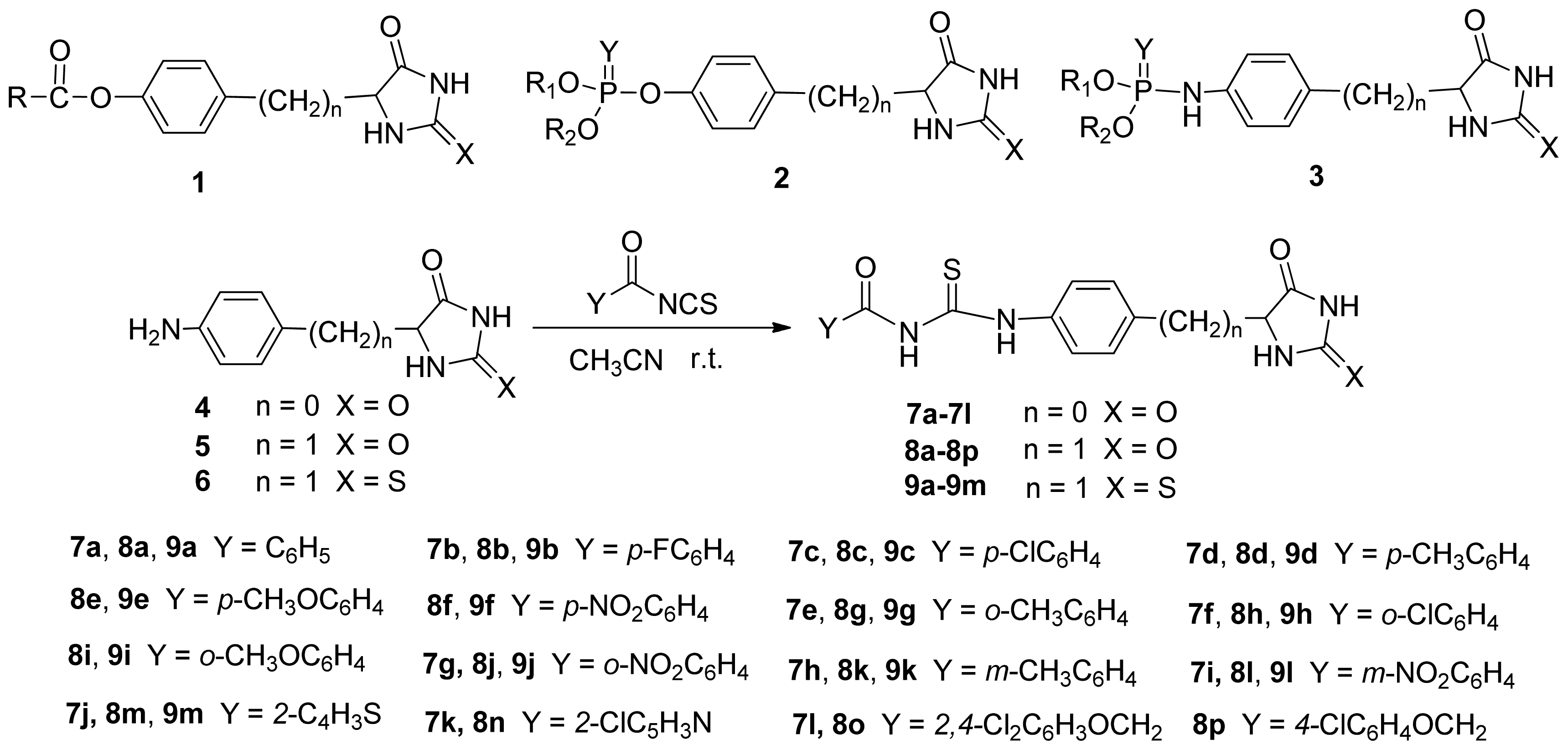
3.2.1. Synthesis of 5-(4-Aminobenzyl)-, 5-(4-Aminophenyl)-Hydantoin (4 and 5), 5-(4-Aminobenzyl)-Thiohydantoin (6) and Acyl Isothiocyanate Derivatives
3.2.2. General Procedure for the Synthesis of Compounds 7, 8 and 9
3.3. Bioassay of Herbicidal and Fungicidal Activity
4. Conclusions
| Compd. | B. campestris | E. crus-galli | Compd. | B. campestris | E. crus-galli | Compd. | B. campestris | E. crus-galli |
|---|---|---|---|---|---|---|---|---|
| 7a | 20 | 10 | 8a | 2 | 15 | 9a | 3 | 10 |
| 7b | 38 | 0 | 8b | 12 | 15 | 9b | 44 | 10 |
| 7c | 0 | 15 | 8c | 16 | 20 | 9c | 40 | 5 |
| 7d | 39 | 0 | 8d | 14 | 5 | 9d | 33 | 15 |
| 7e | 29 | 10 | 8e | 0 | 5 | 9e | 0 | 15 |
| 7f | 0 | 5 | 8f | 25 | 0 | 9f | 0 | 10 |
| 7g | 49 | 10 | 8g | 4 | 10 | 9g | 6 | 25 |
| 7h | 23 | 15 | 8h | 10 | 10 | 9h | 29 | 25 |
| 7i | 0 | 0 | 8i | 11 | 15 | 9i | 18 | 25 |
| 7j | 14 | 5 | 8j | 25 | 0 | 9j | 6 | 10 |
| 7k | 6 | 20 | 8k | 0 | 10 | 9k | 0 | 5 |
| 7l | 91 | 0 | 8l | 0 | 0 | 9l | 0 | 10 |
| 8m | 0 | 0 | 9m | 33 | 5 | |||
| 8n | 7 | 10 | ||||||
| 8o | 94 | 10 | ||||||
| 8p | 87 | 10 |
| Compd. | B. campestris | E. crus-galli | ||
|---|---|---|---|---|
| Pre-emergence | Post-emergence | Pre-emergence | Post-emergence | |
| 7l | 23 | 100 | 0 | 0 |
| 8o | 46 | 100 | 0 | 10 |
| 8p | 31 | 95 | 10 | 15 |
| Compd. | F. oxysporum | A. Solani | B. berengeriana | C. arachidcola | F. graminearum |
|---|---|---|---|---|---|
| 7a | 74 | 20 | 32 | 0 | 25 |
| 7b | 79 | 24 | 23 | 0 | 54 |
| 7c | 79 | 24 | 23 | 0 | 40 |
| 7d | 71 | 21 | 7 | 6 | 44 |
| 7e | 59 | 17 | 30 | 6 | 25 |
| 7f | 24 | 27 | 28 | 6 | 10 |
| 7g | 24 | 27 | 37 | 6 | 18 |
| 7h | 71 | 3 | 20 | 0 | 40 |
| 7i | 24 | 13 | 25 | 6 | 16 |
| 7j | 24 | 27 | 37 | 6 | 18 |
| 7k | 41 | 20 | 23 | 0 | 40 |
| 7l | 15 | 17 | 40 | 0 | 12 |
| Carbendazin | 100 | 44 | 97 | 8 | 100 |
Acknowledgments
Conflicts of Interest
References
- Li, K.; Shi, D.Q. Synthesis and herbicidal activity of 3-aryl-1-[2-(aryloxy)propanoyl]imidazolidine- 2,4-diones. J. Heterocycl. Chem 2009, 46, 544–547. [Google Scholar]
- Thenmozhiyal, J.C.; Wong, P.T.-H.; Chui, W.K. Anticonvulsant activity of phenylmethylene-hydantoins: A structure-activity relationship study. J. Med. Chem 2004, 47, 1527–1535. [Google Scholar]
- Elokdah, H.; Abou-Gharbia, M.; Hennan, J.K.; Mcfarlane, G.; Mugford, C.P.; Krishnamurthy, G.; Crandall, D.L. Tiplaxtinin, a novel orally efficacious inhibitor of plasminogen activator inhibitor-1: Design, synthesis, and preclinical characterization. J. Med. Chem 2004, 47, 3491–3494. [Google Scholar]
- Brady, S.F.; Bauer, J.D.; Clarke-Pearson, M.F.; Daniels, R. Natural products from isnA-containing biosynthetic gene clusters recovered from the genomes of cultured and uncultured bacteria. J. Am. Chem. Soc 2007, 129, 12102–12103. [Google Scholar]
- Nakajima, M.; Itoi, K.; Takamatsu, Y.; Kinoshita, T.; Okazaki, T.; Kawakubo, K.; Shindo, M.; Honma, T.; Tohjigamori, M.; Haneishi, T. Hydantocidin: A new compound with herbicidal activity from Streptomyes hygroscopicus. J. Antibiot 1991, 44, 293–300. [Google Scholar]
- Zhao, B.G.; Du, H.F.; Shi, Y.A. A Cu(I)-catalyzed C-H α-Amination of esters. Direct synthesis of hydantoins. J. Am. Chem. Soc 2008, 130, 7220–7221. [Google Scholar]
- Lei, J.P.; Han, J.T.; Xu, Z.H.; Dong, H.B.; Wang, M.A. Synthesis and fungicidal activity of 5-cyclohexylidene-2-aminoimidazolin-4-one derivatives. Chin. J. Org. Chem 2012, 32, 1993–1998. [Google Scholar]
- Kiec-Kononowicz, K.; Szymanska, E. Antimycobacterial activity of 5-arylidene derivatives of hydantoin. Farmaco 2002, 57, 909–916. [Google Scholar]
- Kiec-Kononowicz, K.; Karolak-Wojicechowska, J.; Muller, C.E.; Schumacher, B.; Pekela, E.; Szymanska, E. Imidazo-thiazine, -diazinone and -diazepinone derivatives. Synthesis, structure and benzodiazepine receptor binding. Eur. J. Med. Chem 2001, 36, 407–419. [Google Scholar]
- Han, J.T.; Dong, H.B.; Xu, Z.H.; Lei, J.P.; Wang, M.A. Facile synthesis of 5-arylidene thiohydantoin by sequential sulfonylation/desulfination reaction. Int. J. Mol. Sci 2013, 14, 12484–12495. [Google Scholar]
- Han, J.T.; Wang, J.M.; Dong, H.B.; Xu, Z.H.; Liu, B.; Wang, M.A. Synthesis and biological activity of novel phosphoramidate with hydantoin. Chin. J. Org. Chem 2013, 33, 596–601. [Google Scholar]
- Xu, Z.H.; Wang, J.M.; Han, J.T.; Liu, B.; Wang, M.A. Synthesis and biological activity of novel (thio)phosphates with hydantoin. Chin. J. Org. Chem 2012, 32, 2134–2140. [Google Scholar]
- Han, J.T.; Dong, H.B.; Wang, J.M.; Lei, J.P.; Wang, M.A.; Fang, J.X. Synthesis and herbicidal activity of 5-(4-hydroxybenzyl)-2-thioxoimidazolidin-4-one esters. Molecules 2011, 16, 2833–2845. [Google Scholar]
- Zhang, J.F.; Liu, C.; Ma, Y.; Wang, B.L.; Xiong, L.X.; Yu, S.J.; Li, Z.M. Synthesis of novel 3-chloropyridin-2-yl-pyrazole derivatives and their insecticidal, fungicidal activities and QSAR Study. Lett. Drug Des. Discov 2013, 10, 497–506. [Google Scholar]
- Zhang, J.F.; Xu, J.Y.; Wang, B.L.; Li, Y.X.; Xiong, L.X.; Li, Y.Q.; Ma, Y.; Li, Z.M. Synthesis and insecticidal activities of novel anthranilic diamides containing acylthiourea and acylurea. J. Agric. Food Chem 2012, 60, 7565–7572. [Google Scholar]
- Limban, C.; Marutescu, L.; Chifiriuc, M.C. Synthesis, spectroscopic properties and antipatho-genic activity of new thiourea derivatives. Molecules 2011, 16, 7593–7607. [Google Scholar]
- Limban, C.; Missir, A.V.; Chirita, I.C.; Nitulescu, G.M.; Caproiu, M.T.; Chifiriuc, M.C.; Israil, A.M. Synthesis and antimicrobial properties of new 2-((4-ethylphenoxy)methyl)benzoylthioureas. Chem. Pap 2011, 65, 60–69. [Google Scholar]
- Rao, X.P.; Wu, Y.; Song, Z.Q.; Shang, S.B.; Wang, Z.D. Synthesis and antitumor activities of unsymmetrically disubstitutedacylthioureas fused with hydrophenanthrene structure. Med. Chem. Res 2011, 20, 333–338. [Google Scholar]
- Sun, R.F.; Zhang, Y.L.; Chen, L.; Li, Y.Q.; Li, Q.S.; Song, H.B.; Huang, R.Q.; Bi, F.C.; Wang, Q.M. Design, synthesis, bioactivity, and structure−activity relationship (SAR) studies of novel benzoylphenylureas containing oxime ether group. J. Agric. Food Chem 2008, 56, 11376–11391. [Google Scholar]
- Hallur, G.; Jimeno, A.; Dalrymple, S.; Zhu, T.; Jung, M.K.; Hidalgo, M.; Isaacs, J.T.; Sukumar, S.; Hamel, E.; Khan, S.R. Benzoylphenylurea sulfur analogues with potent antitumor activity. J. Med. Chem 2006, 49, 2357–2360. [Google Scholar]
- Sun, C.W.; Huang, H.; Feng, M.Q.; Shi, X.L.; Zhang, X.D.; Zhou, P. A novel class of potent influenza virus inhibitors: Polysubstitutedacylthiourea and its fused heterocycle derivatives. Bioorg. Med. Chem. Lett 2006, 16, 162–166. [Google Scholar]
- Junejo, J.; Ghosh, S.K.; Shaikh, M.; Gahtori, P.; Singh, U.P. Facile synthesis, antibacterial activity and molecular properties prediction of some new 1,3-dihydroimidazol-2-thione derivatives. Lett. Drug Des. Discov 2011, 8, 763–768. [Google Scholar]
- Aamer, S.; Mahira, B. Synthesis and bioactivity of some new 1-tolyl-3-aryl-4-Methyl imidazole-2-thiones. Med. Chem. Res 2007, 16, 143–154. [Google Scholar]
- Wang, X.C.; Wang, F.; Quan, Z.J.; Wang, M.G.; Li, Z. An efficient and clean synthesis of 1-aroyl-3-aryl-4-substituted imidazole-2-thiones in water. J. Chem. Res 2005, 11, 689–690. [Google Scholar]
- Zeng, R.S.; Zou, J.P.; Zhi, S.J.; Chen, J.; Shen, Q. Novel synthesis of 1-aroyl-3-aryl-4-substituted imidazole-2-thiones. Org. Lett 2003, 5, 1657–659. [Google Scholar]
- Majid, M.H.; Setareh, M. An efficient synthesis of thiazol-2-imine derivatives via a one-pot, three-component reaction. Tetrahedron Lett 2012, 53, 392–394. [Google Scholar]
- Wang, L.; Pan, X.Q.; Rao, W.D.; Zou, J.P. Synthesis of 1,3-disubstituted-2-thioxo-4- imidazolidinones. Chin. J. Org. Chem 2011, 31, 1939–1942. [Google Scholar]
- Aamer, S.; Uzma, S.; Abdul, H.; Faiza, K. Synthesis and antimicrobial activity of some novel 2-(substituted fluorobenzoylimino)-3-(substituted fluorophenyl)-4-methyl-1,3-thiazolines. J. Fluorine Chem 2010, 131, 333–339. [Google Scholar]
- Murru, S.; Singh, C.B.; Kavala, V.; Patel, B.K. A convenient one-pot synthesis of thiazol-2- imines: Application in the construction of pifithrin analogues. Tetrahedron 2008, 64, 1931–1942. [Google Scholar]
- Aamer, S.; Sabah, Z.; Michael, B. Synthesis and crystal structure of some novel 2-aroylimino-3- aryl-4-phenyl-1,3-thiazolines. Synth. Commun 2008, 38, 2185–2199. [Google Scholar]
- Singh, C.B.; Siva, M.; Veerababurao, K.; Bhisma, K.P. 3-Aryl-1-benzoylthioureas with α-bromoketones in water form 2-N-benzoyl-3-arylthiazol-2(3H)-imines, not 3-aryl-1-benzoylimidazoline- 2-thiones. J. Chem. Res 2007, 3, 136–137. [Google Scholar]
- Aamer, S.; Madood, P. Synthesis, crystal structure of Some new 2-(4-methylbenzoyl-imino)-3- aryl-4-methyl-1,3-thiazolines. J. Heterocycl. Chem 2006, 43, 1027–1030. [Google Scholar]
- Singh, C.B.; Siva, M.; Veerababurao, K.; Bhisma, K.P. It is “thiazolidene-2-imine” and not imi-dazole-2-thione as the reaction product of 1-benzoyl-3-phenylthiourea with Br2/enolizable ketone. Org. Lett 2006, 23, 5397–5399. [Google Scholar]
- Walters, E.W.; Lee, S.F.; Niderman, T.; Bemasconi, P.; Subramanian, M.V.; Siehl, D.L. Adenylosuccinate synthetase from maize. Purification, properties, and mechanism of inhibition by 5’-phosphohydantocidin. Plant Physiol 1997, 114, 549–555. [Google Scholar]
- Cseke, C.; Gerwick, B.C.; Crouse, G.D.; Murdoch, M.G.; Green, S.B.; Heim, D.R. 2-Phosphohydantocidin: the in vivo adenylosuccinate synthetase inhibitor responsible for hydantocidin phytotoxicity. Pestic. Biochem. Physiol 1996, 55, 210–217. [Google Scholar]
- Fonne-Pfister, R.; Chemla, P.; Ward, E.; Girardet, M.; Kreuz, K.E.; Hinzatko, R.B.; Fromm, H.; Schar, H.P.; Grutter, M.G.; Cowan-Jacob, S.W. The mode of action and the structure of a herbicide in complex with its target: Binding of activated hydantocidin to the feedback regulation site of adenylosuccinate synthetase. Proc. Natl. Acad. Sci. USA 1996, 93, 9431–9436. [Google Scholar]
- Wang, J.M.; Xu, Z.H.; Han, J.T.; Dong, H.B.; Liu, B.; Wang, M.A. Synthesis and biological activity of novel phosphoramide with hydantoin. Chin. J. Org. Chem 2013. [Google Scholar] [CrossRef]
- Chen, N.C. The Bioassay Technologies for Pesticides; Beijing Agricultural University Press: Beijing, China, 1991; pp. 161–162. [Google Scholar]
- Chen, W.Y. The Research & Development of New Pesticide—Methods & Progress; Chemical Industry Press: Beijing, China, 2007; pp. 132–134. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Han, J.; Dong, H.; Xu, Z.; Wang, J.; Wang, M. Synthesis and Activity of Novel Acylthiourea with Hydantoin. Int. J. Mol. Sci. 2013, 14, 19526-19539. https://doi.org/10.3390/ijms141019526
Han J, Dong H, Xu Z, Wang J, Wang M. Synthesis and Activity of Novel Acylthiourea with Hydantoin. International Journal of Molecular Sciences. 2013; 14(10):19526-19539. https://doi.org/10.3390/ijms141019526
Chicago/Turabian StyleHan, Jintao, Hongbo Dong, Zhihong Xu, Jinmin Wang, and Mingan Wang. 2013. "Synthesis and Activity of Novel Acylthiourea with Hydantoin" International Journal of Molecular Sciences 14, no. 10: 19526-19539. https://doi.org/10.3390/ijms141019526
APA StyleHan, J., Dong, H., Xu, Z., Wang, J., & Wang, M. (2013). Synthesis and Activity of Novel Acylthiourea with Hydantoin. International Journal of Molecular Sciences, 14(10), 19526-19539. https://doi.org/10.3390/ijms141019526





