The Role of Free Radicals in the Aging Brain and Parkinson’s Disease: Convergence and Parallelism
Abstract
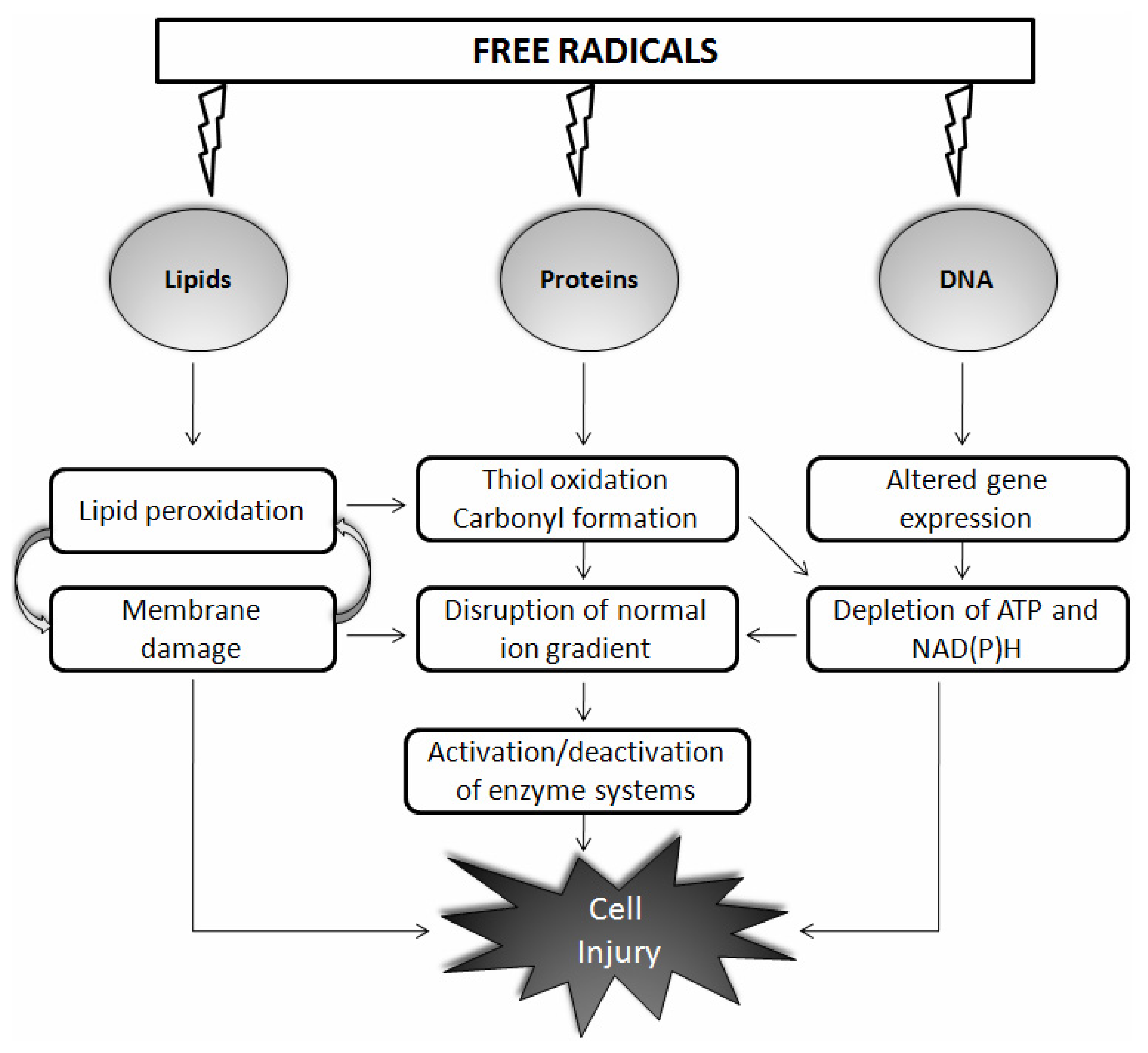
:1. Introduction
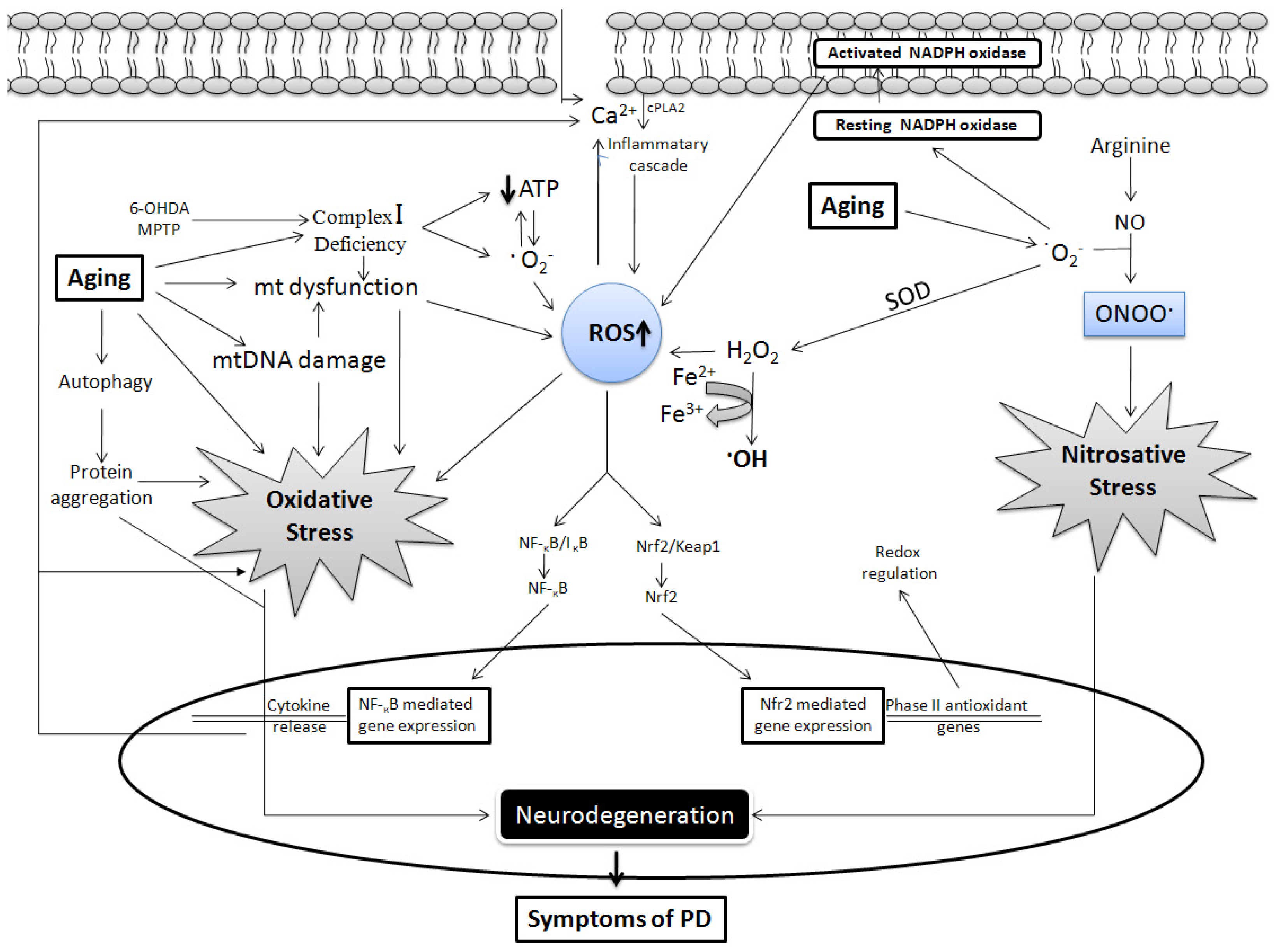
2. Common Link between PD and Aging: Role of Free Radicals
2.1. Mitochondrial Dysfunction
2.2. Role of Dopamine
2.3. α-Synuclein
2.4. Glial Cells
2.4.1. Microglia
2.4.2. Astrocytes
2.5. Regulation of Calcium
2.6. Iron
2.7. Defective Mechanism
3. Conclusions
Acknowledgments
References
- Brand, M.D.; Affourtit, C.; Esteves, T.C.; Green, K.; Lambert, A.J.; Miwa, S.; Pakay, J.L.; Parker, N. Mitochondrial superoxide: Production, biological effects, and activation of uncoupling proteins. Free Radic. Biol. Med 2004, 37, 755–767. [Google Scholar]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol 2003, 552, 335–344. [Google Scholar]
- Thannickal, V.J.; Fanburg, B.L. Reactive oxygen species in cell signaling. Am. J. Physiol. Lung. Cell. Mol. Physiol 2000, 279, L1005–L1028. [Google Scholar]
- Forman, H.J.; Torres, M. Reactive oxygen species and cell signaling: Respiratory burst in macrophage signaling. Am. J. Respir. Crit. Care Med 2002, 166, S4–S8. [Google Scholar]
- Rhee, S.G. Redox signaling: Hydrogen peroxide as intracellular messenger. Exp. Mol. Med 1999, 31, 53–59. [Google Scholar]
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev 2007, 87, 245–313. [Google Scholar]
- Kaul, N.; Forman, H.J. Reactive Oxygen Species in Physiology and Toxicology: From Lipid Peroxidation to Transcriptional Activation; Rhodes, C.R., Ed.; Taylor and Francis: New York, NY, USA, 2000; pp. 310–335. [Google Scholar]
- Nordberg, J.; Arner, E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med 2001, 31, 1287–1312. [Google Scholar]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact 2006, 160, 1–40. [Google Scholar]
- Finkel, T. Oxidant signals and oxidative stress. Curr. Opin. Cell Biol 2003, 15, 247–254. [Google Scholar]
- Lindahl, T.; Nyberg, B. Rate of depurination of native deoxyribonucleic acid. Biochemistry 1972, 11, 3610–3618. [Google Scholar]
- Lindahl, T.; Karlstrom, O. Heat-induced depyrimidination of deoxyribonucleic acid in neutral solution. Biochemistry 1973, 12, 5151–5154. [Google Scholar]
- Devasagayam, T.P.; Steenken, S.; Obendorf, M.S.; Schulz, W.A.; Sies, H. Formation of 8-hydroxy(deoxy)guanosine and generation of strand breaks at guanine residues in DNA by singlet oxygen. Biochemistry 1991, 30, 6283–6289. [Google Scholar]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol 1956, 11, 298–300. [Google Scholar]
- Harman, D. The biologic clock: The mitochondria? J. Am. Geriatr. Soc 1972, 20, 145–147. [Google Scholar]
- Dei, R.; Takeda, A.; Niwa, H.; Li, M.; Nakagomi, Y.; Watanabe, M.; Inagaki, T.; Washimi, Y.; Yasuda, Y.; Horie, K.; et al. Lipid peroxidation and advanced glycation end products in the brain in normal aging and in Alzheimer’s disease. Acta Neuropathol 2002, 104, 113–122. [Google Scholar]
- Lovell, M.A.; Ehmann, W.D.; Mattson, M.P.; Markesbery, W.R. Elevated 4-hydroxynonenal in ventricular fluid in Alzheimer’s disease. Neurobiol. Aging 1997, 18, 457–461. [Google Scholar]
- Starke-Reed, P.E.; Oliver, C.N. Protein oxidation and proteolysis during aging and oxidative stress. Arch. Biochem. Biophys 1989, 275, 559–567. [Google Scholar]
- Parkes, T.L.; Elia, A.J.; Dickinson, D.; Hilliker, A.J.; Phillips, J.P.; Boulianne, G.L. Extension of Drosophila lifespan by overexpression of human SOD1 in motorneurons. Nat. Genet 1998, 19, 171–174. [Google Scholar]
- Magwere, T.; West, M.; Riyahi, K.; Murphy, M.P.; Smith, R.A.; Partridge, L. The effects of exogenous antioxidants on lifespan and oxidative stress resistance in Drosophila melanogaster. Mech. Ageing Dev 2006, 127, 356–370. [Google Scholar]
- Sohal, R.S. Role of oxidative stress and protein oxidation in the aging process. Free Radic. Biol. Med 2002, 33, 37–44. [Google Scholar]
- Muller, F.L.; Lustgarten, M.S.; Jang, Y.; Richardson, A.; van Remmen, H. Trends in oxidative aging theories. Free Radic. Biol. Med 2007, 43, 477–503. [Google Scholar]
- Przedborski, S.; Ischiropoulos, H. Reactive oxygen and nitrogen species: Weapons of neuronal destruction in models of Parkinson’s disease. Antioxid. Redox Signal 2005, 7, 685–693. [Google Scholar]
- Schapira, A.H. Pathogenesis of Parkinson’s disease. Baillieres Clin. Neurol 1997, 6, 15–36. [Google Scholar]
- Alam, Z.I.; Daniel, S.E.; Lees, A.J.; Marsden, D.C.; Jenner, P.; Halliwell, B. A generalised increase in protein carbonyls in the brain in Parkinson’s but not incidental Lewy body disease. J. Neurochem 1997, 69, 1326–1329. [Google Scholar]
- Dexter, D.; Carter, C.; Agid, F.; Agid, Y.; Lees, A.J.; Jenner, P.; Marsden, C.D. Lipid peroxidation as cause of nigral cell death in Parkinson’s disease. Lancet 1986, 2, 639–640. [Google Scholar]
- Hamilton, M.L.; van Remmen, H.; Drake, J.A.; Yang, H.; Guo, Z.M.; Kewitt, K.; Walter, C.A.; Richardson, A. Does oxidative damage to DNA increase with age? Proc. Natl. Acad. Sci. USA 2001, 98, 10469–10474. [Google Scholar]
- Oliver, C.N.; Ahn, B.W.; Moerman, E.J.; Goldstein, S.; Stadtman, E.R. Age-related changes in oxidized proteins. J. Biol. Chem 1987, 262, 5488–5491. [Google Scholar]
- Calne, D.B.; Langston, J.W. Aetiology of Parkinson’s disease. Lancet 1983, 2, 1457–1459. [Google Scholar]
- Morens, D.M.; Davis, J.W.; Grandinetti, A.; Ross, G.W.; Popper, J.S.; White, L.R. Epidemiologic observations on Parkinson’s disease: Incidence and mortality in a prospective study of middle-aged men. Neurology 1996, 46, 1044–1050. [Google Scholar]
- De Lau, L.M.; Giesbergen, P.C.; de Rijk, M.C.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M. Incidence of parkinsonism and Parkinson disease in a general population: The Rotterdam Study. Neurology 2004, 63, 1240–1244. [Google Scholar]
- De Rijk, M.C.; Tzourio, C.; Breteler, M.M.; Dartigues, J.F.; Amaducci, L.; Lopez-Pousa, S.; Manubens-Bertran, J.M.; Alperovitch, A.; Rocca, W.A. Prevalence of parkinsonism and Parkinson’s disease in Europe: The Europarkinson collaborative study. European Community Concerted Action on the Epidemiology of Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1997, 62, 10–15. [Google Scholar]
- Fearnley, J.M.; Lees, A.J. Ageing and Parkinson’s disease: Substantia nigra regional selectivity. Brain 1991, 114, 2283–2301. [Google Scholar]
- Mann, D.M.; Yates, P.O. Possible role of neuromelanin in the pathogenesis of Parkinson’s disease. Mech. Ageing Dev 1983, 21, 193–203. [Google Scholar]
- Dorsey, E.R.; Constantinescu, R.; Thompson, J.P.; Biglan, K.M.; Holloway, R.G.; Kieburtz, K.; Marshall, F.J.; Ravina, B.M.; Schifitto, G.; Siderowf, A.; et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 2007, 68, 384–386. [Google Scholar]
- Marsden, C.D. Parkinson’s disease. Lancet 1990, 335, 948–952. [Google Scholar]
- Beal, M.F. Therapeutic approaches to mitochondrial dysfunction in Parkinson’s disease. Parkinsonism Relat. Disord 2009, 15, S189–S194. [Google Scholar]
- Kushnareva, Y.; Murphy, A.N.; Andreyev, A. Complex I-mediated reactive oxygen species generation: Modulation by cytochrome c and NAD(P)+ oxidation-reduction state. Biochem. J 2002, 368, 545–553. [Google Scholar]
- Fasano, M.; Bergamasco, B.; Lopiano, L. Modifications of the iron-neuromelanin system in Parkinson’s disease. J. Neurochem 2006, 96, 909–916. [Google Scholar]
- Ischiropoulos, H.; Beckman, J.S. Oxidative stress and nitration in neurodegeneration: Cause, effect, or association? J. Clin. Invest 2003, 111, 163–169. [Google Scholar]
- Linnane, A.W.; Marzuki, S.; Ozawa, T.; Tanaka, M. Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet 1989, 1, 642–645. [Google Scholar]
- Kraytsberg, Y.; Kudryavtseva, E.; McKee, A.C.; Geula, C.; Kowall, N.W.; Khrapko, K. Mitochondrial DNA deletions are abundant and cause functional impairment in aged human substantia nigra neurons. Nat. Genet 2006, 38, 518–520. [Google Scholar]
- Bender, A.; Krishnan, K.J.; Morris, C.M.; Taylor, G.A.; Reeve, A.K.; Perry, R.H.; Jaros, E.; Hersheson, J.S.; Betts, J.; Klopstock, T.; et al. High levels of mitochondrial DNA deletions in substantia nigra neurons in aging and Parkinson disease. Nat. Genet 2006, 38, 515–517. [Google Scholar]
- Anderson, S.; Bankier, A.T.; Barrell, B.G.; de Bruijn, M.H.; Coulson, A.R.; Drouin, J.; Eperon, I.C.; Nierlich, D.P.; Roe, B.A.; Sanger, F.; et al. Sequence and organization of the human mitochondrial genome. Nature 1981, 290, 457–465. [Google Scholar]
- Pinton, P.; Rimessi, A.; Marchi, S.; Orsini, F.; Migliaccio, E.; Giorgio, M.; Contursi, C.; Minucci, S.; Mantovani, F.; Wieckowski, M.R.; et al. Protein kinase C beta and prolyl isomerase 1 regulate mitochondrial effects of the life-span determinant p66Shc. Science 2007, 315, 659–663. [Google Scholar]
- Ruan, H.; Tang, X.D.; Chen, M.L.; Joiner, M.L.; Sun, G.; Brot, N.; Weissbach, H.; Heinemann, S.H.; Iverson, L.; Wu, C.F.; et al. High-quality life extension by the enzyme peptide methionine sulfoxide reductase. Proc. Natl. Acad. Sci. USA 2002, 99, 2748–2753. [Google Scholar]
- Kujoth, G.C.; Hiona, A.; Pugh, T.D.; Someya, S.; Panzer, K.; Wohlgemuth, S.E.; Hofer, T.; Seo, A.Y.; Sullivan, R.; Jobling, W.A.; et al. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science 2005, 309, 481–484. [Google Scholar]
- Trifunovic, A.; Wredenberg, A.; Falkenberg, M.; Spelbrink, J.N.; Rovio, A.T.; Bruder, C.E.; Bohlooly, Y.M.; Gidlof, S.; Oldfors, A.; Wibom, R.; et al. Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 2004, 429, 417–423. [Google Scholar]
- Davidzon, G.; Greene, P.; Mancuso, M.; Klos, K.J.; Ahlskog, J.E.; Hirano, M.; DiMauro, S. Early-onset familial parkinsonism due to POLG mutations. Ann. Neurol 2006, 59, 859–862. [Google Scholar]
- Ahluwalia, N.; Gordon, M.A.; Handte, G.; Mahlon, M.; Li, N.Q.; Beard, J.L.; Weinstock, D.; Ross, A.C. Iron status and stores decline with age in Lewis rats. J. Nutr 2000, 130, 2378–2383. [Google Scholar]
- Bonifati, V.; Rizzu, P.; van Baren, M.J.; Schaap, O.; Breedveld, G.J.; Krieger, E.; Dekker, M.C.; Squitieri, F.; Ibanez, P.; Joosse, M.; et al. Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science 2003, 299, 256–259. [Google Scholar]
- Van Duijn, C.M.; Dekker, M.C.; Bonifati, V.; Galjaard, R.J.; Houwing-Duistermaat, J.J.; Snijders, P.J.; Testers, L.; Breedveld, G.J.; Horstink, M.; Sandkuijl, L.A.; et al. Park7, a novel locus for autosomal recessive early-onset parkinsonism, on chromosome 1p36. Am. J. Hum. Genet 2001, 69, 629–634. [Google Scholar]
- Lesage, S.; Brice, A. Role of Mendelian genes in “sporadic” Parkinson’s disease. Parkinsonism Relat. Disord 2012, 18, S66–S70. [Google Scholar]
- Horowitz, M.P.; Greenamyre, J.T. Gene-environment interactions in Parkinson’s disease: The importance of animal modeling. Clin. Pharmacol. Ther 2010, 88, 467–474. [Google Scholar]
- Meulener, M.C.; Xu, K.; Thomson, L.; Ischiropoulos, H.; Bonini, N.M. Mutational analysis of DJ-1 in Drosophila implicates functional inactivation by oxidative damage and aging. Proc. Natl. Acad. Sci. USA 2006, 103, 12517–12522. [Google Scholar]
- Yamaguchi, H.; Shen, J. Absence of dopaminergic neuronal degeneration and oxidative damage in aged DJ-1-deficient mice. Mol. Neurodegener 2007, 2. [Google Scholar] [CrossRef]
- Kawajiri, S.; Saiki, S.; Sato, S.; Hattori, N. Genetic mutations and functions of PINK1. Trends Pharmacol. Sci 2011, 32, 573–580. [Google Scholar]
- Moore, D.J.; West, A.B.; Dawson, V.L.; Dawson, T.M. Molecular pathophysiology of Parkinson’s disease. Annu. Rev. Neurosci 2005, 28, 57–87. [Google Scholar]
- Todd, A.M.; Staveley, B.E. Pink1 suppresses alpha-synuclein-induced phenotypes in a Drosophila model of Parkinson’s disease. Genome 2008, 51, 1040–1046. [Google Scholar]
- Haywood, A.F.; Staveley, B.E. Parkin counteracts symptoms in a Drosophila model of Parkinson’s disease. BMC Neurosci 2004, 5. [Google Scholar] [CrossRef] [Green Version]
- Palacino, J.J.; Sagi, D.; Goldberg, M.S.; Krauss, S.; Motz, C.; Wacker, M.; Klose, J.; Shen, J. Mitochondrial dysfunction and oxidative damage in parkin-deficient mice. J. Biol. Chem 2004, 279, 18614–18622. [Google Scholar]
- Perez, F.A.; Palmiter, R.D. Parkin-deficient mice are not a robust model of parkinsonism. Proc. Natl. Acad. Sci. USA 2005, 102, 2174–2179. [Google Scholar]
- Periquet, M.; Corti, O.; Jacquier, S.; Brice, A. Proteomic analysis of parkin knockout mice: Alterations in energy metabolism, protein handling and synaptic function. J. Neurochem 2005, 95, 1259–1276. [Google Scholar]
- Kitada, T.; Asakawa, S.; Hattori, N.; Matsumine, H.; Yamamura, Y.; Minoshima, S.; Yokochi, M.; Mizuno, Y.; Shimizu, N. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 1998, 392, 605–608. [Google Scholar]
- Hedrich, K.; Eskelson, C.; Wilmot, B.; Marder, K.; Harris, J.; Garrels, J.; Meija-Santana, H.; Vieregge, P.; Jacobs, H.; Bressman, S.B.; et al. Distribution, type, and origin of Parkin mutations: Review and case studies. Mov. Disord 2004, 19, 1146–1157. [Google Scholar]
- Lucking, C.B.; Durr, A.; Bonifati, V.; Vaughan, J.; de Michele, G.; Gasser, T.; Harhangi, B.S.; Meco, G.; Denefle, P.; Wood, N.W.; et al. Association between early-onset Parkinson’s disease and mutations in the parkin gene. N. Engl. J. Med 2000, 342, 1560–1567. [Google Scholar]
- Periquet, M.; Latouche, M.; Lohmann, E.; Rawal, N.; de Michele, G.; Ricard, S.; Teive, H.; Fraix, V.; Vidailhet, M.; Nicholl, D.; et al. Parkin mutations are frequent in patients with isolated early-onset parkinsonism. Brain 2003, 126, 1271–1278. [Google Scholar]
- Lohmann, E.; Periquet, M.; Bonifati, V.; Wood, N.W.; de Michele, G.; Bonnet, A.M.; Fraix, V.; Broussolle, E.; Horstink, M.W.; Vidailhet, M.; et al. How much phenotypic variation can be attributed to parkin genotype? Ann. Neurol 2003, 54, 176–185. [Google Scholar]
- Foroud, T.; Uniacke, S.K.; Liu, L.; Pankratz, N.; Rudolph, A.; Halter, C.; Shults, C.; Marder, K.; Conneally, P.M.; Nichols, W.C. Heterozygosity for a mutation in the parkin gene leads to later onset Parkinson disease. Neurology 2003, 60, 796–801. [Google Scholar]
- Sun, M.; Latourelle, J.C.; Wooten, G.F.; Lew, M.F.; Klein, C.; Shill, H.A.; Golbe, L.I.; Mark, M.H.; Racette, B.A.; Perlmutter, J.S.; et al. Influence of heterozygosity for parkin mutation on onset age in familial Parkinson disease: The GenePD study. Arch. Neurol 2006, 63, 826–832. [Google Scholar]
- Broussolle, E.; Lucking, C.B.; Ginovart, N.; Pollak, P.; Remy, P.; Durr, A. [18 F]-dopa PET study in patients with juvenile-onset PD and parkin gene mutations. Neurology 2000, 55, 877–879. [Google Scholar]
- Shulman, J.M.; de Jager, P.L.; Feany, M.B. Parkinson’s disease: Genetics and pathogenesis. Annu. Rev. Pathol 2011, 6, 193–222. [Google Scholar]
- Slivka, A.; Cohen, G. Hydroxyl radical attack on dopamine. J. Biol. Chem 1985, 260, 15466–15472. [Google Scholar]
- Fornstedt, B.; Pileblad, E.; Carlsson, A. In vivo autoxidation of dopamine in guinea pig striatum increases with age. J. Neurochem 1990, 55, 655–659. [Google Scholar]
- Sulzer, D.; Zecca, L. Intraneuronal dopamine-quinone synthesis: A review. Neurotox. Res 2000, 1, 181–195. [Google Scholar]
- Mann, J.J.; Stanley, M. Postmortem monoamine oxidase enzyme kinetics in the frontal cortex of suicide victims and controls. Acta Psychiatr. Scand 1984, 69, 135–139. [Google Scholar]
- Jossan, S.S.; Gillberg, P.G.; d’Argy, R.; Aquilonius, S.M.; Langstrom, B.; Halldin, C.; Oreland, L. Quantitative localization of human brain monoamine oxidase B by large section autoradiography using l-[3H]deprenyl. Brain Res 1991, 547, 69–76. [Google Scholar]
- Strolin Benedetti, M.; Dostert, P. Monoamine oxidase, brain ageing and degenerative diseases. Biochem. Pharmacol 1989, 38, 555–561. [Google Scholar]
- Thomas, B.B.M. Parkinson’s disease. Hum. Mol. Genet 2007, 16, R183–R194. [Google Scholar]
- Morgan, D.G.; May, P.C.; Finch, C.E. Dopamine and serotonin systems in human and rodent brain: Effects of age and neurodegenerative disease. J. Am. Geriatr. Soc 1987, 35, 334–345. [Google Scholar]
- Kish, S.J.; Shannak, K.; Rajput, A.; Deck, J.H.; Hornykiewicz, O. Aging produces a specific pattern of striatal dopamine loss: Implications for the etiology of idiopathic Parkinson’s disease. J. Neurochem 1992, 58, 642–648. [Google Scholar]
- Thannickal, T.C.; Lai, Y.Y.; Siegel, J.M. Hypocretin (orexin) and melanin concentrating hormone loss and the symptoms of Parkinson’s disease. Brain 2008, 131, e87. [Google Scholar]
- Cruz-Muros, I.; Afonso-Oramas, D.; Abreu, P.; Barroso-Chinea, P.; Rodriguez, M.; Gonzalez, M.C.; Hernandez, T.G. Aging of the rat mesostriatal system: Differences between the nigrostriatal and the mesolimbic compartments. Exp. Neurol 2007, 204, 147–161. [Google Scholar]
- Goudsmit, E.; Feenstra, M.G.; Swaab, D.F. Central monoamine metabolism in the male Brown-Norway rat in relation to aging and testosterone. Brain Res. Bull 1990, 25, 755–763. [Google Scholar]
- Volkow, N.D.; Ding, Y.S.; Fowler, J.S.; Wang, G.J.; Logan, J.; Gatley, S.J.; Hitzemann, R.; Smith, G.; Fields, S.D.; Gur, R. Dopamine transporters decrease with age. J. Nucl. Med 1996, 37, 554–559. [Google Scholar]
- Caudle, W.M.; Richardson, J.R.; Wang, M.Z.; Taylor, T.N.; Guillot, T.S.; McCormack, A.L.; Colebrooke, R.E.; di Monte, D.A.; Emson, P.C.; Miller, G.W. Reduced vesicular storage of dopamine causes progressive nigrostriatal neurodegeneration. J. Neurosci 2007, 27, 8138–8148. [Google Scholar]
- Gonzalez-Hernandez, T.; Barroso-Chinea, P.; de La Cruz Muros, I.; del Mar Perez-Delgado, M.; Rodriguez, M. Expression of dopamine and vesicular monoamine transporters and differential vulnerability of mesostriatal dopaminergic neurons. J. Comp. Neurol 2004, 479, 198–215. [Google Scholar]
- Cantuti-Castelvetri, I.; Shukitt-Hale, B.; Joseph, J.A. Dopamine neurotoxicity: Age-dependent behavioral and histological effects. Neurobiol. Aging 2003, 24, 697–706. [Google Scholar]
- De Keyser, J.; Ebinger, G.; Vauquelin, G. Age-related changes in the human nigrostriatal dopaminergic system. Ann. Neurol 1990, 27, 157–161. [Google Scholar]
- Irwin, I.; DeLanney, L.E.; McNeill, T.; Chan, P.; Forno, L.S.; Murphy, G.M., Jr; di Monte, D.A.; Sandy, M.S.; Langston, J.W. Aging and the nigrostriatal dopamine system: A non-human primate study. Neurodegeneration 1994, 3, 251–265. [Google Scholar]
- Borges, C.R.; Geddes, T.; Watson, J.T.; Kuhn, D.M. Dopamine biosynthesis is regulated by S-glutathionylation. Potential mechanism of tyrosine hydroxylast inhibition during oxidative stress. J. Biol. Chem 2002, 277, 48295–48302. [Google Scholar]
- De La Cruz, C.P.; Revilla, E.; Venero, J.L.; Ayala, A.; Cano, J.; Machado, A. Oxidative inactivation of tyrosine hydroxylase in substantia nigra of aged rat. Free Radic. Biol. Med 1996, 20, 53–61. [Google Scholar]
- Ischiropoulos, H. Biological tyrosine nitration: A pathophysiological function of nitric oxide and reactive oxygen species. Arch. Biochem. Biophys 1998, 356, 1–11. [Google Scholar]
- Goedert, M. Familial Parkinson’s disease. The awakening of alpha-synuclein. Nature 1997, 388, 232–233. [Google Scholar]
- Yu, S.; Ueda, K.; Chan, P. Alpha-synuclein and dopamine metabolism. Mol. Neurobiol 2005, 31, 243–254. [Google Scholar]
- Perez, R.G.; Hastings, T.G. Could a loss of alpha-synuclein function put dopaminergic neurons at risk? J. Neurochem 2004, 89, 1318–1324. [Google Scholar]
- Kim, K.S.; Choi, S.Y.; Kwon, H.Y.; Won, M.H.; Kang, T.C.; Kang, J.H. Aggregation of alpha-synuclein induced by the Cu,Zn-superoxide dismutase and hydrogen peroxide system. Free Radic. Biol. Med 2002, 32, 544–550. [Google Scholar]
- Conway, K.A.; Rochet, J.C.; Bieganski, R.M.; Lansbury, P.T., Jr. Kinetic stabilization of the alpha-synuclein protofibril by a dopamine-alpha-synuclein adduct. Science 2001, 294, 1346–1349. [Google Scholar]
- Xu, J.; Kao, S.Y.; Lee, F.J.; Song, W.; Jin, L.W.; Yankner, B.A. Dopamine-dependent neurotoxicity of alpha-synuclein: A mechanism for selective neurodegeneration in Parkinson disease. Nat. Med 2002, 8, 600–606. [Google Scholar]
- Borghi, R.; Marchese, R.; Negro, A.; Marinelli, L.; Forloni, G.; Zaccheo, D.; Abbruzzese, G.; Tabaton, M. Full length alpha-synuclein is present in cerebrospinal fluid from Parkinson’s disease and normal subjects. Neurosci. Lett 2000, 287, 65–67. [Google Scholar]
- Chu, Y.; Kordower, J.H. Age-associated increases of alpha-synuclein in monkeys and humans are associated with nigrostriatal dopamine depletion: Is this the target for Parkinson’s disease? Neurobiol. Dis 2007, 25, 134–149. [Google Scholar]
- Jellinger, K.A. Lewy body-related alpha-synucleinopathy in the aged human brain. J. Neural. Transm 2004, 111, 1219–1235. [Google Scholar]
- Li, W.; Lesuisse, C.; Xu, Y.; Troncoso, J.C.; Price, D.L.; Lee, M.K. Stabilization of alpha-synuclein protein with aging and familial parkinson’s disease-linked A53T mutation. J. Neurosci 2004, 24, 7400–7409. [Google Scholar]
- Buchman, A.S.; Shulman, J.M.; Nag, S.; Leurgans, S.E.; Arnold, S.E.; Morris, M.C.; Schneider, J.A.; Bennett, D.A. Nigral pathology and parkinsonian signs in elders without Parkinson disease. Ann. Neurol 2012, 71, 258–266. [Google Scholar]
- Adamczyk, A.; Kazmierczak, A.; Strosznajder, J.B. Alpha-synuclein and its neurotoxic fragment inhibit dopamine uptake into rat striatal synaptosomes. Relationship to nitric oxide. Neurochem. Int 2006, 49, 407–412. [Google Scholar]
- Saito, Y.; Kawashima, A.; Ruberu, N.N.; Fujiwara, H.; Koyama, S.; Sawabe, M.; Arai, T.; Nagura, H.; Yamanouchi, H.; Hasegawa, M.; et al. Accumulation of phosphorylated alpha-synuclein in aging human brain. J. Neuropathol. Exp. Neurol 2003, 62, 644–654. [Google Scholar]
- Junn, E.; Mouradian, M.M. Human alpha-synuclein over-expression increases intracellular reactive oxygen species levels and susceptibility to dopamine. Neurosci. Lett 2002, 320, 146–150. [Google Scholar]
- Hashimoto, M.; Hsu, L.J.; Xia, Y.; Takeda, A.; Sisk, A.; Sundsmo, M.; Masliah, E. Oxidative stress induces amyloid-like aggregate formation of NACP/alpha-synuclein in vitro. Neuroreport 1999, 10, 717–721. [Google Scholar]
- Turnbull, S.; Tabner, B.J.; El-Agnaf, O.M.; Moore, S.; Davies, Y.; Allsop, D. Alpha-Synuclein implicated in Parkinson’s disease catalyses the formation of hydrogen peroxide in vitro. Free Radic. Biol. Med 2001, 30, 1163–1170. [Google Scholar]
- Hartl, F.U. Molecular chaperones in cellular protein folding. Nature 1996, 381, 571–579. [Google Scholar]
- Neumann, M.; Kahle, P.J.; Giasson, B.I.; Ozmen, L.; Borroni, E.; Spooren, W.; Muller, V.; Odoy, S.; Fujiwara, H.; Hasegawa, M.; et al. Misfolded proteinase K-resistant hyperphosphorylated alpha-synuclein in aged transgenic mice with locomotor deterioration and in human alpha-synucleinopathies. J. Clin. Invest 2002, 110, 1429–1439. [Google Scholar]
- Beach, T.G.; Walker, D.G.; Sue, L.I.; Newell, A.; Adler, C.C.; Joyce, J.N. Substantia nigra Marinesco bodies are associated with decreased striatal expression of dopaminergic markers. J. Neuropathol. Exp. Neurol 2004, 63, 329–337. [Google Scholar]
- Braak, H.; del Tredici, K.; Rub, U.; de Vos, R.A.; Jansen Steur, E.N.; Braak, E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol. Aging 2003, 24, 197–211. [Google Scholar]
- Cuervo, A.M.; Stefanis, L.; Fredenburg, R.; Lansbury, P.T.; Sulzer, D. Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science 2004, 305, 1292–1295. [Google Scholar]
- Lee, H.J.; Khoshaghideh, F.; Patel, S.; Lee, S.J. Clearance of alpha-synuclein oligomeric intermediates via the lysosomal degradation pathway. J. Neurosci 2004, 24, 1888–1896. [Google Scholar]
- Webb, J.L.; Ravikumar, B.; Atkins, J.; Skepper, J.N.; Rubinsztein, D.C. Alpha-Synuclein is degraded by both autophagy and the proteasome. J. Biol. Chem 2003, 278, 25009–25013. [Google Scholar]
- Perry, V.H.; Nicoll, J.A.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol 2010, 6, 193–201. [Google Scholar]
- Xu, H.; Chen, M.; Forrester, J.V. Para-inflammation in the aging retina. Prog. Retin. Eye Res 2009, 28, 348–368. [Google Scholar]
- Price, N.E.; Wadzinski, B.; Mumby, M.C. An anchoring factor targets protein phosphatase 2A to brain microtubules. Brain Res. Mol. Brain Res 1999, 73, 68–77. [Google Scholar]
- Klegeris, A.; McGeer, E.G.; McGeer, P.L. Therapeutic approaches to inflammation in neurodegenerative disease. Curr. Opin. Neurol 2007, 20, 351–357. [Google Scholar]
- Beach, T.G.; Sue, L.I.; Walker, D.G.; Lue, L.F.; Connor, D.J.; Caviness, J.N.; Sabbagh, M.N.; Adler, C.H. Marked microglial reaction in normal aging human substantia nigra: Correlation with extraneuronal neuromelanin pigment deposits. Acta Neuropathol 2007, 114, 419–424. [Google Scholar]
- Roy, A.; Jana, A.; Yatish, K.; Freidt, M.B.; Fung, Y.K.; Martinson, J.A.; Pahan, K. Reactive oxygen species up-regulate CD11b in microglia via nitric oxide: Implications for neurodegenerative diseases. Free Radic. Biol. Med 2008, 45, 686–699. [Google Scholar]
- McGeer, P.L.; Itagaki, S.; Boyes, B.E.; McGeer, E.G. Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 1988, 38, 1285–1291. [Google Scholar]
- Imamura, K.; Hishikawa, N.; Sawada, M.; Nagatsu, T.; Yoshida, M.; Hashizume, Y. Distribution of major histocompatibility complex class II-positive microglia and cytokine profile of Parkinson’s disease brains. Acta Neuropathol 2003, 106, 518–526. [Google Scholar]
- Gerhard, A.; Pavese, N.; Hotton, G.; Turkheimer, F.; Es, M.; Hammers, A.; Eggert, K.; Oertel, W.; Banati, R.B.; Brooks, D.J. In vivo imaging of microglial activation with [11C](R)-PK11195 PET in idiopathic Parkinson’s disease. Neurobiol. Dis 2006, 21, 404–412. [Google Scholar]
- Aloisi, F. Cytokine Production. In Neuroglia; Kettenmann, H., Ransom, B.R., Eds.; Oxford University Press: New York, NY, USA, 2005; pp. 285–301. [Google Scholar]
- Sierra, A.; Gottfried-Blackmore, A.C.; McEwen, B.S.; Bulloch, K. Microglia derived from aging mice exhibit an altered inflammatory profile. Glia 2007, 55, 412–424. [Google Scholar]
- Sawada, M.; Sawada, H.; Nagatsu, T. Effects of aging on neuroprotective and neurotoxic properties of microglia in neurodegenerative diseases. Neurodegener. Dis 2008, 5, 254–256. [Google Scholar]
- Godbout, J.P.; Chen, J.; Abraham, J.; Richwine, A.F.; Berg, B.M.; Kelley, K.W.; Johnson, R.W. Exaggerated neuroinflammation and sickness behavior in aged mice following activation of the peripheral innate immune system. FASEB J 2005, 19, 1329–1331. [Google Scholar]
- Damani, M.R.; Zhao, L.; Fontainhas, A.M.; Amaral, J.; Fariss, R.N.; Wong, W.T. Age-related alterations in the dynamic behavior of microglia. Aging Cell 2011, 10, 263–276. [Google Scholar]
- Gregory, A.; Polster, B.J.; Hayflick, S.J. Clinical and genetic delineation of neurodegeneration with brain iron accumulation. J. Med. Genet 2009, 46, 73–80. [Google Scholar]
- Colton, C.A.; Gilbert, D.L. Production of superoxide anions by a CNS macrophage, the microglia. FEBS Lett 1987, 223, 284–288. [Google Scholar]
- Biemond, P.; van Eijk, H.G.; Swaak, A.J.; Koster, J.F. Iron mobilization from ferritin by superoxide derived from stimulated polymorphonuclear leukocytes. Possible mechanism in inflammation diseases. J. Clin. Invest 1984, 73, 1576–1579. [Google Scholar]
- Agrawal, R.; Sharma, P.K.; Rao, G.S. Release of iron from ferritin by metabolites of benzene and superoxide radical generating agents. Toxicology 2001, 168, 223–230. [Google Scholar]
- Tanaka, M.; Sotomatsu, A.; Yoshida, T.; Hirai, S.; Nishida, A. Detection of superoxide production by activated microglia using a sensitive and specific chemiluminescence assay and microglia-mediated PC12h cell death. J. Neurochem 1994, 63, 266–270. [Google Scholar]
- Chen, Y.; Swanson, R.A. Astrocytes and brain injury. J. Cereb. Blood Flow Metab 2003, 23, 137–149. [Google Scholar]
- Nichols, N.R. Glial responses to steroids as markers of brain aging. J. Neurobiol 1999, 40, 585–601. [Google Scholar]
- Porchet, R.; Probst, A.; Bouras, C.; Draberova, E.; Draber, P.; Riederer, B.M. Analysis of glial acidic fibrillary protein in the human entorhinal cortex during aging and in Alzheimer’s disease. Proteomics 2003, 3, 1476–1485. [Google Scholar]
- Peinado, M.A.; Quesada, A.; Pedrosa, J.A.; Torres, M.I.; Martinez, M.; Esteban, F.J.; del Moral, M.L.; Hernandez, R.; Rodrigo, J.; Peinado, J.M. Quantitative and ultrastructural changes in glia and pericytes in the parietal cortex of the aging rat. Microsc. Res. Tech 1998, 43, 34–42. [Google Scholar]
- Pilegaard, K.; Ladefoged, O. Total number of astrocytes in the molecular layer of the dentate gyrus of rats at different ages. Anal. Quant. Cytol. Histol 1996, 18, 279–285. [Google Scholar]
- Rozovsky, I.; Finch, C.E.; Morgan, T.E. Age-related activation of microglia and astrocytes: In vitro studies show persistent phenotypes of aging, increased proliferation, and resistance to down-regulation. Neurobiol. Aging 1998, 19, 97–103. [Google Scholar]
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem 2009, 390, 191–214. [Google Scholar]
- Rice, M.E.; Russo-Menna, I. Differential compartmentalization of brain ascorbate and glutathione between neurons and glia. Neuroscience 1998, 82, 1213–1223. [Google Scholar]
- Gegg, M.E.; Beltran, B.; Salas-Pino, S.; Bolanos, J.P.; Clark, J.B.; Moncada, S.; Heales, S.J. Differential effect of nitric oxide on glutathione metabolism and mitochondrial function in astrocytes and neurones: Implications for neuroprotection/neurodegeneration? J. Neurochem 2003, 86, 228–237. [Google Scholar]
- Sian, J.; Dexter, D.T.; Lees, A.J.; Daniel, S.; Agid, Y.; Javoy-Agid, F.; Jenner, P.; Marsden, C.D. Alterations in glutathione levels in Parkinson’s disease and other neurodegenerative disorders affecting basal ganglia. Ann. Neurol 1994, 36, 348–355. [Google Scholar]
- O’Callaghan, J.P.; Miller, D.B. The concentration of glial fibrillary acidic protein increases with age in the mouse and rat brain. Neurobiol. Aging 1991, 12, 171–174. [Google Scholar]
- Uchida, K.; Kihara, N.; Hashimoto, K.; Nakayama, H.; Yamaguchi, R.; Tateyama, S. Age-related histological changes in the canine substantia nigra. J. Vet. Med. Sci 2003, 65, 179–185. [Google Scholar]
- Gu, X.L.; Long, C.X.; Sun, L.; Xie, C.; Lin, X.; Cai, H. Astrocytic expression of Parkinson’s disease-related A53T alpha-synuclein causes neurodegeneration in mice. Mol. Brain 2010, 3. [Google Scholar] [CrossRef]
- Giasson, B.I.; Duda, J.E.; Quinn, S.M.; Zhang, B.; Trojanowski, J.Q.; Lee, V.M. Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein. Neuron 2002, 34, 521–533. [Google Scholar]
- Lee, M.K.; Stirling, W.; Xu, Y.; Xu, X.; Qui, D.; Mandir, A.S.; Dawson, T.M.; Copeland, N.G.; Jenkins, N.A.; Price, D.L. Human alpha-synuclein-harboring familial Parkinson’s disease-linked Ala-53 → Thr mutation causes neurodegenerative disease with alpha-synuclein aggregation in transgenic mice. Proc. Natl. Acad. Sci. USA 2002, 99, 8968–8973. [Google Scholar]
- Park, S.J.; Lee, J.H.; Kim, H.Y.; Choi, Y.H.; Park, J.S.; Suh, Y.H.; Park, S.M.; Joe, E.H.; Jou, I. Astrocytes, but not microglia, rapidly sense H2O2 via STAT6 phosphorylation, resulting in cyclooxygenase-2 expression and prostaglandin release. J. Immunol 2012, 188, 5132–5141. [Google Scholar]
- Khachaturian, Z.S. The role of calcium regulation in brain aging: Reexamination of a hypothesis. Aging (Milano) 1989, 1, 17–34. [Google Scholar]
- Bean, B.P. The action potential in mammalian central neurons. Nat. Rev. Neurosci 2007, 8, 451–465. [Google Scholar]
- Bonci, A.; Grillner, P.; Mercuri, N.B.; Bernardi, G. L-Type calcium channels mediate a slow excitatory synaptic transmission in rat midbrain dopaminergic neurons. J. Neurosci 1998, 18, 6693–6703. [Google Scholar]
- Puopolo, M.; Raviola, E.; Bean, B.P. Roles of subthreshold calcium current and sodium current in spontaneous firing of mouse midbrain dopamine neurons. J. Neurosci 2007, 27, 645–656. [Google Scholar]
- Chan, C.S.; Guzman, J.N.; Ilijic, E.; Mercer, J.N.; Rick, C.; Tkatch, T.; Meredith, G.E.; Surmeier, D.J. “Rejuvenation” protects neurons in mouse models of Parkinson’s disease”. Nature 2007, 447, 1081–1086. [Google Scholar]
- Pignatelli, A.; Kobayashi, K.; Okano, H.; Belluzzi, O. Functional properties of dopaminergic neurones in the mouse olfactory bulb. J. Physiol 2005, 564, 501–514. [Google Scholar]
- German, D.C.; Manaye, K.F.; Sonsalla, P.K.; Brooks, B.A. Midbrain dopaminergic cell loss in Parkinson’s disease and MPTP-induced parkinsonism: Sparing of calbindin-D28k-containing cells. Ann. N. Y. Acad. Sci 1992, 648, 42–62. [Google Scholar]
- Belzunegui, S.; San Sebastian, W.; Garrido-Gil, P.; Izal-Azcarate, A.; Vazquez-Claverie, M.; Lopez, B.; Marcilla, I.; Lanciego, J.L.; Luquin, M.R. The number of dopaminergic cells is increased in the olfactory bulb of monkeys chronically exposed to MPTP. Synapse 2007, 61, 1006–1012. [Google Scholar]
- Rizzuto, R.; Pozzan, T. Microdomains of intracellular Ca2+: Molecular determinants and functional consequences. Physiol. Rev 2006, 86, 369–408. [Google Scholar]
- Verkhratsky, A. Physiology and pathophysiology of the calcium store in the endoplasmic reticulum of neurons. Physiol. Rev 2005, 85, 201–279. [Google Scholar]
- Roveri, A.; Coassin, M.; Maiorino, M.; Zamburlini, A.; van Amsterdam, F.T.; Ratti, E.; Ursini, F. Effect of hydrogen peroxide on calcium homeostasis in smooth muscle cells. Arch. Biochem. Biophys 1992, 297, 265–270. [Google Scholar]
- Wang, H.; Joseph, J.A. Mechanisms of hydrogen peroxide-induced calcium dysregulation in PC12 cells. Free Radic. Biol. Med 2000, 28, 1222–1231. [Google Scholar]
- Thibault, O.; Landfield, P.W. Increase in single L-type calcium channels in hippocampal neurons during aging. Science 1996, 272, 1017–1020. [Google Scholar]
- Altamura, S.; Muckenthaler, M.U. Iron toxicity in diseases of aging: Alzheimer’s disease, Parkinson’s disease and atherosclerosis. J. Alzheimers Dis 2009, 16, 879–895. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem. J 1984, 219, 1–14. [Google Scholar]
- Dexter, D.T.; Sian, J.; Jenner, P.; Marsden, C.D. Implications of alterations in trace element levels in brain in Parkinson’s disease and other neurological disorders affecting the basal ganglia. Adv. Neurol 1993, 60, 273–281. [Google Scholar]
- Connor, J.R.; Menzies, S.L.; St Martin, S.M.; Mufson, E.J. Cellular distribution of transferrin, ferritin, and iron in normal and aged human brains. J. Neurosci. Res 1990, 27, 595–611. [Google Scholar]
- Roskams, A.J.; Connor, J.R. Iron, transferrin, and ferritin in the rat brain during development and aging. J. Neurochem 1994, 63, 709–716. [Google Scholar]
- Bartzokis, G.; Tishler, T.A.; Lu, P.H.; Villablanca, P.; Altshuler, L.L.; Carter, M.; Huang, D.; Edwards, N.; Mintz, J. Brain ferritin iron may influence age- and gender-related risks of neurodegeneration. Neurobiol. Aging 2007, 28, 414–423. [Google Scholar]
- Berg, D.; Youdim, M.B.; Riederer, P. Redox imbalance. Cell Tissue Res 2004, 318, 201–213. [Google Scholar]
- Seo, A.Y.; Xu, J.; Servais, S.; Hofer, T.; Marzetti, E.; Wohlgemuth, S.E.; Knutson, M.D.; Chung, H.Y.; Leeuwenburgh, C. Mitochondrial iron accumulation with age and functional consequences. Aging Cell 2008, 7, 706–716. [Google Scholar]
- Aquino, D.; Bizzi, A.; Grisoli, M.; Garavaglia, B.; Bruzzone, M.G.; Nardocci, N.; Savoiardo, M.; Chiapparini, L. Age-related iron deposition in the basal ganglia: Quantitative analysis in healthy subjects. Radiology 2009, 252, 165–172. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Oxygen free radicals and iron in relation to biology and medicine: Some problems and concepts. Arch. Biochem. Biophys 1986, 246, 501–514. [Google Scholar]
- Martin, W.R.; Ye, F.Q.; Allen, P.S. Increasing striatal iron content associated with normal aging. Mov. Disord 1998, 13, 281–286. [Google Scholar]
- Zecca, L.; Stroppolo, A.; Gatti, A.; Tampellini, D.; Toscani, M.; Gallorini, M.; Giaveri, G.; Arosio, P.; Santambrogio, P.; Fariello, R.G.; et al. The role of iron and copper molecules in the neuronal vulnerability of locus coeruleus and substantia nigra during aging. Proc. Natl. Acad. Sci. USA 2004, 101, 9843–9848. [Google Scholar]
- Leibold, E.A.; Gahring, L.C.; Rogers, S.W. Immunolocalization of iron regulatory protein expression in the murine central nervous system. Histochem. Cell Biol 2001, 115, 195–203. [Google Scholar]
- Han, J.; Cheng, F.C.; Yang, Z.; Dryhurst, G. Inhibitors of mitochondrial respiration, iron (II), and hydroxyl radical evoke release and extracellular hydrolysis of glutathione in rat striatum and substantia nigra: Potential implications to Parkinson’s disease. J. Neurochem 1999, 73, 1683–1695. [Google Scholar]
- Lan, J.; Jiang, D.H. Desferrioxamine and vitamin E protect against iron and MPTP-induced neurodegeneration in mice. J. Neural. Transm 1997, 104, 469–481. [Google Scholar]
- Shachar, D.B.; Kahana, N.; Kampel, V.; Warshawsky, A.; Youdim, M.B. Neuroprotection by a novel brain permeable iron chelator, VK-28, against 6-hydroxydopamine lession in rats. Neuropharmacology 2004, 46, 254–263. [Google Scholar]
- Nguyen, T.; Sherratt, P.J.; Nioi, P.; Yang, C.S.; Pickett, C.B. Nrf2 controls constitutive and inducible expression of ARE-driven genes through a dynamic pathway involving nucleocytoplasmic shuttling by Keap1. J. Biol. Chem 2005, 280, 32485–32492. [Google Scholar]
- Moi, P.; Chan, K.; Asunis, I.; Cao, A.; Kan, Y.W. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc. Natl. Acad. Sci. USA 1994, 91, 9926–9930. [Google Scholar]
- Ramsey, C.P.; Glass, C.A.; Montgomery, M.B.; Lindl, K.A.; Ritson, G.P.; Chia, L.A.; Hamilton, R.L.; Chu, C.T.; Jordan-Sciutto, K.L. Expression of Nrf2 in neurodegenerative diseases. J. Neuropathol. Exp. Neurol 2007, 66, 75–85. [Google Scholar]
- Von Otter, M.; Landgren, S.; Nilsson, S.; Celojevic, D.; Bergstrom, P.; Hakansson, A.; Nissbrandt, H.; Drozdzik, M.; Bialecka, M.; Kurzawski, M.; et al. Association of Nrf2-encoding NFE2L2 haplotypes with Parkinson’s disease. BMC Med. Genet 2010, 11. [Google Scholar] [CrossRef]
- Suh, J.H.; Shenvi, S.V.; Dixon, B.M.; Liu, H.; Jaiswal, A.K.; Liu, R.M.; Hagen, T.M. Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc. Natl. Acad. Sci. USA 2004, 101, 3381–3386. [Google Scholar]
- Rojo, A.I.; Sagarra, M.R.; Cuadrado, A. GSK-3beta down-regulates the transcription factor Nrf2 after oxidant damage: Relevance to exposure of neuronal cells to oxidative stress. J. Neurochem 2008, 105, 192–202. [Google Scholar]
- Ikeyama, S.; Kokkonen, G.; Shack, S.; Wang, X.T.; Holbrook, N.J. Loss in oxidative stress tolerance with aging linked to reduced extracellular signal-regulated kinase and Akt kinase activities. FASEB J 2002, 16, 114–116. [Google Scholar]
- Kozikowski, A.P.; Gaisina, I.N.; Petukhov, P.A.; Sridhar, J.; King, L.T.; Blond, S.Y.; Duka, T.; Rusnak, M.; Sidhu, A. Highly potent and specific GSK-3beta inhibitors that block tau phosphorylation and decrease alpha-synuclein protein expression in a cellular model of Parkinson’s disease. Chem. Med. Chem 2006, 1, 256–266. [Google Scholar]
- Chen, G.; Bower, K.A.; Ma, C.; Fang, S.; Thiele, C.J.; Luo, J. Glycogen synthase kinase 3beta (GSK3beta) mediates 6-hydroxydopamine-induced neuronal death. FASEB J 2004, 18, 1162–1164. [Google Scholar]
- Chen, C.; Pung, D.; Leong, V.; Hebbar, V.; Shen, G.; Nair, S.; Li, W.; Kong, A.N. Induction of detoxifying enzymes by garlic organosulfur compounds through transcription factor Nrf2: Effect of chemical structure and stress signals. Free Radic. Biol. Med 2004, 37, 1578–1590. [Google Scholar]
- Silva, R.M.; Kuan, C.Y.; Rakic, P.; Burke, R.E. Mixed lineage kinase-c-jun N-terminal kinase signaling pathway: A new therapeutic target in Parkinson’s disease. Mov. Disord 2005, 20, 653–664. [Google Scholar]
- Dagda, R.K.; Zhu, J.; Chu, C.T. Mitochondrial kinases in Parkinson’s disease: Converging insights from neurotoxin and genetic models. Mitochondrion 2009, 9, 289–298. [Google Scholar]
- Shih, P.H.; Yen, G.C. Differential expressions of antioxidant status in aging rats: The role of transcriptional factor Nrf2 and MAPK signaling pathway. Biogerontology 2007, 8, 71–80. [Google Scholar]
- Hsieh, C.C.; Rosenblatt, J.I.; Papaconstantinou, J. Age-associated changes in SAPK/JNK and p38 MAPK signaling in response to the generation of ROS by 3-nitropropionic acid. Mech. Ageing Dev 2003, 124, 733–746. [Google Scholar]
- Hsieh, C.C.; Papaconstantinou, J. Dermal fibroblasts from long-lived Ames dwarf mice maintain their in vivo resistance to mitochondrial generated reactive oxygen species (ROS). Aging (Albany NY) 2009, 1, 784–802. [Google Scholar]
- Helenius, M.; Hanninen, M.; Lehtinen, S.K.; Salminen, A. Changes associated with aging and replicative senescence in the regulation of transcription factor nuclear factor-kappa B. Biochem. J 1996, 318, 603–608. [Google Scholar]
- Korhonen, P.; Helenius, M.; Salminen, A. Age-related changes in the regulation of transcription factor NF-kappa B in rat brain. Neurosci. Lett 1997, 225, 61–64. [Google Scholar]
- Dehmer, T.; Lindenau, J.; Haid, S.; Dichgans, J.; Schulz, J.B. Deficiency of inducible nitric oxide synthase protects against MPTP toxicity in vivo. J. Neurochem 2000, 74, 2213–2216. [Google Scholar]
- Mogi, M.; Harada, M.; Kondo, T.; Riederer, P.; Inagaki, H.; Minami, M.; Nagatsu, T. Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from parkinsonian patients. Neurosci. Lett 1994, 180, 147–150. [Google Scholar]
- Sriram, K.; Matheson, J.M.; Benkovic, S.A.; Miller, D.B.; Luster, M.I.; O’Callaghan, J.P. Mice deficient in TNF receptors are protected against dopaminergic neurotoxicity: Implications for Parkinson’s disease. FASEB J 2002, 16, 1474–1476. [Google Scholar]
- Hayden, M.S.; Ghosh, S. Signaling to NF-kappaB. Genes Dev 2004, 18, 2195–2224. [Google Scholar]
- Saggu, H.; Cooksey, J.; Dexter, D.; Wells, F.R.; Lees, A.; Jenner, P.; Marsden, C.D. A selective increase in particulate superoxide dismutase activity in parkinsonian substantia nigra. J. Neurochem 1989, 53, 692–697. [Google Scholar]
- Tsay, H.J.; Wang, P.; Wang, S.L.; Ku, H.H. Age-associated changes of superoxide dismutase and catalase activities in the rat brain. J. Biomed. Sci 2000, 7, 466–474. [Google Scholar]
- Pearce, R.K.; Owen, A.; Daniel, S.; Jenner, P.; Marsden, C.D. Alterations in the distribution of glutathione in the substantia nigra in Parkinson’s disease. J. Neural. Transm 1997, 104, 661–677. [Google Scholar]
- Sofic, E.; Lange, K.W.; Jellinger, K.; Riederer, P. Reduced and oxidized glutathione in the substantia nigra of patients with Parkinson’s disease. Neurosci. Lett 1992, 142, 128–130. [Google Scholar]
- Huang, C.S.; Anderson, M.E.; Meister, A. Amino acid sequence and function of the light subunit of rat kidney gamma-glutamylcysteine synthetase. J. Biol. Chem 1993, 268, 20578–20583. [Google Scholar]
- Huang, C.S.; Chang, L.S.; Anderson, M.E.; Meister, A. Catalytic and regulatory properties of the heavy subunit of rat kidney gamma-glutamylcysteine synthetase. J. Biol. Chem 1993, 268, 19675–19680. [Google Scholar]
- Lu, S.C.; Huang, Z.Z.; Yang, H.; Tsukamoto, H. Effect of thioacetamide on the hepatic expression of gamma-glutamylcysteine synthetase subunits in the Rat. Toxicol. Appl. Pharmacol 1999, 159, 161–168. [Google Scholar]
- Zhu, Y.; Carvey, P.M.; Ling, Z. Age-related changes in glutathione and glutathione-related enzymes in rat brain. Brain Res 2006, 1090, 35–44. [Google Scholar]
- Vermulst, M.; Bielas, J.H.; Kujoth, G.C.; Ladiges, W.C.; Rabinovitch, P.S.; Prolla, T.A.; Loeb, L.A. Mitochondrial point mutations do not limit the natural lifespan of mice. Nat. Genet 2007, 39, 540–543. [Google Scholar]
- Schriner, S.E.; Linford, N.J.; Martin, G.M.; Treuting, P.; Ogburn, C.E.; Emond, M.; Coskun, P.E.; Ladiges, W.; Wolf, N.; van Remmen, H.; et al. Extension of murine life span by overexpression of catalase targeted to mitochondria. Science 2005, 308, 1909–1911. [Google Scholar]
- Perier, C.; Bove, J.; Dehay, B.; Jackson-Lewis, V.; Rabinovitch, P.S.; Przedborski, S.; Vila, M. Apoptosis-inducing factor deficiency sensitizes dopaminergic neurons to parkinsonian neurotoxins. Ann. Neurol 2010, 68, 184–192. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kumar, H.; Lim, H.-W.; More, S.V.; Kim, B.-W.; Koppula, S.; Kim, I.S.; Choi, D.-K. The Role of Free Radicals in the Aging Brain and Parkinson’s Disease: Convergence and Parallelism. Int. J. Mol. Sci. 2012, 13, 10478-10504. https://doi.org/10.3390/ijms130810478
Kumar H, Lim H-W, More SV, Kim B-W, Koppula S, Kim IS, Choi D-K. The Role of Free Radicals in the Aging Brain and Parkinson’s Disease: Convergence and Parallelism. International Journal of Molecular Sciences. 2012; 13(8):10478-10504. https://doi.org/10.3390/ijms130810478
Chicago/Turabian StyleKumar, Hemant, Hyung-Woo Lim, Sandeep Vasant More, Byung-Wook Kim, Sushruta Koppula, In Su Kim, and Dong-Kug Choi. 2012. "The Role of Free Radicals in the Aging Brain and Parkinson’s Disease: Convergence and Parallelism" International Journal of Molecular Sciences 13, no. 8: 10478-10504. https://doi.org/10.3390/ijms130810478
APA StyleKumar, H., Lim, H.-W., More, S. V., Kim, B.-W., Koppula, S., Kim, I. S., & Choi, D.-K. (2012). The Role of Free Radicals in the Aging Brain and Parkinson’s Disease: Convergence and Parallelism. International Journal of Molecular Sciences, 13(8), 10478-10504. https://doi.org/10.3390/ijms130810478






