Immobilization of Laccase for Oxidative Coupling of Trans-Resveratrol and Its Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Comparison of Immobilized TVL with Free TVL
2.2. Effect of Buffer Type
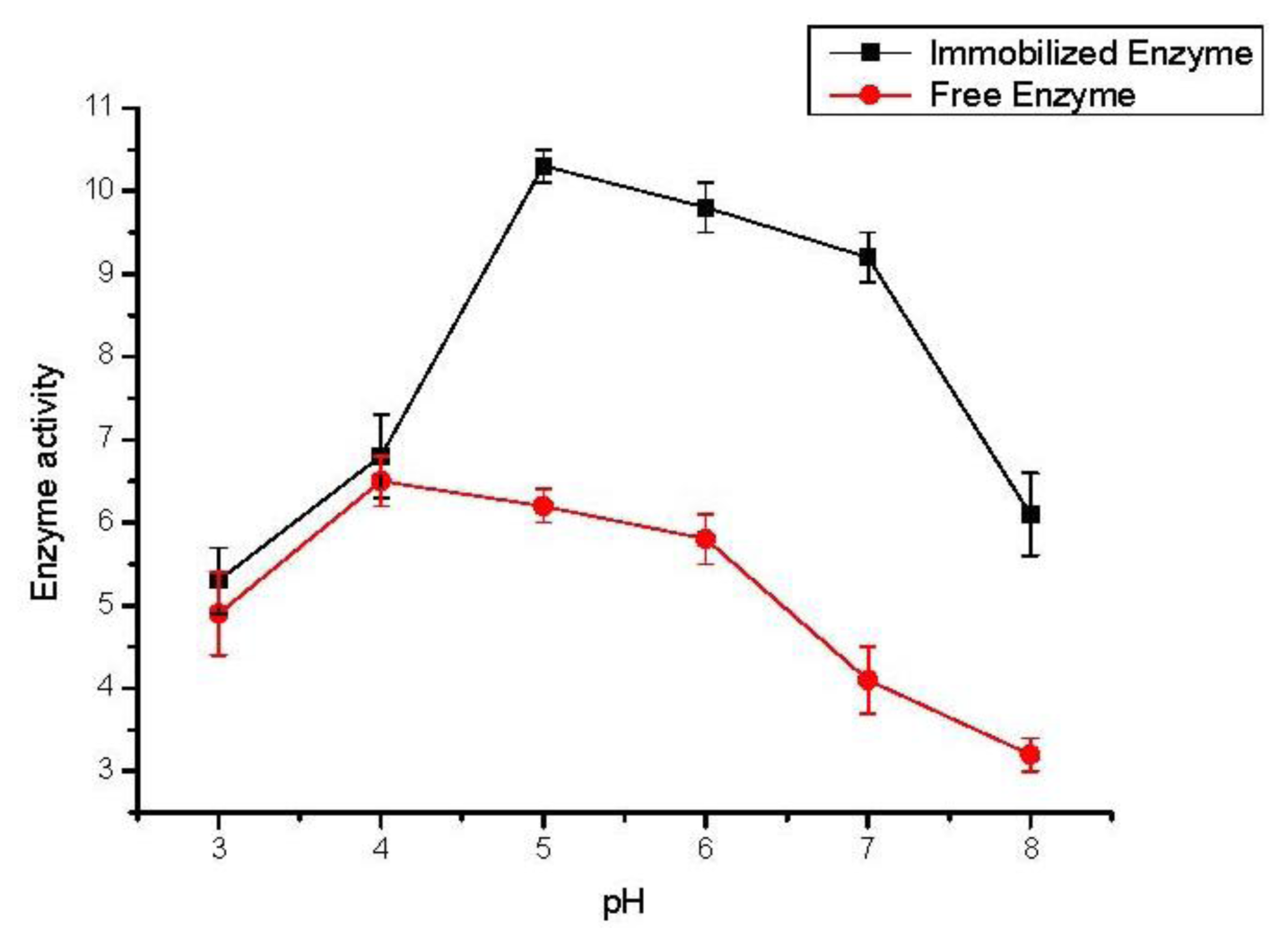
2.3. Effect of Buffer pH
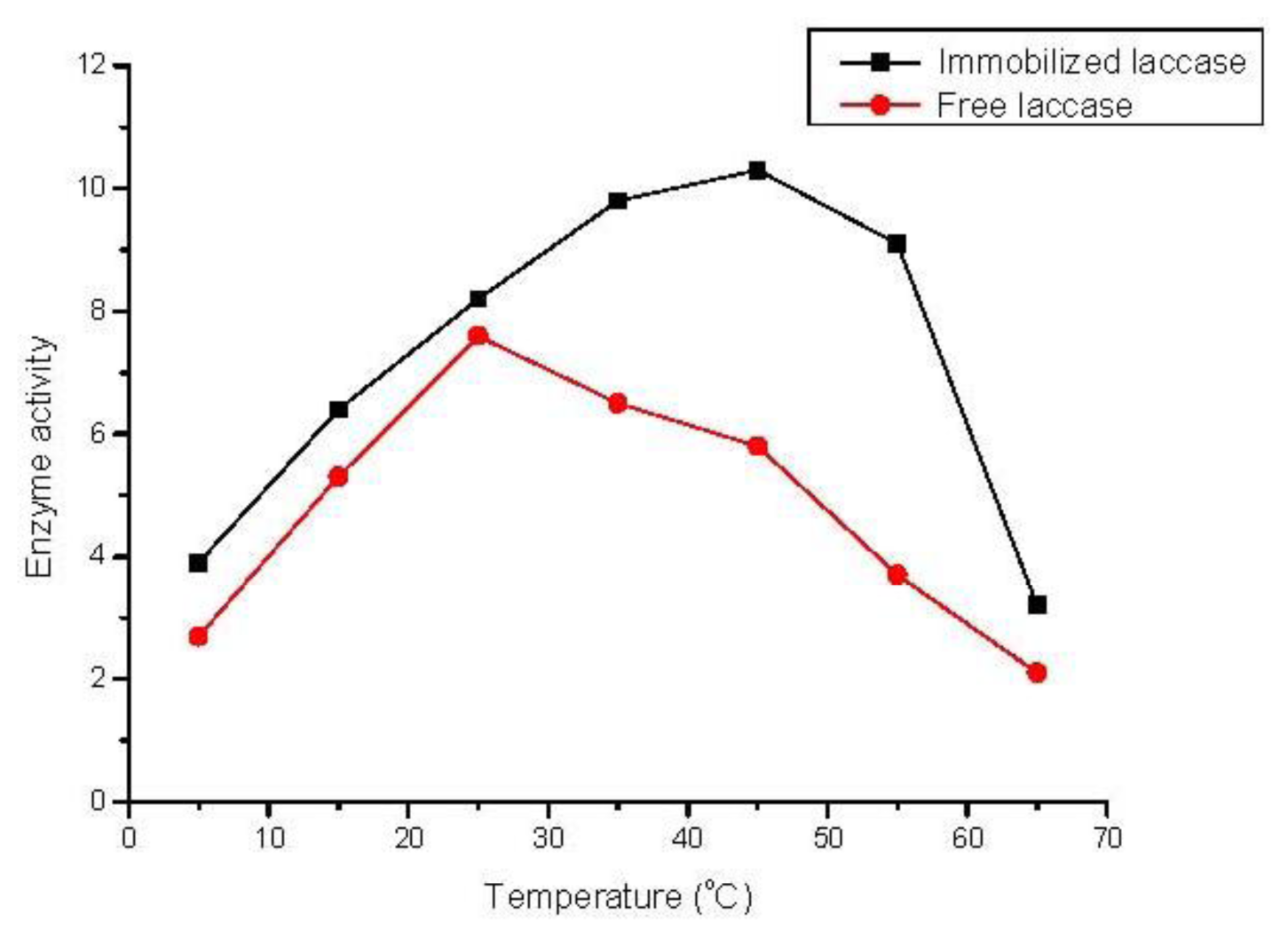
2.4. Effect of Temperature
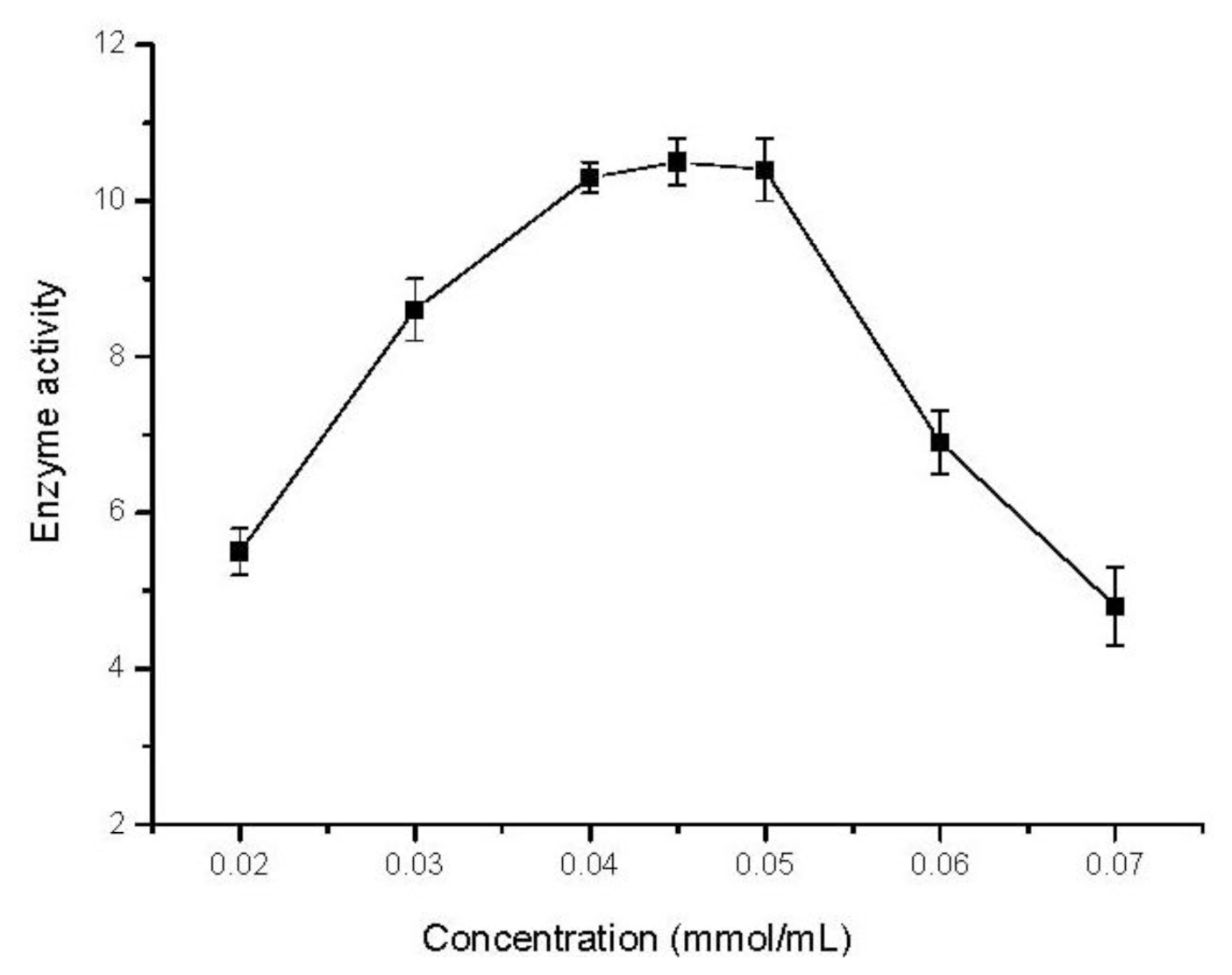
2.5. Effect of Substrate Concentration
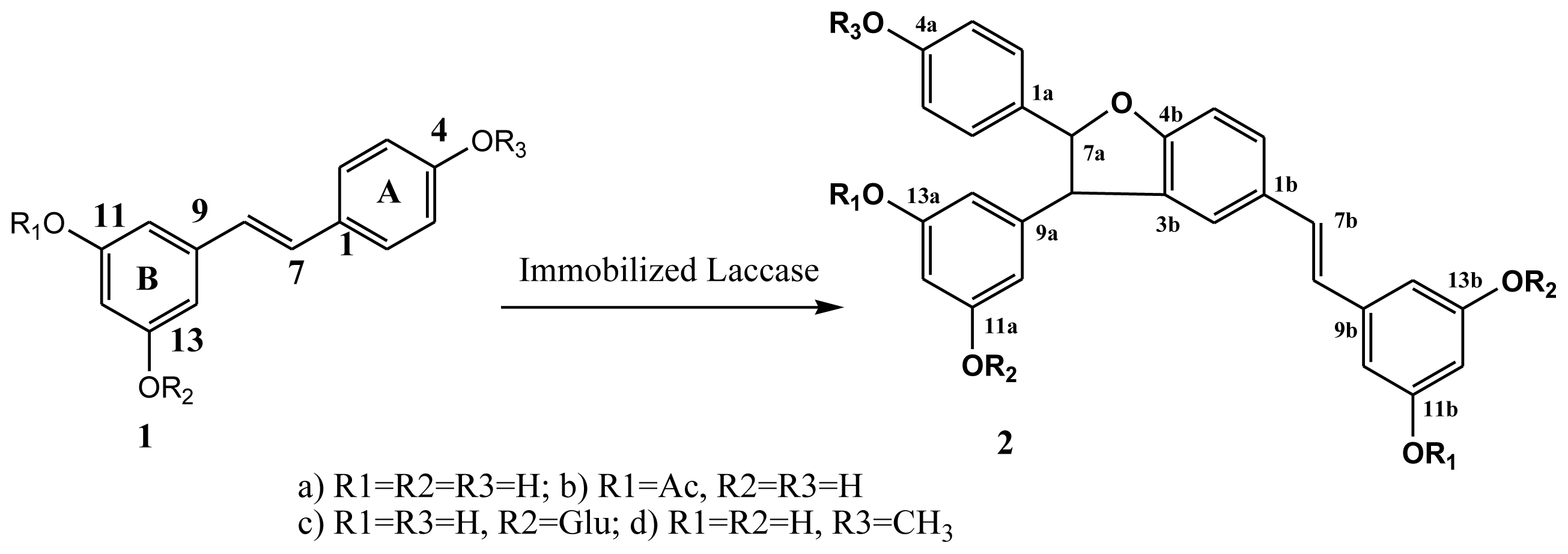
2.6. Oxidative Coupling of Resveratrol Derivatives
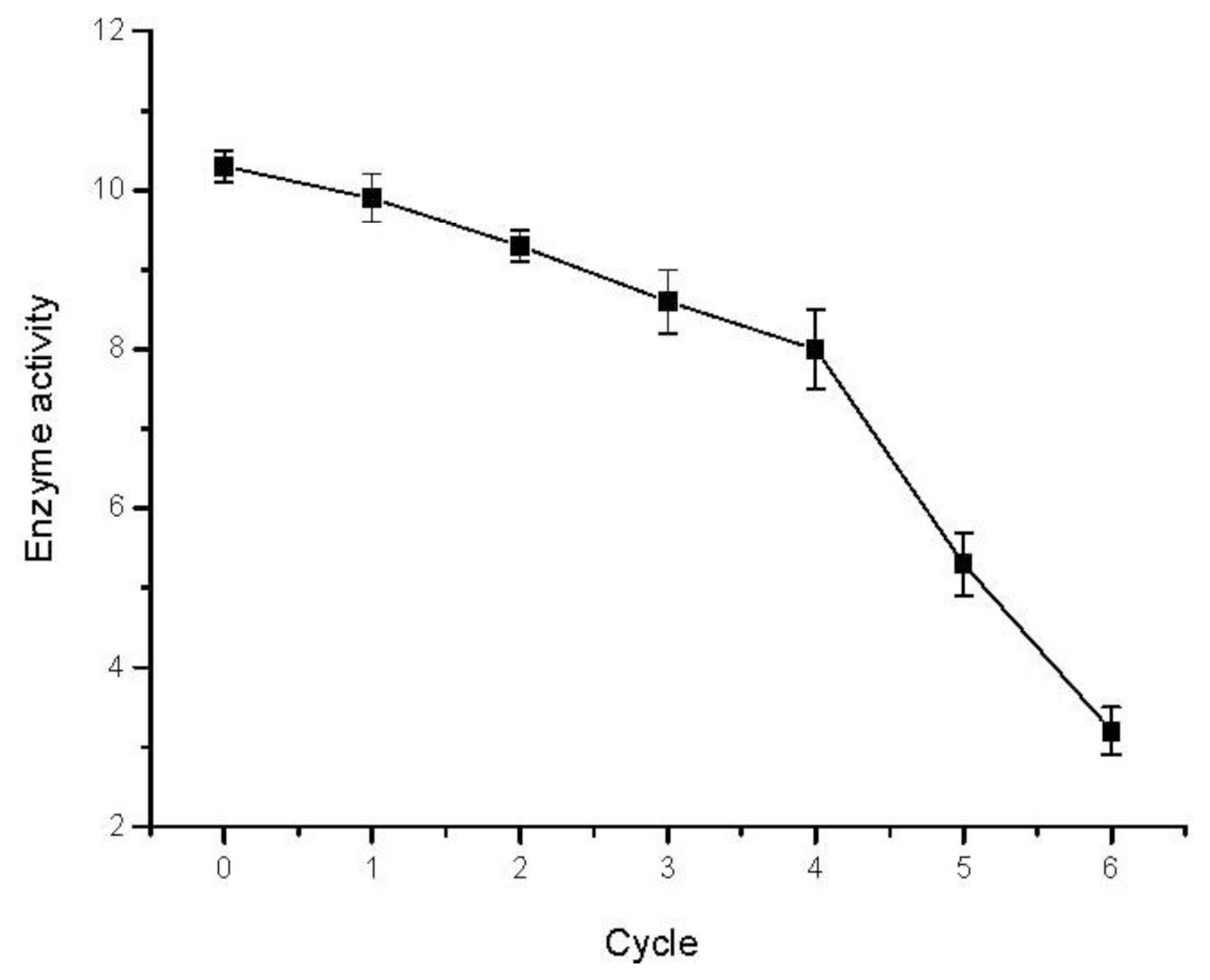
2.7. Reusability of Immobilized Enzyme
3. Experimental Section
3.1. Materials
3.2. Immobilization of Laccase Trametes Villosa
3.3. Enzymatic Oxidation of Trans-Resveratrol and Its Derivatives
3.4. Analytical Methods
3.5. Reusability
4. Conclusions
Acknowledgements
Reference
- Sotheewaran, S.; Pasupathy, V. Distribution of resveratrol oligomers in plants. Phytochemistry 1993, 35, 1083–1092. [Google Scholar]
- Dai, J.R.; Hallock, Y.F.; Cardellina, J.H., II; Boyd, M. Hiv-Inhibitory and cytotoxic oligostilbenes from the leaves of hopea malibato. J. Nat. Prod 1998, 61, 351–353. [Google Scholar]
- Iliya, I.; Tanaka, T.; Linuma, M.; Ali, Z.; Furasawa, M.; Nakaya, K.; Matsuura, N.; Ubukata, K. Four dimeric stilbenes in stem lianas of Gnetum africanum. Heterocycles 2002, 57, 1507–1512. [Google Scholar]
- Aggarwal, B.B.; Bhardwaj, A.; Aggarwal, R.S.; Seeram, N.P.; Shishodia, S.; Takada, Y. Role of resveratrol in prevention and therapy of cancer: Preclinical and clinical studies. Anticancer Res 2004, 24, 2783–2840. [Google Scholar]
- Bradamante, S.; Barenghi, L.; Villa, A. Cardiovascular protective effects of resveratrol. Cardiovasc. Drug Rev 2004, 22, 169–188. [Google Scholar]
- Cal, C.; Garban, H.; Jazirehi, A.; Yeh, C.; Mizutani, Y.; Bonavida, B. Resveratrol and cancer: Chemoprevention, apoptosis, and chemoimmunosensitizing activities. Curr. Med. Chem. Anti-Cancer Agent 2003, 3, 77–93. [Google Scholar]
- Jeffery, T.; Ferber, B. One-pot palladium-catalyzed highly chemo-, regio-, and stereoselective synthesis of trans-stilbene derivatives. A concise and convenient synthesis of resveratrol. Tetrahedron Lett 2003, 44, 193–197. [Google Scholar]
- Andrus, M.B.; Liu, J.; Meredith, E.L.; Nartey, E. Synthesis of resveratrol using a direct decarbonylative Heck approach from resorcylic acid. Tetrahedron Lett 2003, 44, 4819–4822. [Google Scholar]
- Park, E.J.; Min, H.Y.; Ahn, Y.H.; Bae, C.M.; Pyeeb, J.H.; Lee, S.K. Synthesis and inhibitoryeffects of pinosylvinderivatives on prostaglandin E2 production in lipopolysaccharide-induced mouse macrophage cells. Bioorg. Med. Chem. Lett 2004, 14, 5895–5898. [Google Scholar]
- Maliakel, B.P.; Schmid, W. Chemo-enzymatic synthesis of natural products: Synthesis of sphydrofuran. Tetrahedron Lett 1992, 33, 3297–3300. [Google Scholar]
- Shafiee, A.; Tsipouras, A.; Bouffard, A.; Conishi, J.; Guan, Z.Q.; Motamedi, H. Enzymatic deglycosylation of enfumafungin, a triterpene glycoside natural product, and its chemically synthesized analogues. J. Mol. Catal. B 2001, 16, 27–32. [Google Scholar]
- Gianfreda, L.; Xu, F.; Bollag, J.M. Laccases: A useful group of oxidoreductive enzymes. Bioremediat. J 1999, 3, 1–25. [Google Scholar]
- Solomon, E.I.; Sundaram, U.M.; Machonkin, T.E. Multicopper oxidases and oxygenases. Chem. Rev 1996, 96, 2563–2605. [Google Scholar]
- Nicotra, S.; Cramarossa, M.R.; Mucci, A.; Pagnoni, U.M.; Rivaa, S.; Fortib, L. Biotransformation of resveratrol: Synthesis of trans-dehydrodimers catalyzed by laccases from Myceliophtora thermophyla and from Trametes pubescens. Tetrahedron 2004, 60, 595–600. [Google Scholar]
- Goradia, D.; Cooney, J.; Hodnett, B.K.; Magner, E. The adsorption characteristics, activity and stability of trypsin onto mesoporous silicates. J. Mol. Catal. B 2005, 32, 231–239. [Google Scholar]
- Du, C.; Zhao, B.; Li, C.; Wang, P.; Wang, Z.; Tang, J.; Wang, L. Improvement of the enantioselectivity and activity of lipase from Pseudomonas sp. via adsorption on a hydrophobic support: Kinetic resolution of 2-octanol. Biocatal. Biotransfor 2009, 27, 340–347. [Google Scholar]
- Klibanov, A.M. Enzyme stabilization by immobilization. Anal. Biochem 1979, 93, 1–25. [Google Scholar]
- Kim, J.; Grate, J.W.; Wang, P. Nanostructures for enzyme stabilization. Chem. Eng. Sci 2006, 61, 1017–1026. [Google Scholar]
- Peralta-Zamora, P.; Pereira, C.M.; Tiburtius, E.R.L.; Moraes, S.G.; Rosa, M.A.; Minussi, R.C.; Durán, N. Decolorization of reactive dyes by immobilized laccase. Appl. Catal. B Environ 2003, 42, 131–144. [Google Scholar]
- Yu, D.H.; Wang, Z.; Zhao, L.F.; Cheng, Y.M.; Cao, S.G. Resolution of 2-octanol by SBA-15 immobilized Pseudomonas sp. Lipase. J. Mol. Catal. B 2007, 48, 64–69. [Google Scholar]
- Salis, A.; Pisano, M.; Monduzzi, M.; Solinas, V.; Sanjust, E. Laccase from Pleurotus sajor-caju on functionalised SBA-15 mesoporous silica: Immobilisation and use for the oxidation of phenolic compounds. J. Mol. Catal. B 2009, 58, 175–180. [Google Scholar]
- Yiu, H.H.P.; Wright, P.A. Enzymes supported on ordered mesoporous solids: A special case of an inorganic-organic hybrid. J. Mater. Chem 2005, 15, 3690–3700. [Google Scholar]
- Cheison, S.C.; Leeb, E.; Letzel, T.; Kulozik, U. Influence of buffer type and concentration on the peptide composition of trypsin hydrolysates of b-lactoglobulin. Food Chem 2011, 125, 121–127. [Google Scholar]
- Mohy Eldin, M.S.; Bencivenga, U.; Rossi, S.; Canciglia, P.; Gaeta, F.S.; Tramper, J.; Mita, D.G. Characterization of the activity of penicillin G acylase immobilized onto nylon membranes grafted with different acrylic monomers by means of γ-radiation. J. Mol. Catal. B 2000, 8, 233–244. [Google Scholar]
- Liu, J.G.; Cong, W.; Wang, S.; Ouyang, F. Studies of poly(vinyl acetate-co-diviyl benzene) beads as a carrier for the immobilization of penicillin acylase and the kinetics of immobilized penicillin acylase. React. Funct. Polym 2001, 48, 75–84. [Google Scholar]
- Leena, K.; Rohini, A.; Gadre, S.B.; Rajan, C.; Raman, S.P. Immobilization of penicillin G acylase on methacrylate polymers. J. Chem. Technol. Biotechnol 1990, 49, 173–182. [Google Scholar]
- Wang, F.; Guo, C.; Yang, L.R.; Liu, C.Z. Magnetic mesoporous silica nanoparticles: Fabrication and their laccase immobilization performance. Bioresour. Technol 2010, 101, 8931–8935. [Google Scholar]
- Hollmann, F.; Gumulya, Y.; Tölle, C.; Liese, A.; Thum, O. Evaluation of the Laccase from Myceliophthora thermophila as industrial biocatalyst for polymerization reactions. Macromolecules 2008, 41, 8520–8524. [Google Scholar]
- Luigi, C.; Michele, G.; Enrico, M.; Laura, S.; Giorgio, Z.; Takeshi, S. Enzymatic and spectroscopic studies on the activation or inhibition effects by substituted phenolic compounds in the oxidation of aryldiamines and catechols catalyzed by Rhus vernicifera laccase. J. Inorg. Biochem 2006, 100, 2127–2139. [Google Scholar]
- Santalla, E.; Serra, E.; Mayoral, A.; Losada, J.; Blanco, R.M.; Díaz, I. In situ immobilization of enzymes in mesoporous silicas. Solid State Sci 2011, 13, 691–697. [Google Scholar]
- Riva, S. Laccases: Blue enzymes for green chemistry. Trends Biotechnol 2006, 24, 219–216. [Google Scholar]
- Ye, P.; Xu, Z.K.; Wu, J.; Innocent, C.; Seta, P. Nanofibrous poly(acrylonitrile-co-maleic acid) membranes functionalized with gelatin and chitosan for lipase immobilization. Biomaterials 2006, 27, 4169–4176. [Google Scholar]
- Kiiskinen, L.L.; Kruus, V.K. Purification and characterisation of a novel laccase from the ascomycete Melanocarpus albomyces. Appl. Microbiol. Biotechnol 2002, 59, 198–204. [Google Scholar]
| Support | Free Enzyme | Glass Beads | Al2O3 | SBA-15 |
|---|---|---|---|---|
| bound protein amount (mg/g) | - | 34.9 ± 0.3 | 49.6 ± 0.2 | 95.5 ± 0.4 |
| Enzyme activity (μmol/g·h) | 5.8 ± 0.4 | 4.5 ± 0.3 | 6.2 ± 0.3 | 10.3 ± 0.2 |
| Buffer Type | Enzyme Activity (μmol/g·h) |
|---|---|
| citric acid–sodium citrate 0.1 M | 8.6 ± 0.2 |
| Na2HPO4–sodium citrate 0.1 M | 10.3 ± 0.2 |
| NaH2PO4-Na2HPO4 0.1 M | 5.6 ± 0.4 |
| Tris-HCl 0.1 | M 6.9 ± 0.2 |
| Substrate | 1A | 1B | 1C | 1D |
|---|---|---|---|---|
| R1 | H | Ac | H | H |
| R2 | H | H | Glu | H |
| R3 | H | H | H | CH3 |
| Enzyme activity (μmol/g·h) | 10.3 ± 0.2 | 9.5 ± 0.4 | 15.6 ± 0.3 | 0 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, H.; Xun, E.; Wang, J.; Chen, G.; Cheng, T.; Wang, Z.; Ji, T.; Wang, L. Immobilization of Laccase for Oxidative Coupling of Trans-Resveratrol and Its Derivatives. Int. J. Mol. Sci. 2012, 13, 5998-6008. https://doi.org/10.3390/ijms13055998
Zhang H, Xun E, Wang J, Chen G, Cheng T, Wang Z, Ji T, Wang L. Immobilization of Laccase for Oxidative Coupling of Trans-Resveratrol and Its Derivatives. International Journal of Molecular Sciences. 2012; 13(5):5998-6008. https://doi.org/10.3390/ijms13055998
Chicago/Turabian StyleZhang, Hong, Erna Xun, Jiaxin Wang, Ge Chen, Tiexin Cheng, Zhi Wang, Tengfei Ji, and Lei Wang. 2012. "Immobilization of Laccase for Oxidative Coupling of Trans-Resveratrol and Its Derivatives" International Journal of Molecular Sciences 13, no. 5: 5998-6008. https://doi.org/10.3390/ijms13055998
APA StyleZhang, H., Xun, E., Wang, J., Chen, G., Cheng, T., Wang, Z., Ji, T., & Wang, L. (2012). Immobilization of Laccase for Oxidative Coupling of Trans-Resveratrol and Its Derivatives. International Journal of Molecular Sciences, 13(5), 5998-6008. https://doi.org/10.3390/ijms13055998






