Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Composition in the Essential Oil
2.2. Antimicrobial Activity
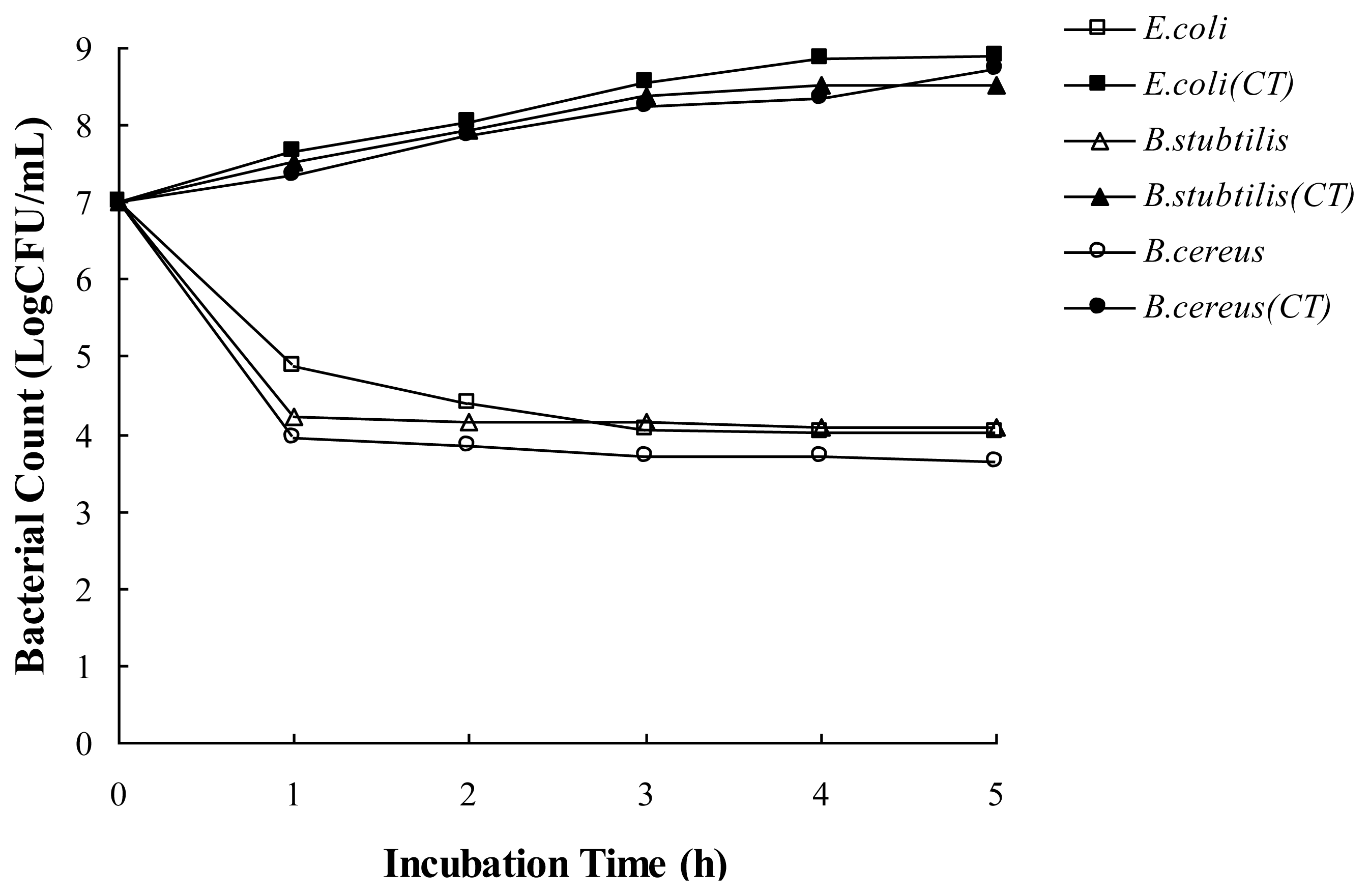
2.3. Antibacterial Activity the Essential Oil in a Food Model Media
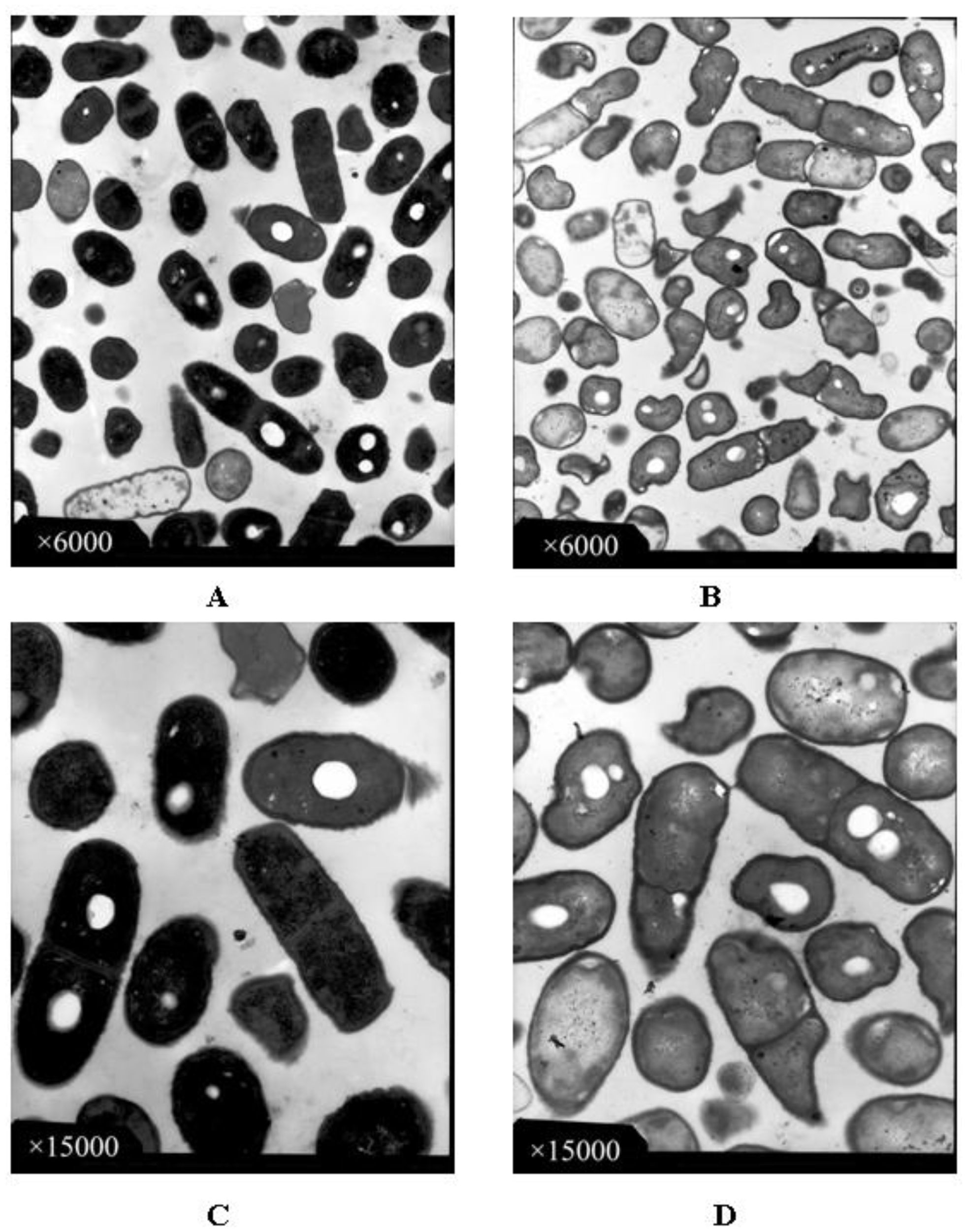
2.4. Observation with Transmission Electron Microscope (TEM)
3. Materials and Methods
3.1. Isolation of the Essential Oil
3.2. Gas Chromatography-Mass Spectroscopy (GC-MS) Analysis
3.3. Test Microorganisms
3.4. Antimicrobial Activity
3.5. Antibacterial Activity of the Essential Oil in a Food Model
3.6. Observation with Transmission Electron Microscope (TEM)
3.7. Statistical Analysis
4. Conclusion
Acknowledgement
References
- Koyasako, A.; Bernhard, R.A. Volatile constituents of essential oils of kumquat. J. Food Sci 1983, 48, 1807–1810. [Google Scholar]
- Ogawa, K.; Kawasaki, A.; Omura, M.; Yoshida, T.; Ikoma, Y.; Yano, M. 3′,5′-Di-C-β-glucopyranosylphloretin, a flavonoid characteristic of the genus Fortunella. Phytochemistry 2001, 57, 737–742. [Google Scholar]
- Kondo, S.; Katayama, R.; Uchino, K. Antioxidant activity in meiwa kumquat as affected by environmental and growing factors. Environ. Exp. Bot 2005, 54, 60–68. [Google Scholar]
- Schirra, M.; Palma, A.; D’Aquino, S.; Anggioni, A.; Minello, E.V.; Melis, M.; Cabras, P. Influence of postharvest hot water treatment on nutritional and functional properties of kumquat (Fortunella japonica Lour, Swingle cv. Ovale) fruits. J. Agric. Food Chem 2008, 56, 455–460. [Google Scholar]
- Subba, M.S.; Soumithri, T.C.; Suryanarayana Rao, R. Antimicrobial action of Citrus oils. J. Food Sci 1967, 32, 225–227. [Google Scholar]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol 2008, 19, 156–164. [Google Scholar]
- Mkaddem, M.; Bouajila, J.; Ennajar, M.; Lebrihi, A.; Mathieu, F.; Romdhane, M. Chemical composition and antimicrobial and antioxidant activities of Mentha (longifolia L. J. Food Sci 2009, 74, 358–363. [Google Scholar]
- Zeng, W.C.; Zhu, R.X.; Jia, L.R.; Gao, H.; Zheng, Y.; Sun, Q. Chemical composition, antimicrobial and antioxidant activities of essential oil from Gnaphlium affine. Food Chem. Toxicol 2011, 49, 1322–1328. [Google Scholar]
- Salleh, W.M.N.H.W.; Ahmad, F.; Yen, K.H.; Sirat, H.M. Chemical compositions, antioxidant and antimicrobial activities of essential oils of Piper caninum Blume. Int. J. Mol. Sci 2011, 12, 7720–7731. [Google Scholar]
- Moufida, S.; Marzouk, B. Biochemical characterization of blood orange, sweet orange, lemon, bergamot and bitter orange. Phytochemistry 2003, 62, 1283–1289. [Google Scholar]
- Choi, H.S. Characteristic odor components of kumquat (Fortunella japonica Swingle) peel oil. J. Agric. Food Chem 2005, 53, 1642–1647. [Google Scholar]
- Cosentino, S.; Tuberoso, C.I.G.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas, F. In-vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Appl. Microbiol 1999, 29, 130–135. [Google Scholar]
- Schelz, Z.; Molnar, J.; Hohmann, J. Antimicrobial and antiplasmid activities of essential oils. Fitoterapia 2006, 77, 279–285. [Google Scholar]
- Fisher, K.; Phillips, C. The effect of lemon, orange and bergamot essential oils and their components on the survival of Campylobacter jejuni, Escherichia coli O157, Listeria monocytogenes, Bacillus cereus and Staphylococcus aureus in vitro and in food systems. J. Appl. Microbiol 2006, 101, 1232–1240. [Google Scholar]
- Matasyoh, J.C.; Maiyo, Z.C.; Ngure, R.M.; Chepkorir, R. Chemical composition and antimicrobial activity of the essential oil of Coriandrum sativum. Food Chem 2009, 113, 526–529. [Google Scholar]
- Liolios, C.C.; Gortzi, O.; Lalas, S.; Tsaknis, J.; Chinou, I. Liposomal incorporation of carvacrol and thymol isolated from the essential oil of Origanum dictamnus L. and in vitro antimicrobial activity. Food Chem 2009, 112, 77–83. [Google Scholar]
- Sikkema, J.; de Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev 1995, 59, 201–222. [Google Scholar]
- Matasyoh, J.C.; Kiplimo, J.J.; Karubiu, N.M.; Hailstorks, T.P. Chemical composition and antimicrobial activity of essential oil of Tarchonanthus camphorates. Food Chem 2007, 101, 1183–1187. [Google Scholar]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J. Antifungal activity of lemon (Citrus lemon L.), mandarin (Citrus reticulate L.), grapefruit (Citrus paradise L.) and orange (Citrus sinesis L.) essential oils. Food Control 2008, 19, 1130–1138. [Google Scholar]
- Hussain, A.I.; Anwar, F.; Sherazi, S.T.H.; Przybylski, R. Chemical composition, antioxidant and antimicrobial and activities of basil (Ocimun basilicum) essential oils depends on seasonal variations. Food Chem 2008, 108, 986–995. [Google Scholar]
- Vagionas, K.; Graikou, K.; Ngassapa, O.; Runyoro, D.; Chinou, I. Composition and antimicrobial activity of the essential oils of three Satureja species growing in Tanzania. Food Chem 2007, 103, 319–324. [Google Scholar]
- Oke, F.; Aslim, B.; Ozturk, S.; Altundag, S. Essential oil composition, antimicrobial and antioxidant activities of Satureja cuneifolia Ten. Food Chem 2009, 112, 874–879. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol 2004, 94, 223–253. [Google Scholar]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antifungal effects of Melaleuca alternifolia (tea tree) oil and its components on Candida albicans, Candida glabrata and Saccharomyces cerevisiae. J. Antimicrob. Chemother 2004, 53, 1081–1085. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Quadrupole Mass Spectroscopy; Allured Publishing Corporation: Carol Stream, IL, USA, 2001. [Google Scholar]
- Beverlya, R.L.; Janes, M.E.; Prinyawiwatkula, W.; No, H.K. Edible chitosan films on ready-to-eat roast beef for the control of Listeria monocytogenes. Food Microbiol 2008, 25, 534–537. [Google Scholar]
- Sokmen, A.; Gulluce, M.; Akpulat, H.A.; Daferera, D.; Tepe, B.; Polissiou, M.; Sokmen, M.; Sahin, F. The in vitro antimicrobial and antioxidant activities of the essential oils and methanol extracts of endemic Thymus spathulifolius. Food Control 2004, 15, 627–634. [Google Scholar]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol 2008, 124, 91–97. [Google Scholar]
| NO. | RI a | Compound | Identification b | (%) c |
|---|---|---|---|---|
| 1 | 936 | α-Pinene | MS, RI | 0.34 |
| 2 | 955 | Camphene | MS, RI | 1.43 |
| 3 | 993 | Myrcene | MS, RI | 7.11 |
| 4 | 1021 | p-Cymene | MS, RI | 0.01 |
| 5 | 1035 | Limonene | MS, RI | 74.79 |
| 6 | 1064 | γ-Terpinene | MS, RI | 0.09 |
| 7 | 1072 | 2,7-Dimethyl-1,6-octadione | MS | 0.02 |
| 8 | 1091 | 3,4-Dimethyl styrene | MS | 0.32 |
| 9 | 1097 | Linalool | MS, RI | 0.30 |
| 10 | 1138 | Carveol | MS, RI | 0.13 |
| 11 | 1146 | p-Mentha-2,8-dien-1-ol | MS, RI | 1.00 |
| 12 | 1164 | p-Mentha-1,5-dien-8-ol | MS, RI | 1.65 |
| 13 | 1209 | Octyl acetate | MS, RI | 0.62 |
| 14 | 1245 | Carvone | MS, RI | 1.58 |
| 15 | 1365 | Piperitenone | MS | 0.22 |
| 16 | 1371 | Perillaldehyde | MS | 0.18 |
| 17 | 1376 | α-Copaene | MS, RI | 0.18 |
| 18 | 1381 | cis-Myrtanyl acetate | MS, RI | 0.15 |
| 19 | 1383 | trans-Myrtanyl acetate | MS, RI | 0.62 |
| 20 | 1392 | β-Elemene | MS, RI | 0.29 |
| 21 | 1459 | Perillyl acetate | MS | 0.22 |
| 22 | 1498 | Bicyclogermacrene | MS, RI | 0.08 |
| 23 | 1506 | α-Selinene | MS, RI | 0.70 |
| 24 | 1578 | Spathulenol | MS, RI | 0.22 |
| 25 | 1861 | Isopropyl cinnamate | MS | 0.11 |
| Total | 92.36 |
| Inhibition zone (mm) | Essential oil (μg/mL) | ||||
|---|---|---|---|---|---|
| Microorganisms | Source | Essential oil a | Gentamicin b | MIC c | MBC d |
| Gram negative | |||||
| E. coli | ATCC 25922 | 18.0 ± 1.29 | 26.0 ± 0.10 | 50.0 | 52.5 |
| S. typhimurium | ATCC 14028 | 13.2 ± 0.37 | 23.3 ± 0.47 | 70.0 | 72.5 |
| Gram positive | |||||
| S. aureus | ATCC 25923 | 14.8 ± 0.69 | 27.3 ± 0.47 | 65.0 | 67.5 |
| B. subtilis | ATCC 21216 | 20.2 ± 0.37 | 23.3 ± 0.47 | 47.5 | 50.0 |
| B. cereus | ATCC 10231 | 20.8 ± 0.69 | 22.0 ± 0.40 | 37.5 | 40.0 |
| L. bulgaricus | ATCC 11842 | 18.2 ± 1.07 | 24.7 ± 0.47 | 67.5 | 70.0 |
| B. laterosporus | ATCC 64 | 18.3 ± 0.94 | 20.3 ± 0.45 | 67.5 | 70.0 |
| Fungus | |||||
| C. albicans | ATCC 50013 | 13.7 ± 0.75 | 27.3 ± 0.47 | 70.0 | 72.5 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Y.-W.; Zeng, W.-C.; Xu, P.-Y.; Lan, Y.-J.; Zhu, R.-X.; Zhong, K.; Huang, Y.-N.; Gao, H. Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel. Int. J. Mol. Sci. 2012, 13, 3382-3393. https://doi.org/10.3390/ijms13033382
Wang Y-W, Zeng W-C, Xu P-Y, Lan Y-J, Zhu R-X, Zhong K, Huang Y-N, Gao H. Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel. International Journal of Molecular Sciences. 2012; 13(3):3382-3393. https://doi.org/10.3390/ijms13033382
Chicago/Turabian StyleWang, Yong-Wei, Wei-Cai Zeng, Pei-Yu Xu, Ya-Jia Lan, Rui-Xue Zhu, Kai Zhong, Yi-Na Huang, and Hong Gao. 2012. "Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel" International Journal of Molecular Sciences 13, no. 3: 3382-3393. https://doi.org/10.3390/ijms13033382
APA StyleWang, Y.-W., Zeng, W.-C., Xu, P.-Y., Lan, Y.-J., Zhu, R.-X., Zhong, K., Huang, Y.-N., & Gao, H. (2012). Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel. International Journal of Molecular Sciences, 13(3), 3382-3393. https://doi.org/10.3390/ijms13033382





