First Insights on Organic Cosolvent Effects on FhuA Wildtype and FhuA Δ1-159
Abstract
:1. Introduction
2. Results and Discussion
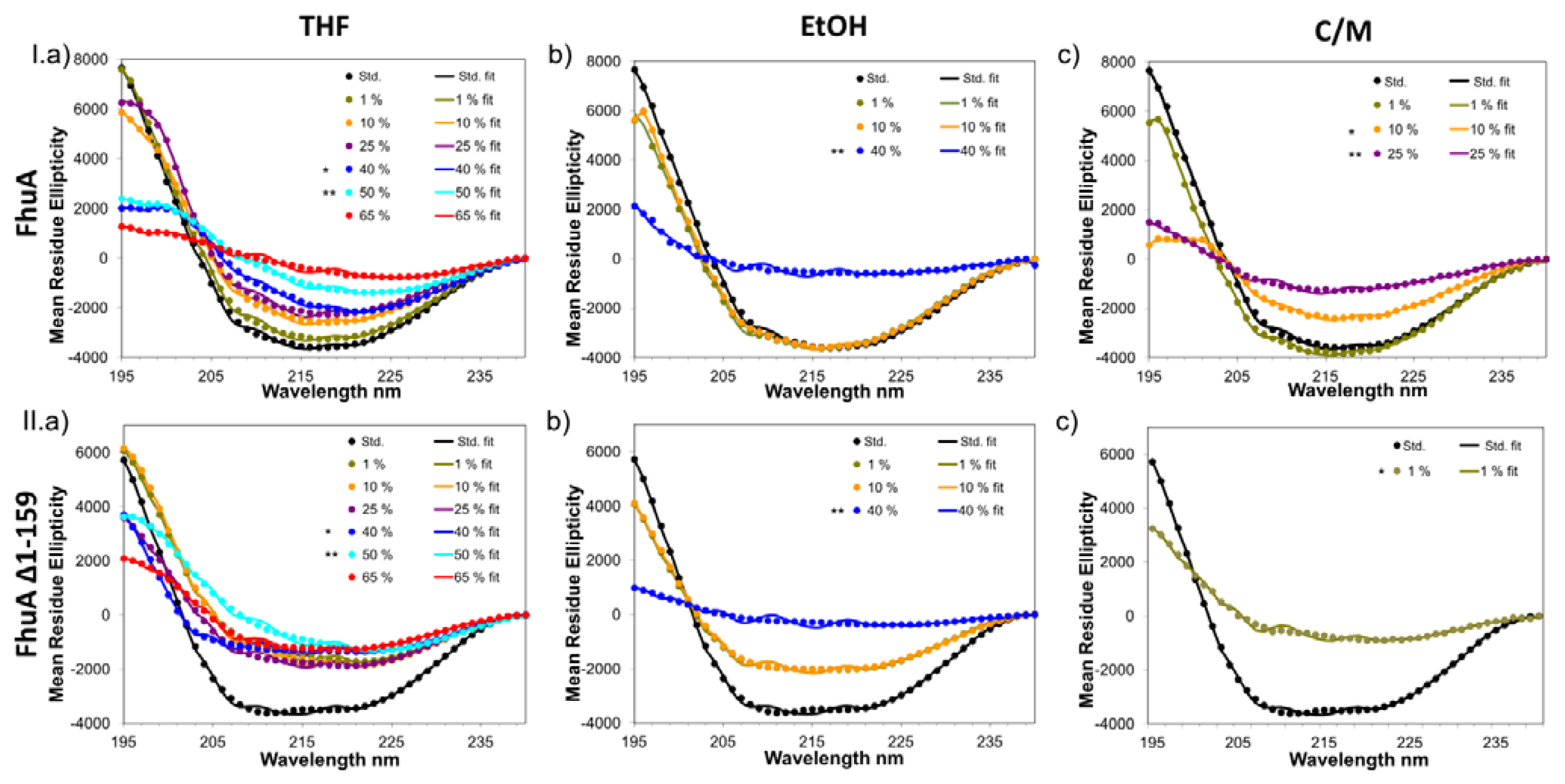
2.1. Results on CD-Measurements
2.2. Secondary Structure Analysis
2.3. Solvent Effects
3. Experimental Section
3.1. Strains
3.2. Expression and Extraction of FhuA and FhuA Δ1-159
3.3. Purification and Concentrating of FhuA and FhuA Δ1-159
3.4. Determination of Protein Purity and Concentration
3.5. Sample Preparation for CD Spectroscopy
3.6. Secondary Structure Determination of FhuA Variants by CD Spectroscopy
4. Conclusions
Supplementary Information
ijms-13-02459-s001.pdfAcknowledgments
Abbreviations
| CD | circular dichroism |
| THF | tetrahydrofuran |
| EtOH | ethanol |
| C/M | chloroform/ methanol |
| FhuA protein | ferric hydroxamate uptake protein component A |
| FhuA Δ1-159 | FhuA deletion variant |
| oPOE | n-octyl-oligo-oxyethylene |
| BCA-assay | bicinchoninic acid assay |
| IPTG | isopropyl-β-d-thiogalactopyranoside |
| Pi | potassium phosphate |
| EDTA | ethylenediaminetetraacetic acid |
| MWCO | molecular weight cut off |
| SDS | sodiumdodecylsulfate |
| α | alpha-helical structure |
| β | beta-sheet structure |
| r | random-coil structure |
| FhuA WT | FhuA wildtype |
| O-atom | oxygen-atom |
| H-bond | hydrogen-bond |
References
- Cramer, W.; Engelman, D.; Von Heijne, G.; Rees, D. Forces involved in the assembly and stabilization of membrane proteins. FASEB J 1992, 6, 3397–3402. [Google Scholar]
- Haltia, T.; Freire, E. Forces and factors that contribute to the structural stability of membrane proteins. Biochim. Biophys. Acta 1995, 1241, 295–322. [Google Scholar]
- Kelly, S.M.; Price, N.C. The use of circular dichroism in the investigation of protein structure and function. Curr. Protein Pept. Sci 2000, 1, 349–384. [Google Scholar]
- Koebnik, R.; Locher, K.P.; Van Gelder, P. Structure and function of bacterial outer membrane proteins: Barrels in a nutshell. Mol. Microbiol 2000, 37, 239–253. [Google Scholar]
- Bonhivers, M.; Desmadril, M.; Moeck, G.S.; Boulanger, P.; Colomer-Pallas, A.; Letellier, L. Stability studies of FhuA, a two-domain outer membrane protein from Escherichia coli. Biochemistry 2001, 40, 2606–2613. [Google Scholar]
- Onaca, O.; Nallani, M.; Ihle, S.; Schenk, A.; Schwaneberg, U. Functionalized nanocompartments (Synthosomes): Limitations and prospective applications in industrial biotechnology. Biotechnol. J 2006, 1, 795–805. [Google Scholar]
- Mohammad, M.M.; Howard, K.R.; Movileanu, L. The redesign of a plugged beta-barrel membrane protein. J. Biol. Chem 2010, 286, 8000–8013. [Google Scholar]
- Ihle, S.; Onaca, O.; Rigler, P.; Hauer, B.; Rodríguez-Ropero, F.; Fioroni, M.; Schwaneberg, U. Nanocompartments with a pH release system based on an engineered OmpF channel protein. Soft Matter 2011, 7, 532–539. [Google Scholar]
- Onaca, O.; Sarkar, P.; Roccatano, D.; Friedrich, T.; Hauer, B.; Grzelakowski, M.; Güven, A.; Fioroni, M.; Schwaneberg, U. Functionalized nanocompartments (synthosomes) with a reduction-triggered release system. Angew. Chem 2008, 47, 7029–7031. [Google Scholar]
- Güven, A.; Dworeck, T.; Fioroni, M.; Schwaneberg, U. Residue K556: A light triggerable gatekeeper to sterically control translocation in FhuA. Adv. Eng. Mater 2011, 13, 324–329. [Google Scholar]
- Muhammad, N.; Dworeck, T.; Fioroni, M.; Schwaneberg, U. Engineering of the E. coli outer membrane protein FhuA to overcome the hydrophobic mismatch in thick polymeric membranes. J. Nanobiotechnol 2011, 9, 1–9. [Google Scholar]
- Ferguson, A.D.; Hofmann, E.; Coulton, J.W.; Diederichs, K.; Welte, W. Siderophore-mediated iron transport: Crystal structure of FhuA with bound lipopolysaccharide. Science 1998, 282, 2215–2220. [Google Scholar]
- Güven, A.; Fioroni, M.; Hauer, B.; Schwaneberg, U. Molecular understanding of sterically controlled compound release through an engineered channel protein (FhuA). J. Nanobiotechnol 2010, 8, 1–9. [Google Scholar]
- Nallani, M.; Onaca, O.; Gera, N.; Hildenbrand, K.; Hoheisel, W.; Schwaneberg, U. A nanophosphor-based method for selective DNA recovery in Synthosomes. Biotechnol. J 2006, 1, 828–834. [Google Scholar]
- Tenne, S.-J.; Dworeck, T.; Güven, A.; Ihle, S.; Fioroni, M.; Schwaneberg, U. Design of Triggered Channel Proteins. Proceedings of PEP Talk Conference, San Diego, CA, USA, 10–14 January 2011.
- Locher, K.P.; Rees, B.; Koebnik, R.; Mitschler, A.; Moulinier, L.; Rosenbusch, J.P.; Moras, D. Transmembrane signaling across the ligand-gated FhuA receptor: Crystal structures of free and ferrichrome-bound states reveal allosteric changes. Cell 1998, 95, 771–778. [Google Scholar]
- Schultz, G.; Ullrich, F.; Heller, K.J.; Braun, V. Export and activity of hybrid Fhua’-’lut receptor proteins and of truncated Fhua’ proteins of the outer membrane of Escherichia coli. Mol. Gen. Genet 1989, 216, 230–238. [Google Scholar]
- Destoumieux-Garzón, D.; Duquesne, S.; Peduzzi, J.; Goulard, C.; Desmadril, M.; Letellier, L.; Rebuffat, S.; Boulanger, P. The iron-siderophore transporter FhuA is the receptor for the antimicrobial peptide microcin J25: Role of the microcin Val11-Pro16 β-hairpin region in the recognition mechanism. Biochem. J 2005, 389, 869–876. [Google Scholar]
- Borisover, M.D.; Zakharychev, D.V.; Solomonov, B.N. Effect of solvent composition on DSC exothermic peak of human serum albumin suspended in pyridine-n-hexane mixtures. J. Therm. Anal. Calorim 1999, 55, 85–92. [Google Scholar]
- Manavalan, P.; Johnson, W.C. Protein secondary structure from circular dichroism spectra. J. Biosci 1985, 8, 141–149. [Google Scholar]
- Pebay-Peyroula, E. Biophysical Analysis of Membrane Proteins: Investigating Structure and Function, 1st ed; Wiley-VCH: Weinheim, Germany, 2008; pp. 55–79. [Google Scholar]
- Wallace, B.A.; Lees, J.G.; Orry, A.J.W.; Lobley, A.; Janes, R.W. Analyses of circular dichroism spectra of membrane proteins. Protein Sci 2003, 12, 875–884. [Google Scholar]
- Boulanger, P.; le Maire, M.; Bonhivers, M.; Dubois, S.; Desmadril, M.; Letellier, L. Purification and structural and functional characterization of FhuA, a transporter of the Escherichia coli outer membrane. Biochemistry 1996, 35, 14216–14224. [Google Scholar]
- Chen, Y.; Wallace, B.A. Secondary solvent effects on the circular dichroism spectra of polypeptides in non-aqueous environments: Influence of polarisation effects on the far ultraviolet spectra of alamethicin. Biophys. Chem 1997, 65, 65–74. [Google Scholar]
- Kabsch, W.; Sander, C. Dictionary of protein secondary structure: Pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 1983, 22, 2577–2637. [Google Scholar]
- Provencher, S. An eigenfunction expansion method for the analysis of exponential decay curves. J. Chem. Phys 1976, 64, 2772–2777. [Google Scholar]
- Dworeck, T.; Petri, A.-K.; Muhammad, N.; Fioroni, M.; Schwaneberg, U. FhuA deletion variant [Delta]1-159 overexpression in inclusion bodies and refolding with Polyethylene-Poly(ethylene glycol) diblock copolymer. Protein Expr. Purif 2011, 77, 75–79. [Google Scholar]
- Pancoska, P.; Blazek, M.; Keiderling, T.A. Relationships between secondary structure fractions for globular proteins. Neural network analyses of crystallographic data sets. Biochemistry 1992, 31, 10250–10257. [Google Scholar]
- Kleinschmidt, J.H.; Tamm, L.K. Secondary and tertiary structure formation of the β-barrel membrane protein OmpA is synchronized and depends on membrane thickness. J. Mol. Biol 2002, 324, 319–330. [Google Scholar]
- Rupley, J.A.; Gratton, E.; Careri, G. Water and globular proteins. Trends Biochem. Sci 1983, 8, 18–22. [Google Scholar]
- Natarajan, K.R. Biocatalysis in organic solvents. J. Chem. Educ 1991, 68, 13–16. [Google Scholar]
- Glew, D.N.; Watts, H. Aqueous nonelectrolyte solutions. Part XII. Enthalpies of mixing of water and deuterium-oxide with tetrahydrofuran. Can. J. Chemi.-Revue Can. Chim 1973, 51, 1933–1940. [Google Scholar]
- Migron, Y.; Marcus, Y. Polarity and hydrogen-bonding ability of some binary aqueous organic mixtures. J. Chem. Soc.-Faraday Trans 1991, 87, 1339–1343. [Google Scholar]
- Laaksonen, A.; Kusalik, P.G.; Svishchev, I.M. Three-dimensional structure in water-methanol mixtures. J. Phys. Chem. A 1997, 101, 5910–5918. [Google Scholar]
- Killmann, H.; Benz, R.; Braun, V. Conversion of the FhuA transport protein into a diffusion channel through the outer membrane of Escherichia coli. EMBO J 1993, 12, 3007–3016. [Google Scholar]
- Nallani, M.; Benito, S.; Onaca, O.; Lindemann, M.; Winterhalter, M.; Meier, W.; Schwaneberg, U. A nanocompartment system (synthosome) designed for biotechnological applications. J. Biotechnol 2006, 123, 50–59. [Google Scholar]
- Prilipov, A.; Phale, P.S.; Van Gelder, P.; Rosenbusch, J.P.; Koebnik, R. Coupling site-directed mutagenesis with high-level expression: Large scale production of mutant porins from E. coli. FEMS Microbiol. Lett 1998, 163, 65–72. [Google Scholar]
- Leammli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
- Provencher, S.; Glöckner, J. Estimation of globular protein secondary structure from circular dichroism. Biochemistry 1981, 20, 33–37. [Google Scholar]
- Deléage, G.; Geourjon, C. An interactive graphic program for calculating the secondary structure content of proteins from circular dichroism. Comput. Appl. Biosci 1993, 9, 197–199. [Google Scholar]
| Secondary structure in %-age | ||||||
|---|---|---|---|---|---|---|
| Sample | FhuA WT | FhuA Δ1-159 | ||||
| Helix | Sheet | Random coil | Helix | Sheet | Random coil | |
| Standard | 4 | 61 | 34 | 3 | 58 | 39 |
| 1 vol% THF | 3 | 64 | 33 | 3 | 64 | 33 |
| 10 vol% THF | 1 | 66 | 33 | 1 | 66 | 33 |
| 25 vol% THF | 0 | 73 | 26 | 0 | 73 | 26 |
| 40 vol% THF | 0 | 60 | 40 | 0 | 60 | 40 |
| 50 vol% THF | 0 | 65 | 35 | 0 | 65 | 35 |
| 65 vol% THF | 0 | 67 | 33 | 0 | 67 | 33 |
| 1 vol% EtOH | 6 | 63 | 31 | 0 | 63 | 37 |
| 10 vol% EtOH | 8 | 73 | 19 | 0 | 63 | 37 |
| 40 vol% EtOH | 0 | 68 | 32 | 0 | 70 | 30 |
| 1 vol% C/M | 8 | 66 | 26 | 0 | 69 | 31 |
| 10 vol% C/M | 3 | 62 | 35 | 0 | 68 | 32 |
| 25 vol% C/M | 0 | 67 | 33 | 0 | 67 | 33 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tenne, S.-J.; Schwaneberg, U. First Insights on Organic Cosolvent Effects on FhuA Wildtype and FhuA Δ1-159. Int. J. Mol. Sci. 2012, 13, 2459-2471. https://doi.org/10.3390/ijms13022459
Tenne S-J, Schwaneberg U. First Insights on Organic Cosolvent Effects on FhuA Wildtype and FhuA Δ1-159. International Journal of Molecular Sciences. 2012; 13(2):2459-2471. https://doi.org/10.3390/ijms13022459
Chicago/Turabian StyleTenne, Stefanie-Joana, and Ulrich Schwaneberg. 2012. "First Insights on Organic Cosolvent Effects on FhuA Wildtype and FhuA Δ1-159" International Journal of Molecular Sciences 13, no. 2: 2459-2471. https://doi.org/10.3390/ijms13022459
APA StyleTenne, S.-J., & Schwaneberg, U. (2012). First Insights on Organic Cosolvent Effects on FhuA Wildtype and FhuA Δ1-159. International Journal of Molecular Sciences, 13(2), 2459-2471. https://doi.org/10.3390/ijms13022459




