Supercritical Carbon Dioxide Extraction of the Oak Silkworm (Antheraea pernyi) Pupal Oil: Process Optimization and Composition Determination
Abstract
:1. Introduction
2. Results and Discussion
2.1. Fitting the Model
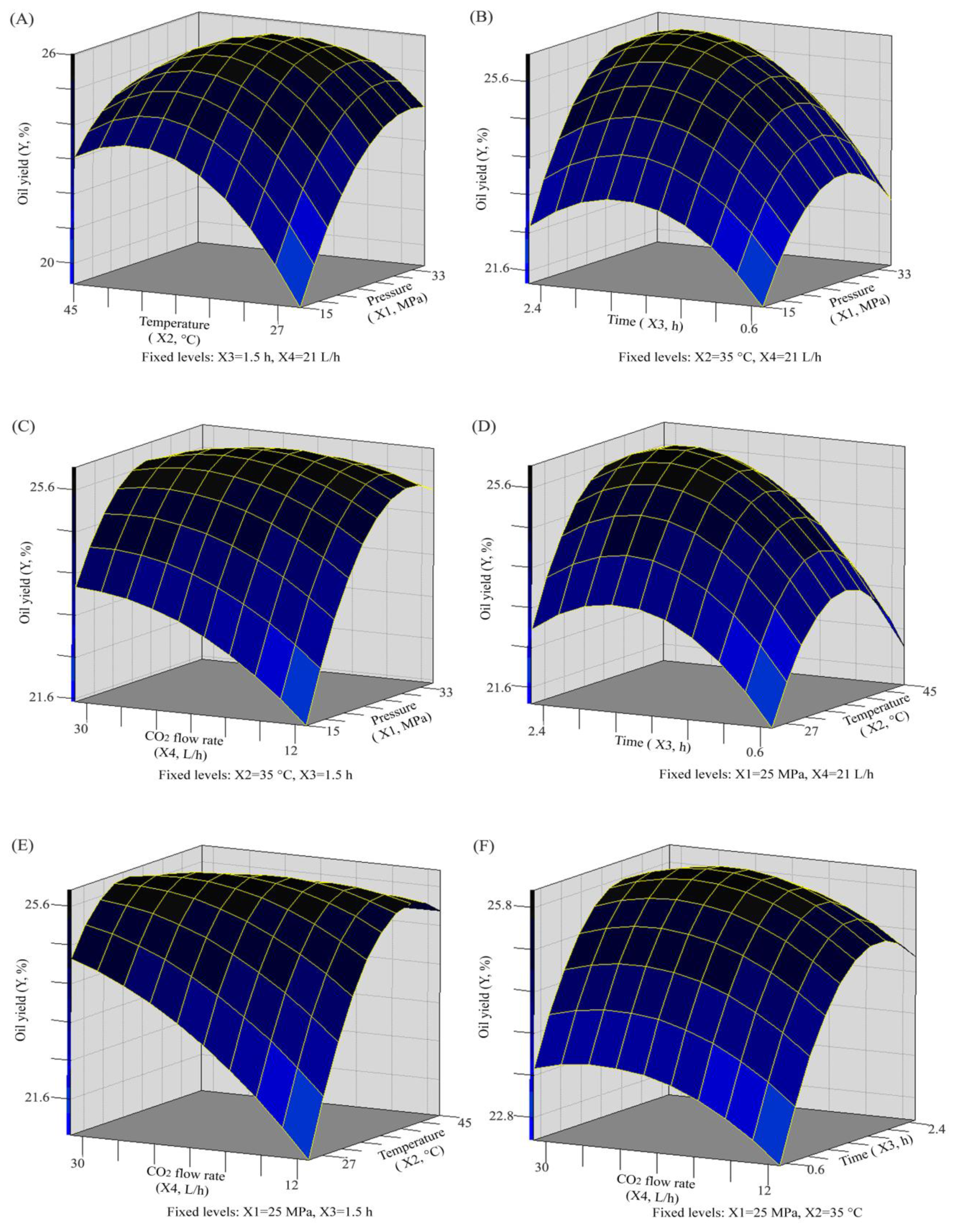
2.2. Analysis of Response Surface
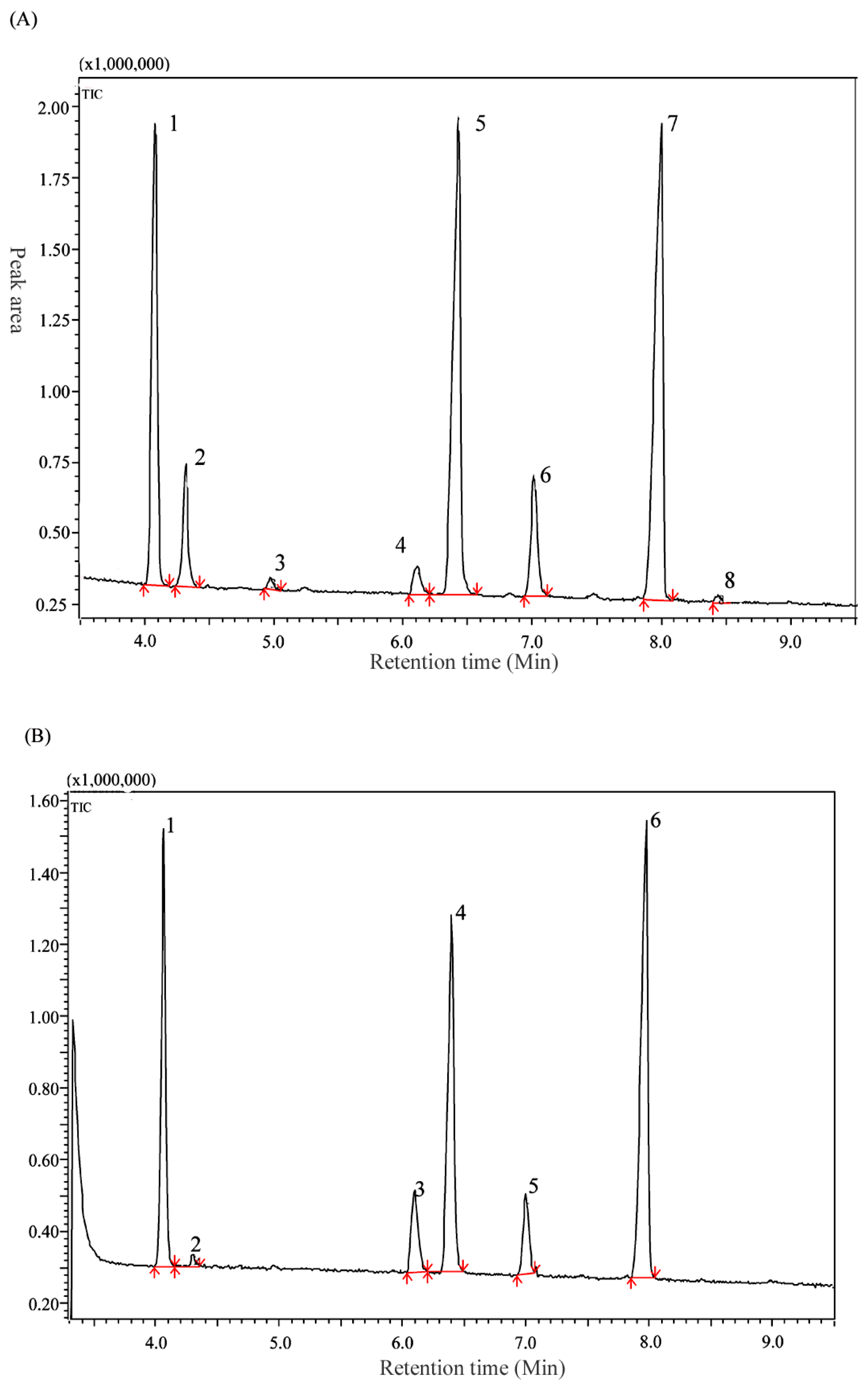
2.3. Determination of Fatty Acid Composition of Extracted Oils
3. Experimental Section
3.1. Materials
3.2. Reagents
3.3. Oil Determination by Soxhlet
3.4. Oil Extraction by Supercritical Carbon Dioxide
3.5. Chromatographic Procedure
3.6. Experimental Design for Response Surface Methodology
4. Conclusions
Acknowledgments
References
- Yokoyama, T. Sericulture. Ann. Rev. Entomol 1963, 8, 287–306. [Google Scholar]
- Singh, K.C.; Suryanarayna, N. Eri pupae: A popular cuisine too. Indian Silk 2003, 41, 57–58. [Google Scholar]
- Zhou, J.; Han, D.X. Proximate, amino acid and mineral composition of pupae of the silkworm Antheraea pernyi in China. J. Food Compos. Anal 2006, 19, 850–853. [Google Scholar]
- Usub, T.; Lertsatitthanakorn, C.; Poomsa-ad, N.; Wiset, L.; Yang, L.F.; Siriamornpun, S. Experimental performance of a solar tunnel dryer for drying silkworm pupae. Biosyst. Eng 2008, 101, 209–216. [Google Scholar]
- Mishra, N.; Hazarika, N.C.; Narain, K.; Mahanta, J. Nutritive value of non-mulberry and mulberry silkworm pupae and consumption pattern in Assam, India. Nutr. Res 2003, 23, 1303–1311. [Google Scholar]
- Wei, Z.J.; Liao, A.M.; Zhang, H.X.; Liu, J.; Jiang, S.T. Optimization of supercritical carbon dioxide extraction of silkworm pupal oil applying the response surface methodology. Bioresour. Technol 2009, 100, 4214–4219. [Google Scholar]
- Zhou, J.; Han, D. Safety evaluation of protein of silkworm (Antheraea pernyi) pupae. Food Chem. Toxicol 2006, 44, 1123–1130. [Google Scholar]
- Herrero, M.; Mendiola, J.A.; Cifuentes, A.; Ibanez, E. Supercritical fluid extraction: Recent advances and applications. J. Chromatogr. A 2009, 1217, 2495–2511. [Google Scholar]
- Wang, Y.; Ying, L.; Sun, D.; Zhang, S.; Zhu, Y.; Xu, P. Supercritical carbon dioxide extraction of bioactive compounds from Ampelopsis grossedentata Stems: Process optimization and antioxidant activity. Int. J. Mol. Sci 2011, 12, 6856–6870. [Google Scholar]
- Sajfrtová, M.; Ličková, I.; Wimmerová, M.; Sovová, H.; Wimmer, Z. β-Sitosterol: Supercritical carbon dioxide extraction from sea buckthorn (Hippophae rhamnoides L.) seeds. Int. J. Mol. Sci 2010, 11, 1842–1850. [Google Scholar]
- Vidovic, S.; Mujic, I.; Zekovic, Z.; Lepojevic, Z.; Milosevic, S.; Jokic, S. Extraction of fatty acids from Boletus edulis by subcritical and supercritical carbon dioxide. J. Am. Oil Chem. Soc 2011, 88, 1189–1196. [Google Scholar]
- Herrero, M.; Cifuentes, A.; Ibanez, E. Sub-and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae: A review. Food Chem 2006, 98, 136–148. [Google Scholar]
- Krichnavaruk, S.; Shotipruk, A.; Goto, M.; Pavasant, P. Supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis with vegetable oils as co-solvent. Bioresour. Technol 2008, 99, 5556–5560. [Google Scholar]
- Bhattacharjee, P.; Singhal, R.S.; Tiwari, S.R. Supercritical carbon dioxide extraction of cottonseed oil. J. Food Eng 2007, 79, 892–898. [Google Scholar]
- Jokic, S.; Zekovic, Z.; Vidovic, S.; Sudar, R.; Nemet, I.; Bilic, M.; Velic, D. Supercritical CO2 extraction of soybean oil: Process optimisation and triacylglycerol composition. Int. J. Food Sci. Technol 2010, 45, 1939–1946. [Google Scholar]
- Liu, G.; Xu, X.; Hao, Q.; Gao, Y. Supercritical CO2 extraction optimization of pomegranate (Punica granatum L.) seed oil using response surface methodology. LWT Food Sci. Technol 2009, 42, 1491–1495. [Google Scholar]
- Machmudah, S.; Kawahito, Y.; Sasaki, M.; Goto, M. Supercritical CO2 extraction of rosehip seed oil: Fatty acids composition and process optimization. J. Supercrit. Fluid 2007, 41, 421–428. [Google Scholar]
- Mitra, P.; Ramaswamy, H.S.; Chang, K.S. Pumpkin (Cucurbita maxima) seed oil extraction using supercritical carbon dioxide and physicochemical properties of the oil. J. Food Eng 2009, 95, 208–213. [Google Scholar]
- Wang, Y.; Sun, D.; Chen, H.; Qian, L.; Xu, P. Fatty Acid composition and antioxidant activity of tea (Camellia sinensis L.) seed oil extracted by optimized supercritical carbon dioxide. Int. J. Mol. Sci 2011, 12, 7708–7719. [Google Scholar]
- Amin, N.A.S.; Anggoro, D.D. Optimization of direct conversion of methane to liquid fuels over Cu loaded W/ZSM-5 catalyst. Fuel 2004, 83, 487–494. [Google Scholar] [Green Version]
- Xu, X.; Gao, Y.X.; Liu, G.M.; Wang, Q.; Zhao, J. Optimization of supercritical carbon dioxide extraction of sea buckthorn (Hippophae thamnoides L.) oil using response surface methodology. LWT Food Sci. Technol 2008, 41, 1223–1231. [Google Scholar]
- Wei, Z.J.; Zhou, L.C.; Chen, H.; Chen, G.H. Optimization of the fermentation conditions for 1-deoxynojirimycin production by Streptomyces lawendulae applying the response surface methodology. Int. J. Food Eng 2011, 7, 2354. [Google Scholar]
- Liu, S.C.; Yang, F.; Zhang, C.H.; Ji, H.W.; Hong, P.Z.; Deng, C.J. Optimization of process parameters for supercritical carbon dioxide extraction of Passiflora seed oil by response surface methodology. J. Supercrit. Fluid 2009, 48, 9–14. [Google Scholar]
- Zahedi, G.; Azarpour, A. Optimization of supercritical carbon dioxide extraction of Passiflora seed oil. J. Supercrit. Fluid 2011, 58, 40–48. [Google Scholar]
- Zhang, S.; Zu, Y.G.; Fu, Y.J.; Luo, M.; Liu, W.; Li, J.; Efferth, T. Supercritical carbon dioxide extraction of seed oil from yellow horn (Xanthoceras sorbifolia Bunge.) and its anti-oxidant activity. Bioresour. Technol 2010, 101, 2537–2544. [Google Scholar]
- Friedrich, J.P.; Pryde, E.H. Supercritical CO2 extraction of lipid-bearing materials and characterization of the products. J. Am. Oil Chem. Soc 1984, 61, 223–228. [Google Scholar]
- Clifford, T. Fundamentals of Supercritical Fluids; Oxford University Press: New York NY, USA, 1999; pp. 1–65. [Google Scholar]
- Pourmortazavi, S.M.; Hajimirsadeghi, S.S. Supercritical fluid extraction in plant essential and volatile oil analysis. J. Chromatogr. A 2007, 1163, 2–24. [Google Scholar]
- Zhang, Q.A.; Fan, X.H.; Zhang, Z.Q.; Zhang, B.S.; Zhang, Z.Q. Optimization of SC-CO2 extraction of oil from almond pretreated with autoclaving. LWT Food Sci. Technol 2009, 42, 1530–1537. [Google Scholar]
- Shanker, K.S.; Shireesha, K.; Kanjilal, S.; Kumar, S.V.L.N.; Rao, J.V.K.; Prasad, R.B.N. Isolation and characterization of neutral lipids of desilked Eri silkworm pupae grown on castor and tapioca leaves. J. Agric. Food Chem 2006, 54, 3305–3309. [Google Scholar]
- Uauy, R.; Valenzuela, A. Marine oils: The health benefits of n-3 fatty acids. Nutrition 2000, 6, 680–684. [Google Scholar]
- Christie, W.W. The Preparation of Derivatives of Lipids. In Lipid Analysis, 2nd ed; Pergamon Press: Oxford, UK, 1982; pp. 50–100. [Google Scholar]
- Dodds, E.D.; McCoy, M.R.; Rea, L.D.; Kennish, J.M. Gas chromatographic quantification of fatty acid methyl esters: Flame ionization detection vs. electron impact mass spectrometry. Lipids 2005, 40, 419–428. [Google Scholar]
- Ahn, J.; Kwak, H.S. Optimizing cholesterol removal in cream using β-cyclodextrin and response surface methodology. J. Food Sci 1999, 64, 629–632. [Google Scholar]
- SAS Software, Version 9; SAS Institute Inc: Cary, NC, USA, 2002.
| Independent Variables | Symbols | Variable Levels | ||||
|---|---|---|---|---|---|---|
| −2 | −1 | 0 | 1 | 2 | ||
| Pressure (MPa) | X1 | 15 | 20 | 25 | 30 | 35 |
| Temperature (°C) | X2 | 25 | 30 | 35 | 40 | 45 |
| Extraction time (h) | X3 | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 |
| CO2 flow rate (L/h) | X4 | 11 | 16 | 21 | 26 | 31 |
| Coded Variable | Process Variable | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Design Point | X1 | X2 | X3 | X4 | Pressure (MPa) | Temperature (°C) | Time (h) | CO2 Flow Rate (L/h) | Oil Yield (Y, %) |
| 1 | −1 | −1 | −1 | −1 | 20 | 30 | 1.0 | 16 | 22.38 ± 0.20 |
| 2 | −1 | −1 | −1 | 1 | 20 | 30 | 1.0 | 26 | 24.34 ± 0.33 |
| 3 | −1 | −1 | 1 | −1 | 20 | 30 | 2.0 | 16 | 23.73 ± 0.18 |
| 4 | −1 | −1 | 1 | 1 | 20 | 30 | 2.0 | 26 | 24.25 ± 0.45 |
| 5 | −1 | 1 | −1 | −1 | 20 | 40 | 1.0 | 16 | 24.19 ± 0.26 |
| 6 | −1 | 1 | −1 | 1 | 20 | 40 | 1.0 | 26 | 24.00 ± 0.34 |
| 7 | −1 | 1 | 1 | −1 | 20 | 40 | 2.0 | 16 | 24.75 ± 0.15 |
| 8 | −1 | 1 | 1 | 1 | 20 | 40 | 2.0 | 26 | 24.62 ± 0.38 |
| 9 | 1 | −1 | −1 | −1 | 30 | 30 | 1.0 | 16 | 24.65 ± 0.22 |
| 10 | 1 | −1 | −1 | 1 | 30 | 30 | 1.0 | 26 | 24.98 ± 0.51 |
| 11 | 1 | −1 | 1 | −1 | 30 | 30 | 2.0 | 16 | 25.09 ± 0.16 |
| 12 | 1 | −1 | 1 | 1 | 30 | 30 | 2.0 | 26 | 26.04 ± 0.32 |
| 13 | 1 | 1 | −1 | −1 | 30 | 40 | 1.0 | 16 | 24.54 ± 0.21 |
| 14 | 1 | 1 | −1 | 1 | 30 | 40 | 1.0 | 26 | 23.33 ± 0.43 |
| 15 | 1 | 1 | 1 | −1 | 30 | 40 | 2.0 | 16 | 25.87 ± 0.18 |
| 16 | 1 | 1 | 1 | 1 | 30 | 40 | 2.0 | 26 | 24.67 ± 0.15 |
| 17 | −2 | 0 | 0 | 0 | 15 | 35 | 1.5 | 21 | 23.01 ± 0.20 |
| 18 | 2 | 0 | 0 | 0 | 35 | 35 | 1.5 | 21 | 24.98 ± 0.32 |
| 19 | 0 | −2 | 0 | 0 | 25 | 25 | 1.5 | 21 | 22.68 ± 0.66 |
| 20 | 0 | 2 | 0 | 0 | 25 | 45 | 1.5 | 21 | 24.37 ± 0.19 |
| 21 | 0 | 0 | −2 | 0 | 25 | 35 | 0.5 | 21 | 23.36 ± 0.48 |
| 22 | 0 | 0 | 2 | 0 | 25 | 35 | 2.5 | 21 | 25.09 ± 0.13 |
| 23 | 0 | 0 | 0 | −2 | 25 | 35 | 1.5 | 11 | 24.58 ± 0.27 |
| 24 | 0 | 0 | 0 | 2 | 25 | 35 | 1.5 | 31 | 25.68 ± 0.37 |
| 25 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 26.00 ± 0.10 |
| 26 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 25.98 ± 0.17 |
| 27 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 25.19 ± 0.15 |
| 28 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 26.09 ± 0.09 |
| 29 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 26.01 ± 0.20 |
| 30 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 26.18 ± 0.12 |
| 31 | 0 | 0 | 0 | 0 | 25 | 35 | 1.5 | 21 | 25.95 ± 0.17 |
| Source | Degrees of Freedom | Sum of Square | Mean Square | F-value | Probability (p) |
|---|---|---|---|---|---|
| Model | 14 | 30.9368 | 2.2098 | 12.7669 | 0.0001 |
| Lack of fit | 10 | 2.1212 | 0.2121 | 1.9636 | 0.2112 |
| Pure error | 6 | 0.6482 | 0.1080 | ||
| Cor total | 30 | 33.7062 |
| Source | Degrees of Freedom | Sum of Square | Mean Square | F-value | Probability (p) |
|---|---|---|---|---|---|
| X1 | 1 | 4.9051 | 4.9051 | 28.3391 | 0.0001 |
| X2 | 1 | 0.6305 | 0.6305 | 3.6427 | 0.07442 |
| X3 | 1 | 4.2252 | 4.2252 | 24.4110 | 0.000148 |
| X4 | 1 | 0.4347 | 0.4347 | 2.51149 | 0.1326 |
| X1 × X1 | 1 | 5.5081 | 5.5081 | 31.8228 | 0.0001 |
| X1 × X2 | 1 | 1.6965 | 1.6965 | 9.8015 | 0.006451 |
| X1 × X3 | 1 | 0.1871 | 0.1871 | 1.0807 | 0.3140 |
| X1 × X4 | 1 | 0.6765 | 0.6765 | 3.9085 | 0.06554 |
| X2 × X2 | 1 | 8.8522 | 8.8522 | 51.1432 | 0.0001 |
| X2 × X3 | 1 | 0.07426 | 0.07426 | 0.4290 | 0.5218 |
| X2 × X4 | 1 | 2.6325 | 2.6325 | 15.2092 | 0.001274 |
| X3 × X3 | 1 | 4.1594 | 4.1594 | 24.0306 | 0.00016 |
| X3 × X4 | 1 | 0.03516 | 0.03516 | 0.2031 | 0.6583 |
| X4 × X4 | 1 | 0.6882 | 0.6882 | 3.9761 | 0.06348 |
| Composition | Relative Content/% | |
|---|---|---|
| Oak Silkworm Pupal Oil | Mulberry Silkworm Pupal Oil | |
| Palmitic acid | 19.92 | 22.77 |
| Palmitoleic acid | 4.77 | 0.60 |
| Heptadecanoic acid | 0.60 | ND |
| Stearic acid | 1.99 | 6.69 |
| Oleic acid | 30.97 | 26.01 |
| Linoleic acid | 6.89 | 5.90 |
| α-linolenic acid | 34.27 | 38.02 |
| 0(z),13(z),16(z)-nonadecatrienoic acid | 0.39 | ND |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pan, W.-J.; Liao, A.-M.; Zhang, J.-G.; Dong, Z.; Wei, Z.-J. Supercritical Carbon Dioxide Extraction of the Oak Silkworm (Antheraea pernyi) Pupal Oil: Process Optimization and Composition Determination. Int. J. Mol. Sci. 2012, 13, 2354-2367. https://doi.org/10.3390/ijms13022354
Pan W-J, Liao A-M, Zhang J-G, Dong Z, Wei Z-J. Supercritical Carbon Dioxide Extraction of the Oak Silkworm (Antheraea pernyi) Pupal Oil: Process Optimization and Composition Determination. International Journal of Molecular Sciences. 2012; 13(2):2354-2367. https://doi.org/10.3390/ijms13022354
Chicago/Turabian StylePan, Wen-Juan, Ai-Mei Liao, Jian-Guo Zhang, Zeng Dong, and Zhao-Jun Wei. 2012. "Supercritical Carbon Dioxide Extraction of the Oak Silkworm (Antheraea pernyi) Pupal Oil: Process Optimization and Composition Determination" International Journal of Molecular Sciences 13, no. 2: 2354-2367. https://doi.org/10.3390/ijms13022354
APA StylePan, W.-J., Liao, A.-M., Zhang, J.-G., Dong, Z., & Wei, Z.-J. (2012). Supercritical Carbon Dioxide Extraction of the Oak Silkworm (Antheraea pernyi) Pupal Oil: Process Optimization and Composition Determination. International Journal of Molecular Sciences, 13(2), 2354-2367. https://doi.org/10.3390/ijms13022354






