Primary, Secondary Metabolites, Photosynthetic Capacity and Antioxidant Activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) Exposed to Potassium Fertilization under Greenhouse Conditions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Phenolics and Flavonoids Profiling
2.2. Ascorbic Acid and Their Profiling
2.3. Total Soluble Sugar, Starch and Total Non-Structural Carbohydrate (TNC) and Their Profiling
2.4. Phenylalanine-Ammonia-Lyase; PAL Activity and Soluble Protein
2.5. Leaf Gas Exchange Properties
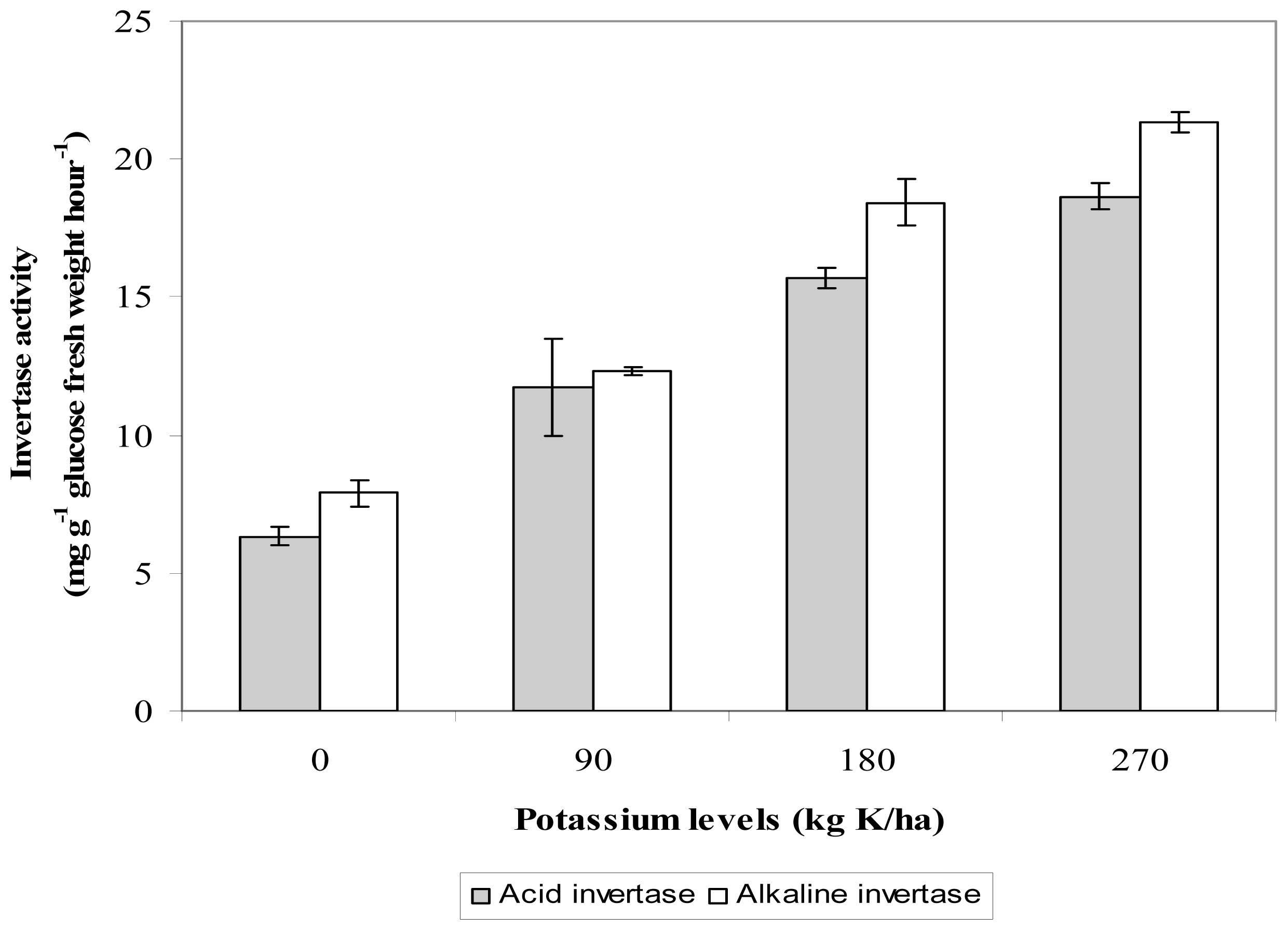
2.6. Leaf Invertase Activity
2.7. Antioxidant Enzyme Activities
3. Experimental
3.1. Experimental Location, Plant Materials and Treatments
3.2. Total Phenolics and Flavonoids Quantification
3.3. Ascorbic Acid Content
3.4. Total Soluble Sugar Determination
3.5. Starch Determination
3.6. Total Non-Structural Carbohydrate (TNC)
3.7. Phenylalanine Ammonia-Lyase (PAL)
3.8. Protein Determination
3.9. Leaf Gas Exchange Measurement
3.10. Invertase Determination
3.11. Antioxidant Enzyme Activity
3.11.1. Preparation of Enzyme Extracts
3.11.2. Assay of Ascorbate Peroxidase (APX) Activity
3.11.3. Assay of Catalase (Cat) Activity
3.11.4. Assay of Superoxide Dismutase (Sod) Activity
3.12. Statistical Analysis
4. Conclusions
Acknowledgements
References
- Robak, J.; Gryglewski, R.J. Flavonoids are scavengers of superoxide anions. Biochem. Pharmacol 1988, 37, 837–841. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci 1997, 2, 152–159. [Google Scholar]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Bolwell, G.P.; Rice-Evans, C.A. Polyphenolic flavanols as scavengers of aqeous phase radicals and as chain-breaking antioxidants. Arch. Biochem. Biophys 1995, 322, 339–346. [Google Scholar]
- Stewart, A.J.; Bozonnet, S.; Mullen, W.; Jenkins, G.I.; Lean, M.E.J.; Crozier, A. Occurrence of flavonols in tomatoes and tomato-based products. J. Agric. Food Chem 2000, 48, 2663–2669. [Google Scholar]
- Yamamoto, Y.; Gaynor, R.B. Therapeutic potential of inhibition of the NF-JB pathway in the treatment of inflammation and cancer. J. Clin. Invest 2001, 107, 135–142. [Google Scholar]
- Tim Cushnie, T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar]
- Shahidi, F.; Wanasundara, P.K. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr 1992, 32, 67–103. [Google Scholar]
- Wei, H.; Tye, L.; Bresnick, E.; Birt, D.F. Inhibitory effect of epigenin, a plant flavonoid, on epidermal ornithine decarboxylase and skin tumor promotion in mice. Cancer Res 1990, 50, 499–502. [Google Scholar]
- Migliaccio, S.; Anderson, J.B. Isoflavones and skeletal health: Are these molecules ready for clinical application. Osteoporos. Int 2003, 14, 361–368. [Google Scholar]
- Karimi, E.; Jaafar, H.Z.; Ahmad, S. Phytochemical analysis and antimicrobial activities of methanolic extracts of leaf, stem and root from different varieties of Labisa pumila Benth. Molecules 2011, 16, 4438–4450. [Google Scholar]
- Fazliana, M.; Wan Nazaimoon, W.M.; Gu, H.F. Labisia pumila extract regulates body weight and adipokines in ovariectomized rats. Maturitas 2009, 62, 91–97. [Google Scholar]
- Ali, Z.; Khan, I.A. Chemical constituents of Labisia pumila (Kacip Fatimah). Planta Med 2009, 75, 40–45. [Google Scholar]
- Avula, B.; Wang, Y.H.; Ali, Z.; Smillie, T.J.; Khan, I.A. Quantitative determination of triterpene saponins and alkenated-phenolics from Labisia pumila by LCUV/ELSD method and confirmation by LC-ESI-TOF. Planta Med 2010, 76, 25–29. [Google Scholar]
- Schneider, J.G.; Tompkins, C.; Blumenthal, R.S.; Mora, S. The metabolic syndrome in women. Cardiol. Rev 2006, 14, 286–291. [Google Scholar]
- Perkins-Veazie, P.; Kalt, W. Postharvest storage of black-berry fruit does not increase antioxidant levels. Acta Hortic 2002, 585, 521–524. [Google Scholar]
- Thomas, R.H.; Woods, F.M.; Dozier, W.A.; Ebel, R.C.; Nesbitt, M.; Wilkins, B.; Himelrick, D.G. Cultivar variation in physicochemical and antioxidant activity of Alabama-grown blackberries. Small Fruits Rev 2005, 4, 57–71. [Google Scholar]
- Watson, R.; Wright, C.J.; McBurney, T.; Taylor, A.J.; Linforth, R.S.T. Influence of harvest date and light integral on the development of strawberry flavour compounds. J. Exp. Bot 2002, 53, 2121–2129. [Google Scholar]
- Reyes-Carmona, J.; Yousef, G.G.; Martınez-Peniche, R.A.; Lila, M.A. Antioxidant capacity of fruit extracts of blackberry (Rubus sp.) produced in different climatic regions. J. Food Sci 2005, 70, 497–503. [Google Scholar]
- Prange, R.K.; Dell, J.R. Preharvest factors affecting post-harvest quality of berry crops. HortScience 1997, 32, 824–830. [Google Scholar]
- Strik, B.C. A review of nitrogen nutrition of Rubus. Proceedings of the IXth International Rubus and Ribes Symposium, Pucon, Chile, 1–7 November 2008; pp. 403–410.
- Cassman, K.G.; Kerby, T.A.; Roberts, B.A.; Bryant, D.C.; Higashi, S.L. Potassium nutrition effects on lint yield and fiber quality of acala cotton. Crop Sci 1990, 30, 672–677. [Google Scholar]
- Pettigrew, M. Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiol. Plant 2008, 133, 670–681. [Google Scholar]
- Cakmak, I. Potassium for better crop production and quality. Plant Soil 2010, 335, 1–2. [Google Scholar]
- Gulick, S.H.; Cassman, K.G.; Grattan, S.R. Exploitation of soil potassium in layered profiles by root systems of cotton and barley. Soil Sci. Soc. Am. J 1989, 53, 146–153. [Google Scholar]
- Hartt, C.E. Effect of potassium deficiency upon translocation of 14C in detached blades of sugarcane. Plant Physiol 1970, 45, 183–187. [Google Scholar]
- Li, R.; Volence, J.J.; Joem, B.C.; Cunningham, S.M. Potassium and nitrogen effects on carbohydrate and protein metabolism in alfalfa roots. J. Plant Nutr 2007, 32, 511–529. [Google Scholar]
- Farley, R.F.; Draycott, P.A. Growth and yield of sugar beet in relation to potassium and sodium supply. J. Sci. Food Agric 1975, 26, 385–392. [Google Scholar]
- Bednarz, C.W.; Oosterhuis, D.M. Physiological changes associated with potassium deficiency in cotton. J. Plant Nutr 1999, 22, 303–313. [Google Scholar]
- Huber, S.C. Biochemical basis for effects of K-deficiency on assimilate export rate and accumulation of soluble sugars in soybean leaves. Plant Physiol 1984, 76, 424–430. [Google Scholar]
- Ward, G.M. Potassium in plant metabolism. III. Some carbohydrate changes in the wheat seedling associated with varying rates of potassium supply. Can. J. Plant Sci 1960, 40, 729–735. [Google Scholar]
- Cakmak, I.; Hengeler, C.; Marschner, H. Partitioning of shoot and root dry matter and carbohydrates in bean plants suffering from phosphorus, potassium and magnesium deficiency. J. Exp. Bot 1994, 45, 1245–1250. [Google Scholar]
- Koch, K.; Mengel, K. The influence of the level of potassium supply to young tobacco plants (Nicotiana tabacum L.) on short-term uptake and utilization of nitrate nitrogen (15N). J. Sci. Food Agric 1974, 25, 465–471. [Google Scholar]
- Helal, H.; Mengel, K. Nitrogen metabolism of young barley plants as affected by NaCl-salinity and potassium. Plant Soil 1979, 51, 457–462. [Google Scholar]
- Liaqat, A.; Beatrix, W.A.; Anna, K.R.; Birgitta, S.; Tim, N.; Marie, E.O. Effects of nutrition strategy on the levels of nutrients and bioactive compounds in blackberries. Eur. Food Res. Technol 2012, 234, 33–44. [Google Scholar]
- Lehman, R.H.; Rice, E.L. Effects of deficiencies of nitrogen, potassium and sulfur on chlorogenic acid and scopolin in sunflower. Am. Midl. Nat. 1972, 87, 71–80. [Google Scholar]
- Troufflard, S.; Mullen, W.; Larson, T.R.; Graham, I.A.; Crozier, A.; Amtmann, A.; Armengaurd, P. Potassium deficiency induces the biosynthesis of oxylipins and glucosinolates in Arabidopsis thaliana. BMC Plant Biol 2010, 10, 172–182. [Google Scholar]
- Lubbe, A.; Choi, Y.H.; Vreeburg, P.; Verpoorte, R. Effects of fertilizers on galanthamine and metabolites profiles in Narcissus Bulbs by 1H-NMR. J. Agric. Food Chem 2011, 59, 3155–3161. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. The relationship of nitrogen and C/N ratio with secondary metabolites levels and antioxidant activities in three varieties of malaysian kacip fatimah (Labisia pumila Blume). Molecules 2011, 16, 5514–5526. [Google Scholar]
- Jaafar, H.Z.E.; Mohamed, H.N.B.; Rahmat, A. Accumulation and partitioning of total phenols in two varieties of Labisia pumila Benth. under manipulation of greenhouse irradiance. Acta Hortic 2008, 797, 387–392. [Google Scholar]
- Armengaud, P.; Sulpice, R.; Miller, A.J.; Stitt, M.; Amtmann, A.; Gibon, Y. Multilevel analysis of primary metabolism provides new insights into the role of potassium nutrition for glycolysis and nitrogen assimilation in Arabidopsis roots. Plant Physiol 2009, 150, 772–785. [Google Scholar]
- Pettigrew, W.T. Potassium deficiency increases specific leaf weights and leaf glucose levels in field-grown cotton. Agron. J 1999, 91, 962–968. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.; Rahmat, A.; Rahman, Z.A. Effects of nitrogen fertilization on synthesis of primary and secondary metabolites in three varieties of Kacip Fatimah (Labisia Pumila Blume). Int. J. Mol. Sci 2011, 12, 5238–5254. [Google Scholar]
- Norhaiza, M.; Maziah, M.; Hakiman, M. Antioxidative properties of leaf extracts of popular Malaysian herb, Labisia pumila. J. Med. Plant Res 2009, 3, 217–223. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A.; Wahab, P.E.M.; Halim, M.R.A. Effect of Different Light Intensities on Total Phenolics and Flavonoids Synthesis and Anti-oxidant Activities in Young Ginger Varieties (Zingiber officinale Roscoe). Int. J. Mol. Sci 2010, 11, 3885–3897. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E. Effect of CO2 Enrichment on Synthesis of Some Primary and Secondary Metabolites in Ginger (Zingiber officinale Roscoe). Int. J. Mol. Sci 2011, 12, 1101–1114. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Synthesis of Phenolics and Flavonoids in Ginger (Zingiber officinale Roscoe) and Their Effects on Photosynthesis Rate. Int. J. Mol. Sci 2010, 11, 4539–4555. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties. Molecules 2010, 15, 7907–7922. [Google Scholar]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Identification and Concentration of Some Flavonoid Components in Malaysian Young Ginger (Zingiber officinale Roscoe) Varieties by a High Performance Liquid Chromatography Method. Molecules 2010, 15, 6231–6243. [Google Scholar]
- Wong, C.C.; Li, H.B.; Cheng, K.W.; Chen, F. A systematic survey of antioxidant activity of 30 Chinese medicinal plants using the ferric reducing antioxidant power assay. Food Chem 2006, 97, 705–711. [Google Scholar]
- Smirnoff, N. Ascorbic acid: Metabolism and functions of a multifacetted molecule. Curr. Opin. Plant Biol 2000, 3, 229–235. [Google Scholar]
- Prasad, M.; Spiers, T.M. The effects of nutrition on the storage quality of kiwifruit. Acta Hortic 1991, 297, 79–85. [Google Scholar]
- De Tullio, M.C.; Arrigoni, O. Hopes, disillusions and more hopes from vitamin C. Cell. Mol. Life Sci 2004, 61, 209–219. [Google Scholar]
- Hancock, R.D.; Viola, R. Biosynthesis and catabolism of l-ascorbic acid in plants. Crit. Rev. Plant Sci 2005, 24, 167–188. [Google Scholar]
- Blaustein, J.; Lips, S.H. Potassium, nitrogen, ammonium/nitrate ratio, and sodium chloride effects on wheat growth. J. Plant Nutr 1991, 14, 756–773. [Google Scholar]
- Peng, Y.; Thomas, S.C. Influence of Non-nitrogenous Soil Amendments on Soil CO2 Efflux and Fine Root Production in an N-Saturated Northern Hardwood Forest. Ecosystems 2010, 13, 1145–115. [Google Scholar]
- Sarkar, D.; Pandey, S.K.; Sharma, S. High K+ does not affect potato (Solanum tuberosum L.) tuber induction, but represses its development in vitro. In Vitro Cell. Dev. Biol. Plant 2010, 46, 569–577. [Google Scholar]
- Liang, X.; Yu, Z. Effect of potassium application stage on photosynthetic characteristics of winter wheat flag leaves and on starch accumulation in wheat grains. Chin. J. App. Ecol 2004, 15, 1349–1355. [Google Scholar]
- Liu, D.H.; Guo, L.P.; Zhu, D.W.; Liu, W.; Jin, H. Characteristics of accumulation and distribution of nitrogen, phosphorus, potassium, calcium and magnesium in Chrysanthemum morifolium. Chin. J. Chin. Mat. Med 2009, 34, 2444–2448. [Google Scholar]
- Nakamura, K.; Ohto, M.; Yoshida, N.; Nakamura, K. Sucrose induced accumulation of amylase occurs concomitant with the accumulation of starch and sporamin in leaf petiole cuttings of sweet potato. Plant Physiol 1991, 96, 902–909. [Google Scholar]
- Murata, T.; Akazawa, T. Stimulative effect of potassium ion on starch synthetase of different plant origins. Plant Cell Physiol 1968, 10, 457–460. [Google Scholar]
- Lasnitzki, A. Potassium and carbohydrate metabolism. Nature 1964, 146, 99–100. [Google Scholar]
- Shui, Y.C.; Feng, X.; Yan, W. Advances in the study of flavonoids in Gingko biloba leaves. J. Med. Plant Res 2009, 3, 1248–1252. [Google Scholar]
- Li, W.; He, P.; Jin, J. Potassium influenced phenylalanine ammonia-lyase, peroxidases and polyphenol oxidases in Fusarium graminearum infected maize (Zea mays L.). Proceedings of the International Plant Nutrition Colloquium XVI, Davis, CA, USA, 1 January 2009.
- Ibrahim, M.H.; Jaafar, H.Z.E.; Rahmat, A.; Abdul Rahman, Z. The relationship between phenolics and flavonoids production with total non structural carbohydrate and photosynthetic rate in Labisia pumila Benth. under high CO2 and nitrogen fertilization. Molecules 2011, 16, 162–174. [Google Scholar]
- Mohammed, S.; Singh, D.; Ahlawat, V.P. Growth, yield and quality of grapes as affected by pruning and basal application of potassium. J. Hort. Sci 1993, 22, 179–192. [Google Scholar]
- Devi, B.S.R.; Kim, Y.J.; Selvi, S.K.; Gayathri, S.; Altanzul, K.; Parvin, S.; Yang, D.U.; Yang, D.C. Influence of potassium nitrate on antioxidant level and secondary metabolite genes under cold stress in Panax ginseng. Russ. J. Plant Physiol 2012, 59, 318–332. [Google Scholar]
- Delahunt, J.; Lindeman, T. Review of the safety of potassium and potassium oxides, including deactivation by introduction into water. J. Chem. Health Saf 2007, 14, 21–32. [Google Scholar]
- Cooper, R.B.; Blaser, R.E.; Brown, R.H. Potassium nutrition effects on net photosynthesis and morphology of alfalfa. Soil Sci 1967, 31, 231–235. [Google Scholar]
- Peoples, T.R.; Koch, D.W. Role of potassium in carbon dioxide assimilation in Medicago sativa L. Plant. Physiol 1979, 63, 878–881. [Google Scholar]
- Peaslee, D.E.; Moss, D.N. Stomatal conductivities in K deficient leaves of maize (Zea mays L.). Crop Sci 1968, 8, 427–430. [Google Scholar]
- Ozbun, J.L.; Volk, R.J.; Jackson, W.A. Effect of potassium deficiency on photosynthesis, respiration and the utilization of photosynthetic reductant by immature bean leaves. Crop Sci 1965, 5, 69–75. [Google Scholar]
- Lam, H.M.; Coschigano, K.T.; Oliveira, I.C.; Melo-Oliveira, R.; Coruzzi, G.M. The molecular genetics of nitrogen assimilation into amino acids in higher plants. Ann. Rev. Plant Physiol. Plant Mol. Biol 1996, 47, 569–593. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z. Enhancement of leaf gas exchange and primary metabolites under carbon dioxide enrichment upregulates the production of secondary metabolites in Labisia pumila seedlings. Molecules 2011, 16, 3761–3777. [Google Scholar]
- Benlloch-Gonzalezm, M.; Javier, R.; Simona, R.; Fran, H.; Jose, M.F.; Manuel, B. K+ starvation inhibits water-stress-induced stomatal closure via ethylene synthesis in sunflower plants. J. Plant Physiol 2008, 165, 623–630. [Google Scholar]
- Oddo, E.; Inzerillo, S.; la Bella, F.; Grisafi, F.; Salleo, S.; Nardini, A. Short-term effects of potassium fertilization on the hydraulic conductance of Laurus nobilis L. Tree Physiol 2011, 31, 131–138. [Google Scholar]
- Basile, B.; Reidel, E.J.; Weinbaum, S.A.; DeJong, T.M. Leaf potassium concentration, CO2 exchange and light interception in almond trees (Prunus dulcis (Mill) D.A. Webb). Sci. Hortic 2003, 98, 185–194. [Google Scholar]
- Lei, M.; Yan, S. Effects of potassium fertilizer on physiological and biochemical index of Stevia rebaudiana Bertoni. Energy Procedia 2011, 5, 581–586. [Google Scholar]
- Bhandal, I.S.; Malik, C.P. Potassium estimation uptake and its role in the physiology and metabolism of flowering plants. Int. Rev. Cytol 1988, 110, 205–254. [Google Scholar]
- Thomas, T.C.; Thomas, A.C. The vital role of potassium in the osmotic mechanism of stomata aperture modulation and its link with potassium deficiency. Plant Signal. Behav 2009, 4, 240–243. [Google Scholar]
- Jones, R.G.; Pollard, A. Proteins Enzymes and Inorganics Ions. In Inorganic Plant Nutrition; Lauchli, A., Bileski, R.L., Eds.; Springer: New York, NY, USA, 1983; pp. 528–562. [Google Scholar]
- Tripathi, B.N.; Bhatt, I.; Dietz, K.J. Peroxiredoxins: A less studied component of hydrogen peroxide detoxification in photosynthetic organism. Protoplasma 2009, 235, 3–15. [Google Scholar]
- Foyer, C.H.; Decourviers, P.; Kenerk, K.J. Protection against oxygen radicals: An important defense mechanism studied in transgenic plants. Plant Cell Environ 1994, 17, 507–523. [Google Scholar]
- Chaitanya, K.; Sundar, D.; Masilamani, S.; Ramachandra, R.A. Variation in heat stress induced antioxidant enzyme activities among three mulberry cultivars. Plant Growth Regul 2002, 36, 175–180. [Google Scholar]
- Cakmak, I.; Horst, W.J. Effects of aluminium and lipid peroxidation, superoxide dismutase, catalase and peroxidase activities in root tips of soybean (Glycine max). Physiol. Plant 1991, 83, 463–468. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z. Reduced photoinhibition under low irradiance enhanced Kacip Fatimah (Labisia pumila Benth) secondary metabolites, phenyl alanine lyase and antioxidant activity. Int. J. Mol. Sci 2012, 13, 5290–5306. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E.; Asmah, R.; Zaharah, A.R. Involvement of nitrogen on flavonoids, glutathione, anthocyanin, ascorbic acid and antioxidant activities of Malaysian Medicinal Plant Labisia pumila Blume (Kacip Fatimah). Int. J. Mol. Sci 2012, 13, 393–408. [Google Scholar]
- Davies, S.H.R.; Masten, S.J. Spectrophotometric method for ascorbic acid using dichlorophenolindophenol: Elimination of the interference due to iron. Anal. Chim. Acta 1991, 248, 225–227. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. Primary, secondary metabolites, H2O2, malondialdehyde and photosynthetic responses of Orthosiphon stimaneus Benth. to different irradiance levels. Molecules 2012, 17, 1159–1176. [Google Scholar]
- Thayumanam, B.; Sidasivam, S. Carbohydrate chemistry. Qual. Plant Foods Hum. Nutr 1984, 34, 253–254. [Google Scholar]
- Tognetti, R.; Johnson, J.D. The effect of elevated atmospheric CO2 concentration and nutrient supply on gas exchange, carbohydrates and foliar phenolics concentration in live oak (Quercus virginiana Mill.) seedlings. Ann. For. Sci 1999, 56, 379–389. [Google Scholar]
- Martinez-Tellez, M.A.; Lafuente, M.T. Effects of high temperature conditioning on ethylene, phenylalanine ammonia lyase, peroxidase and polyphenol oxidase in flavedo of chilled “Fortune” mandarin fruit. J. Plant Physiol 1997, 150, 674–678. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. Carbon dioxide fertilization enhanced antioxidant compounds in Malaysian Kacip Fatimah (Labisia pumila Blume). Molecules 2011, 16, 6068–6081. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E.; Haniff, M.H.; Raffi, M.Y. Changes in growth and photosynthetic patterns of oil palm seedling exposed to short term CO2 enrichment in a closed top chamber. Acta Physiol. Plant 2010, 32, 305–313. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. Impact of Elevated Carbon Dioxide on Primary, Secondary Metabolites and Antioxidant Responses of Eleais guineensis Jacq. (Oil Palm) Seedlings. Molecules 2012, 17, 5195–5211. [Google Scholar]
- Schaffer, A.A.; Sagee, E.E.; Goldschmidt, A.; Goren, R. Invertase and sucrose synthase activity, carbohydrate status and endogenous IAA levels during citrus leaf development. Physiol. Plant 1987, 69, 151–155. [Google Scholar]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplast. Plant Cell Physiol 1981, 22, 867–880. [Google Scholar]
- Aebi, H. Catalase. In Methods of Enzymatic Analysis, 3rd ed; Bergmeyer, H., Verlag, C., Weinheim, A., Eds.; Verlag Chemie: Weinheim, Germany, 1983; pp. 273–277. [Google Scholar]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutase: Occurance in higher plants. Plant Physiol 1977, 59, 309–314. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. Photosynthetic capacity, photochemical efficiency and chlorophyll content of three varieties of Labisia pumila benth. exposed to open field and greenhouse growing conditions. Acta Physiol. Plant 2011, 33, 2179–2185. [Google Scholar]
- Jaafar, H.Z.; Ibrahim, M.H.; Karimi, E. Phenolics and Flavonoids Compounds, Phenylanine Ammonia Lyase and Antioxidant Activity Responses to Elevated CO2 in Labisia pumila (Myrisinaceae). Molecules 2012, 17, 6331–6347. [Google Scholar]
- Jaafar, H.Z.E.; Ibrahim, M.H.; Mohamad Fakri, N.F. Impact of Soil Field Water Capacity on Secondary Metabolites, Phenylalanine Ammonia-lyase (PAL), Maliondialdehyde (MDA) and Photosynthetic Responses of Malaysian Kacip Fatimah (Labisia pumila Benth). Molecules 2012, 17, 7305–7322. [Google Scholar]
- Karimi, E.; Jaafar, H.Z. HPLC and GC-MS Determination of Bioactive Compounds in Microwave Obtained Extracts of Three Varieties of Labisia pumila Benth. Molecules 2011, 16, 6791–6805. [Google Scholar]
- Karimi, E.; Ehsan, O.; Rudi, H.; Jaafar, H.Z.E. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules 2010, 15, 6244–6256. [Google Scholar]
| Plant parts | Potassium levels (Kg K/ha) | Total phenolics (mg g−1 gallic acid dry weight) | Total flavonoid (mg g−1 rutin dry weight) | Ascorbic acid (mg g−1 dry weight) |
|---|---|---|---|---|
| 0 | 1.22 ± 0.44 d | 0.68 ± 0.05 d | 0.030 ± 0.006 d | |
| Leaves | 90 | 1.40 ± 0.01 c | 0.75 ± 0.23 c | 0.047 ± 0.001 c |
| 180 | 1.65 ± 0.15 b | 0.82 ± 0.12 b | 0.062 ± 0.003 b | |
| 270 | 1.82 ± 0.61 a | 0.94 ± 0.25 a | 0.083 ± 0.003 a | |
| 0 | 0.62 ± 0.23 d | 0.35 ± 0.15 c | 0.018 ± 0.004 d | |
| Stems | 90 | 0.70 ± 0.44 c | 0.42 ± 0.32 b | 0.046 ± 0.005 c |
| 180 | 0.85 ± 0.24 b | 0.59 ± 0.08 a | 0.057 ± 0.008 b | |
| 270 | 0.96 ± 0.34 a | 0.61 ± 0.14 a | 0.074 ± 0.004 a | |
| 0 | 0.40 ± 0.11 d | 0.18 ± 0.05 c | 0.010 ± 0.003 d | |
| Roots | 90 | 0.42 ± 0.12 c | 0.21 ± 0.31 b | 0.040 ± 0.002 c |
| 180 | 0.58 ± 0.05 b | 0.25 ± 0.13 b | 0.051 ± 0.001 b | |
| 270 | 0.62 ± 0.21 a | 0.41 ± 0.03 a | 0.073 ± 0.001 a |
| Parameters | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Phenolics | 1.000 | |||||||||||||||||
| 2. Flavonoids | 0.987 * | 1.000 | ||||||||||||||||
| 3. Ascorbic acid | 0.902 * | 0.765 * | 1.000 | |||||||||||||||
| 4. Soluble sugar | 0.879 * | 0.887 * | 0.809 * | 1.000 | ||||||||||||||
| 5. Starch | 0.884 * | 0.873 * | 0.805 * | 0.879 * | 1.000 | |||||||||||||
| 6. TNC | 0.985 * | 0.812 * | 0.765 * | 0.806* | 0.777 * | 1.000 | ||||||||||||
| 7. Protein | 0.678 * | 0.879 * | 0.768 * | 0.777 | 0.677 | 0.776 * | 1.000 | |||||||||||
| 8. PAL | 0.945 * | 0.987 * | 0.915 * | 0.954* | 0.876 * | 0.778 * | 0.877 * | 1.000 | ||||||||||
| 9. Photosynthesis | 0.998 * | 0.897 * | 0.789 * | 0.776 * | 0.778 * | 0.887 * | 0.556 | 0.778 * | 1.000 | |||||||||
| 10. gs | 0.956 * | 0.804* | 0.667 | 0.765 * | 0.667 | 0.885 * | 0.446 | 0.667 * | 0.987 * | 1.000 | ||||||||
| 11. Ci | 0.887 * | 0.702 * | 0.556 | 0.644 | 0.554 | 0.345 | 0.334 | 0.443 | 0.908 * | 0.667 * | 1.000 | |||||||
| 12. Rd | −0.567 | −0.445 | −0.567 | −0.563 | 0.445 | −0.065 | −0.084 | 0.443 | 0.098 | 0.098 | 0.098 | 1.000 | ||||||
| 13. APY | 0.786 * | 0.654 | 0.576 | 0.445 | 0.334 | 0.087 | 0.087 | 0.223 | 0.765 * | 0.657 * | 0.045 | 0.078 | 1.000 | |||||
| 14. Acid invertase | 0.987 * | 0.879 * | 0.876 * | 0.778 * | 0.767 * | 0.764 * | 0.786 * | 0.889 * | 0.879 * | 0.765 * | 0.776 * | 0.067 | 0.010 | 1.000 | ||||
| 15. Alkaline invertase | 0.912 * | 0.889 * | 0.776 * | 0.887 * | 0.879 * | 0.776 * | 0.665 * | 0.897 * | 0.986 * | 0.778 * | 0.786 * | 0.086 | 0.010 | 0.987 * | 1.000 | |||
| 16. APX | −0.786 * | −0.879 * | 0.067 | 0.045 | 0.213 | 0.012 | 0.221 | 0.312 | 0.124 | 0.432 | 0.341 | 0.311 | 0.080 | 0.123 | 0.034 | 1.000 | ||
| 17. CAT | −0.675 * | −0.897 * | 0.456 | 0.043 | 0.321 | 0.123 | 0.123 | 0.034 | 0.012 | 0.123 | 0.113 | 0.123 | 0.070 | 0.234 | 0.076 | 0.021 | 1.000 | |
| 18. SOD | −0.876 * | −0.961 * | 0.452 | 0.021 | 0.111 | 0.211 | 0.321 | 0.112 | 0.345 | 0.111 | 0.213 | 0.051 | 0.330 | 0.211 | 0.765 | 0.065 | 0.231 | 1.000 |
| Plant parts | Potassium levels (Kg K/ha) | Soluble sugar (mg g−1 sucrose dry weight) | Starch (mg g−1 glucose dry weight) | Total non structural carbohydrate (mg g−1 dry weight) |
|---|---|---|---|---|
| 0 | 48.23 ± 0.23 c | 87.11 ± 0.24 d | 133.42 ± 11.34 d | |
| Leaves | 90 | 42.11 ± 2.46 d | 91.17 ± 3.12 c | 140.21 ± 12.31 c |
| 180 | 52.31 ± 0.90 b | 96.11 ± 1.66b | 148.11 ± 10.23 b | |
| 270 | 54.13 ± 3.42 a | 99.04 ± 1.13 a | 152.43 ± 10.14 a | |
| 0 | 55.13 ± 3.25 d | 78.34 ± 0.48 c | 117.31 ± 10.01 d | |
| Stems | 90 | 42.22 ± 2.16 c | 80.12 ± 2.14 b | 121.40 ± 8.35 c |
| 180 | 43.11 ± 0.36 b | 80.92 ± 2.21 b | 122.41 ± 7.19 b | |
| 270 | 47.81 ± 2.47 a | 86.01 ± 1.43 a | 134.61 ± 9.02 a | |
| 0 | 29.61 ± 2.12 d | 52.21 ± 1.13 c | 82.21 ± 6.89 d | |
| Roots | 90 | 34.21 ± 3.13 c | 57.21 ± 1.23 d | 93.21 ± 4.16 c |
| 180 | 35.42 ± 0.73 b | 60.17 ± 2.07 b | 96.27 ± 12.14 b | |
| 270 | 37.22 ± 1.07 a | 77.48 ± 4.13 a | 115.18 ± 7.35 a |
| Potassium levels (Kg K/ha) | PAL activity (nM transcinnamic mg−1 protein−1 hour−1) | Soluble Protein (mg g−1 fresh weight) |
|---|---|---|
| 0 | 8.24 ± 0.23 d | 1.17 ± 0.34 d |
| 90 | 19.28 ± 0.05 c | 3.28 ± 0.08 c |
| 180 | 25.61 ± 0.34 b | 6.97 ± 0.11 b |
| 270 | 37.28 ± 2.11 a | 12.38 ± 0.21 a |
| Parameters | 0 kg K/ha | 90 kg K/ha | 180 kg K/ha | 270 kg K/ha |
|---|---|---|---|---|
| Net photosynthesis, A (μmol m−2 s−1) | 3.45 ± 0.24 d | 6.75 ± 0.31 c | 8.45 ± 0.04 b | 12.72 ± 0.12 a |
| Stomatal conductance, gs (mmol m−2 s−1) | 12.32 ± 0.12 d | 19.21 ± 0.25 c | 28.72 ± 0.31 b | 40.25 ± 0.14a |
| Intercellular CO2, Ci (μmol m−2 s−1) | 250 ± 10.32 d | 292 ± 23.34 c | 312 ± 25.11 c | 325 ± 30.12 a |
| Dark respiration, Rd (μmol m−2 s−1) | 8.24 ± 3.14 a | 6.23 ± 3.93 b | 4.11 ± 1.07 c | 2.17 ± 0.91 d |
| Apparent quantum yield, AQY (μmol m−2 s−1) | 0.032 ± 0.001 d | 0.043 ± 0.005 c | 0.078 ± 0.003 b | 0.097 ± 0.004 a |
| Potassium levels (Kg K/ha) | Ascorbate peroxidase activity (APX; mg protein−1 min−1) | Catalase activity (CAT; μmol mg protein−1 min−1) | Superoxide dismutase activity (SOD; mg protein−1 min−1) |
|---|---|---|---|
| 0 | 10.72 ± 0.21 a | 171.24 ± 2.44 a | 230.45 ± 2.36 a |
| 90 | 8.74 ± 0.76 b | 160.32 ± 1.23 b | 196.72 ± 1.34 b |
| 180 | 6.23 ± 0.45 c | 110.34 ± 0.12 c | 172.51 ± 1.87 c |
| 270 | 3.44 ± 1.32 d | 101.24 ± 0.23 d | 136.24 ± 2.23 d |
| Characteristics | Results |
|---|---|
| pH | 5.8 |
| EC (ds/m) | 0.6 |
| Nitrogen (%) | 1.0% |
| Phosphorous (%) | 0.9% |
| Potassium (%) | 1.6% |
| Sulfur (%) | 0.7% |
| Calcium (%) | 0.8% |
| Magnesium (%) | 0.4% |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibrahim, M.H.; Jaafar, H.Z.E.; Karimi, E.; Ghasemzadeh, A. Primary, Secondary Metabolites, Photosynthetic Capacity and Antioxidant Activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) Exposed to Potassium Fertilization under Greenhouse Conditions. Int. J. Mol. Sci. 2012, 13, 15321-15342. https://doi.org/10.3390/ijms131115321
Ibrahim MH, Jaafar HZE, Karimi E, Ghasemzadeh A. Primary, Secondary Metabolites, Photosynthetic Capacity and Antioxidant Activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) Exposed to Potassium Fertilization under Greenhouse Conditions. International Journal of Molecular Sciences. 2012; 13(11):15321-15342. https://doi.org/10.3390/ijms131115321
Chicago/Turabian StyleIbrahim, Mohd Hafiz, Hawa Z. E. Jaafar, Ehsan Karimi, and Ali Ghasemzadeh. 2012. "Primary, Secondary Metabolites, Photosynthetic Capacity and Antioxidant Activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) Exposed to Potassium Fertilization under Greenhouse Conditions" International Journal of Molecular Sciences 13, no. 11: 15321-15342. https://doi.org/10.3390/ijms131115321
APA StyleIbrahim, M. H., Jaafar, H. Z. E., Karimi, E., & Ghasemzadeh, A. (2012). Primary, Secondary Metabolites, Photosynthetic Capacity and Antioxidant Activity of the Malaysian Herb Kacip Fatimah (Labisia Pumila Benth) Exposed to Potassium Fertilization under Greenhouse Conditions. International Journal of Molecular Sciences, 13(11), 15321-15342. https://doi.org/10.3390/ijms131115321






