Dual Bioactivities of Essential Oil Extracted from the Leaves of Artemisia argyi as an Antimelanogenic versus Antioxidant Agent and Chemical Composition Analysis by GC/MS
Abstract
:1. Introduction
2. Results and Discussion
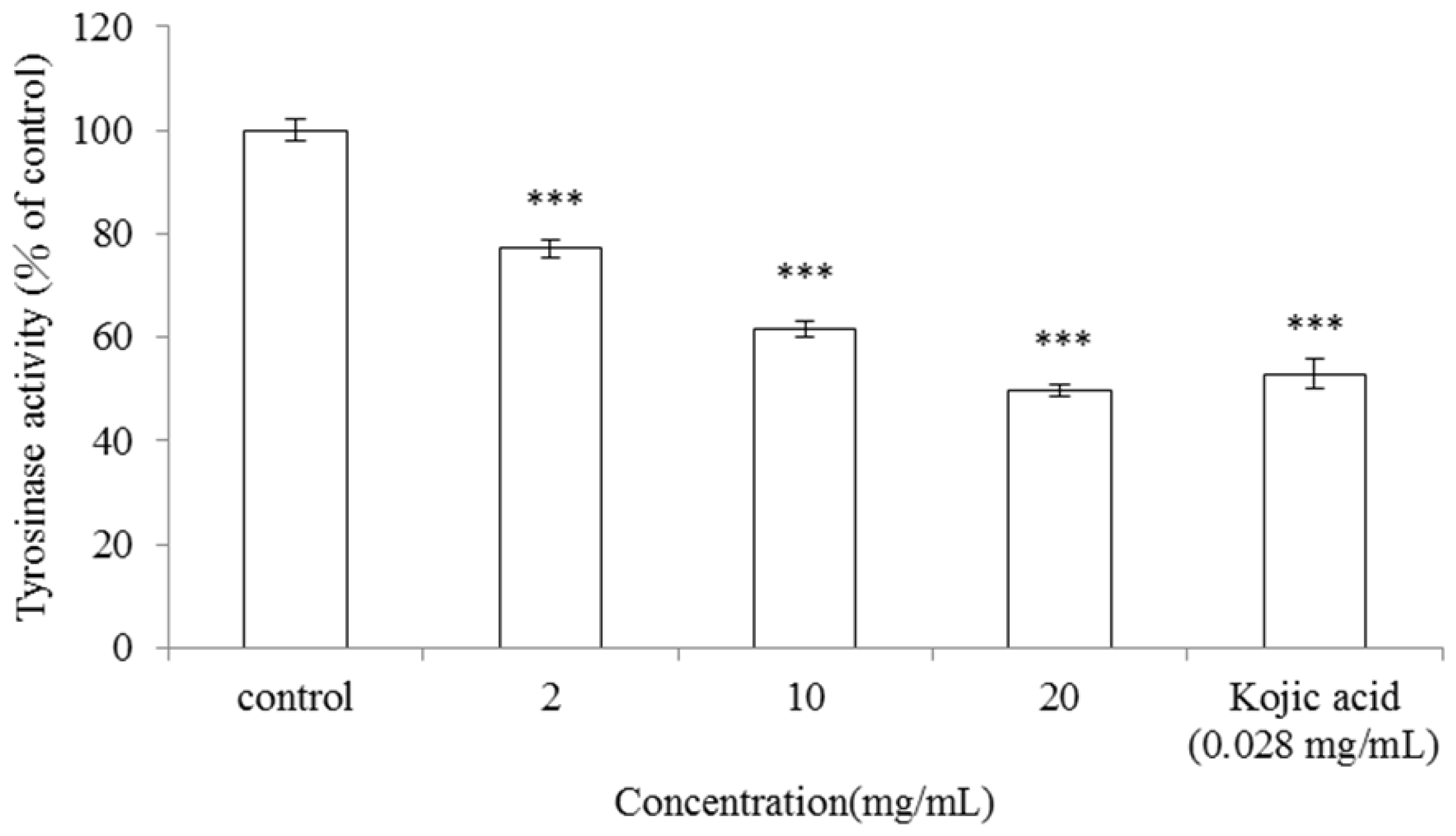
2.1. Inhibitory Effect of A. argyi Essential Oil on Mushroom Tyrosinase Activity
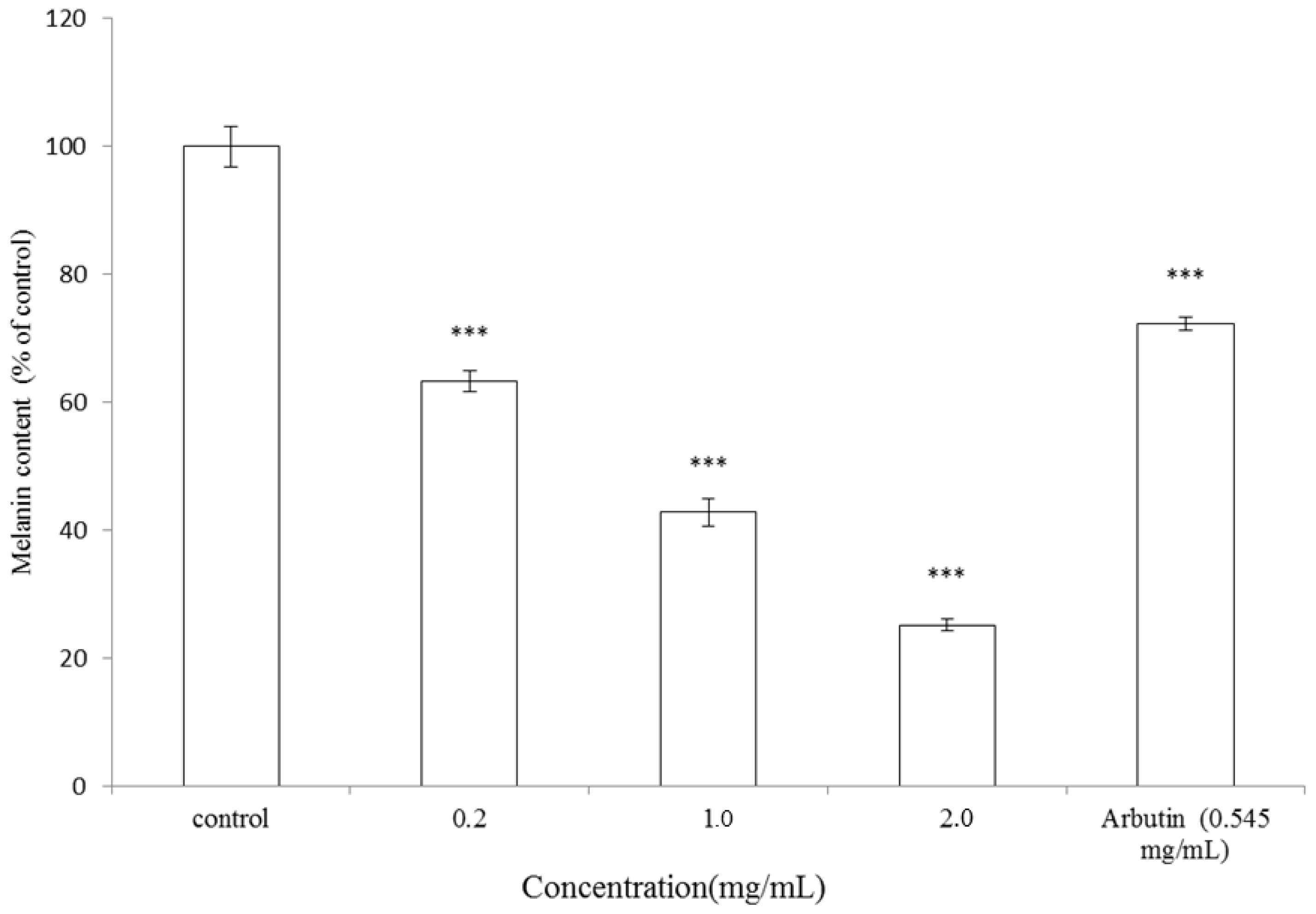
2.2. Effect of A. argyi Essential Oil on Melanogenesis in B16F10 Cells
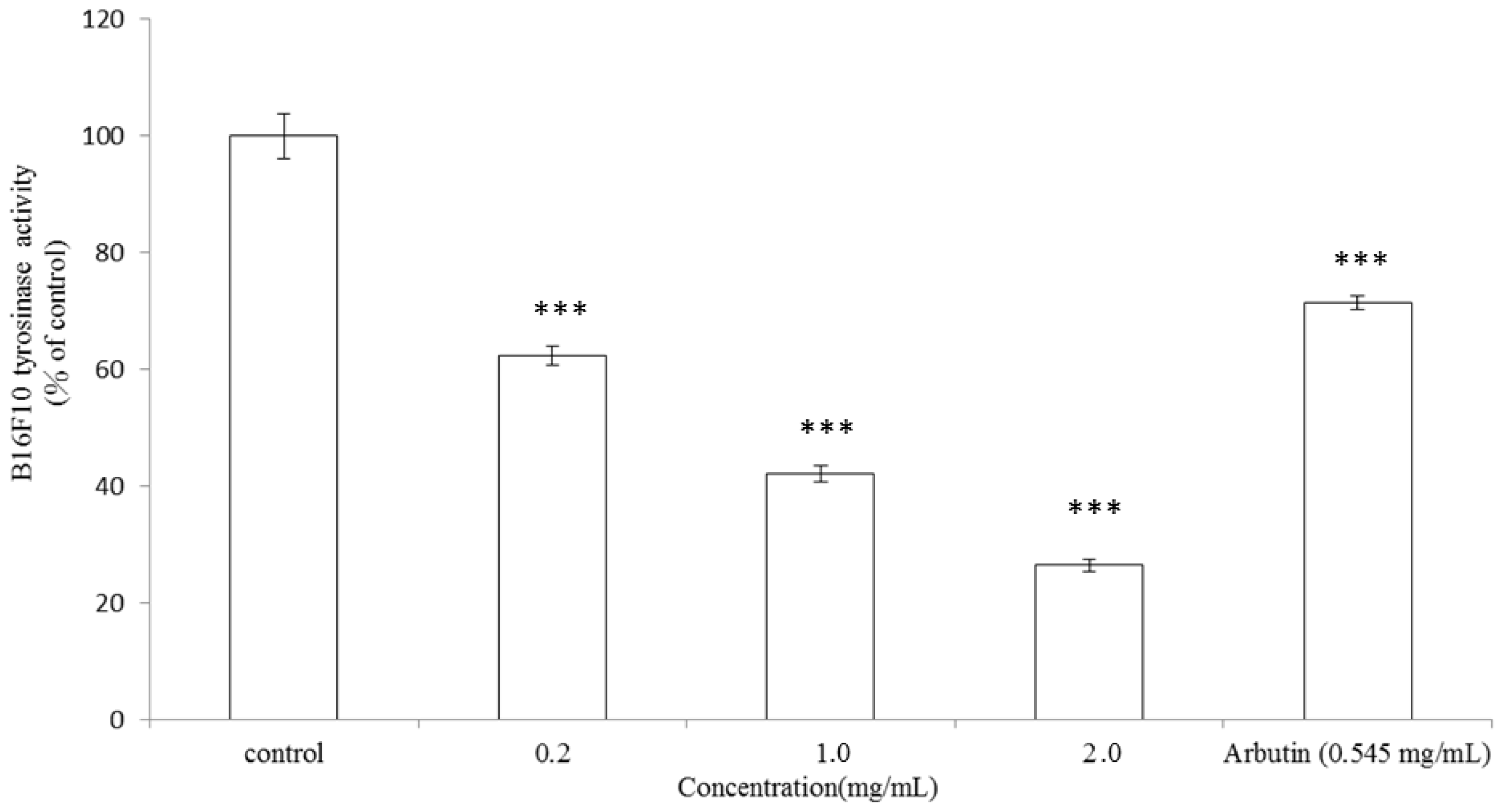
2.3. Inhibitory Effect of A. argyi Essential Oil on Intracellular Tyrosinase Activity in B16F10 Cells
2.4. DPPH Scavenging Capacity Assay
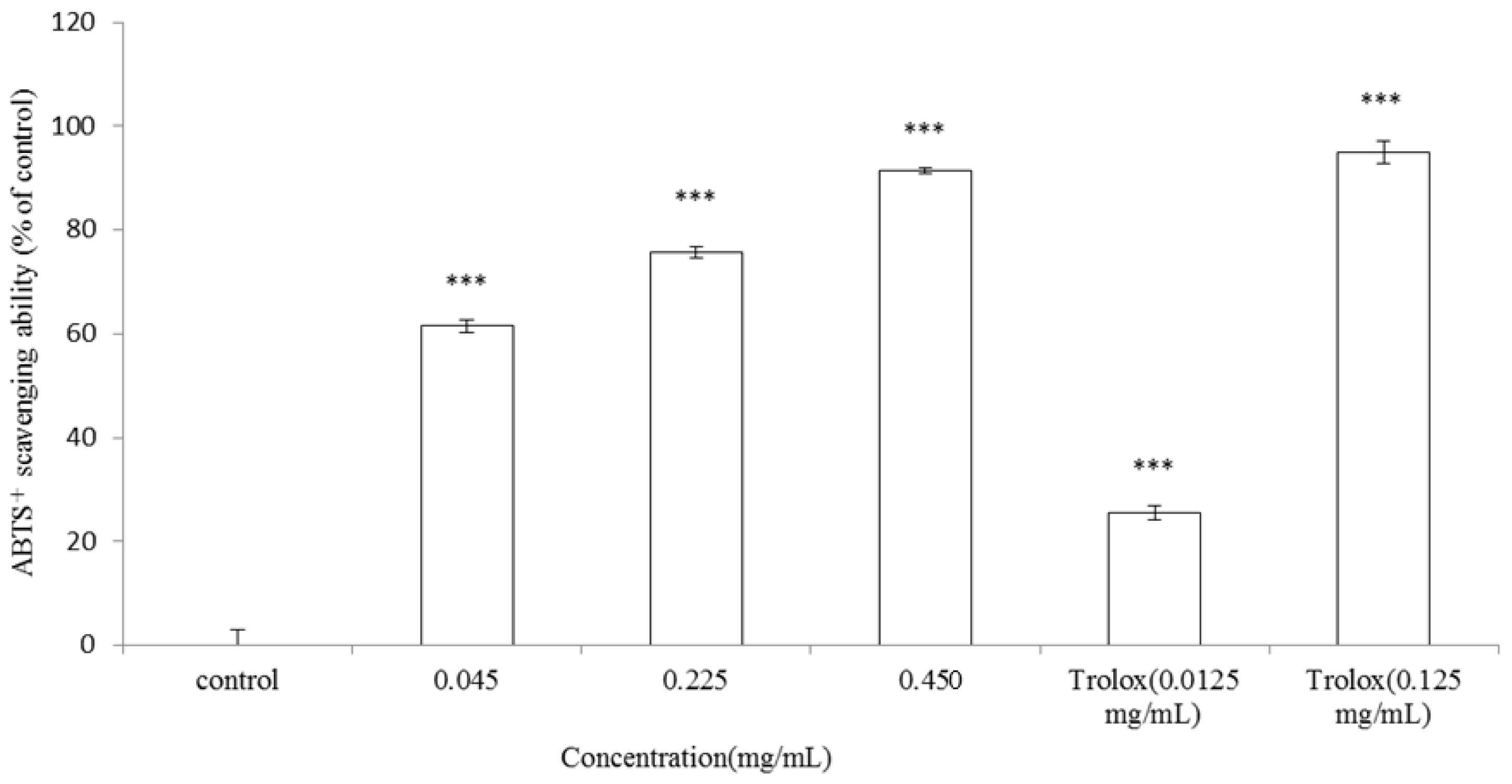
2.5. ABTS Scavenging Ability Assay
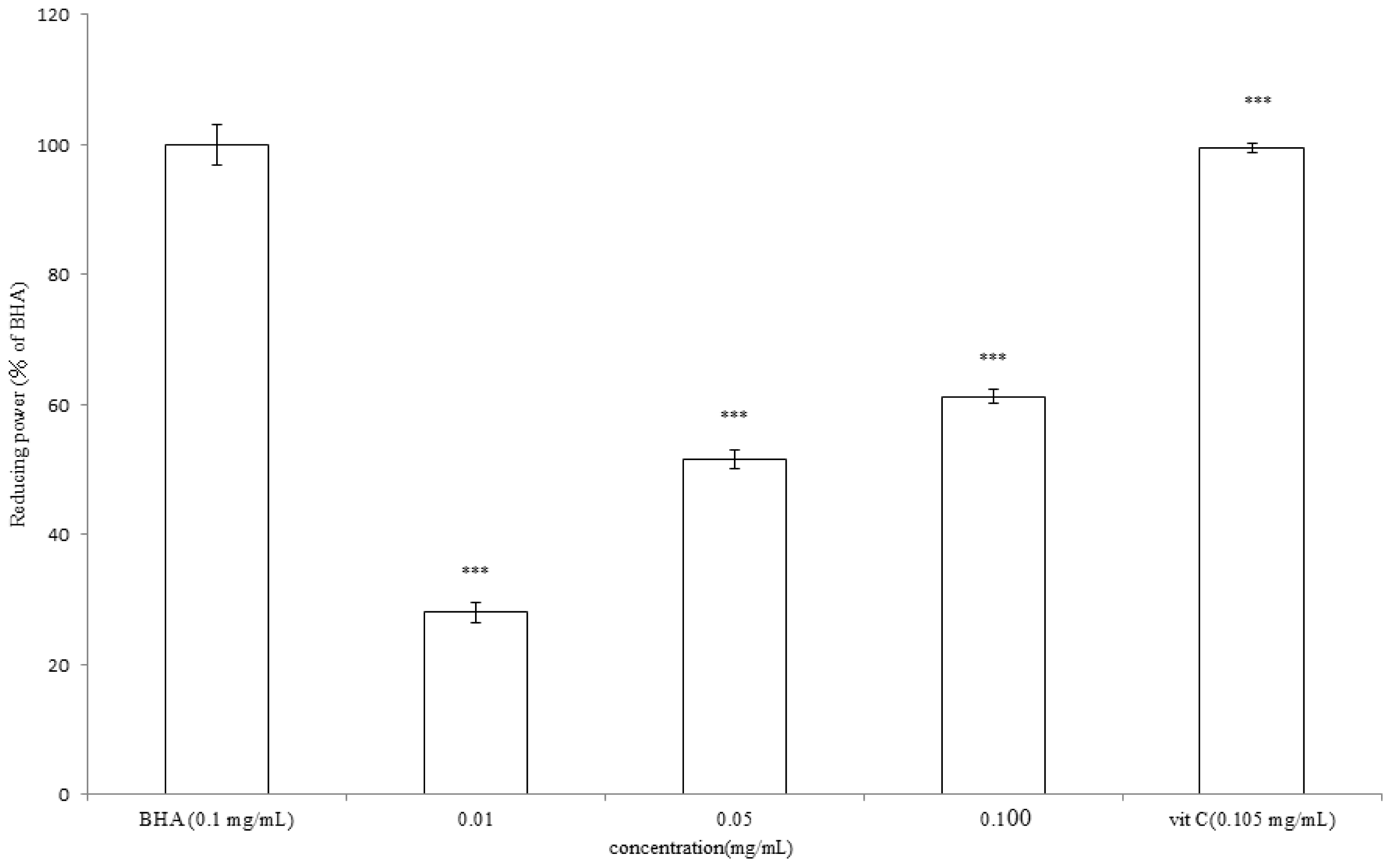
2.6. Determination of Reducing Power of A. argyi Essential Oil
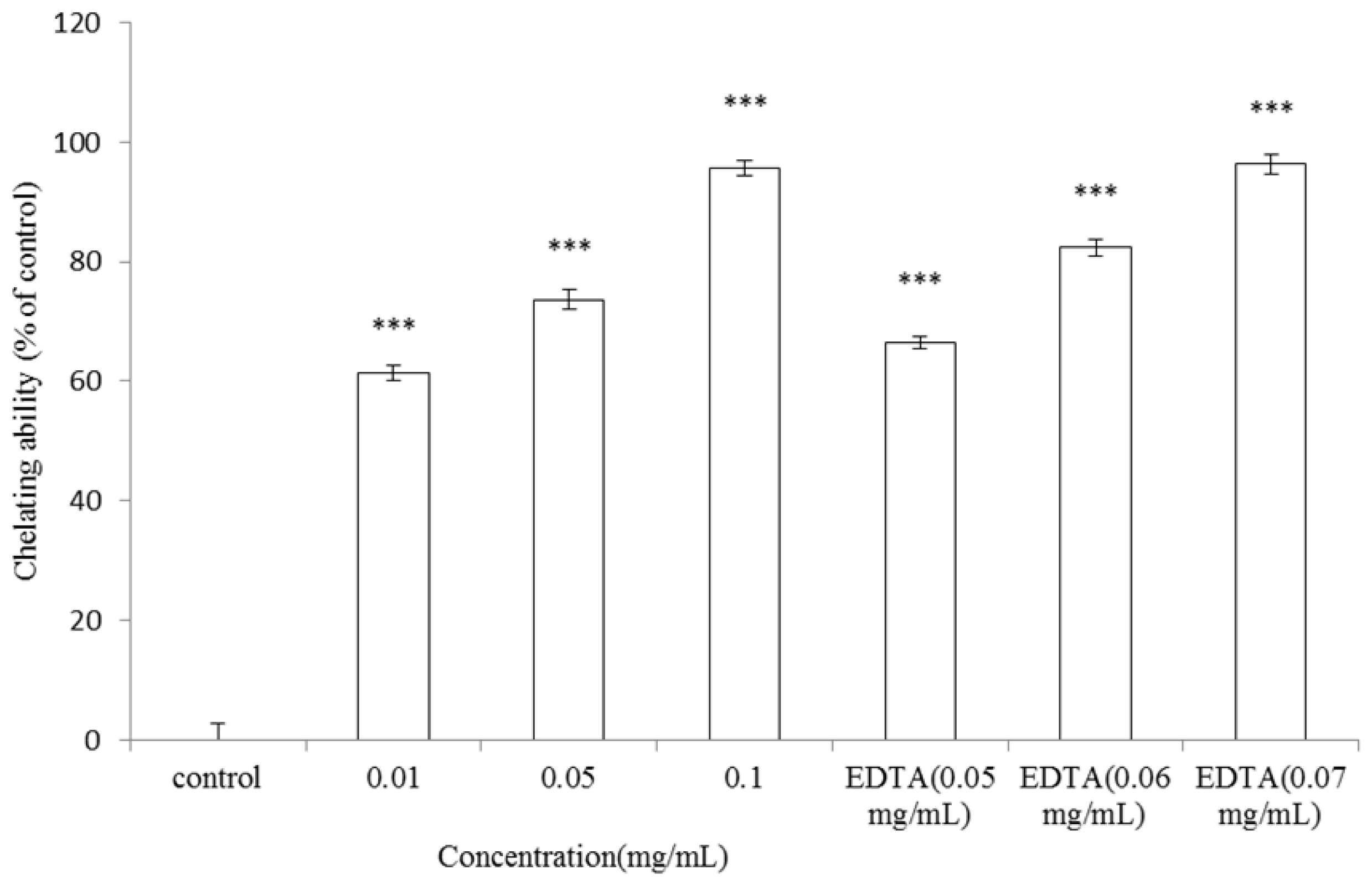
2.7. Metal-ion Chelating Activity of A. argyi Essential Oil
2.8. Chemical Composition of A. argyi Essential Oil
3. Experimental Section
3.1. Plant Material and Extraction of Essential Oils
3.2. Mushroom Tyrosinase Actvity Measurement
3.3. B16F10 Intracellular Melanin Content Measurement
3.4. B16F10 Intracellular Tyrosinase Activity Assay
3.5. DPPH Scavenging Activity Assay
3.6. ABTS+ Scavenging Capacity Assay
3.7. Determination of Reducing Power
3.8. Measurement of Metal-Ion Chelating Capacity
3.9. Gas Chromatography-Mass Spectrometry (GC/MS)
3.10. Statistical Analysis
4. Conclusions
Acknowledgements
- Conflict of InterestThe authors declare no conflict of interest.
References
- Panich, U.; Tangsupa-a-nan, V.; Onkoksoong, T.; Kongtaphan, K.; Kasetsinsombat, K.; Akarasereenont, P.; Wongkajornsilp, A. Inhibition of UVA-mediated melanogenesis by ascorbic acid through modulation of antioxidant defense and nitric oxide system. Arch. Pharma. Res 2011, 34, 811–820. [Google Scholar]
- Briganti, S.; Camera, E.; Picardo, M. Chemical and instrumental approaches to treat hyperpigmentation. Pigment Cell. Res 2003, 16, 101–110. [Google Scholar]
- Slominski, A.; Zmijewski, M.A.; Pawelek, J. l-tyrosine and l-dihydroxyphenylalanine as hormone-like regulators of melanocyte functions. Pigment Cell. Melanoma Res 2012, 25, 14–27. [Google Scholar]
- Garcia, A.; Fulton, J.E., Jr. The combination of glycolic acid and hydroquinone or kojic acid for the treatment of melasma and related conditions. Dermatol. Surg. 1996, 22, 443–447. [Google Scholar]
- Virador, V.M.; Kobayashi, N.; Matsunaga, J.; Hearing, V.J. A standardized protocol for assessing regulators of pigmentation. Anal. Biochem 1999, 270, 207–219. [Google Scholar]
- Sarkar, R.; Bhalla, M.; Kanwar, A.J. A comparative study of 20% azelaic acid cream monotherapy versus a sequential therapy in the treatment of melasma in dark-skinned patients. Dermatology 2002, 205, 249–254. [Google Scholar]
- Rendon, M.I.; Gaviria, J.I. Review of skin-lightening agents. Dermatol. Surg 2005, 31, 886–889. [Google Scholar]
- Cheng, S.L.; Liu, R.H.; Sheu, J.N.; Chen, S.T.; Sinchaikul, S.; Tsay, G.J. Toxicogenomics of A375 human malignant melanoma cells treated with arbutin. J. Biomed. Sci 2007, 14, 87–105. [Google Scholar]
- Garcia-Gavin, J.; Gonzalez-Vilas, D.; Fernandez-Redondo, V.; Toribio, J. Pigmented contact dermatitis due to kojic acid. A paradoxical side effect of a skin lightener. Contact Dermatitis 2010, 62, 63–64. [Google Scholar]
- Balina, L.M.; Graupe, K. The treatment of melasma. 20% azelaic acid versus 4% hydroquinone cream. Int J. Dermatol 1991, 30, 893–895. [Google Scholar]
- Halliwell, B. Free radicals, antioxidants, and human disease: Curiosity, cause, or consequence? Lancet 1994, 344, 721–724. [Google Scholar]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar]
- Sardy, M. Role of matrix metalloproteinases in skin ageing. Connect. Tissue Res 2009, 50, 132–138. [Google Scholar]
- Wlaschek, M.; Briviba, K.; Stricklin, G.P.; Sies, H.; Scharffetter-Kochanek, K. Singlet oxygen may mediate the ultraviolet A-induced synthesis of interstitial collagenase. J. Invest. Dermatol 1995, 104, 194–198. [Google Scholar]
- Buyukokuroglu, M.E.; GulcIn, I.; Oktay, M.; Kufrevioglu, O.I. In vitro antioxidant properties of dantrolene sodium. Pharmacol. Res 2001, 44, 491–494. [Google Scholar]
- Shahidul Alam, M.; Quader, M.A.; Rashid, M.A. HIV-inhibitory diterpenoid from Anisomeles indica. Fitoterapia 2000, 71, 574–576. [Google Scholar]
- Gulcin, I.; Oktay, M.; Kufrevioglu, O.I.; Aslan, A. Determination of antioxidant activity of lichen Cetraria islandica (L.) Ach. J. Ethnopharmacol 2002, 79, 325–329. [Google Scholar]
- Guo, C.H.; Liu, P.J.; Lin, K.P.; Chen, P.C. Nutritional supplement therapy improves oxidative stress, immune response, pulmonary function, and quality of life in allergic asthma patients: An open-label pilot study. Altern. Med. Rev 2012, 17, 42–56. [Google Scholar]
- Ito, N.; Hirose, M.; Fukushima, S.; Tsuda, H.; Shirai, T.; Tatematsu, M. Studies on antioxidants: Their carcinogenic and modifying effects on chemical carcinogenesis. Food Chem. Toxicol 1986, 24, 1071–1082. [Google Scholar]
- Romero-Graillet, C.; Aberdam, E.; Clement, M.; Ortonne, J.P.; Ballotti, R. Nitric oxide produced by ultraviolet-irradiated keratinocytes stimulates melanogenesis. J. Clin. Invest 1997, 99, 635–642. [Google Scholar]
- Sasaki, M.; Horikoshi, T.; Uchiwa, H.; Miyachi, Y. Up-regulation of tyrosinase gene by nitric oxide in human melanocytes. Pigment Cell. Res 2000, 13, 248–252. [Google Scholar]
- Chakraborty, A.K.; Funasaka, Y.; Slominski, A.; Ermak, G.; Hwang, J.; Pawelek, J.M.; Ichihashi, M. Production and release of proopiomelanocortin (POMC) derived peptides by human melanocytes and keratinocytes in culture: Regulation by ultraviolet B. Biochim. Biophys. Acta 1996, 1313, 130–138. [Google Scholar]
- Sasaki, M.; Kizawa, K.; Igarashi, S.; Horikoshi, T.; Uchiwa, H.; Miyachi, Y. Suppression of melanogenesis by induction of endogenous intracellular metallothionein in human melanocytes. Exp. Dermatol 2004, 13, 465–471. [Google Scholar]
- Yasui, H.; Sakurai, H. Age-dependent generation of reactive oxygen species in the skin of live hairless rats exposed to UVA light. Exp. Dermatol 2003, 12, 655–661. [Google Scholar]
- Funasaka, Y.; Komoto, M.; Ichihashi, M. Depigmenting effect of alpha-tocopheryl ferulate on normal human melanocytes. Pigment Cell. Res 2000, 13, 170–174. [Google Scholar]
- Nakasugi, T.; Nakashima, M.; Komai, K. Antimutagens in gaiyou (Artemisia argyi levl. et vant.). J. Agric. Food Chem 2000, 48, 3256–3266. [Google Scholar]
- Adams, M.; Efferth, T.; Bauer, R. Activity-guided isolation of scopoletin and isoscopoletin, the inhibitory active principles towards CCRF-CEM leukaemia cells and multi-drug resistant CEM/ADR5000 cells, from Artemisia argyi. Planta Med 2006, 72, 862–864. [Google Scholar]
- Jeong, M.A.; Lee, K.W.; Yoon, D.Y.; Lee, H.J. Jaceosidin, a pharmacologically active flavone derived from Artemisia argyi, inhibits phorbol-ester-induced upregulation of COX-2 and MMP-9 by blocking phosphorylation of ERK-1 and -2 in cultured human mammary epithelial cells. Ann. N. Y. Acad. Sci 2007, 1095, 458–466. [Google Scholar]
- Seo, J.M.; Kang, H.M.; Son, K.H.; Kim, J.H.; Lee, C.W.; Kim, H.M.; Chang, S.I.; Kwon, B.M. Antitumor activity of flavones isolated from Artemisia argyi. Planta Med 2003, 69, 218–222. [Google Scholar]
- Lee, H.G.; Yu, K.A.; Oh, W.K.; Baeg, T.W.; Oh, H.C.; Ahn, J.S.; Jang, W.C.; Kim, J.W.; Lim, J.S.; Choe, Y.K.; et al. Inhibitory effect of jaceosidin isolated from Artemisiaargyi on the function of E6 and E7 oncoproteins of HPV 16. J. Ethnopharmacol 2005, 98, 339–343. [Google Scholar]
- Bian, R.L. Pharmacological studies on the antiasthmatic principle of essential oil of Artemisia argyi--terpinenol-4 (author’s transl). Zhonghua Jie He He Hu Xi Xi Ji Bing Za Zhi 1981, 4, 203–206. [Google Scholar]
- Wenqiang, G.; Shufen, L.; Ruixiang, Y.; Yanfeng, H. Comparison of composition and antifungal activity of Artemisia argyi Levl. et Vant inflorescence essential oil extracted by hydrodistillation and supercritical carbon dioxide. Nat. Prod. Res 2006, 20, 992–998. [Google Scholar]
- Li, N.; Mao, Y.; Deng, C.; Zhang, X. Separation and identification of volatile constituents in Artemisia argyi flowers by GC-MS with SPME and steam distillation. J. Chromatogr. Sci 2008, 46, 401–405. [Google Scholar]
- Huang, H.C.; Chang, T.Y.; Chang, L.Z.; Wang, H.F.; Yih, K.H.; Hsieh, W.Y.; Chang, T.M. Inhibition of melanogenesis versus antioxidant properties of essential oil extracted from leaves of Vitex negundo Linn and chemical composition analysis by GC-MS. Molecules 2012, 17, 3902–3916. [Google Scholar]
- Busca, R.; Ballotti, R. Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell. Res 2000, 13, 60–69. [Google Scholar]
- Oyaizu, M. Studies on products of browing reaction: Antioxidative activity of product of browing reaction preapared from glucosamine. Jpn. J. Nutr 1986, 44, 307–315. [Google Scholar]
- Emerit, I. Free radicals and aging of the skin. EXS 1992, 62, 328–341. [Google Scholar]
- Wlaschek, M.; Tantcheva-Poor, I.; Naderi, L.; Ma, W.; Schneider, L.A.; Razi-Wolf, Z.; Schuller, J.; Scharffetter-Kochanek, K. Solar UV irradiation and dermal photoaging. J. Photochem. Photobiol. B 2001, 63, 41–51. [Google Scholar]
- Dinis, T.C.; Maderia, V.M.; Almeida, L.M. Action of phenolic derivatives (acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys 1994, 315, 161–169. [Google Scholar]
- Yu, L.; Haley, S.; Perret, J.; Harris, M.; Wilson, J.; Qian, M. Free radical scavenging properties of wheat extracts. J. Agric. Food Chem 2002, 50, 1619–1624. [Google Scholar]
- Battaini, G.; Monzani, E.; Casella, L.; Santagostini, L.; Pagliarin, R. Inhibition of the catecholase activity of biomimetic dinuclear copper complexes by kojic acid. J. Biol. Inorg. Chem 2000, 5, 262–268. [Google Scholar]
- Klabunde, T.; Eicken, C.; Sacchettini, J.C.; Krebs, B. Crystal structure of a plant catechol oxidase containing a dicopper center. Nat. Struct. Biol 1998, 5, 1084–1090. [Google Scholar]
- Wood, J.M.; Schallreuter, K.U. Studies on the reactions between human tyrosinase, superoxide anion, hydrogen peroxide and thiols. Biochim. Biophys. Acta 1991, 1074, 378–385. [Google Scholar]
- Karg, E.; Odh, G.; Wittbjer, A.; Rosengren, E.; Rorsman, H. Hydrogen peroxide as an inducer of elevated tyrosinase level in melanoma cells. J. Invest. Dermatol 1993, 100, 209S–213S. [Google Scholar]
- Ebanks, J.P.; Wickett, R.R.; Boissy, R.E. Mechanisms regulating skin pigmentation: The rise and fall of complexion coloration. Int. J. Mol. Sci 2009, 10, 4066–4087. [Google Scholar]
- Ciftci, O.; Ozdemir, I.; Tanyildizi, S.; Yildiz, S.; Oguzturk, H. Antioxidative effects of curcumin, beta-myrcene and 1,8-cineole against 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced oxidative stress in rats liver. Toxicol. Ind. Health 2011, 27, 447–453. [Google Scholar]
- Sud'ina, G.F.; Mirzoeva, O.K.; Pushkareva, M.A.; Korshunova, G.A.; Sumbatyan, N.V.; Varfolomeev, S.D. Caffeic acid phenethyl ester as a lipoxygenase inhibitor with antioxidant properties. FEBS Lett 1993, 329, 21–24. [Google Scholar]
- Loreto, F.; Pinelli, P.; Manes, F.; Kollist, H. Impact of ozone on monoterpene emissions and evidence for an isoprene-like antioxidant action of monoterpenes emitted by Quercus ilex leaves. Tree Physiol 2004, 24, 361–367. [Google Scholar]
- Hussain, T.; Siddiqui, H.L.; Zia-ur-Rehman, M.; Masoom Yasinzai, M.; Parvez, M. Anti-oxidant, anti-fungal and anti-leishmanial activities of novel 3-[4-(1H-imidazol-1-yl) phenyl]prop-2-en-1- ones. Eur. J. Med. Chem 2009, 44, 4654–4660. [Google Scholar]
- Cheng, J.H.; Hung, C.F.; Yang, S.C.; Wang, J.P.; Won, S.J.; Lin, C.N. Synthesis and cytotoxic, anti-inflammatory, and anti-oxidant activities of 2′,5′-dialkoxylchalcones as cancer chemopreventive agents. Bioorg. Med. Chem 2008, 16, 7270–7276. [Google Scholar]
- Yoshikawa, K.; Kawahara, Y.; Arihara, S.; Hashimoto, T. Aromatic compounds and their antioxidant activity of Acer saccharum. J. Nat. Med 2011, 65, 191–193. [Google Scholar]
- Kumar, P.K.S.; Lalitha, C. Screening of antioxidant activity, total phenolics and GC-MS study of Vitex negundo. Afri. J. Biochem. Res 2010, 4, 191–195. [Google Scholar]
- Hussain, A.I.; Anwar, F.; Chatha, S.A.S.; Jabbar, A.; Mahboob, S.; Nigam, P.S. Rosmarinus officinalis essential oil: Antiproliferative, antioxidant and antibacterial activities. Braz. J. Microbiol 2010, 41, 1070–1078. [Google Scholar]
- Abad, M.J.; Bedoya, L.M.; Apaza, L.; Bermejo, P. The Artemisia L. Genus: A review of bioactive essential oils. Molecules 2012, 17, 2542–2566. [Google Scholar]
- Marzoug, H.N.B.; Romdhane, M.; Lebrihi, A.; Mathieu, F.; Couderc, F.; Abderraba, M.; Khouja, M.L.; Bouajila, J. Eucalyptus oleosa essential oils: Chemical composition and antimicrobial and antioxidant activities of the oils from different plant parts (stems, leaves, flowers and fruits). Molecules 2011, 16, 1695–1709. [Google Scholar]
- Huang, H.-C.; Wang, H.-F.; Yih, K.-H.; Chang, L.-Z.; Chang, T.-M. The dual antimelanogenic and antioxidant activities of the essential oil extracted from the leaves of Acorus macrospadiceus (Yamamoto) F. N. Wei et Y. K. Li. Evid.-Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Bilodeau, M.L.; Greulich, J.D.; Hullinger, R.L.; Bertolotto, C.; Ballotti, R.; Andrisani, O.M. BMP-2 stimulates tyrosinase gene expression and melanogenesis in differentiated melanocytes. Pigment Cell. Res 2001, 14, 328–336. [Google Scholar]
- Tsuboi, T.; Kondoh, H.; Hiratsuka, J.; Mishima, Y. Enhanced melanogenesis induced by tyrosinase gene-transfer increases boron-uptake and killing effect of boron neutron capture therapy for amelanotic melanoma. Pigment Cell. Res 1998, 11, 275–282. [Google Scholar]
- Yang, J.Y.; Koo, J.H.; Song, Y.G.; Kwon, K.B.; Lee, J.H.; Sohn, H.S.; Park, B.H.; Jhee, E.C.; Park, J.W. Stimulation of melanogenesis by scoparone in B16 melanoma cells. Acta Pharmacol. Sin 2006, 27, 1467–1473. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol 1995, 28, 25–30. [Google Scholar]
- Sánchez-Moreno, C.; Larrauri, J.A.; Saura-Calixto, F. A procedure to measure the antiradical efficiency of polyphenols. J. Sci. Food Agric 1998, 76, 270–276. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med 1999, 26, 1231–1237. [Google Scholar]
- Adama, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy; Allured Publishing Co.: Carol Stream, IL, USA, 1995; pp. 57–332. [Google Scholar]
- Vandendool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr 1963, 11, 463–471. [Google Scholar]

| Rta | Compound b | M. f. c | Peak area (%) | Classification |
|---|---|---|---|---|
| 20.22 | 3-Carene | C10H16 | 4.64 | monoterpene |
| 21.52 | Camphene | C10H16 | 0.48 | monoterpene |
| 24.03 | (−)-β-Pinene | C10H16 | 5.62 | monoterpene |
| 25.35 | 3-Octanone | C8H16O | 0.25 | ketone |
| 25.54 | Myrcene | C10H16 | 0.19 | monoterpene |
| 26.82 | Dihydrocarveol | C10H18O | 0.55 | alcohol |
| 27.25 | o-Cymene | C10H14 | 5.01 | aromatic compound |
| 27.55 | Eucalyptol | C10H18O | 23.66 | ether |
| 28.18 | 1R-α-Pinene | C10H16 | 0.34 | monoterpene |
| 29.04 | τ-Terpinene | C10H16 | 0.36 | monoterpene |
| 30.94 | Linalool | C10H18O | 3.31 | alcohol |
| 32.59 | Camphor | C10H16O | 1.53 | ketone |
| 33.03 | Menthone | C10H18O | 4.18 | ketone |
| 33.58 | (−)-Borneol | C10H18O | 6.55 | alcohol |
| 33.97 | (−)-Menthol | C10H20O | 1.19 | alcohol |
| 34.15 | (−)-Terpinen-4-ol | C10H18O | 1.40 | alcohol |
| 34.89 | α-Terpieol | C10H18O | 0.17 | alcohol |
| 38.05 | (+)-Carvone | C10H14O | 0.13 | ketone |
| 39.07 | Bergamiol | C12H20O2 | 3.91 | ester |
| 41.31 | Bornyl acetate | C12H20O2 | 3.62 | ester |
| 44.59 | α-Gurjunene | C15H24 | 0.74 | Sesquiterpene |
| 45.56 | Nerol acetate | C12H20O2 | 0.99 | ester |
| 46.93 | Cedryl acetate | C17H28O2 | 2.28 | ester |
| 47.48 | Geranyl acetate | C12H20O2 | 0.88 | ester |
| 48.83 | Caryophyllene | C15H24 | 10.19 | Sesquiterpene |
| 49.54 | α-Bergamotene | C15H24 | 0.41 | Sesquiterpene |
| 50.15 | Humulene | C15H24 | 1.40 | Sesquiterpene |
| 50.37 | (Z)-β-Farnesene | C15H24 | 0.59 | Sesquiterpene |
| 51.15 | β-Cubebene | C15H24 | 1.29 | Sesquiterpene |
| 51.68 | Geranyl isovalerate | C15H26O2 | 0.1 | ester |
| 52.11 | Nerolidol | C15H26O | 0.88 | alcohol |
| 52.55 | δ-Cadinene | C15H24 | 0.59 | Sesquiterpene |
| 54.29 | Longiborneol | C15H26O | 2.67 | alcohol |
| Unknown | 9.90 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, H.-C.; Wang, H.-F.; Yih, K.-H.; Chang, L.-Z.; Chang, T.-M. Dual Bioactivities of Essential Oil Extracted from the Leaves of Artemisia argyi as an Antimelanogenic versus Antioxidant Agent and Chemical Composition Analysis by GC/MS. Int. J. Mol. Sci. 2012, 13, 14679-14697. https://doi.org/10.3390/ijms131114679
Huang H-C, Wang H-F, Yih K-H, Chang L-Z, Chang T-M. Dual Bioactivities of Essential Oil Extracted from the Leaves of Artemisia argyi as an Antimelanogenic versus Antioxidant Agent and Chemical Composition Analysis by GC/MS. International Journal of Molecular Sciences. 2012; 13(11):14679-14697. https://doi.org/10.3390/ijms131114679
Chicago/Turabian StyleHuang, Huey-Chun, Hsiao-Fen Wang, Kuang-Hway Yih, Long-Zen Chang, and Tsong-Min Chang. 2012. "Dual Bioactivities of Essential Oil Extracted from the Leaves of Artemisia argyi as an Antimelanogenic versus Antioxidant Agent and Chemical Composition Analysis by GC/MS" International Journal of Molecular Sciences 13, no. 11: 14679-14697. https://doi.org/10.3390/ijms131114679
APA StyleHuang, H.-C., Wang, H.-F., Yih, K.-H., Chang, L.-Z., & Chang, T.-M. (2012). Dual Bioactivities of Essential Oil Extracted from the Leaves of Artemisia argyi as an Antimelanogenic versus Antioxidant Agent and Chemical Composition Analysis by GC/MS. International Journal of Molecular Sciences, 13(11), 14679-14697. https://doi.org/10.3390/ijms131114679





