Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes
Abstract
:1. Introduction
2. Experimental Section
2.1. Wheat Samples
2.2 Chemical Analysis
2.2.1. Osborne Fractionation Method
Albumin-Globulin extraction
Gliadin extraction
Soluble glutenin extraction
Insoluble glutenin
2.2.2. SDS-PAGE Gel Electrophoresis
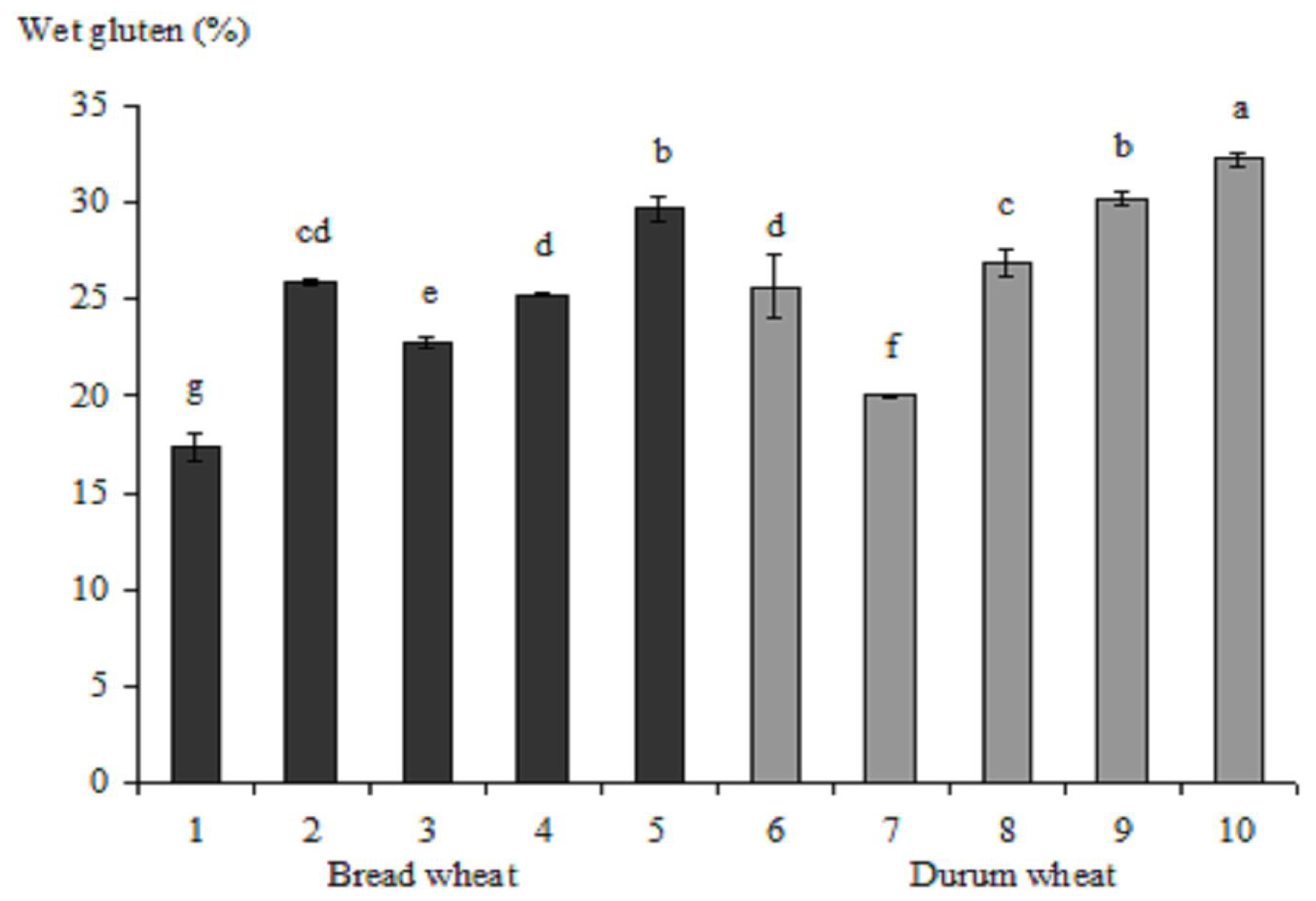
2.2.3. Wet Gluten Content
2.2.4. Tryptophan Content
2.2.5. Statistical Analyses
3. Results and Discussion
3.1. Results
3.2. Discussion
4. Conclusions
Acknowledgments
References
- Food and Agriculture Organization (FAO). FAOSTAT database. Available online: http://www.faostat.fao.org accessed on 10 December 2010.
- Shewry, PR; Tatham, AS; Forde, J; Kreis, M; Miflin, BJ. The classification and nomenclature of wheat gluten proteins: a reassessment. J Cereal Sci 1986, 4, 97–106. [Google Scholar]
- Belderok, B; Mesdag, J; Donner, DA. Bread-Making Quality of Wheat: A Century of Breeding in Europe; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2000; pp. 30–31. [Google Scholar]
- Merlino, M; Leroy, P; Chambon, C; Branlard, G. Mapping and proteomic analysis of albumin and globulin proteins in hexaploid wheat kernels (Triticum aestivum L.). Theor Appl Genet 2009, 18, 1321–1337. [Google Scholar]
- Carbonero, P; Salcedo, G; Sánchez-Monge, R; Garcia-Maroto, F; Royo, J; Gomez, L; Mena, M; Diaz, L. A multigene Family from Cereals Which Encodes Inhibitors of Trypsin and Heterologous-Amylases. In Innovations of Proteases and Their Inhibitors; Aviles, FX, Ed.; Walter de Gruyter: Berlin, Germany, 1993; pp. 333–348. [Google Scholar]
- Posch, A; Weiss, W; Wheeler, C; Dunn, MJ; Görg, A. Sequence analysis of wheat grain allergens separated by two-dimensional electrophoresis with immobilized gradients. Electrophoresis 1995, 18, 1115–1119. [Google Scholar]
- Jiménez, T; Martínez-Anaya, MA. Amylases and hemicellulases in breadmaking. Degradation by-products and potential relationship with functionality. Food Sci Technol Int 2001, 7, 5–14. [Google Scholar]
- Toyosaki, T. Effect of hydroperoxide in lipid peroxidation on dough fermentation. Food Chem 2007, 104, 680–685. [Google Scholar]
- Uthayakumaran, S; Newberry, M; Phan-Tien, N; Tanner, R. Small and large strain rheology of wheat gluten. Rheol Acta 2002, 41, 162–172. [Google Scholar]
- MacRitchie, F. Physico-chemical properties of wheat proteins in relation to functionality. Adv Food Nutr Res 1992, 36, 1–87. [Google Scholar]
- Abdel-Aal, E-SM; Salama, DA; Hucl, P; Sosulski, FW; Cao, W. Electrophoretic characterization of spring spelt wheat gliadins. J Agric Food Chem 1996, 44, 2117–2123. [Google Scholar]
- Kirkman, MA; Shewry, PR; Miflin, BJ. The effect of nitrogen nutrition on the lysine content and protein composition of barley seeds. J Sci Food Agric 1982, 33, 115–127. [Google Scholar]
- Shewry, PR. Improving the protein content and composition of cereal grain. J Cereal Sci 2007, 46, 239–250. [Google Scholar]
- Kasarda, DD; Autran, JC; Lew, EJL; Nimmo, CC; Shewry, PR. N-terminal amino acid sequences of ω-gliadins and ω-secalins: Implications for the evolution of prolamin genes. Biochim Biophys Acta 1983, 747, 138–150. [Google Scholar]
- Skerritt, JH; Devery, JM; Hill, AS. Gluten intolerance: Chemistry, celiac-toxicity, and detection of prolamins in foods. Cereal Foods World 1990, 35, 638–644. [Google Scholar]
- Vader, W; Kooy, Y; Van Veelen, P; De Ru, A; Harris, D; Benckhuijsen, W; Peña, S; Mearin, L; Drijfhout, JW; Koning, F. The gluten response in children with celiac disease is directed toward multiple gliadin and glutenin peptides. Gastroenterology 2002, 122, 1729–1737. [Google Scholar]
- Bietz, JA; Simpson, DG. Electrophoresis and chromatography of wheat proteins: Available methods, and procedures for statistical evaluation of the data. J Chromatogr 1992, 624, 53–80. [Google Scholar]
- Wrigley, CW. Giant proteins with flour power. Nature 1996, 381, 738–739. [Google Scholar]
- Kasarda, DD. Glutenin polymers: The in vitro to in vivo transition. Cereal Foods World 1999, 44, 566–571. [Google Scholar]
- Müller, S; Vensel, WH; Kasarda, DD; Köler, P; Wieser, H. Disulphide bonds of adjacent cysteine residues in low molecular weight subunits of wheat glutenin. J Cereal Sci 1998, 27, 109–116. [Google Scholar]
- Payne, PI. Endosperm Proteins. In Plant Gene Research: A Genetic Approach to Plant Biochemistry; Blenstein, AD, King, PJ, Eds.; Springer: New York, NY, USA, 1986; pp. 207–301. [Google Scholar]
- Lookhart, G; Bean, S. Separation and characterization of wheat protein fractions by high-performance capillary electrophoresis. Cereal Chem 1995, 72, 527–532. [Google Scholar]
- Vereijken, JM; Klostermann, VLC; Beckers, FHR; Spekking, WTJ; Graveland, A. Intercultivar variation in the proportions of wheat protein fractions and relation to mixing behaviour. J Cereal Sci 2000, 32, 159–167. [Google Scholar]
- Fling, SP; Gregerson, DS. Peptide and protein molecular weight determination by electrophoresis using a high-molarity tris-buffer system without urea. Anal Biochem 1986, 155, 83–88. [Google Scholar]
- American Association of Cereal Chemists (AACC), Approved Methods of the American Association of Cereal Chemists, 10th ed; AACC methods 38-12.02; AACC: St. Paul, MN, USA, 2000.
- Nurit, E; Tiessen, A; Pixley, KV; Palacios-Rojas, N. Reliable and inexpensive colorimetric method for determining protein-bound tryptophan in maize kernels. J Agric Food Chem 2009, 57, 7233–7238. [Google Scholar]
- Vogel, KP; Johnson, VA; Mattern, PJ. Protein and lysine contents of endosperm and bran of the parents and progenies of crosses of common wheat. Crop Sci 1978, 18, 751–754. [Google Scholar]
- Weegels, PL; Van de Pijpekamp, AM; Graveland, A; Hamer, RJ; Schofield, JD. Depolymerisation and re-polymerisation of wheat glutenin during dough processing. Relationships between glutenin macropolymer content and quality parameters. J Cereal Sci 1996, 23, 103–111. [Google Scholar]
- Emanuelson, J; Wollenweber, B; Jorgensen, JR; Andersen, SBF; Jensen, CR. Wheat Grain Composition and Implications for Bread Quality. In DIAS report Plant Production no 92; Danish Institute of Agricultural Sciences: Tjele, Denmark, 2003. [Google Scholar]
- Stehno, Z; Dvořáček, V; Dotlačil,, L. Wheat Protein Fractions in Relation to Grain Quality Characters of the Cultivars Registered in the Czech Republic 2004-2006. Proceedings of 11th International Wheat Genetics Symposium, Apples, R, Eastwood, R, Lagudah, E, Langridge, P, Mackay, M, McIntyre, L, Sharp, P, Eds.; Sydney University Press: Brisbane, Australia, 2008; pp. 556–559. [Google Scholar]
- Abdelrahman, E; Elagib, A; Bureng, PL; Mohamed, BE. Proteins and baking quality of three Sudanese wheat cultivars 1. the relationship between protein soluble fractions and breadmaking properties. Univ Khartoum J Agric Sci 2004, 12, 391–404. [Google Scholar]
- Kovacs, MIP; Howes, NK; Leisle, D; Zawistowski, J. Effect of two different Low Molecular Weight glutenin subunits on durum wheat pasta quality parameters. Cereal Chem 1995, 72, 85–87. [Google Scholar]
- Cornish, GB; Bekes, F; Eagles, HA; Payne, PI. Prediction of Dough Properties for Bread Wheat. In Gliadin and Glutenin—the Unique Balance of Wheat Quality; Wrigley, C, Bekes, F, Bushuk, W, Eds.; American Association of Cereal Chemistry: St. Paul, MN, USA, 2006; pp. 152–160. [Google Scholar]
- Kohler, P; Keck, B; Muller, S; Wieser, H. Disulphide Bonds in Wheat Gluten. In Proceedings of the International Meeting on Wheat Kernel Proteins Molecular and Functional Aspects; University of Tuscia: Viterbo, Italy, 1994; pp. 45–54. [Google Scholar]
- Janssen, AM; Van Vliet, T; Vereijken, JM. Rheological behaviour of wheat glutens at small and large deformations. Effect of gluten composition. J Cereal Sci 1996, 23, 33–42. [Google Scholar]
- Uthayakumaran, S; Gras, PW; Stoddard, FL; Békés, F. Effect of varying protein content and glutenin-to-gliadin ratio on the functional properties of wheat dough. Cereal Chem 1999, 76, 389–394. [Google Scholar]
- Peruffo, ADB; Pogna, NE; Curioni, C. Evidence for the Presence of Disulfide Bonds between Beta-Amylase and Low Molecular Weight Glutenin Subunits. Proceedings of 6th International Gluten Workshop “Gluten 96”, Sydney, Australia, 2–4 September 1996, O’Brien, L, Blakeney, AB, Ross, AS, Wrigley, CW, Eds.; Royal Australian Chemical Institute: Melbourne, Australia, 1996; pp. 45–54. [Google Scholar]
- Wieser, H. Chemistry of gluten proteins. Food Microbiol 2007, 24, 115–119. [Google Scholar]
- Federmann, GR; Goecke, EU; Steiner, AM. Detection of adulteration of flour of spelt (Triticum spelta L.) with flour of wheat (Triticum aestivum L. emend. Fiori et Paol.) by electrophoresis. Plant Var Seeds 1992, 5, 123–125. [Google Scholar]
- Chakraborty, K; Khan, K. Biochemical and breadmaking properties of wheat protein components. Compositional differences revealed through quantitation and polyacrylamide gel electrophoresis of protein fractions from various isolation procedures. Cereal Chem 1988, 65, 333–340. [Google Scholar]
- Grosch, W; Wieser, H. Redox reactions in wheat dough as affected by ascorbic acid. J Cereal Sci 1999, 29, 1–16. [Google Scholar]
- Sapirstein, HD; David, P; Preston, KR; Dexter, JE. Durum wheat breadmaking quality: Effects of gluten strength, protein composition, semolina particle size and fermentation time. J Cereal Sci 2007, 45, 150–161. [Google Scholar]
- Schaechter, JD; Wurtman, RJ. Serotonin release varies with brain tryptophan levels. Brain Res 1990, 532, 203–210. [Google Scholar]
- Gafurova, DA; Tursunkhodzhaev, PM; Kasymova, TD; Yuldashev, PK. Fractional and amino-acid composition of wheat grain cultivated in Uzbekistan. Chem Nat Compd 2002, 38, 462–465. [Google Scholar]
- Comai, S; Bertazzo, A; Bailoni, L; Zancato, M; Costa, VLC; Allegri, G. The content of proteic and nonproteic (free and protein-bound) tryptophan in quinoa and cereal flours. Food Chem 2007, 100, 1350–1355. [Google Scholar]

| Genotypes | Parents (Origin) | Country | Growth Type |
|---|---|---|---|
| Bread wheat | |||
| ZP 87/I | L-99 (SRB) × Pobeda (SRB) | SRB | winter |
| ZP Zemunska rosa | Skopljanka (MKD) × Proteinka (SRB) | SRB | winter |
| ZP 224 | L-4 (SRB) × Dulus/Metso (CIMMYT) | SRB | facultative |
| ZP Zlatna | Jasenica (SRB) × Rodna (SRB) | SRB | winter |
| Apache | FRA | winter | |
| Durum wheat | |||
| ZP 34/I | SOD 55 (SVK) × Korifla (ICARDA) | SRB | facultative |
| ZP 10/I | Windur (DEU) × Rodur (ROU) | SRB | winter |
| ZP DSP/01 | Windur (DEU) × SOD 64 (SVK) | SRB | winter |
| ZP 7858 | Mina (MKD) × Mexicali 75 (CIMMYT) | SRB | facultative |
| Varano | ITA | facultative | |
| Varieties | Protein | Albumins + Globulins | Gliadins | Soluble glutenins | Insoluble glutenins | Sum Gli + Glu | Ratio Gli/Glu | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (1) | (1) | (2) | (1) | (2) | (1) | (2) | (1) | (2) | (2) | ||
| Bread wheat | |||||||||||
| ZP 87/I | 9.51 d | 3.51 e | 36.93 c | 2.31 c | 24.31 b | 1.16 d | 12.20 b | 2.53 c | 26.56 b | 63.07 b | 0.62 b |
| Apache | 9.26 d | 3.87 d | 41.79 a | 2.71 b | 29.27 a | 1.38 a | 14.91 a | 1.29 d | 13.99 d | 58.17 d | 1.01 a |
| ZP Zemunska rosa | 12.64 a | 4.39 c | 34.73 d | 2.71 b | 21.44 c | 1.20 c,d | 9.49 d | 4.34 a | 34.33 a | 65.26 a | 0.49 c |
| ZP 224 | 11.76 c | 4.62 b | 39.28 b | 2.59 b | 21.93 c | 1.30 a,b | 11.01 c | 3.25 b | 27.63 b | 60.57 c | 0.57 b,c |
| ZP Zlatna | 12.22 b | 4.83 a | 39.52 b | 3.63 a | 29.84 a | 1.26 b,c | 10.31 c,d | 2.50 c | 20.45 c | 60.66 c | 0.97 a |
| F-test | *** | *** | *** | *** | *** | * | *** | *** | *** | *** | *** |
| CV (%) | 13.54 | 12.11 | 6.67 | 16.84 | 14.88 | 6.73 | 17.28 | 38.14 | 29.92 | 13.85 | 31.81 |
| Durum wheat | |||||||||||
| ZP 34/I | 12.15 a | 4.79 ab | 39.44 b | 2.67 c | 21.98 c | 0.88 e | 7.24 d | 3.81 a | 31.33 a | 60.55 c | 0.57 c |
| ZP 10/I | 11.12 b | 3.95 d | 35.52 d | 2.87 b | 25.81 b | 1.30 a | 11.69 a | 3.00 d | 26.98 c | 64.48 a | 0.67 b |
| ZP DSP/01 | 11.04 b | 4.83 a | 43.75 a | 3.19 a | 28.90 a | 1.06 d | 9.55 bc | 1.96 e | 17.80 d | 56.25 d | 1.06 a |
| Varano | 12.36 a | 4.59 c | 37.15 c | 3.15 a | 25.76 b | 1.12 c | 9.16 c | 3.50 c | 28.29 c | 63.21 a,b | 0.68 b |
| ZP 7858 | 12.40 a | 4.62 b,c | 37.28 c | 2.87 b | 23.16 c | 1.26 b | 10.04 b | 3.64 b | 29.41 b | 62.61 b | 0.58 c |
| F-test | * | *** | *** | *** | *** | *** | *** | *** | *** | *** | *** |
| CV (%) | 5.71 | 7.43 | 7.86 | 7.07 | 10.17 | 14.22 | 15.97 | 21.94 | 18.50 | 15.35 | 27.69 |
| Mean (bread wheat) | 11.08 b | 4.24 a | 38.45 a | 2.79 a | 25.36 a | 1.26 a | 11.58 a | 2.78 b | 24.59 b | 61.53 a | 0.73 a |
| Mean (durum wheat) | 11.81 a | 4.56 a | 38.63 a | 2.95 a | 25.12 a | 1.12 b | 9.54 b | 3.16 a | 26.76 a | 61.42 a | 0.69 a |
| Polypeptides Mw (kDa) | Bread wheat | Durum wheat | LSD0.05 | CV (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ZP 87/I | Apache | ZP Zemunska rosa | ZP 224 | ZP Zlatna | ZP 34/I | ZP 10/I | ZP DSP/01 | Varano | ZP 7858 | |||
| 111.3 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.81 b | 1.23 a | 0.99 b | 0.81 b | 0.056 | 57.26 |
| 101.2 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.79 b | 1.51 a | 0.085 | 88.56 |
| 93.55 | 0.76 c | 1.38 b | 0.54 e | n.d. | 0.67 d | n.d. | 1.51 a | n.d. | n.d. | n.d. | 0.072 | 66.25 |
| 90.5 | n.d. | n.d. | n.d. | n.d. | n.d. | 1.23 b | n.d. | 2.81 a | n.d. | n.d. | 0.148 | 93.71 |
| 87.7 | 0.97 e | 1.08 d | 1.15 d | 2.06 c | 1.04 e | n.d. | 2.91 b | 1.07 d | 3.69 a | n.d. | 0.101 | 71.06 |
| 85.7 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 4.14 a | - | - |
| 82.3 | n.d. | n.d. | n.d. | 0.64 b | n.d. | n.d. | n.d. | n.d. | n.d. | 1.40 a | 0.101 | 92.73 |
| 80.3 | n.d. | n.d. | n.d. | n.d. | n.d. | 0.57 d | 1.52 b,c | 1.40 c | 1.71 a | 1.39 c | 0.138 | 58.17 |
| 77.2 | 1.26 c | 1.43 b | 1.15 d | 0.59 e | 1.32 c | 0.36 f | n.d. | n.d. | n.d. | 1.64 a | 0.072 | 58.27 |
| 73.6 | n.d. | n.d. | n.d. | n.d. | 0.14 c | 0.56 b | 2.09 a | 2.05 a | 2.17 a | n.d. | 0.202 | 85.33 |
| 62.4 | 0.60 g | 0.50g | 0.83f | 1.28 e | 1.81 c | 1.77 c | 2.46 a | 2.29 b | 1.51 d | 2.24 b | 0.124 | 45.92 |
| 55.7 | n.d. | n.d. | n.d. | 1.25 e | n.d. | 1.32 e | 2.30 c | 2.65 b | 2.02 d | 3.24 a | 0.101 | 56.88 |
| 43.9 | 0.51 | n.d. | n.d. | 16.27 a | n.d. | 13.70 b | 9.95 | 12.92 c | 13.21 b,c | 13.64 b | 0.613 | 60.43 |
| 42.7 | n.d. | n.d. | n.d. | 0.76 c | n.d. | 3.17 a | 3.25 a | 1.38 b | 1.43 b | 1.50 b | 0.160 | 69.65 |
| 40.2 | 5.89 b | 17.19 a | 5.17 b | n.d. | 5.37 b | n.d. | n.d. | n.d. | n.d. | n.d. | 0.529 | 88.67 |
| 39.5 | 10.87 d | 7.31 e | 2.36 g | 12.37 b | 3.84 f | 13.44 a | 11.63 c | 10.70 d | 10.76 d | 11.41 c | 0.515 | 38.75 |
| 37.6–34.8 | 41.88 c | 32.43 d | 47.22 a | 31.81 d | 46.12 b | 24.03 e | 20.34 h | 22.59 f | 21.97 g | 23.15 e,f | 0.896 | 32.73 |
| 31.4 | 4.26 f | 6.43 b | n.d. | 5.88 c | 3.78 g | 6.44 b | 7.39 a | 5.03 | 5.58 d | 5.28 e | 0.202 | 40.07 |
| 29.7 | 0.70 f | 0.87 f | 4.03 a | 1.51 e | 1.86 d | 2.43 b,c | 2.37 c | 2.71 b | n.d. | 2.78 b | 0.263 | 59.71 |
| 29.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 1.77 a | n.d. | - | - |
| 26.6 | n.d. | 0.75 g | 1.22 f | 3.08 d | 0.81 g | 3.74 c | 2.71 e | 4.68 b | 4.87 b | 5.71 a | 0.252 | 70.45 |
| 20.6 | n.d. | n.d. | n.d. | n.d. | 0.14 f | 0.58 e | 2.65 b | 2.45 c | 3.13 a | 1.92 d | 0.189 | 80.08 |
| 16.2 | 0.48 g | 12.32 c | 13.45 b | 6.23 f | 14.14 a | 6.39 f | 5.89 f | 8.17 d | 7.02 e | 6.60 e,f | 0.529 | 50.19 |
| 15.0 | 13.39 b | 11.97 c | 15.09 a | 10.45 d | 11.28 | 12.21 c | 10.28 d | 8.85 f | 9.76 e | 7.82 g | 0.515 | 20.18 |
| 14.0 | 12.37 a | 4.67 c | 5.39 b | 2.85 f | 5.39 b | 4.69 c | 5.35 b | 3.69 d | 3.95 d | 3.14 e | 0.409 | 51.10 |
| 11.4 | 6.04 a | 1.67 g | 2.37 f | 2.95 e | 2.26 f | 3.33 d | 4.56 b | 3.32 d | 3.65 c | 0.68 h | 0.160 | 47.51 |
| S-rich subunits (γ- + β- + α-gliadins) | S-poor subunits (ω-gliadins) | Sum (S-poor + S-rich) | S-poor/S-rich ratio | |
|---|---|---|---|---|
| Bread wheat | ||||
| ZP 87/I | 63.41 b | 0.60 d | 64.01 b | 0.017 c |
| Apache | 63.36 b | 0.50 d | 63.86 c | 0.008 d |
| ZP Zemunska rosa | 54.75 d | 0.83 c | 55.58 e | 0.015 c |
| ZP 224 | 67.09 a | 2.53 a | 69.62 a | 0.038 a |
| ZP Zlatna | 59.11 c | 1.95 b | 61.06 d | 0.033 b |
| F-test | *** | *** | *** | *** |
| CV (%) | 7.24 | 57.29 | 7.77 | 54.29 |
| Durum wheat | ||||
| ZP 34/I | 60.78 a | 3.65c | 64.43 a | 0.060 e |
| ZP 10/I | 52.56 d | 6.85 a | 59.41 d | 0.130 b |
| ZP DSP/01 | 55.33 d | 6.99 a | 62.32 b | 0.126 a |
| Varano | 52.95 c | 5.70 b | 58.65 e | 0.108 c |
| ZP 7858 | 54.98 b | 5.48 b | 60.46 c | 0.100 d |
| F-test | *** | *** | *** | ** |
| CV (%) | 5.23 | 39.96 | 6.12 | 26.48 |
| Mean (bread wheat) | 61.54 a | 1.28 b | 62.83 a | 0.022 b |
| Mean (durum wheat) | 55.32 b | 5.73 a | 61.05 a | 0.105 a |
| Tryptophan (% d.w.) | QI (%) | |
|---|---|---|
| Bread wheat | ||
| ZP 87/I | 0.138 d | 1.447 b |
| Apache | 0.150 bc | 1.621 a |
| ZP Zemunska rosa | 0.148 c | 1.171 c |
| ZP 224 | 0.159 a | 1.352 b |
| ZP Zlatna | 0.141 cd | 1.154 c |
| F-test | ** | *** |
| CV (%) | 6.89 | 14.44 |
| Durum wheat | ||
| ZP 34/I | 0.154 d | 1.268 b |
| ZP 10/I | 0.138 e | 1.245 b |
| ZP DSP/01 | 0.172 b | 1.553 a |
| Varano | 0.163 c | 1.319 b |
| ZP 7858 | 0.186 a | 1.496 a |
| F-test | *** | ** |
| CV (%) | 10.66 | 9.94 |
| Mean (bread wheat) | 0.147 b | 1.349 a |
| Mean (durum wheat) | 0.163 a | 1.376 a |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Žilić, S.; Barać, M.; Pešić, M.; Dodig, D.; Ignjatović-Micić, D. Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes. Int. J. Mol. Sci. 2011, 12, 5878-5894. https://doi.org/10.3390/ijms12095878
Žilić S, Barać M, Pešić M, Dodig D, Ignjatović-Micić D. Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes. International Journal of Molecular Sciences. 2011; 12(9):5878-5894. https://doi.org/10.3390/ijms12095878
Chicago/Turabian StyleŽilić, Slađana, Miroljub Barać, Mirjana Pešić, Dejan Dodig, and Dragana Ignjatović-Micić. 2011. "Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes" International Journal of Molecular Sciences 12, no. 9: 5878-5894. https://doi.org/10.3390/ijms12095878
APA StyleŽilić, S., Barać, M., Pešić, M., Dodig, D., & Ignjatović-Micić, D. (2011). Characterization of Proteins from Grain of Different Bread and Durum Wheat Genotypes. International Journal of Molecular Sciences, 12(9), 5878-5894. https://doi.org/10.3390/ijms12095878






