Biodegradable Metals for Cardiovascular Stent Application: Interests and New Opportunities
Abstract
:1. Rational and History
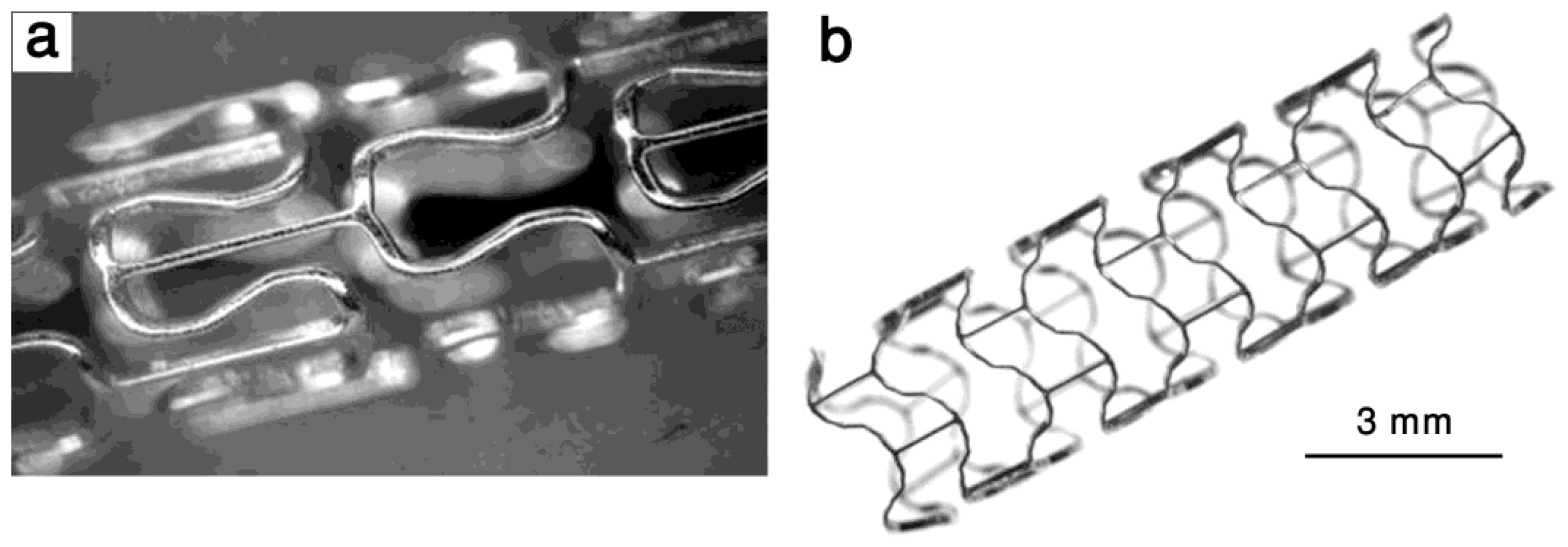
2. Coronary Stents
3. Biodegradable Stents
3.1. Iron-Based Biodegradable Stents
3.2. Magnesium-Based Biodegradable Stents
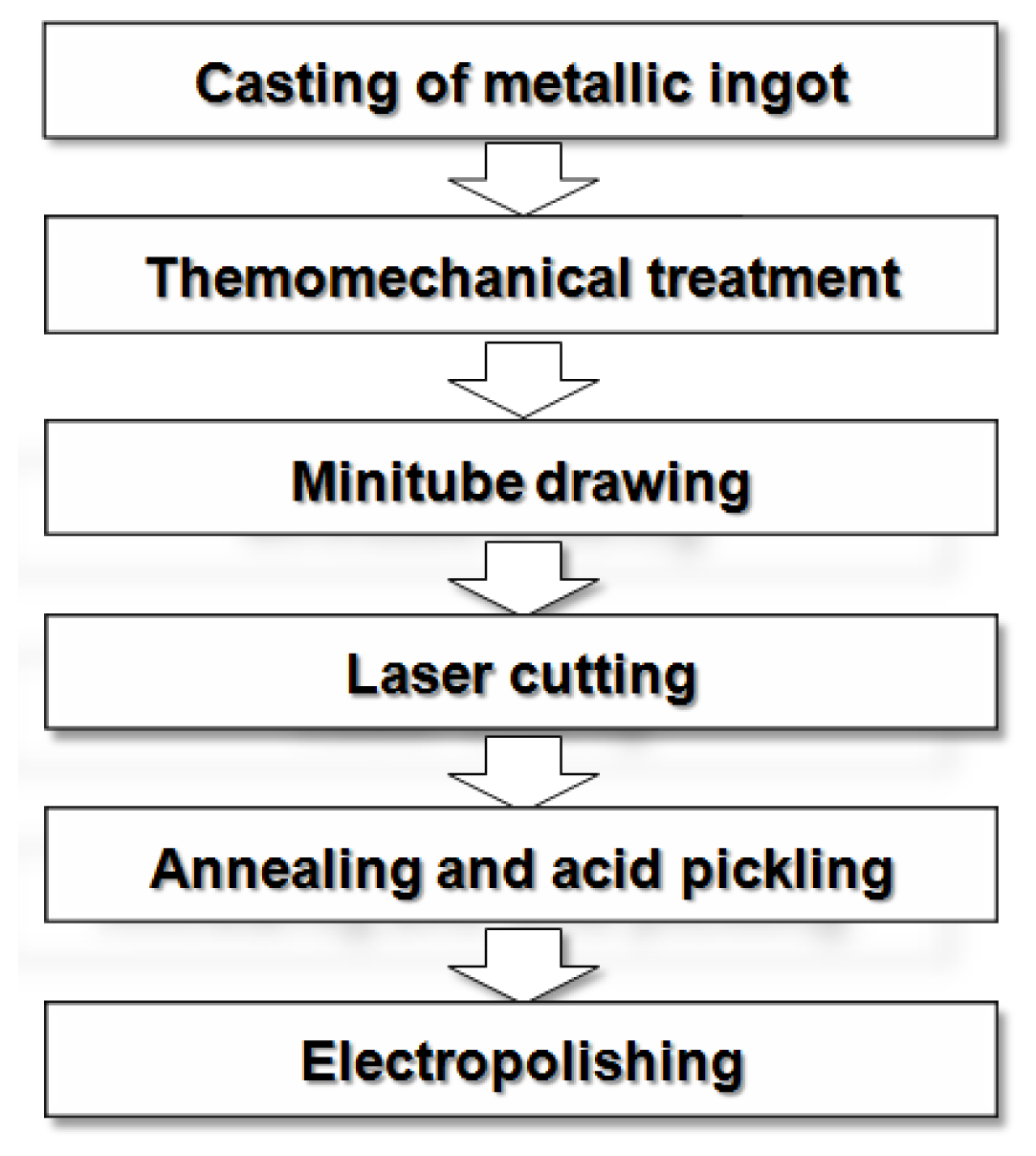
4. Fabrication Process for Biodegradable Stents
4.1. New Fabrication Processes
4.1.1. Powder Metallurgy
4.1.2. Electroforming
5. Conclusive Remarks
Acknowledgement
References
- Bhat, SV. Biomaterials; Kluwer Academic Publishers: Boston, MT, USA, 2002; p. 265. [Google Scholar]
- Park, JB; Lakes, RS. Biomaterials an Introduction, 3rd ed; Springer SpringerLink (Service en ligne): New York, NY, USA, 2007; p. 561. [Google Scholar]
- Witte, F. The history of biodegradable magnesium implants: a review. Acta Biomater 2010, 6, 1680–1692. [Google Scholar]
- Webster, TJ. Nanotechnology Enabled in Situ Sensors for Monitoring Health; Springer Verlag: New York, NY, USA, 2010. [Google Scholar]
- Schulz, MJ; Shanov, VN; Yun, Y. Nanomedicine Design of Particles, Sensors, Motors, Implants, Robots, and Devices; Artech House: Boston, MT, USA, 2009. [Google Scholar]
- Moore, JE; Zouridakis, G. Biomedical Technology and Devices Handbook; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Chan, AW; Moliterno, DJ. In-stent restenosis: update on intracoronary radiotherapy. Cleve. Clin. J. Med 2001, 68, 796–803. [Google Scholar]
- Saito, S. New horizon of bioabsorbable stent. Catheter. Cardiovasc. Interv 2005, 66, 595–596. [Google Scholar]
- Erne, P; Schier, M; Resink, TJ. The road to bioabsorbable stents: reaching clinical reality? Cardiovasc. Interv. Radiol 2006, 29, 11–16. [Google Scholar]
- Peuster, M; Wohlsein, P; Brugmann, M; Ehlerding, M; Seidler, K; Fink, C; Brauer, H; Fischer, A; Hausdorf, G. A novel approach to temporary stenting: degradable cardiovascular stents produced from corrodible metal-results 6–18 months after implantation into New Zealand white rabbits. Heart 2001, 86, 563–569. [Google Scholar]
- Colombo, A; Karvouni, E. Biodegradable stents: “fulfilling the mission and stepping away”. Circulation 2000, 102, 371–373. [Google Scholar]
- Hermawan, H; Dube, D; Mantovani, D. Developments in metallic biodegradable stents. Acta Biomater 2010, 6, 1693–1697. [Google Scholar]
- Mani, G; Feldman, MD; Patel, D; Agrawal, CM. Coronary stents: A materials perspective. Biomaterials 2007, 28, 1689–1710. [Google Scholar]
- Peuster, M; Hesse, C; Schloo, T; Fink, C; Beerbaum, P; von Schnakenburg, C. Long-term biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta. Biomaterials 2006, 27, 4955–4962. [Google Scholar]
- Waksman, R; Pakala, R; Baffour, R; Seabron, R; Hellinga, D; Tio, FO. Short-term effects of biocorrodible iron stents in porcine coronary arteries. J. Interv. Cardiol 2008, 21, 15–20. [Google Scholar]
- Peuster, M; Beerbaum, P; Bach, FW; Hauser, H. Are resorbable implants about to become a reality? Cardiol. Young 2006, 16, 107–116. [Google Scholar]
- ASTM F138-08, Standard Specification for Wrought 18 Chromium-14 Nickel-2.5 Molybdenum Stainless Steel Bar and Wire for Surgical Implants; ASTM International: West Conshohocken, PA, USA, 2008. [CrossRef]
- Hermawan, H; Moravej, M; Dubé, D; Fiset, M; Mantovani, D. Degradation behaviour of metallic biomaterials for degradable stents. Adv Mater Res 2007, 15–17, 113–118. [Google Scholar]
- Hermawan, H; Dube, D; Mantovani, D. Development of degradable Fe-35Mn alloy for biomedical application. Adv. Mater. Res 2007, 15, 107–112. [Google Scholar]
- Hermawan, H; Alamdari, H; Mantovani, D; Dube, D. Iron-manganese: new class of metallic degradable biomaterials prepared by powder metallurgy. Powder Metall 2008, 51, 38–45. [Google Scholar]
- Hermawan, H; Dube, D; Mantovani, D. Degradable metallic biomaterials: design and development of Fe-Mn alloys for stents. J. Biomed. Mater. Res. A 2010, 93, 1–11. [Google Scholar]
- Schinhammer, M; Hanzi, AC; Loffler, JF; Uggowitzer, PJ. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomater 2010, 6, 1705–1713. [Google Scholar]
- Moravej, M; Prima, F; Fiset, M; Mantovani, D. Electroformed iron as new biomaterial for degradable stents: Development process and structure-properties relationship. Acta Biomater 2010, 6, 1726–1735. [Google Scholar]
- Moravej, M; Purnama, A; Fiset, M; Couet, J; Mantovani, D. Electroformed pure iron as a new biomaterial for degradable stents: In vitro degradation and preliminary cell viability studies. Acta Biomater 2010, 6, 1843–1851. [Google Scholar]
- Liu, B; Zheng, Y. Effects of alloying elements (Mn, Co, Al, W, Sn, B, C and S) on biodegradability and in vitro biocompatibility of pure iron. Acta Biomater 2010, 7, 1407–1420. [Google Scholar]
- Liu, B; Zheng, Y; Ruan, L. In vitro investigation of Fe30Mn6Si shape memory alloy as potential biodegradable metallic material. Mater. Lett 2010, 65, 540–543. [Google Scholar]
- Nie, F; Zheng, Y; Wei, S; Hu, C; Yang, G. In vitro corrosion, cytotoxicity and hemocompatibility of bulk nanocrystalline pure iron. Biomed. Mater 2010, 5, 065015. [Google Scholar]
- Hermawan, H; Purnama, A; Dube, D; Couet, J; Mantovani, D. Fe-Mn alloys for metallic biodegradable stents: Degradation and cell viability studies. Acta Biomater 2010, 6, 1852–1860. [Google Scholar]
- ASTM F756-08, Standard Practice for Assessment of Hemolytic Properties of Materials; ASTM International: West Conshohocken, PA, USA, 2008. [CrossRef]
- Moravej, M; Amira, S; Prima, F; Rahem, A; Fiset, M; Mantovani, D. Effect of electrodeposition current density on the microstructure and the degradation of electroformed iron for degradable stents. Mater Sci Eng B 2011. [Google Scholar] [CrossRef]
- Xu, L; Yu, G; Zhang, E; Pan, F; Yang, K. In vivo corrosion behavior of Mg Mn Zn alloy for bone implant application. J. Biomed. Mater. Res. A 2007, 83, 703–711. [Google Scholar]
- Niemeyer, M. Magnesium Alloys as Biodegradable Metallic Implant Materials. Proceedings of 7th Conference on Advanced Materials and Processes, Rimini, Italy; 2001. [Google Scholar]
- Heublein, B; Rohde, R; Niemeyer, M; Kaese, V; Hartung, W; Rocken, C. Degradation of metallic alloys-A new principle in stent technology? J. Am. Coll. Cardiol 2000, 35, 14a–15a. [Google Scholar]
- Heublein, B; Rohde, R; Kaese, V; Niemeyer, M; Hartung, W; Haverich, A. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology? Heart 2003, 89, 651–656. [Google Scholar]
- Di Mario, C; Griffiths, H; Goktekin, O; Peeters, N; Verbist, J; Bosiers, M; Deloose, K; Heublein, B; Rohde, R; Kasese, V; Ilsley, C; Erbel, R. Drug-eluting bioabsorbable magnesium stent. J. Interv. Cardiol 2004, 17, 391–395. [Google Scholar]
- Peeters, P; Bosiers, M; Verbist, J; Deloose, K; Heublein, B. Preliminary results after application of absorbable metal stents in patients with critical limb ischemia. J. Endovasc. Ther 2005, 12, 1–5. [Google Scholar]
- Bosiers, M. AMS INSIGHT—absorbable metal stent implantation for treatment of below-the-knee critical limb ischemia: 6-month analysis. Cardiovasc. Interv. Radiol 2009, 32, 424–435. [Google Scholar]
- Zartner, P; Cesnjevar, R; Singer, H; Weyand, M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter. Cardiovasc. Interv 2005, 66, 590–594. [Google Scholar]
- Waksman, R; Pakala, R; Kuchulakanti, PK; Baffour, R; Hellinga, D; Seabron, R; Tio, FO; Wittchow, E; Hartwig, S; Harder, C; Rohde, R; Heublein, B; Andreae, A; Waldmann, KH; Haverich, A. Safety and efficacy of bioabsorbable magnesium alloy stents in porcine coronary arteries. Catheter. Cardiovasc. Interv 2006, 68, 607–617. [Google Scholar]
- Erbel, R; Di Mario, C; Bartunek, J; Bonnier, J; de Bruyne, B; Eberli, FR; Erne, P; Haude, M; Heublein, B; Horrigan, M. Temporary scaffolding of coronary arteries with bioabsorbable magnesium stents: a prospective, non-randomised multicentre trial. Lancet 2007, 369, 1869–1875. [Google Scholar]
- Gu, XN; Zheng, YF. A review on magnesium alloys as biodegradable materials. Front. Mater. Sci. China 2010, 4, 111–115. [Google Scholar]
- Levesque, J; Hermawan, H; Dube, D; Mantovani, D. Design of a pseudo-physiological test bench specific to the development of biodegradable metallic biomaterials. Acta Biomater 2008, 4, 284–295. [Google Scholar]
- Hanzi, AC; Sologubenko, AS; Uggowitzer, PJ. Design strategy for microalloyed ultra-ductile magnesium alloys for medical applications. Mater Sci Forum 2009, 618–619, 75–82. [Google Scholar]
- Hänzi, A; Gunde, P; Schinhammer, M; Uggowitzer, P. On the biodegradation performance of an Mg-Y-RE alloy with various surface conditions in simulated body fluid. Acta Biomater 2009, 5, 162–171. [Google Scholar]
- Hänzi, AC; Gerber, I; Schinhammer, M; Löffler, JF; Uggowitzer, PJ. On the in vitro and in vivo degradation performance and biological response of new biodegradable Mg-Y-Zn alloys. Acta Biomater 2010, 6, 1824–1833. [Google Scholar]
- Lu, P; Fan, H; Liu, Y; Cao, L; Wu, X; Xu, X. Controllable biodegradability; drug release behavior and hemocompatibility of PTX-eluting magnesium stents. Colloids Surf. B Biointerfaces 2010, 83, 23–28. [Google Scholar]
- Klocke, B; Diener, T; Fringes, M; Harder, C. Degradable metal stent having agent-containing coating. US Patent 20090030507, January 2008. [Google Scholar]
- Orlowski, M; Rubben, A. Bioresorbable metal stent with controlled resopption. WO Patent 2008092436, March 2011. [Google Scholar]
- Asgari, S. Biodegradable porous stent. WO Patent 2008098922, August 2008. [Google Scholar]
- Waksman, R. Current state of the absorbable metallic (magnesium) stent. Euro. Interv. Suppl 2009, 5, F94–F98. [Google Scholar]
- Kondyurin, A; Kondyurina, I; Bilek, M. Biodegradable drug eluting coating of cardiovascular stents dewets and can cause thrombosis. Avaiable online: http://arxiv.org/abs/1101.0659 accessed on 23 June 2011.
- Feyerabend, F; Fischer, J; Holtz, J; Witte, F; Willumeit, R; Drucker, H; Vogt, C; Hort, N. Evaluation of short-term effects of rare earth and other elements used in magnesium alloys on primary cells and cell lines. Acta Biomater 2010, 6, 1834–1842. [Google Scholar]
- Ratner, BD. Biomaterials Science: An Introduction to Materials in Medicine; Elsevier Academic Press: Amsterdam, The Netherlands; Boston, MT, USA, 2004. [Google Scholar]
- Callister, WD; Rethwisch, DG. Materials Science and Engineering: An Introduction, 8th ed; John Wiley Press: Hoboken, NJ, USA, 2009. [Google Scholar]
- Mitchell, BS. An Introduction to Materials Engineering and Science for Chemical and Materials Engineers; John Wiley Press: Hoboken, NJ, USA, 2004. [Google Scholar]
- Poncin, P; Proft, J. Stent Tubing: Understanding the Desired Attributes. Proceedings of Materials & Processes for Medical Devices Conference; The American Society For Microbiology: Anaheim, CA, USA, 2003. [Google Scholar]
- Wu, MH. Fabrication of nitinol materials and components. Shape Mem Mater Appl-Mater Sci Forum 2002, 394–395, 285–292. [Google Scholar]
- Meyer-Kobbe, C; Hinrichs, B. Why the annealing of 316LVM-Stents is so important. Available online: http://www.meko.de/downloads/316LVM.pdf accessed on 5 October 2010.
- Haidopoulos, M; Turgeon, S; Sarra-Bournet, C; Laroche, G; Mantovani, D. Development of an optimized electrochemical process for subsequent coating of 316 stainless steel for stent applications. J. Mater. Sci. Mater. M 2006, 17, 647–657. [Google Scholar]
- Hermawan, H. Conception, Développement et Validation d’Alliages Métalliques Biodégradables pour Emploi dans le Domaine de la Chirurgie Endovasculaire. PhD Thesis, Université Laval, Quebec, Canada, May 2009. [Google Scholar]
- Schlesinger, M; Paunovic, M. Modern Electroplating, 4th ed; Wiley Press: New York, NY, USA, 2000; p. 868. [Google Scholar]
- Schlesinger, M. Electrochemistry and Medical Devices Friend or Foe? Electrochem. Soc. Interface 2003, 12, 20–24. [Google Scholar]
- Koch, CC. Nanostructured Materials Processing, Properties, and Applications; William Andrew Publisher: Norwich, NY, USA, 2007. [Google Scholar]
- MacGeough, JA; Leu, MC; Rajurkar, KP; De Silva, AKM; Liu, Q. Electroforming process and application to micro/macro manufacturing. CIRP Ann. Manuf. Technol 2001, 50, 499–514. [Google Scholar]
- Hart, T; Watson, A. Electroforming. Met. Finish 2007, 105, 331–341. [Google Scholar]
- Colombo, A; Stankovic, G; Moses, J. Selection of coronary stents. J. Am. Coll. Cardiol 2002, 40, 1021–1033. [Google Scholar]
- Stöver, M; Renke-Gluszko, M; Schratzenstaller, T; Will, J; Klink, N; Behnisch, B; Kastrati, A; Wessely, R; Hausleiter, J; Schömig, A. Microstructuring of stainless steel implants by electrochemical etching. J. Mater. Sci 2006, 41, 5569–5575. [Google Scholar]
- Zhao, H; Humbeeck, J; Sohier, J; Scheerder, I. Electrochemical polishing of 316L stainless steel slotted tube coronary stents. J. Mater. Sci. Mater. Med 2002, 13, 911–916. [Google Scholar]





| Material | Yield Strength (MPa) | Tensile Strength (MPa) | Elongation (%) | In vitro Degradation Rate (mm y−1) * | Average Grain Size (μm) |
|---|---|---|---|---|---|
| 316L SS: annealed (ASTM F138) [17] | 190 | 490 | 40 | - | 12–30 |
| Armco® Fe: annealed [13,18] | 150 | 200 | 40 | 0.19 | 40 |
| Fe-35Mn alloy: annealed [19–21] | 230 | 430 | 30 | 0.44 | <100 |
| Fe-10Mn-1Pd alloy: heat treated [22] | 850–950 | 1450–1550 | 2–8 | - | - |
| Electroformed Fe: annealed at 550 °C [23,24] | 270 | 290 | 18 | 0.46–1.22 | 2–8 |
| Fe alloyed by different elements (Mn, Co, Al, W, Sn, B, C and S): as cast [25] | 100–220 | 190–360 | 12–23 | 0.10–0.17 | 100–400 |
| Fe-30Mn-6Si alloy: solution treated [26] | 180 | 450 | 16 | 0.30 | <100 |
| Nanocrystalline Fe: ECAP, 8 passes [27] | - | 250–450 | - | 0.09–0.2 | 0.08–0.20 |
| Material | Yield Strength (MPa) | Tensile Strength (MPa) | Elongation (%) | In vitro Degradation Rate (mm y−1) * | Average Grain Size (μm) |
|---|---|---|---|---|---|
| 316L SS: annealed (ASTM F138) [17] | 190 | 490 | 40 | - | 12–30 |
| Pure Mg: as cast [18,41] | 20 | 86 | 13 | 407 | - |
| WE43 alloy: extruded T5 [41] | 195 | 280 | 2 | 1.35 | 10 |
| AM60B-F: die cast [18,41,42] | - | 220 | 6–8 | 8.97 | 25 |
| ZW21: extruded [43–45] | 200 | 270 | 17 | - | 4 |
| WZ21: extruded [43–45] | 140 | 250 | 20 | - | 7 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moravej, M.; Mantovani, D. Biodegradable Metals for Cardiovascular Stent Application: Interests and New Opportunities. Int. J. Mol. Sci. 2011, 12, 4250-4270. https://doi.org/10.3390/ijms12074250
Moravej M, Mantovani D. Biodegradable Metals for Cardiovascular Stent Application: Interests and New Opportunities. International Journal of Molecular Sciences. 2011; 12(7):4250-4270. https://doi.org/10.3390/ijms12074250
Chicago/Turabian StyleMoravej, Maryam, and Diego Mantovani. 2011. "Biodegradable Metals for Cardiovascular Stent Application: Interests and New Opportunities" International Journal of Molecular Sciences 12, no. 7: 4250-4270. https://doi.org/10.3390/ijms12074250
APA StyleMoravej, M., & Mantovani, D. (2011). Biodegradable Metals for Cardiovascular Stent Application: Interests and New Opportunities. International Journal of Molecular Sciences, 12(7), 4250-4270. https://doi.org/10.3390/ijms12074250




