Use of Brassica Plants in the Phytoremediation and Biofumigation Processes
Abstract
:1. Introduction
- the dynamic growth of industry and transport;
- the non-rational use of pesticides in agriculture, of communal and industrial wastes, and of wastes for de-acidifying soils;
- human causes.
2. Sources of Emission and Pathways of Environmental Contamination by Heavy Metals and Phytoremediation
- the contamination is frequently heterogeneous at the macroscale and microscale;
- the metals cannot be degraded biologically, but only transformed from one oxidation state or organic complex to another;
- the variability of metal forms and the soil matrix influence the environmental risk assessment and the soil treatment feasibility [8].
- reduce an existing or potential environmental hazard;
- lower the potential threat from an unacceptable level to so-called ‘safe levels’.
- ➢ the concentration of heavy metals in the shoots should be 50–100 times greater than in ‘normal’ plants [19];
- ➢ the bioaccumulation coefficient (the ratio of the concentration of a toxic substance in the tissues of an organism to its concentration in the living environment of that organism) must have a value greater than 1 [20];
- ➢ metal concentrations in the shoots should be higher than in the roots [19];
- ➢ fast growth and high accumulating biomass [6];
- ➢ easily grown as an agricultural crop and fully harvestable [6].
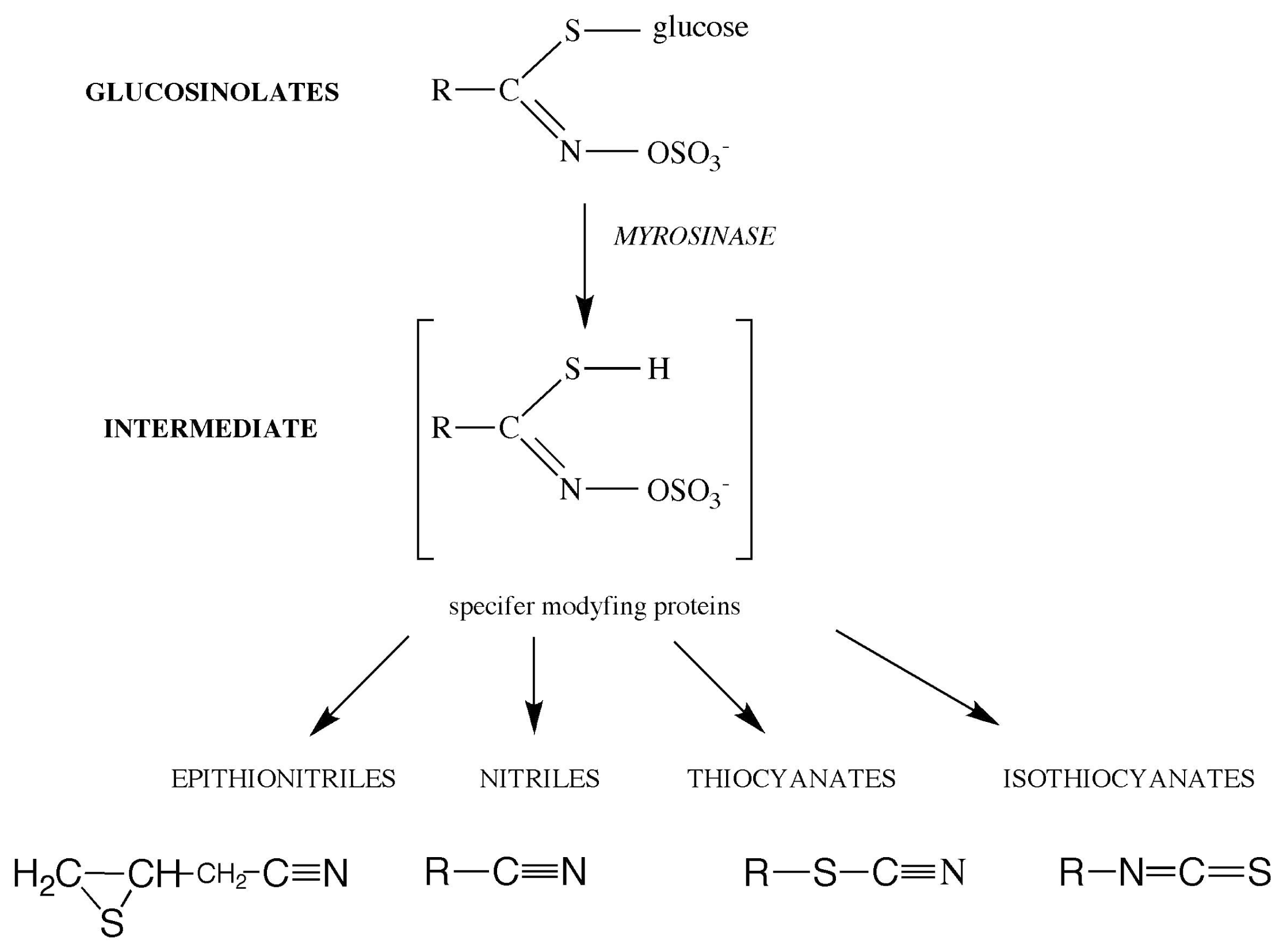
3. Biofumigation as an Alternative Method of Crop Protection
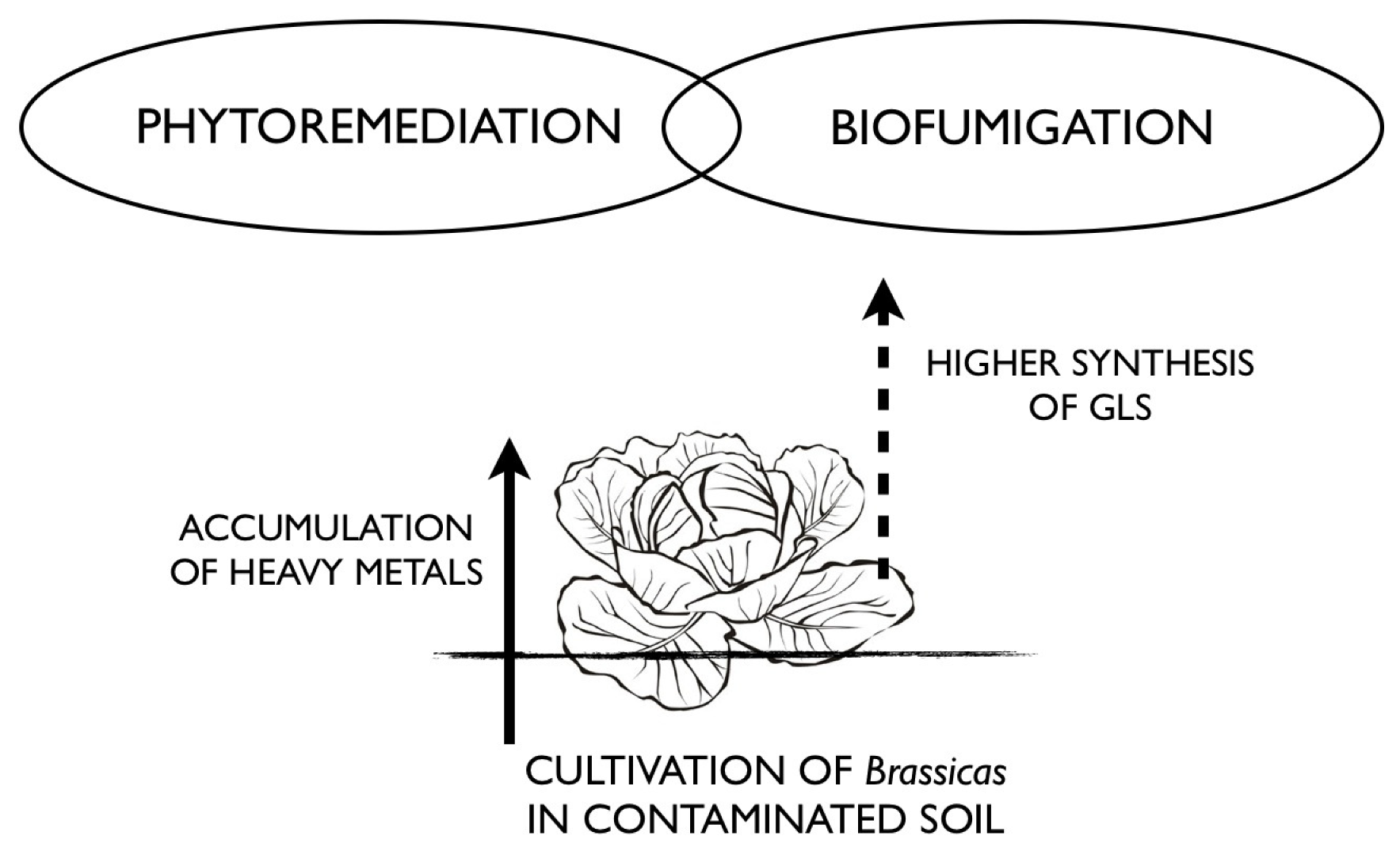
4. Possibilty of Joining The Phytoremediation and Biofumigation Processes
- grows quickly to produce a large biomass;
- is tolerant to environmental contaminants;
- contains metal-binding compounds such as glutathion, phytochelatin and metallothionein proteins;
- has an extensive root system;
- is an undemanding crop plant;
- is easy to harvest;
- has a large biomass enclosed in a small, compact spherical head, where the accumulated contaminants are safely stored until disposal. Even if a cabbage withers, it retains its structure and the dead leaves do not crumble, so there is no danger of the accumulated heavy metals returning to the environment;
- contains large amounts of glucosinolate sinigrin [47], the hydrolysis product of which is allyl isothiocyanate, a very effective biocide important in the biofumigation process.
5. Conclusions
Acknowledgements
References
- Gruca-Królikowska, S.; Wacławek, W. Metale w środowisku. Chemia Dydaktyka Ekologia Metrologia 2006, 11, 41–55. [Google Scholar]
- Chhotu, J.D.; Fulekar, M.H. Phytoremediation of heavy metals: Recent techniques. Afr. J. Biotechnol 2009, 8, 921–928. [Google Scholar]
- Salt, D.E.; Smith, R.D.; Raskin, I. Phytoremediation. Annu. Rev. Plant Physiol. Plant Mol. Biol 1998, 49, 643–668. [Google Scholar]
- Pilon-Smits, E. Phytoremediation. Annu. Rev. Plant Biol 2005, 56, 15–39. [Google Scholar]
- Beyer, A.; Bizuik, M. Przegląd metod oznaczania pozostałości pestycydów i polichlorowanych bifenyli w próbkach żwyności. Ecol. Chem. Eng 2007, 14, 291–313. [Google Scholar]
- Marchiol, L.; Assolari, S.; Sacco, P.; Zerbi, G. Phytoextraction of heavy metals by canola (Brassica napus) and radish (Raphanus sativus) grown on multicontaminated soil. Environ. Pollut 2004, 132, 21–27. [Google Scholar]
- Yang, X.; Feng, Y.; He, Z.; Stoffella, P.J. Molecular mechanisms of heavy metals hyperaccumulation and phytoremediation. J. Trace Elem. Med. Biol 2005, 18, 339–353. [Google Scholar]
- Dermont, G.; Bergeron, M.; Mercier, G.; Richer-Laflèche, M. Metal-contaminated soils: Eemediation practices and treatment technologies. J. Hazard. Toxic Radioact. Waste 2008, 12, 188–209. [Google Scholar]
- Lebeau, T.; Braud, A.; Jézéquel, K. Performance of bioaugmentation-assisted phytoextraction applied to metal contaminated soils: A review. Environ. Pollut 2008, 153, 497–522. [Google Scholar]
- Rascio, N.; Navari-Izzo, F. Heavy metal hyperaccumulating plants: How and why do they do it? And what makes them so interesting? Plant Sci 2011, 180, 169–181. [Google Scholar]
- Pulford, I.D.; Watson, C. Phytoremediation of heavy metal-contaminated land by trees—A review. Environ. Int 2003, 29, 529–540. [Google Scholar]
- Pilon-Smits, E.; Pilon, M. Breeding mercury-breathing plants for environmental cleanup. Trends Plant. Sci 2000, 5, 235–236. [Google Scholar]
- Garbisu, C.; Alkorta, I. Phytoextraction: A cost-effective plant-based technology for the removal of metals from the environment. Bioresour. Technol 2001, 77, 229–236. [Google Scholar]
- Alkorta, I.; Hernandez-Allica, J.; Becerril, J.M.; Amezaga, I.; Albizu, I.; Garbisu, C. Recent findings on the phytoremediation of soils contaminated with environmentally toxic heavy metals and metalloids such as zinc, cadmium, lead, and arsenic. Rev. Environ. Sci. Biotech 2004, 3, 71–90. [Google Scholar]
- Gardea-Torresdey, J.; Peralta-Videa, J.; Parsons, J.G. Phytoremediation of heavy metals and sudy of the metal coordination by X-ray absorption spectroscopy. Coord. Chem. Rev 2005, 249, 1797–1810. [Google Scholar]
- Evangelou, M.; Mathias-Ebel, M.; Andreas-Schaeffer, A. Chelate assisted phytoextraction of heavy metals from soil. Effect, mechanism, toxicity, and fate of chelating agents. Chemosphere 2007, 68, 989–1003. [Google Scholar]
- Nouairi, I.; Wided Ben Ammar, W.; Youssef, N.; Douja Ben Miled Daoud, D.B.; Habib Ghorbal, M.; Zarrouk, M. Comparative study of cadmium effects on membrane lipid composition of Brassica juncea and Brassica napus leaves. Plant Sci 2006, 170, 511–519. [Google Scholar]
- Zhao, F.J.; McGrath, S.P. Biofortification and phytoremediation. Curr. Opin. Plant Biol 2009, 12, 373–380. [Google Scholar]
- Jabeen, R.; Ahmad, A.; Iqbal, M. Phytoremediation of heavy metals: Physiological and molecular mechanisms. Bot. Rev 2009, 75, 339–364. [Google Scholar]
- McGrath, S.P.; Zhao, F.J. Phytoextraction of metals and metalloids from contaminated soils. Curr. Opin. Biotechnol. 2003, 14, 277–282. [Google Scholar]
- Vamerali, T.; Bandiera, M.; Mosca, D. Field crops for phytoremediation of metal-contaminated land. A review. Environ Chem Lett 2010, 8, 1–17. [Google Scholar]
- Gisbert, C.; Clemente, R.; Navarro-Aviñó, J.; Carlos Baixauli, C.; Ginér, A.; Serrano, R.; Walker, D.J.; Pilar Bernal, M.P. Tolerance and accumulation of heavy metals by Brassicaceae species grown in contaminated soils from Mediterranean regions of Spain. Environ. Exp. Bot 2006, 56, 19–26. [Google Scholar]
- Clarke, D. Glucosinolates, structure and analysis in food. Anal. Methods 2010, 4, 301–416. [Google Scholar]
- Kirkegaard, J.; Sarwar, M. Biofumigation potential of brassicas. Plant Soil 1998, 201, 71–89. [Google Scholar]
- Fahey, J.; Zalcmann, A.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar]
- Smolińska, U.; Morra, M.; Knudsen, G.; James, R. Isothiocyanates produced by Brassicaceae species as inhibitors of Fusarium oxysporum. Plant Dis 2003, 8, 407–412. [Google Scholar]
- Manici, L.; Lazzeri, L.; Baruzzi, G.; Leoni, O.; Galletti, S.; Palmieri, S. Suppressive activity of some glucosinolate enzyme degradation products on Pythium irregulare and Rhizoctonia solani in sterile soil. Pest Manag. Sci 2000, 56, 921–926. [Google Scholar]
- Manici, L.; Lazzeri, L.; Palmieri, S. In vitro fungitoxic activity of some glucosinolates and their enzyme-derived products toward plant pathogenic fungi. J. Agric. Food Chem 1997, 45, 2768–2773. [Google Scholar]
- Mari, M.; Leoni, O.; Bernardi, R.; Neri, F.; Palmieri, S. Control of brown rot on stonefruit by synthetic and glucosinolate-derived isothiocyanates. Postharvest Biol. Technol 2008, 47, 61–67. [Google Scholar]
- Mayton, H.; Olivier, C.; Vaughn, S.; Loria, R. Correlation of fungicidal activity of Brassica species with allyl isothicyanate production in macerated leaf tissue. Dis. Control Pest Manag 1996, 86, 267–271. [Google Scholar]
- Lazzeri, L.; Leoni, O.; Manici, L. Biocidal plant dried pellets for biofumigation. Ind. Crops Prod 2004, 20, 59–65. [Google Scholar]
- Larkin, R.; Griffin, T. Control of soilborne potato diseases using Brassica green manure. Crop Prot 2007, 26, 1067–1077. [Google Scholar]
- Lord, J.; Lazzeri, L.; Atkinson, H.; Urwin, P. Biofumigation for control of pale potato cyst nematodes: Activity of Brassica leaf extracts and green manures on Globodera pallida in vitro and in soil. J. Agric. Food Chem 2011, 59, 7882–7890. [Google Scholar]
- Aires, A.; Carvalho, R.; Barbosa, M.; Rosa, E. Suppressing potato cyst nematode, Globodera rostochiensis, with extracts of Brassicaceae plants. Am. J. Potato Res 2009, 86, 327–333. [Google Scholar]
- Lin, C.; Preston, J.; Wei, C. Antibacterial mechanism of allyl isothiocyanate. J. Food Prot 2000, 6, 727–734. [Google Scholar]
- Ward, S.; Delaquis, P.; Holleya, R.; Mazza, G. Inhibition of spoilage and pathogenic bacteria on agar and pre-cooked roast beef by volatile horseradish distillates. Food Res. Int 1998, 31, 19–26. [Google Scholar]
- Lazzeri, L.; Curto, G.; Leoni, O.; Dallavalle, E. Effects of glucosinolates and their enzymatic hydrolysis products via myrosinase on the root-knot nematode Meloidogyne incognita (Kofoid et White) Chitw. J. Agric. Food Chem 2004, 52, 6703–6707. [Google Scholar]
- Lazzeri, L.; Tacconi, R.; Palmieri, S. In vitro activity of some glucosinolates and their reaction products toward a population of the nematode Heterodera schachtii. J. Agric. Food Chem 1993, 41, 825–829. [Google Scholar]
- Lugauskas, A.; Repeškienė, J.; Uselis, N.; Račinskienė, A. Problems on a longtime strawberry growing in one plot. Hortorum Cultus 2003, 2, 59–68. [Google Scholar]
- Mattner, S.; Porter, I.; Gounder, R.; Shanks, A.; Wren, D.; Allen, D. Factors that impact on the ability of biofumigants to suppress fungal pathogens and weeds of strawberry. Crop Prot 2008, 27, 1165–1173. [Google Scholar]
- Gimsing, A.l.; Kirkegaard, J.A. Glucosinolates and biofumigation: Fate of glucosinolates and their hydrolysis products in soil. Phytochem. Rev 2009, 8, 299–310. [Google Scholar]
- Vig, A.P.; Rampa, G.; Thind, T.S.; Arora, S. Bio-protective effects of glucosinolates—A review. LWT 2009, 42, 1561–1572. [Google Scholar]
- Lazzeri, L.; Curto, G.; Dallavalle, E.; D’Avino, L.; Malaguti, I.; Santi, R.; Patalano, G. Nematicidal efficacy of biofumigation by defatted Brassicaceae meal for control of Meloidogyne incognita (Kofoid et White) Chitw. On a full field zucchini crop. J. Sustain. Agric 2009, 33, 349–358. [Google Scholar]
- Williams-Woodward, J.; Pfleger, F.; Fritz, V.; Allmaras, R. Green manures of oat, rape and sweet corn for reducing common root rot in pea (Pisum sativum) caused by Aphanomyces euteiches. Plant Soil 1997, 188, 43–48. [Google Scholar]
- Burgieł, Z. Badania nad możliwością wykorzystania wybranych roślin z rodziny Brassicaceae wochronie ogórka przed zgorzelą siewek powodowaną przez Rhizoctonia solani Kuhn i Fusarium culmorum (W.G. Smith). Sacc. Acta Agrobot 2005, 2, 171–178. [Google Scholar]
- Yulianti, T.; Sivasithamparam, K.; Turner, D. Saprophytic and pathogenic behaviour of R. solani AG2-1(ZG-5) in a soil amended with Diplotaxis tenuifolia or Brassica nigra manures and incubated at different temperatures and soil water content. Plant Soil 2007, 294, 277–289. [Google Scholar]
- Penas, E.; Frias, J.; Martinez-Villaluenga, C.; Vidal-Valverde, C. Bioactive compounds, myrosinase activity and antioxidant capacity of white cabbages grown in different locations of Spain. J. Agric. Food Chem 2011, 59, 3772–3779. [Google Scholar]
- Pongrac, P.; Vogel-Mikus, K.; Regvar, M.; Tolrà, R.; Poschenrieder, C.; Barceló, J. Glucosinolate profiles change during the life cycle and mycorrhizal colonisation in a Cd/Zn hyperaccumulator Thlaspi praecox (Brassicaceae). J. Chem. Ecol 2008, 34, 1038–1044. [Google Scholar]
- Schiavon, M.; Malagoli, M. Role of sulphate and S-rich compounds in heavy metal tolerance and accumulation. In Sulfur Assimilation and Abiotic Stress in Plants; Khan, N.A., Singh, S., Umar, S., Eds.; Springer-Verlag: Berlin, Germany, 2008; pp. 253–269. [Google Scholar]
- Tolrà, R.; Pongrac, P.; Poschenrieder, C.; Vogel-Mikus, K.; Regvar, M.; Barceló, J. Distinctive effects of cadmium on glucosinolate profiles in Cd hyperaccumulator Thlaspi praecox and non-hyperaccumulator Thlaspi arvense. Plant Soil 2006, 288, 333–341. [Google Scholar]
- Tolrà, R.; Poschenrieder, C.; Alonso, R.; Barceló, D.; Barceló, J. Influence of zinc hyperaccumulation on glucosinolates in Thlaspi caerulescens. New Phytol 2001, 151, 621–626. [Google Scholar]
- Coolong, T.W.; Randle, W.M.; Toler, H.D.; Sams, C.E. Zinc availability in hydroponic culture influences glucosinolate concentrations in Brassica rapa. Hortscience 2004, 39, 84–86. [Google Scholar]
- Davis, M.A.; Boyd, R.S. Dynamics of Ni-based defence and organic defences in the Ni hyper- accumulator, Streptanthus polygaloides (Brassicaceae). New Phytol 2000, 146, 211–217. [Google Scholar]
| Advantages | Limitations |
|---|---|
| Applicable to both inorganic and organic contaminants. | Not accessing elements below the root depth. |
| It can be applied in situ. | Management of plant matter after phytoremediation. |
| Reduces the amount of waste going to landfills. | Low bioma. |
| Does not require expensive equipment or highly specialized personnel. | The bioavailability of the pollutants. |
| Phytoremediation is cheaper than conventional remediation methods. | Restricted to sites with low contaminant concentration. |
| Easy to implement and maintain. Plants are a cheap and renewable resource, easily available. | Introduction of inappropriate or invasive plant species should be avoided (non-native species may affect biodiversity). |
| Environmentally friendly, socially accepted. | High concentrations of hazardous materials can be toxic to plans. |
| Less noisy than other remediation methods. | Possibility for contaminants to enter food chain through animal and plant consumption. |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Szczygłowska, M.; Piekarska, A.; Konieczka, P.; Namieśnik, J. Use of Brassica Plants in the Phytoremediation and Biofumigation Processes. Int. J. Mol. Sci. 2011, 12, 7760-7771. https://doi.org/10.3390/ijms12117760
Szczygłowska M, Piekarska A, Konieczka P, Namieśnik J. Use of Brassica Plants in the Phytoremediation and Biofumigation Processes. International Journal of Molecular Sciences. 2011; 12(11):7760-7771. https://doi.org/10.3390/ijms12117760
Chicago/Turabian StyleSzczygłowska, Marzena, Anna Piekarska, Piotr Konieczka, and Jacek Namieśnik. 2011. "Use of Brassica Plants in the Phytoremediation and Biofumigation Processes" International Journal of Molecular Sciences 12, no. 11: 7760-7771. https://doi.org/10.3390/ijms12117760
APA StyleSzczygłowska, M., Piekarska, A., Konieczka, P., & Namieśnik, J. (2011). Use of Brassica Plants in the Phytoremediation and Biofumigation Processes. International Journal of Molecular Sciences, 12(11), 7760-7771. https://doi.org/10.3390/ijms12117760