Metal-Induced Oxidative Stress and Plant Mitochondria
Abstract
:1. Introduction
2. Metal-Induced ROS Production: Interplay between Cytosol and Mitochondria
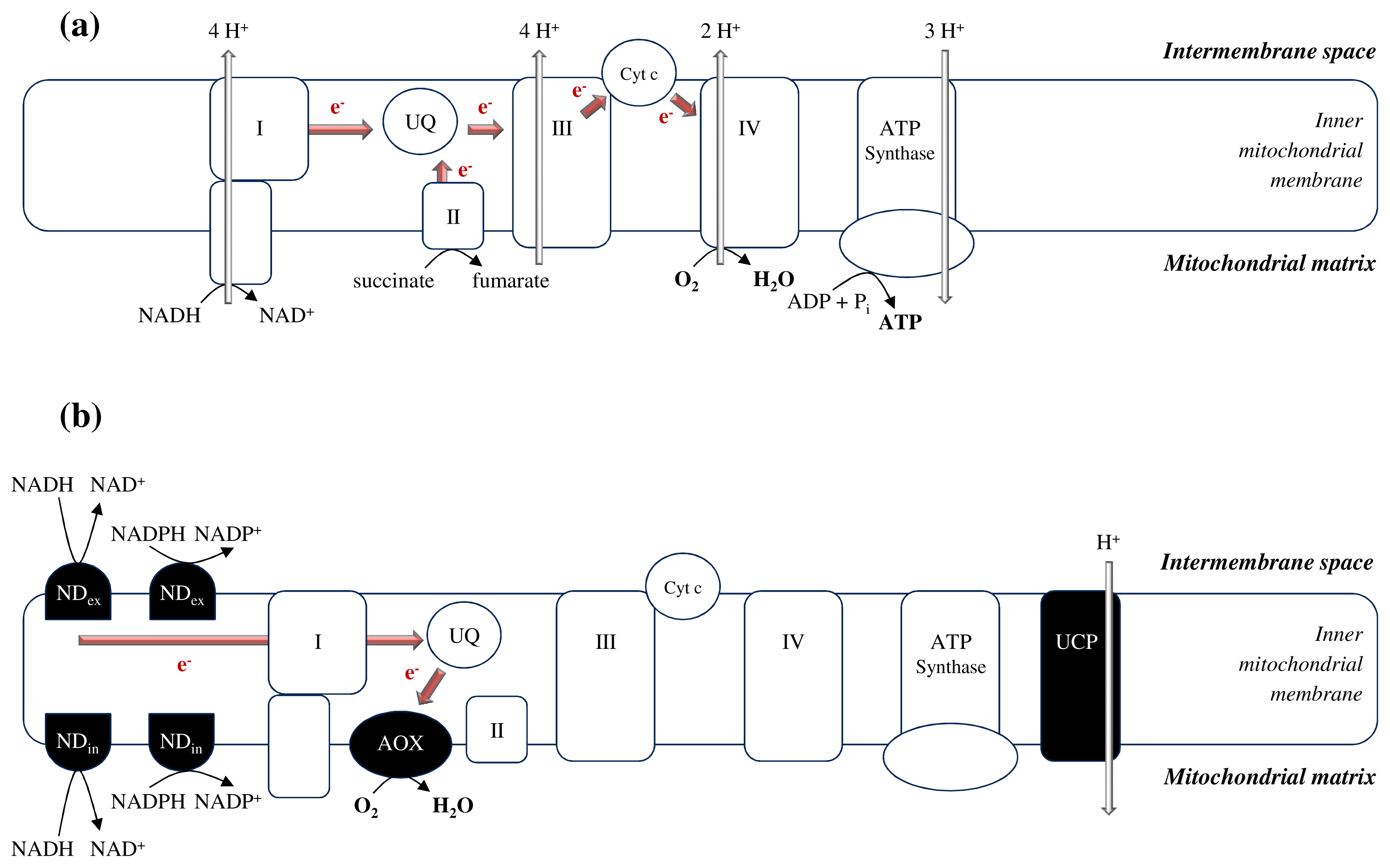
2.1. The Unique Demands Placed on Plant Mitochondria
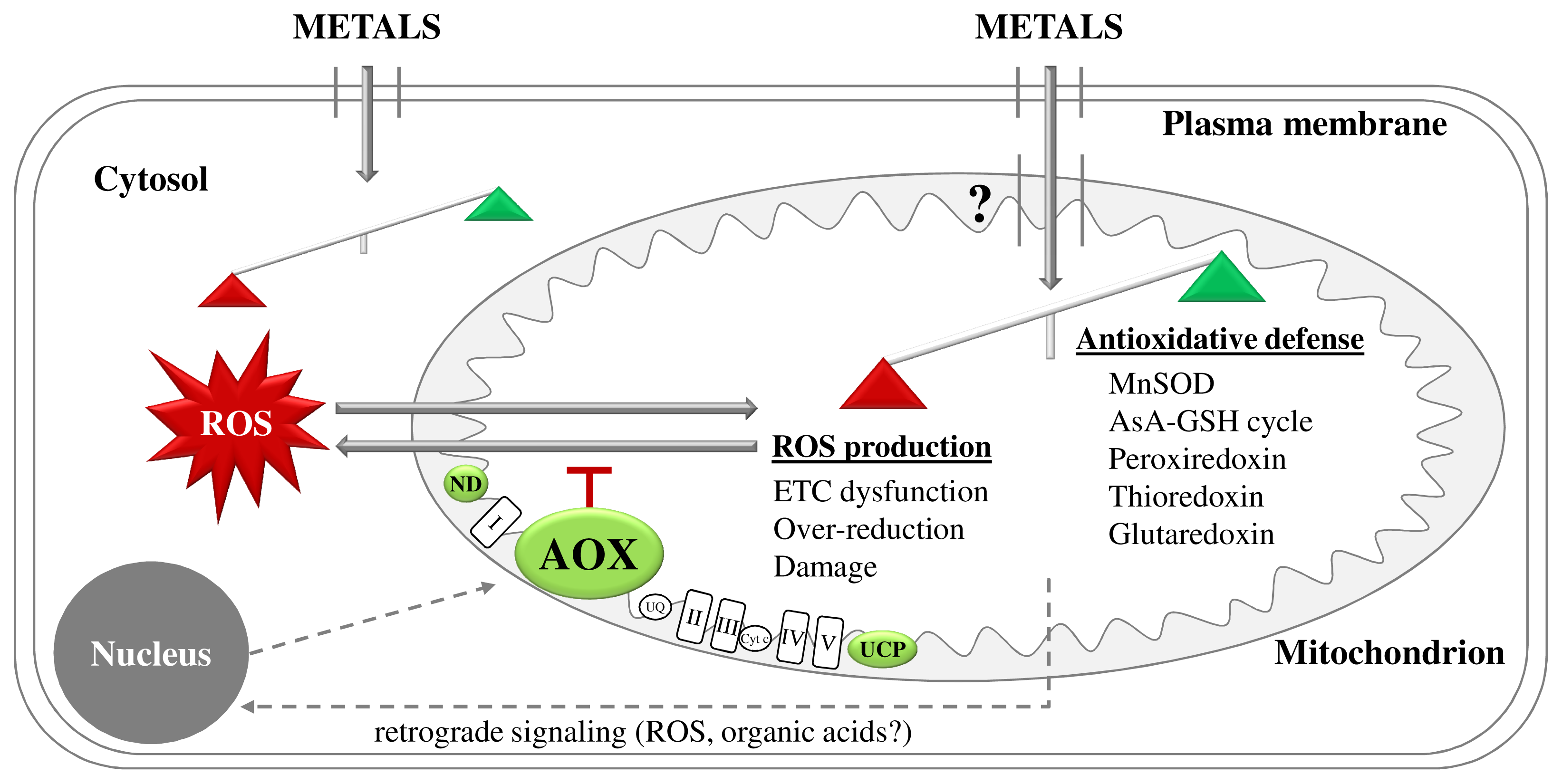
2.2. Metal Exposure Increases ROS Generation in Plant Mitochondria
3. Mechanisms to Control Mitochondrial ROS Production under Metal Stress
3.1. Avoidance of Mitochondrial ROS Production at the ETC Level as a First Line of Defense
3.2. Mitochondrial Enzymes and Metabolites Involved in the Detoxification of Mitochondrial ROS
4. Cellular Acclimation to Metal Exposure in Relation to Plant Mitochondria
4.1. Mitochondrial ROS-Induced Damage
4.2. The Role of Plant Mitochondria in Metal-Induced Programmed Cell Death
4.3. Mitochondrial Retrograde Signaling during Metal Stress in Plants
4.4. Metal Tolerance Mediated by Mitochondrial Organic Acids
5. Conclusions
Acknowledgments
References
- Nriagu, JO; Pacyna, JM. Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 1988, 333, 134–139. [Google Scholar]
- Marschner, H. Functions of Mineral Nutrients: Micronutrients. In Mineral Nutrition of Higher Plants, 2nd ed; Marschner, H, Ed.; Academic Press: London, UK, 1995; pp. 313–404. [Google Scholar]
- Broadley, MR; White, PJ; Hammond, JP; Zelko, I; Lux, A. Zinc in plants. New Phytol 2007, 173, 677–702. [Google Scholar]
- Leonard, SS; Bower, JJ; Shi, X. Metal-induced toxicity, carcinogenesis, mechanisms and cellular responses. Mol. Cell. Biochem 2004, 255, 3–10. [Google Scholar]
- Hogervorst, J; Plusquin, M; Vangronsveld, J; Nawrot, T; Cuypers, A; Van Hecke, E; Roels, HA; Carleer, R; Staessen, JA. House dust as possible route of environmental exposure to cadmium and lead in the adult general population. Environ. Res 2007, 103, 30–37. [Google Scholar]
- Nawrot, T; Plusquin, M; Hogervorst, J; Roels, HA; Celis, H; Thijs, L; Vangronsveld, J; Van Hecke, E; Staessen, JA. Environmental exposure to cadmium and risk of cancer: A prospective population-based study. Lancet Oncol 2006, 7, 119–226. [Google Scholar]
- Thijssen, S; Cuypers, A; Maringwa, J; Smeets, K; Horemans, N; Lambrichts, I; Van Kerkhove, E. Low cadmium exposure triggers a biphasic oxidative stress response in mice kidneys. Toxicology 2007, 236, 29–41. [Google Scholar]
- Chary, NS; Kamala, CT; Raj, DSS. Assessing risk of heavy metals from consuming food grown on sewage irrigated soils and food chain transfer. Ecotoxicol. Environ. Saf 2008, 69, 513–524. [Google Scholar]
- Sharma, SS; Dietz, KJ. The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 2009, 14, 43–50. [Google Scholar]
- Schützendübel, A; Polle, A. Plant responses to abiotic stresses: Heavy metal-induced oxidative stress and protection by mycorrhization. J. Exp. Bot 2002, 53, 1351–1365. [Google Scholar]
- Halliwell, B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 2006, 141, 312–322. [Google Scholar]
- Hao, F; Wang, XC; Chen, J. Involvement of plasma-membrane NADPH oxidase in nickel-induced oxidative stress in roots of wheat seedlings. Plant Sci 2006, 170, 151–158. [Google Scholar]
- Remans, T; Opdenakker, K; Smeets, K; Mathijsen, D; Vangronsveld, J; Cuypers, A. Metal-specific and NADPH oxidase dependent changes in lipoxygenase and NADPH oxidase gene expression in Arabidopsis thaliana exposed to cadmium or excess copper. Funct. Plant Biol 2010, 37, 532–544. [Google Scholar]
- Seth, CS; Remans, T; Keunen, E; Jozefczak, M; Gielen, H; Opdenakker, K; Weyens, N; Vangronsveld, J; Cuypers, A. Phytoextraction of toxic metals: A central role for glutathione. Plant Cell Environ 2011. [Google Scholar] [CrossRef]
- Kučera, T; Horáková, H; Šonská, A. Toxic metal ions in photoautotrophic organisms. Photosynthetica 2008, 46, 481–489. [Google Scholar]
- Noctor, G; De Paepe, R; Foyer, CH. Mitochondrial redox biology and homeostasis in plants. Trends Plant Sci 2007, 12, 125–134. [Google Scholar]
- Sweetlove, LJ; Fait, A; Nunes-Nesi, A; Williams, T; Fernie, AR. The mitochondrion: An integration point of cellular metabolism and signalling. Crit. Rev. Plant Sci 2007, 26, 17–43. [Google Scholar]
- Van Belleghem, F; Cuypers, A; Semane, B; Smeets, K; Vangronsveld, J; d’Haen, J; Valcke, R. Subcellular localization of cadmium in roots and leaves of Arabidopsis thaliana. New Phytol 2007, 173, 495–508. [Google Scholar]
- Nouet, C; Motte, P; Hanikenne, M. Chloroplastic and mitochondrial metal homeostasis. Trends Plant Sci 2011, 16, 395–404. [Google Scholar]
- Dutilleul, C; Garmier, M; Noctor, G; Mathieu, C; Chétrit, P; Foyer, CH; De Paepe, R. Leaf mitochondria modulate whole cell redox homeostasis, set antioxidant capacity, and determine stress resistance through altered signaling and diurnal regulation. Plant Cell 2003, 15, 1212–1226. [Google Scholar]
- Rhoads, DM; Umbach, AL; Subbaiah, CC; Siedow, JN. Mitochondrial reactive oxygen species. Contribution to oxidative stress and interorganellar signaling. Plant Physiol 2006, 141, 357–366. [Google Scholar]
- Van Aken, O; Giraud, E; Clifton, R; Whelan, J. Alternative oxidase: a target and regulator of stress responses. Physiol. Plant 2009, 137, 354–361. [Google Scholar]
- Møller, IM. Plant mitochondria and oxidative stress: electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu. Rev. Plant Physiol. Plant Mol. Biol 2001, 52, 561–591. [Google Scholar]
- Amirsadeghi, S; Robson, CA; Vanlerberghe, GC. The role of the mitochondrion in plant responses to biotic stress. Physiol. Plant 2007, 129, 253–266. [Google Scholar]
- Blokhina, O; Fagerstedt, KV. Reactive oxygen species and nitric oxide in plant mitochondria: origin and redundant regulatory systems. Physiol. Plant 2010, 138, 447–462. [Google Scholar]
- De Gara, L; Locato, V; Dipierro, S; de Pinto, MC. Redox homeostasis in plants. The challenge of living with endogenous oxygen production. Respir. Physiol. Neurobiol 2010, 173, S13–S19. [Google Scholar]
- Tan, YF; O’Toole, N; Taylor, NL; Millar, AH. Divalent metal ions in plant mitochondria and their role in interactions with proteins and oxidative stress-induced damage to respiratory function. Plant Physiol 2010, 152, 747–761. [Google Scholar]
- Lösch, R. Plant Mitochondrial Respiration under the Influence of Heavy Metals. In Heavy Metal Stress in Plants From Biomolecules to Ecosystems, 2nd ed; Prasad, MNV, Ed.; Springer-Verlag: Berlin, Germany, 2004; pp. 182–200. [Google Scholar]
- Kampfenkel, K; Van Montagu, M; Inzé, D. Effects of iron excess on Nicotiana plumbaginifolia plants (implications to oxidative stress). Plant Physiol 1995, 107, 725–735. [Google Scholar]
- Pádua, M; Aubert, S; Casimiro, A; Bligny, R; Millar, AH; Day, DA. Induction of alternative oxidase by excess copper in sycamore cell suspensions. Plant Physiol. Biochem 1999, 37, 131–137. [Google Scholar]
- Dixit, V; Pandey, V; Shyam, R. Chromium ions inactivate electron transport and enhance superoxide generation in vivo in pea (Pisum sativum L. cv. Azad) root mitochondria. Plant Cell Environ 2002, 25, 687–693. [Google Scholar]
- Yamamoto, Y; Kobayashi, Y; Rama Devi, S; Rikiishi, S; Matsumoto, H. Aluminum toxicity is associated with mitochondrial dysfunction and the production of reactive oxygen species in plant cells. Plant Physiol 2002, 128, 63–72. [Google Scholar]
- Plaxton, WC; Podestá, FE. The functional organization and control of plant respiration. Crit. Rev. Plant Sci 2006, 25, 159–198. [Google Scholar]
- Clark, RB. Effect of aluminum on growth and mineral elements of Al-tolerant and Al-intolerant corn. Plant Soil 1977, 47, 653–662. [Google Scholar]
- Cuypers, A; Plusquin, M; Remans, T; Jozefczak, M; Keunen, E; Gielen, H; Opdenakker, K; Ravindran Nair, A; Munters, E; Artois, TJ; et al. Cadmium stress: An oxidative challenge. Biometals 2010, 23, 927–940. [Google Scholar]
- Heyno, E; Klose, C; Krieger-Liszkay, A. Origin of cadmium-induced reactive oxygen species production: mitochondrial electron transfer versus plasma membrane NADPH oxidase. New Phytol 2008, 179, 687–699. [Google Scholar]
- Bi, YH; Chen, WL; Zhang, WN; Zhou, Q; Yun, LJ; Xing, D. Production of reactive oxygen species, impairment of photosynthetic function and dynamic changes in mitochondria are early events in cadmium-induced cell death in Arabidopsis thaliana. Biol. Cell 2009, 101, 629–643. [Google Scholar]
- Verbruggen, N; Hermans, C; Schat, H. Mechanisms to cope with arsenic or cadmium excess in plants. Curr. Opin. Plant Biol 2009, 12, 364–372. [Google Scholar]
- Smiri, M; Chaoui, A; Rouhier, N; Kamel, C; Gelhaye, E; Jacquot, JP; El Ferjani, E. Cadmium induced mitochondrial redox changes in germinating pea seed. Biometals 2010, 23, 973–984. [Google Scholar]
- Gupta, KJ; Fernie, AR; Kaiser, WM; van Dongen, JT. On the origins of nitric oxide. Trends Plant Sci 2011, 16, 160–168. [Google Scholar]
- De Michele, R; Vurro, E; Rigo, C; Costa, A; Elviri, L; Di Valentin, M; Careri, M; Zottini, M; Sanità di Toppi, L; Lo Schiavo, F. Nitric oxide is involved in cadmium-induced programmed cell death in Arabidopsis suspension cultures. Plant Physiol 2009, 150, 217–228. [Google Scholar]
- Arasimowicz-Jelonek, M; Floryszak-Wieczorek, J; GwóŸdŸ, EA. The message of nitric oxide in cadmium challenged plants. Plant Sci 2011, 181, 612–620. [Google Scholar]
- Panda, SK; Yamamoto, Y; Kondo, H; Matsumoto, H. Mitochondrial alterations related to programmed cell death in tobacco cells under aluminium stress. C. R. Biol 2008, 331, 597–610. [Google Scholar]
- Li, Z; Xing, D. Mechanistic study of mitochondria-dependent programmed cell death induced by aluminium phytotoxicity using fluorescence techniques. J. Exp. Bot 2011, 62, 331–343. [Google Scholar]
- Małecka, A; Derba-Maceluch, M; Kaczorowska, K; Piechalak, A; Tomaszewska, B. Reactive oxygen species production and antioxidative defense system in pea root tissues treated with lead ions: Mitochondrial and peroxisomal level. Acta Physiol. Plant 2009, 31, 1065–1075. [Google Scholar]
- Małecka, A; Jarmuszkiewicz, W; Tomaszewska, B. Antioxidative defence to lead stress in subcellular compartments of pea root cells. Acta Biochim. Pol 2001, 48, 687–698. [Google Scholar]
- Sarry, JE; Kuhn, L; Ducruix, C; Lafaye, A; Junot, C; Hugouvieux, V; Jourdain, A; Bastien, O; Fievet, JB; Vailhen, D; et al. The early responses of Arabidopsis thaliana cells to cadmium exposure explored by protein and metabolite profiling analyses. Proteomics 2006, 6, 2180–2198. [Google Scholar]
- Garnier, L; Simon-Plas, F; Thuleau, P; Agnel, JP; Blein, JP; Ranjeva, R; Montillet, JL. Cadmium affects tobacco cells by a series of three waves of reactive oxygen species that contribute to cytotoxicity. Plant Cell Environ 2006, 29, 1956–1969. [Google Scholar]
- Basu, U; Good, AG; Taylor, GJ. Transgenic Brassica napus plants overexpressing aluminium-induced mitochondrial manganese superoxide dismutase cDNA are resistant to aluminium. Plant Cell Environ 2001, 24, 1269–1278. [Google Scholar]
- Garmash, EV; Golovko, TK. Effect of cadmium on growth and respiration of barley plants grown under two temperature regimes. Russ. J. Plant Physiol 2009, 56, 343–347. [Google Scholar]
- Prado, C; Rodríguez-Montelongo, L; González, JA; Pagano, EA; Hilal, M; Prado, FE. Uptake of chromium by Salvinia minima: effect on plant growth, leaf respiration and carbohydrate metabolism. J. Hazard. Mater 2010, 177, 546–553. [Google Scholar]
- Cuypers, A; Smeets, K; Ruytinx, J; Opdenakker, K; Keunen, E; Remans, T; Horemans, N; Vanhoudt, N; Van Sanden, S; Van Belleghem, F; et al. The cellular redox state as a modulator in cadmium and copper responses in Arabidopsis thaliana seedlings. J. Plant Physiol 2011, 168, 309–316. [Google Scholar]
- Chang, HB; Lin, CW; Huang, HJ. Zinc-induced cell death in rice (Oryza sativa L.) roots. Plant Growth Regul 2005, 46, 261–266. [Google Scholar]
- Millar, H; Considine, MJ; Day, DA; Whelan, J. Unraveling the role of mitochondria during oxidative stress in plants. IUBMB Life 2001, 51, 201–205. [Google Scholar]
- Sweetlove, LJ; Heazlewood, JL; Herald, V; Holtzapffel, R; Day, DA; Leaver, CJ; Millar, AH. The impact of oxidative stress on Arabidopsis mitochondria. Plant J 2002, 32, 891–904. [Google Scholar]
- Navrot, N; Rouhier, N; Gelhaye, E; Jacquot, JP. Reactive oxygen species generation and antioxidant systems in plant mitochondria. Physiol. Plant 2007, 129, 185–195. [Google Scholar]
- Van Dongen, JT; Gupta, KJ; Ramírez-Aguilar, SJ; Araújo, WL; Nunes-Nesi, A; Fernie, AR. Regulation of respiration in plants: A role for metabolic pathways. J. Plant Physiol 2011, 168, 1434–1443. [Google Scholar]
- Purvis, AC; Shewfelt, RL. Does the alternative pathway ameliorate chilling injury in sensitive plant tissues? Physiol. Plant 1993, 88, 712–718. [Google Scholar]
- Maxwell, DP; Wang, Y; McIntosh, L. The alternative oxidase lowers mitochondrial reactive oxygen production in plant cells. Proc. Natl. Acad. Sci. USA 1999, 96, 8271–8276. [Google Scholar]
- Castro-Guerrero, NA; Rodríguez-Zavala, JS; Marín-Hernández, A; Rodríguez-Enríquez, S; Moreno-Sánchez, R. Enhanced alternative oxidase and antioxidant enzymes under Cd2+ stress in Euglena. J. Bioenerg. Biomembr 2008, 40, 227–235. [Google Scholar]
- Rasmusson, AG; Geisler, DA; Møller, IM. The multiplicity of dehydrogenases in the electron transport chain of plant mitochondria. Mitochondrion 2008, 8, 47–60. [Google Scholar]
- Clifton, R; Lister, R; Parker, KL; Sappl, PG; Elhafez, D; Millar, AH; Day, DA; Whelan, J. Stress-induced co-expression of alternative respiratory chain components in Arabidopsis thaliana. Plant Mol. Biol 2005, 58, 193–212. [Google Scholar]
- Rasmusson, AG; Møller, IM. Mitochondrial Electron Transport and Plant Stress. In Plant Mitochondria Advances in Plant Biology, 1st ed; Kempken, F, Ed.; Springer: New York, NY, USA, 2011; pp. 357–381. [Google Scholar]
- Keunen, E; Vangronsveld, J; Cuypers, A. Hasselt University: Diepenbeek, Belgium, Unpublished work; 2011.
- Vercesi, AE; Borecký, J; de Godoy Maia, I; Arruda, P; Cuccovia, IM; Chaimovich, H. Plant uncoupling mitochondrial proteins. Annu. Rev. Plant Biol 2006, 57, 383–404. [Google Scholar]
- Nogueira, FTS; Sassaki, FT; de Godoy Maia, I. Arabidopsis thaliana Uncoupling Proteins (AtUCPs): Insights into gene expression during development and stress response and epigenetic regulation. J. Bioenerg. Biomembr 2011, 43, 71–79. [Google Scholar]
- Considine, MJ; Goodman, M; Echtay, KS; Laloi, M; Whelan, J; Brand, MD; Sweetlove, LJ. Superoxide stimulates a proton leak in potato mitochondria that is related to the activity of uncoupling protein. J. Biol. Chem 2003, 278, 22298–22302. [Google Scholar]
- Smith, AMO; Ratcliffe, RG; Sweetlove, LJ. Activation and function of mitochondrial uncoupling protein in plants. J. Biol. Chem 2004, 279, 51944–51952. [Google Scholar]
- Laloi, M; Klein, M; Riesmeier, JW; Müller-Röber, B; Fleury, C; Bouillaud, F; Ricquier, D. A plant cold-induced uncoupling protein. Nature 1997, 389, 135–136. [Google Scholar]
- Desikan, R; A-H-Mackerness, S; Hancock, JT; Neill, SJ. Regulation of the Arabidopsis transcriptome by oxidative stress. Plant Physiol 2001, 127, 159–172. [Google Scholar]
- Brandalise, M; de Godoy Maia, I; Borecký, J; Vercesi, AE; Arruda, P. ZmPUMP encodes a maize mitochondrial uncoupling protein that is induced by oxidative stress. Plant Sci 2003, 165, 329–335. [Google Scholar]
- Ozawa, K; Murayama, S; Kobayashi-Uehara, A; Handa, H. Overexpression of wheat mitochondrial uncoupling protein in rice plants confers tolerances to oxidative stress promoted by exogenous hydrogen peroxide and low temperature. Mol. Breed 2006, 18, 51–56. [Google Scholar]
- Sweetlove, LJ; Lytovchenko, A; Morgan, M; Nunes-Nesi, A; Taylor, NL; Baxter, CJ; Eickmeier, I; Fernie, AR. Mitochondrial uncoupling protein is required for efficient photosynthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 19587–19592. [Google Scholar]
- Pastore, D; Trono, D; Laus, MN; Di Fonzo, N; Flagella, Z. Possible plant mitochondria involvement in cell adaptation to drought stress. A case study: durum wheat mitochondria. J. Exp. Bot 2007, 58, 195–210. [Google Scholar]
- Yin, L; Mano, J; Wang, S; Tsuji, W; Tanaka, K. The involvement of lipid peroxide-derived aldehydes in aluminum toxicity of tobacco roots. Plant Physiol 2010, 152, 1406–1417. [Google Scholar]
- Rasmusson, AG; Fernie, AR; van Dongen, JT. Alternative oxidase: a defence against metabolic fluctuations? Physiol. Plant 2009, 137, 371–382. [Google Scholar]
- Fernie, AR; Carrari, F; Sweetlove, LJ. Respiratory metabolism: Glycolysis, the TCA cycle and mitochondrial electron transport. Curr. Opin. Plant Biol 2004, 7, 254–261. [Google Scholar]
- Alscher, RG; Erturk, N; Heath, LS. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J. Exp. Bot 2002, 53, 1331–1341. [Google Scholar]
- Bueno, P; Piqueras, A. Effect of transition metals on stress, lipid peroxidation and antioxidant enzyme activities in tobacco cell cultures. Plant Growth Regul 2002, 36, 161–167. [Google Scholar]
- Mittler, R; Vanderauwera, S; Gollery, M; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci 2004, 9, 490–498. [Google Scholar]
- Jiménez, A; Hernández, JA; del Río, LA; Sevilla, F. Evidence for the presence of the ascorbate glutathione cycle in mitochondria and peroxisomes of pea leaves. Plant Physiol 1997, 114, 275–284. [Google Scholar]
- Chew, O; Whelan, J; Millar, AH. Molecular definition of the ascorbate-glutathione cycle in Arabidopsis mitochondria reveals dual targeting of antioxidant defenses in plants. J. Biol. Chem 2003, 278, 46869–46877. [Google Scholar]
- Bartoli, CG; Pastori, GM; Foyer, CH. Ascorbate biosynthesis in mitochondria is linked to the electron transport chain between complexes III and IV. Plant Physiol 2000, 123, 335–343. [Google Scholar]
- Zhao, ZQ; Cai, YL; Zhu, YG; Kneer, R. Cadmium-induced oxidative stress and protection by L-galactono-1,4-lactone in winter wheat (Triticum aestivum L.). J. Plant Nutr. Soil Sci 2005, 168, 759–763. [Google Scholar]
- Szarka, A; Horemans, N; Kovács, Z; Gróf, P; Mayer, M; Bánhegyi, G. Dehydroascorbate reduction in plant mitochondria is coupled to the respiratory electron transfer chain. Physiol. Plant 2007, 129, 225–232. [Google Scholar]
- Reese, RN; Roberts, LW. Effects of cadmium on whole cell and mitochondrial respiration in tobacco cell suspension cultures (Nicotiana tabacum L. var. xanthi). J. Plant Physiol 1985, 120, 123–130. [Google Scholar]
- Finkemeier, I; Goodman, M; Lamkemeyer, P; Kandlbinder, A; Sweetlove, LJ; Dietz, KJ. The mitochondrial type II peroxiredoxin F is essential for redox homeostasis and root growth of Arabidopsis thaliana under stress. J. Biol. Chem 2005, 280, 12168–12180. [Google Scholar]
- Gelhaye, E; Rouhier, N; Gérard, J; Jolivet, Y; Gualberto, J; Navrot, N; Ohlsson, PI; Wingsle, G; Hirasawa, M; Knaff, DB; et al. A specific form of thioredoxin h occurs in plant mitochondria and regulates the alternative oxidase. Proc. Natl. Acad. Sci. USA 2004, 101, 14545–14550. [Google Scholar]
- Schwarzländer, M; Fricker, MD; Sweetlove, LJ. Monitoring the in vivo redox state of plant mitochondria: effect of respiratory inhibitors, abiotic stress and assessment of recovery from oxidative challenge. Biochim. Biophys. Acta 2009, 1787, 468–475. [Google Scholar]
- Scheibe, R; Backhausen, JE; Emmerlich, V; Holtgrefe, S. Strategies to maintain redox homeostasis during photosynthesis under changing conditions. J. Exp. Bot 2005, 56, 1481–1489. [Google Scholar]
- Yakes, FM; Van Houten, B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc. Natl. Acad. Sci. USA 1997, 94, 514–519. [Google Scholar]
- Taylor, NL; Day, DA; Millar, AH. Targets of stress-induced oxidative damage in plant mitochondria and their impact on cell carbon/nitrogen metabolism. J. Exp. Bot 2004, 55, 1–10. [Google Scholar]
- Bartoli, CG; Gómez, F; Martínez, DE; Guiamet, JJ. Mitochondria are the main target for oxidative damage in leaves of wheat (Triticum aestivum L.). J. Exp. Bot 2004, 55, 1663–1669. [Google Scholar]
- Kristensen, BK; Askerlund, P; Bykova, NV; Egsgaard, H; Møller, IM. Identification of oxidised proteins in the matrix of rice leaf mitochondria by immunoprecipitation and two-dimensional liquid chromatography-tandem mass spectrometry. Phytochemistry 2004, 65, 1839–1851. [Google Scholar]
- Gadjev, I; Stone, JM; Gechev, TS. Programmed cell death in plants: New insights into redox regulation and the role of hydrogen peroxide. Int. Rev. Cell Mol. Biol 2008, 270, 87–144. [Google Scholar]
- Green, DR; Reed, JC. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar]
- Jones, A. Does the plant mitochondrion integrate cellular stress and regulate programmed cell death? Trends Plant Sci 2000, 5, 225–230. [Google Scholar]
- Vianello, A; Zancani, M; Peresson, C; Petrussa, E; Casolo, V; Krajňáková, J; Patui, S; Braidot, E; Macrì, F. Plant mitochondrial pathway leading to programmed cell death. Physiol. Plant 2007, 129, 242–252. [Google Scholar]
- Suzuki, N; Koussevitzky, S; Mittler, R; Miller, G. ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ 2011. [Google Scholar] [CrossRef]
- Galvez-Valdivieso, G; Mullineaux, PM. The role of reactive oxygen species in signalling from chloroplasts to the nucleus. Physiol. Plant 2010, 138, 430–439. [Google Scholar]
- Mittler, R; Vanderauwera, S; Suzuki, N; Miller, G; Tognetti, VB; Vandepoele, K; Gollery, M; Shulaev, V; Van Breusegem, F. ROS signaling: the new wave? Trends Plant Sci 2011, 16, 300–309. [Google Scholar]
- Møller, IM; Kristensen, BK. Protein oxidation in plant mitochondria as a stress indicator. Photochem. Photobiol. Sci 2004, 3, 730–735. [Google Scholar]
- Møller, IM; Sweetlove, LJ. ROS signalling—specificity is required. Trends Plant Sci 2010, 15, 370–374. [Google Scholar]
- Rhoads, DM; Subbaiah, CC. Mitochondrial retrograde regulation in plants. Mitochondrion 2007, 7, 177–194. [Google Scholar]
- Van Aken, O; Zhang, B; Carrie, C; Uggalla, V; Paynter, E; Giraud, E; Whelan, J. Defining the mitochondrial stress response in Arabidopsis thaliana. Mol. Plant 2009, 2, 1310–1324. [Google Scholar]
- Giraud, E; Ho, LHM; Clifton, R; Carroll, A; Estavillo, G; Tan, YF; Howell, KA; Ivanova, A; Pogson, BJ; Millar, AH; Whelan, J. The absence of ALTERNATIVE OXIDASE1a in Arabidopsis results in acute sensitivity to combined light and drought stress. Plant Physiol 2008, 147, 595–610. [Google Scholar]
- Robson, CA; Vanlerberghe, GC. Transgenic plant cells lacking mitochondrial alternative oxidase have increased susceptibility to mitochondria-dependent and -independent pathways of programmed cell death. Plant Physiol 2002, 129, 1908–1920. [Google Scholar]
- Vanlerberghe, GC; Cvetkovska, M; Wang, J. Is the maintenance of homeostatic mitochondrial signaling during stress a physiological role for alternative oxidase? Phys. Plant 2009, 137, 392–406. [Google Scholar]
- Bartoli, CG; Yu, J; Gómez, F; Fernández, L; McIntosh, L; Foyer, CH. Inter-relationships between light and respiration in the control of ascorbic acid synthesis and accumulation in Arabidopsis thaliana leaves. J. Exp. Bot 2006, 57, 1621–1631. [Google Scholar]
- Rauser, WE. Structure and function of metal chelators produced by plants—The case for organic acids, amino acids, phytin, and metallothioneins. Cell Biochem. Biophys 1999, 31, 19–48. [Google Scholar]
- Haydon, MJ; Cobbett, CS. Transporters of ligands for essential metal ions in plants. New Phytol 2007, 174, 499–506. [Google Scholar]
- Zhu, XF; Zheng, C; Hu, YT; Jiang, T; Liu, Y; Dong, NY; Yang, JL; Zheng, SJ. Cadmium-induced oxalate secretion from root apex is associated with cadmium exclusion and resistance in Lycopersicon esulentum. Plant Cell Environ 2011, 34, 1055–1064. [Google Scholar]
- Delhaize, E; Ryan, PR. Aluminum toxicity and tolerance in plants. Plant Physiol 1995, 107, 315–321. [Google Scholar]
- Singh, D; Chauhan, SK. Organic acids of crop plants in aluminium detoxification. Curr. Sci 2011, 100, 1509–1515. [Google Scholar]
- Gray, GR; Maxwell, DP; Villarimo, AR; McIntosh, L. Mitochondria/nuclear signaling of alternative oxidase gene expression occurs through distinct pathways involving organic acids and reactive oxygen species. Plant Cell Rep 2004, 23, 497–503. [Google Scholar]
| A. METAL-INDUCED RESPONSES IN ISOLATED MITOCHONDRIA | ||||||
|---|---|---|---|---|---|---|
| Metal | Concentration | Exposure Time | Setup | Species | Observations | Ref. |
| Al | 50 μM | 18 h | Isolation after exposing cells | N. tabacum | ↑ ROS production (O2°− and H2O2) ↓ O2 consumption ↓ ATP content ↓ cytochrome capacity ↓ AOX capacity opening of mitochondrial permeability transition pore cytochrome c release and nuclear fragmentation ~ PCD distorted mitochondrial membrane architecture | [43] |
| 0.1–0.5–1 mM | 60 min | Exposure after isolation out of mesophyll protoplasts | A. thaliana | ↑ ROS production (O2°− and H2O2) ↓ complex I and III activity | [44] | |
| Cd | 10–30 μM | 30 min | Exposure after isolation out of tubers | S. tuberosum | ↑ ROS production (O2°− and H2O2) | [36] |
| 5 mM | 12 to 120 h | Isolation after exposing germinating seeds | P. sativum | ↓ glutaredoxin, GR, GSH | [39] | |
| Cr | 20 or 200 μM | 7 days | Isolation out of roots after exposing plants | P. sativum | ↑ O2°− lipid peroxidation of mitochondrial membranes altered SOD activity ↓ respiratory complex activity (IV most sensitive) | [31] |
| Cu | 2–20–50 μM | 6 days | Isolation after exposing cells | A. pseudoplatanus | ↑ alternative respiratory pathway (KCN-resistant) ↑ AOX protein content | [30] |
| Pb | 0.1–0.5 mM | Up to 3 days | Isolation out of roots after exposing plants | P. sativum | ↑ H2O2 (mitochondria main site) ↑ AOX transcription and protein content | [45] |
| 0.5–1 mM | 2 to 96 h | Isolation out of roots after exposing plants | P. sativum | ↑ MnSOD activity ↑ alternative respiratory pathway (KCN-resistant) ↑ AOX protein content ↓ number of mitochondrial cristae | [46] | |
| Al | 25–50–75–100 μM | 6 to 24 h | Cell culture | N. tabacum | ↑ ROS production (O2°− and H2O2) ↓ mitochondrial activity ↓ respiration (O2 uptake) ↓ ATP content | [32] |
| 0.5 mM | 60 to 100 min | Protoplasts | A. thaliana | ↑ ROS production (O2°− and H2O2) ↑ AOX1a transcription ↓ mitochondrial transmembrane potential ↑ caspase-3-like protease activity ~ PCD disrupted mitochondrial ultrastructure | [44] | |
| Cd | 20 μM | 5 h | Protoplasts | A. thaliana | ↑ H2O2 in mitochondria prior to chloroplasts mitochondrial clustering and restricted movement | [37] |
| 0.5–2–5–20– 50–200 μM | 24 h | Cell culture | A. thaliana | ↑ MDHAR, peroxiredoxin | [47] | |
| 100 or 150 μM | 3 days | Cell culture | A. thaliana | ↑ PCD | [41] | |
| 3 mM | 1 h | Cell culture | N. tabacum | ↑ O2°− | [48] | |
| Cu | 2–20–50 μM | Up to 6 days | Cell culture | A. pseudoplatanus | ↓ respiration (O2 uptake) ↑ alternative respiratory pathway (KCN-resistant) ↑ AOX1 transcription | [30] |
| Al | 5–10–15– 20 μM | 4 to 24 h | Root apices | P. sativum | ↓ respiration (O2 uptake) ↓ ATP content | [32] |
| 100 μM | 1 to 48 h | Root tips | T. aestivum | ↑ MSD transcription | [49] | |
| Cd | 30–60–100 μM | Up to 10 days | Roots and leaves | H. distichum | ↓ respiration (O2 uptake) ↑ alternative respiratory pathway (SHAM) | [50] |
| Cr | 2–5–10 mg/L | 6 days | Leaves | S. minima | ↑ AOX capacity (SHAM) | [51] |
| Cu | 2 or 5 μM | 24 h | Roots | A. thaliana | ↓ MSD1 transcription | [52] |
| Fe | 100 μM | 12 h | Root cutting exposure | N. plumbaginifolia | ↑ respiration in leaves before (O2 uptake) | [29] |
| Zn | 1–5–10– 25 mM | 10 min to 9 h | Roots | O. sativa | mitochondrial ROS potentially involved in cell death | [53] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Keunen, E.; Remans, T.; Bohler, S.; Vangronsveld, J.; Cuypers, A. Metal-Induced Oxidative Stress and Plant Mitochondria. Int. J. Mol. Sci. 2011, 12, 6894-6918. https://doi.org/10.3390/ijms12106894
Keunen E, Remans T, Bohler S, Vangronsveld J, Cuypers A. Metal-Induced Oxidative Stress and Plant Mitochondria. International Journal of Molecular Sciences. 2011; 12(10):6894-6918. https://doi.org/10.3390/ijms12106894
Chicago/Turabian StyleKeunen, Els, Tony Remans, Sacha Bohler, Jaco Vangronsveld, and Ann Cuypers. 2011. "Metal-Induced Oxidative Stress and Plant Mitochondria" International Journal of Molecular Sciences 12, no. 10: 6894-6918. https://doi.org/10.3390/ijms12106894
APA StyleKeunen, E., Remans, T., Bohler, S., Vangronsveld, J., & Cuypers, A. (2011). Metal-Induced Oxidative Stress and Plant Mitochondria. International Journal of Molecular Sciences, 12(10), 6894-6918. https://doi.org/10.3390/ijms12106894






