All chemicals and solvents were purchased from Merck, Fluka and Sigma-Aldrich and were of commercial quality. They were used as received, except 1,2-dichloroethane (anhydrized over calcium chloride and distilled at normal pressure), pyridine (stored over potassium hydroxide and then distilled), and benzene (kept overnight with sodium and then distilled).
Melting points were uncorrected and determined in open capillary tubes on an Electrothermal 9100 apparatus.
Elemental analyses were performed on a Perkin Elmer CHNS/O Analyser Series II 2400 apparatus, the results being within ±0.4% of the theoretical values.
IR spectra were recorded on a FT-IR Bruker Vertex 70 spectrophotometer.
NMR spectra were recorded on a Varian Unity Inova 400 instrument operating at 400 MHz for 1H, 100 MHz for 13C and 376.279 MHz for 19F-NMR.
3.1.1. Synthesis of the New Compounds
The dibenz[
b,
e]oxepins were synthesized according to the previusly described procedure [
11]. The 2-phenoxymethylbenzoic acid was prepared in the first stage, by refluxing the phtalide with potassium phenoxyde in xylene. This gives the 2-phenoxymethylbenzoic acid potassium salt, which has good solubility in potassium hydroxide aqueous solution allowing its facile separation from xylene. The aforementioned acid was then precipitated using a mineral acid solution. The potassium salt of phenol was obtained using phenol and potassium hydroxide in xylene, the resulting water being removed by azeotropic distillation. The 6,11-dihydro-dibenz[
b,
e]oxepin-11(6
H)-one was synthesized in the second stage by a Friedel-Crafts cyclization of the 2-phenoxymethylbenzoic acid chloride in dry 1,2-dichloroethane. The acid chloride was obtained by refluxing the corresponding acid with thionyl chloride and anhydrous 1,2-dichloroethane as reaction medium. The new compounds were prepared by acylation of the 11-hydroximino-6,11-dihydro-dibenz[
b,
e]oxepin with various substituted benzoic acid chlorides, in dry benzene and in the presence of anhydrous pyridine as a proton fixator. The resulting solid was recrystallized from isopropanol to yield the new
O-acyl-oximino-dibenz[
b,
e]oxepins. The oxime was obtained by treating the 6,11-dihydro-dibenz[
b,
e]oxepin-11(6
H)-one with hydroxylamine hydrochloride in presence of pyridine. The chemical structure of the new compounds is presented in
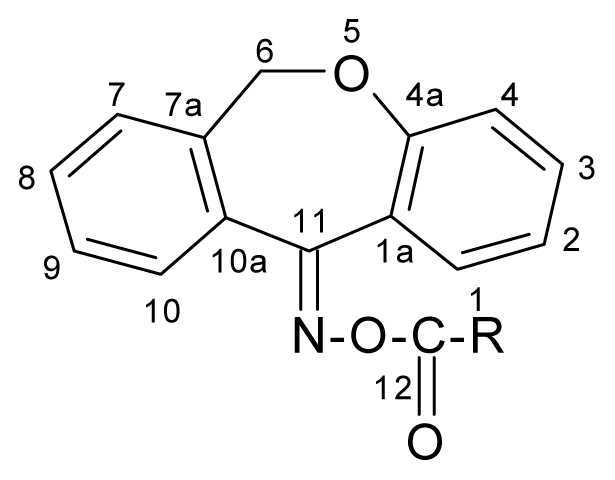
Table 1.
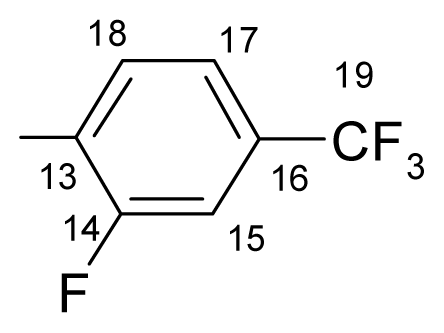
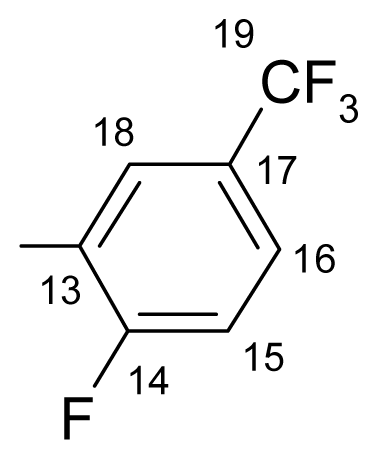
3.1.1.1. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[2-fluoro-3-(trifluoromethyl)- phenyl]methanone (a) (E/Z isomers mixture in ratio 1:2.45 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 532 (w), 572 (w), 630 (w), 705 (m), 748.(vs), 827 (m), 865 (m), 948 (w), 979 (m), 1075 (s), 1098 (s), 1122 (vs), 1159 (m), 1195 (s), 1237 (m), 1269 (s), 1305 (m), 1333 (s), 1443 (m), 1462 (s), 1481 (w), 1595 (m), 1611 (m), 1740 (vs), 2987 (w), 3072 (w), 3104 (w).
19F-NMR (CDCl3): δ-61.97(3F, d, 4J(F-15–F-14) = 13.78 Hz, CF3), δ-109.60 (1F, q, 4J(F-15–F-14) = 13.78 Hz, F-14).
1H-NMR (CDCl3): δ 5.19 (s, H-6 M), 5.25 (s, H-6 m), 6.91 (dd, J = 1.1, 8.4 Hz, H-4 M), 6.98 (dd, J = 1.1, 8.4 Hz, H-4 m), 7.02 (ddd, J = 1.1, 7.0, 8.0 Hz, H-2 M), 7.03 (ddd, J = 1.1, 7.0, 8.0 Hz, H-2 m), 7.29 (1H, m, J(H-17–H-16)= 8.2 Hz, H-17), 7.36 (1H, ddd, J = 1.7, 7.2, 8.4 Hz, H-3), 7.49–7.40 (3H, m, H-7, H-8, H-9), 7.61 (m, H-10 M), 7.66 (m, H-10 m), 7.83–7.73 (m, H-16, H-1 m), 7.87 (dd, J = 1.8, 8.0 Hz, H-1 M), 8.06 (ddd, J(F-H-18)= 6.5 Hz, J(H-17–H-18)= 8.2 Hz, J(H-18–H-16)= 1.7 Hz, H-18 M), 8.13 (ddd, J(F-H-18)= 6.5 Hz, J(H-17–H-18)= 8.2 Hz, J(H-18–H-16)= 1.7 Hz, H-18 m).
13C-NMR (CDCl3): δ 70.4 (C-6 m), 70.5 (C-6 M), 118.4, 119.2, 119.4, 120.0 M, 120.4 m, 120.8 m, 121.5 M, 123.3 (q, J(F-C)= 271.1 Hz, C-19), 124.1 (d, 3J(F-C) = 4.4 Hz, C-18), 127.2, 127.9 m, 128.3 M, 128.4 m, 128.5 M, 129.3 m, 130.7 M, 130.9 M, 131.0 m, 131.7 (m, C-15), 132.6, 132.8 m, 132.9 M, 134.2 (C-10a m), 134.2 (C-10a M), 136.0 (C-16 M), 136.1 (C-16 m), 155.8 (C-4a m), 157.6 (C-4a M), 162.1 (d, J(F-C) = 252.7 Hz, C-14), 165.4 (C-11), 162.7 (C-12).
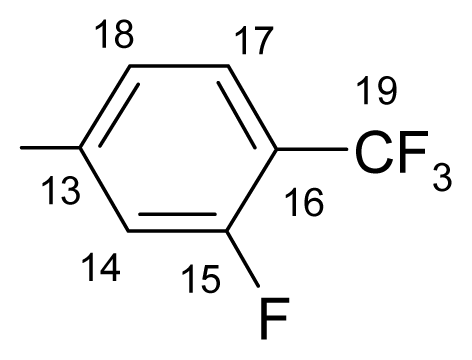
3.1.1.2. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[2-fluoro-4-(trifluoromethyl)-phenyl]methanone (b) (E/Z isomers mixture in ratio 1:4.3 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 545 (w), 602 (w), 634 (w), 698 (w), 756 (s), 835 (w), 882 (m), 916 (w), 977 (s), 1007 (m), 1049 (m), 1084 (vs), 1127 (s), 1176 (s), 1214 (s), 1236 (m), 1277 (m), 1304 (m), 1332(s), 1430 (s), 1481 (w), 1507 (w), 1606 (m), 1745 (vs), 2865 (w), 2917 (w), 2993 (w), 3071 (w).
19F-NMR (CDCl3): δ-63.90 (3F, bs, CF3), δ-105.11 (1F, bs, F-14).
1H-NMR (CDCl3): δ 5.19 (s, H-6 M), 5.25 (s, H-6 m), 6.91 (dd, J = 1.1, 8.4 Hz, H-4 M), 6.98 (dd, J = 1.1, 8.4 Hz, H-4 m), 7.02 (1H, td, J = 8.4, 1.1 Hz, H-2), 7.51–7.33 (6H, m, H-3, H-7, H-8, H-9, H-15, H-18), 7.59 (1H, m, H-10), 7.88 (1H, dd, J = 1.8, 8.0 Hz, H-1), 7.99 (bt, 5J(H-17–F-14) = 7.2 Hz, 3J(H-17–H-18) = 7.2 Hz, H-17 M), 8.08 (bt, 5J(H-17–F-14) = 7.2 Hz, 3J(H-17–H-18) = 7.2 Hz, H-17 m).
13C-NMR (CDCl3): δ 70.4 (C-6 m), 70.5 (C-6 M), 114.7 (dq, J(F-C-15) = 25.6 Hz, J(F3-C-15) = 3.8 Hz, C-15), 119.4 (C-1 a), 120.0, 121.0 (dq, C-17), 121.5, 123.5 (q, J(F-C) = 272.3 Hz, C-19), 127.2 M, 127.9 m, 128.3 (d, J(F-C) = 2.2 Hz, C-18), 128.4 M, 128.5 m, 130.6, 130.9, 132.7, 132.9, 133.2, 134.3, 136.5, 136.8, 157.6 (C-4 a), 162.6 (C-11), 162.7 (d, J(F-C) = 245.8 Hz, C-14), 165.5 (C-12).
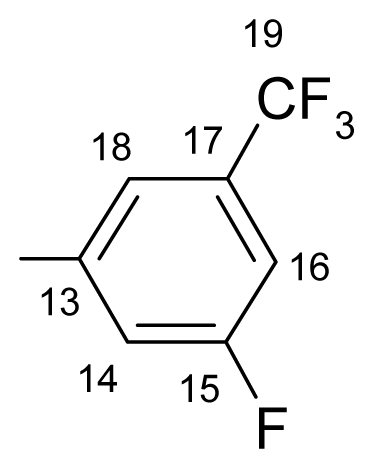
3.1.1.3. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[2-fluoro-5-(trifluoromethyl)- phenyl]methanone (c) (E/Z isomers mixture in ratio 1:2.2 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 548 (w), 616 (w), 681 (w), 753 (s), 794 (w), 847 (m), 895 (m), 977 (s), 1011 (m), 1038 (vs), 1078 (s), 1110 (s), 1161 (m), 1207 (vs), 1271 (w), 1310 (m), 1334 (s), 1445 (m), 1483 (w), 1606 (m), 1764 (vs), 2883 (w), 2986 (w), 3070 (w).
19F-NMR (CDCl3): δ-62.81 (3F, bs, CF3), δ-102.18 (1F, bs, F-14).
1H-NMR (CDCl3): δ 5.19 (s, H-6 M), 5.25(s, H-6 m), 6.91 (dd, J = 1.1, 8.4 Hz, H-4M), 7.06–6.97 (m, H-2, H-4 m), 7.23 (t, J = 7.8 Hz, H-15 M), 7.28 (t, J = 7.8 Hz, H-15 m), 7.51–7.32 (5H, m, H-2, H-3, H-7, H-8, H-9), 7.60 (bd, J = 7.8 Hz, H-16 M), 7.76 (bd, 7.8 Hz, H-16 m), 7.77 (1H, m, H-10), 7.88 (1H, dd, J = 1.8, 8.0 Hz, H-1), 8.25 (1H, bd, J = 4.9 Hz, H-18).
13C-NMR (CDCl3): δ 70.5 (C-6 m), 70.6 (C-6 M), 118.1 (m, C-15), 119.4(C-1a), 120.0 M, 120.5 m, 120.8 M, 121.5 m, 123.1 (q, J(F-C-19) = 270.9 Hz, C-19), 127.2 m, 127.9 M, 128.2 M, 128.5 m, 128.4 M, 128.5 m, 129.4, 130.0 (m, C-18), 130.6 M, 130.7 m, 130.9, 131.9 (m, C-16), 132.7 M, 132.9 m, 134.2 (C-10a m), 134.3 (C-10a M), 136.7, 157.6 (C-4a), 160.3 (q, J(CF3-C-17) = 4.4 Hz, C-17), 163.5 (d, J(F-C) = 264.4 Hz, C-14), 164.53 (C-11), 165.4 (C-12).
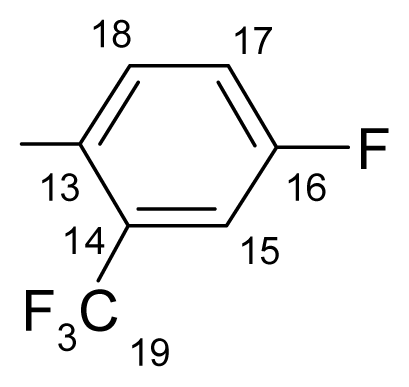
3.1.1.4. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[3-fluoro-4-(trifluoromethyl)- phenyl]methanone (d) (E/Z isomers mixture in ratio 1:2.4 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 561 (w), 591 (w), 609 (w), 641 (w), 673 (w), 705 (w), 727 (m), 754 (s), 774 (m), 796 (w), 824 (m), 843 (w), 878 (m), 936 (s), 988 (m), 1004 (m), 1045 (s), 1085 (vs), 1123 (s), 1139 (vs), 1192 (vs), 1222 (m), 1250 (vs), 1281 (m), 1312 (s), 1328 (s), 1378 (w), 1418 (s), 1443 (m), 1482 (m), 1506 (w), 1583 (m), 1605 (m), 1630 (w), 1761 (vs), 2892 (w), 2965 (w), 3068 (w).
19F-NMR (CDCl3): δ-62.34 (3F, d, 4J(3F-16–F-15) = 12.64 Hz, CF3), δ-112.94 (1F, q, 4J(3F-16–F-15) = 12.64 Hz, F-15).
1H-NMR (CDCl3): δ 5.20 (s, H-6 M), 5.26 (s, H-6 m), 6.92 (dd, J = 1.2, 8.4 Hz, H-4 M), 7.03 (dd, J = 1.2, 8.4 Hz, H-4 m), 7.03 (ddd; J = 1.2, 7.2, 8.2 Hz, H-2 M), 7.08 (ddd, J = 1.2, 7.2, 8.2 Hz, H-2 m), 7.37 (1H, ddd, J = 1.4, 7.2, 8.2 Hz, H-3), 7.58–7.33 (4H, m, H-7-, H-8, H-9, H-10), 7.84–7.64 (m, H-1 m, H-14, H-17, H-18), 7.89 (dd, J = 1.8, 8.0 Hz, H-1 M).
13C-NMR (CDCl3): δ 70.5 (C-6 m), 70.6 (C-6 M), 118.1 (d, J(F-C) = 22.7 Hz, C-14 M), 118.3 (d, J(F-C) = 22.7 Hz, C-14 m,), 119.2 (C-1a), 120.1, 121.6, 122.0 (q, J(F-C-19) = 271.6 Hz, C-19), 125.3 (d, J(F-C-13) = 4.3 Hz, C-13 M), 125.4 (d, J(F-C-13) = 4.3 Hz, C-13 m), 127.2 m, 127.6, 127.8 M, 127.9 m, 128.5 M, 128.8 M, 129.4 m, 130.3, 130.8 (d, J(F-C-18)= 2.8 Hz, C-18), 132.9 m, 133.0 M, 134.1 (C-10a m), 134.2 (C-10a M), 134.5, 136.7, 156.0 (C-4a m), 157.7 (C-4a M), 160.3 (q, J(CF3-C-16) = 4.4 Hz, C-16), 159.6 (d, J(F-C-15) = 254.8 Hz, C-15), 164.6 (C-11), 165.5 (C-12).
3.1.1.5. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[3-fluoro-5-(trifluoromethyl)-phenyl]methanone (e) (E/Z isomers mixture in ratio 1:4.4 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 554 (w), 591 (w), 628 (w), 641 (w), 689 (m), 706 (w), 755 (s), 783 (w), 807 (w), 885 (s), 921 (w), 945 (m), 1002 (m), 1044 (w), 1103 (m), 1137 (vs), 1181 (vs), 1231 (vs), 1250 (s), 1336 (m), 1352 (m), 1443 (w), 1482 (m), 1607 (m), 1763 (s), 3001 (w), 3079 (w), 3081 (w).
19F-NMR (CDCl3): δ-63.51 (3F, bs, CF3), δ-109.19 (1F, bs, F-15).
1H-NMR (CDCl3): δ 5.20 (s, H-6 M), 5.26 (s, H-6 m), 6.92 (dd, J = 1.2, 8.4 Hz, H-4 M), 7.03 (dd, J = 1.2, 8.4 Hz, H-4 m), 7.03 (ddd, J = 8.2, 7.2, 1.2 Hz, H-2 M), 7.09 (ddd, J = 8.2, 7.2, 1.2 Hz, H-2 m), 7.37 (1H, ddd, J = 1.4, 7.2, 8.2 Hz, H-3), 7.61–7.34 (m, H-7, H-8, H-9, H-10 m, H-14), 7.68 (m, H-10 M), 7.79 (1H, ddd, J = 2.4, 1.8 Hz, J(F-H-16) = 8.6 Hz, H-16), 7.89 (1H, dd, J = 1.8, 8.0 Hz, H-1), 7.92 (bs, H-18 M), 8.40 (bs, H-18 m).
13C-NMR (CDCl3): δ 70.6 (C-6 m), 70.6 (C-6M), 117.6 (dq, J(F-C-16) = 22.7.2 Hz, J(F3-C-16) = 3.7Hz, C-16), 119.2 (C-1a), 120.1, 121.6, 121.8 (q, J(F-C-19) = 269.8 Hz, C-19), 122.4 (q, J(F-C) = 3.6 Hz, C-18), 123.0, 127.8, 128.5, 128.8, 130.3, 130.8, 131.9, 132.0 (d, J(F-C) = 8.0 Hz, C-13), 132.9, 133.0, 134.2 (C-10a), 157.7 (C-4a), 161.1 (C-11), 162.3 (d, J(F-C-15) = 250.0 Hz, C-15), 165.5 (C-12).
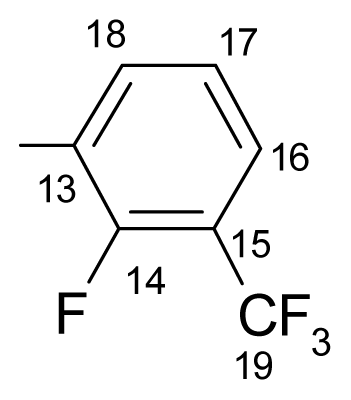
3.1.1.6. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[4-fluoro-2-(trifluoromethyl)- phenyl]methanone (f) (E/Z isomers mixture in ratio 1:3 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 547 (w), 579 (w), 632 (m), 688 (w), 753 (s), 878 (m), 910 (m), 971 (s), 1001 (m), 1037 (s), 1082 (s), 1139 (vs), 1246 (vs), 1310 (s), 1431 (m), 1480 (m), 1604 (m), 1761 (vs), 2888 (w), 2990 (w), 3071 (w).
19F-NMR (CDCl3): δ-60.30 (3F, d, 5J(F-16–3F-19) = 5.74 Hz, CF3), δ-105.30 (1F, q, 4J(3F-19–F-16) = 5.74 Hz, F-16).
1H-NMR (CDCl3): δ 5.19 (s, H-6 M), 5.25 (s, H-6 m), 6.90 (dd, J = 1.4, 8.2 Hz, H-4 M), 6.93 (td, J = 8.2, 1.4 Hz, H-2 m), 6.97 (dd, J = 1.4, 8.2 Hz, H-4 m), 7.01 (td, J = 8.2, 1.4 Hz, H-2 M), 7.22–7.49 (7H, m, H-3, H-7, H-8, H-9, H-10, H-15, H-17), 7.69 (dd, J(F-H-18) = 5.4 Hz, J(H-18-H-17) = 8.6 Hz, H-18 M), 7.81 (dd, J(F-H-18) = 5.4 Hz, J(H-18-H-17) = 8.6 Hz, H-18 m), 7.54 (dd, J = 1.8, 8.0 Hz, H-1 m), 7.87 (dd, J = 1.8, 8.0 Hz, H-1 M).
13C-NMR (CDCl3): δ 70.4 (C-6 m), 70.48 (C-6 M), 115.0 (dq, J(F-C-15)= 25.6 Hz, J(F3-C-15)= 5.1 Hz, C-15), 118.8 (J(F-C-17)= 21.2 Hz, C-17 M), 118.8 (J(F-C-17) = 21.2 Hz, C-17 m), 119.3 (C-1a), 120.0 M, 120.5 m, 120.8 m, 121.5 M, 122.2 (q, J(F3-C-19) = 264.3 Hz, C-19), 127.7 M, 127.9 m, 128.4M, 128.5 m, 129.4, 130.5, 130.5 M, 130.7 m, 130.8, 132.7, 132.9 (dq, J(F-C-14)= 12.4 Hz, J(F3-C-14) = 8 Hz, C-14), 132.9, 134.0 (C-10a m), 134.3 (C-10a M), 136.7, 155.8 (C-4a m), 157.5 (C-4a M), 162.9 (C-11), 163.8 (d, J(F-C-16) = 253.3 Hz, C-16), 164.6 (C-12 M), 165.6 (C-12 m).
3.1.1.7. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[4-fluoro-3-(trifluoromethyl)-phenyl]methanone (g) (E/Z isomers mixture in ratio 1:4.8 estimated by 1H-NMR)
FT-IR (solid in ATR, ν cm−1): 548 (w), 631 (w), 675 (w), 707 (w), 731 (w), 752 (s), 846 (m), 981 (m), 1006 (w), 1051 (m), 1077 (s), 1108 (w), 1133 (m), 1147 (s), 1161 (s), 1223 (s), 1243 (s), 1269 (m), 1305 (m), 1325 (s), 1423 (m), 1441 (m), 1464 (w), 1480 (w), 1601 (m), 1759 (vs), 2897 (w), 2962 (w), 3073 (w).
19F-NMR (CDCl3): δ-62.24(3F, d, 4J(3F-15–F-16) = 12.06 Hz, CF3), δ-106.47(1F, q, 4J(3F-15–F-16) = 12.06 Hz, F-16).
1H-NMR (CDCl3): δ 5.20 (s, H-6 M), 5.26 (s, H-6 m), 6.92 (dd, J = 1.4, 8.2 Hz, H-4 M), 7.03 (dd, J = 1.4, 8.2 Hz, H-4 m), 7.04 (td, J = 8.2, 1.4 Hz, H-2 M), 7.08 (td, J = 8.2, 1.4 Hz, H-2 m), 7.26 (dd, J(H-17-H-18) = 8.2 Hz, J(F-H-17)= 10.4 Hz, H-17 M), 7.29 (dd, J(H-17–H-18) = 8.2 Hz, J(F-H-17) = 10.4 Hz, H-17 m), 7.37 (1H, ddd, J = 1.7, 7.2, 8.0 Hz, H-3), 7.57–7.34 (3H, m, H-7, H-8, H-9), 7.68 (dq, J(F-H-14) = 8.0 Hz, J(F3-H-14) = 1.8 Hz, H-14 M), 7.89 (1H, dd, J = 1.8, 8.0 Hz, H-1), 8.18–8.10 (m, H-10 M, H-18), 8.24 (m, H-14 m), 8.30 (m, H-10 m).
13C-NMR (CDCl3): δ 70.5 (C-6 m), 70.6 (C-6 M), 117.6 (d, J(F3-C-17) = 21.2 Hz, C-17), 119.3 (C-1a), 120.0 (C-4), 121.6 (C-2), 122.0 (q, J(F3-C-19) = 270.3 Hz, C-19), 125.3 (d, J(F-C-13) = 2.9 Hz, C-13), 127.8 (C-10 M), 127.9 (C-10 m), 128.4, 128.7, 129.3 (m, C-15), 130.4, 130.7 (C-1 m), 130.8, 130.8 (C-1 M), 132.8 (C-3 m), 132.9 (C-3 M), 133.0, 134.3 (C-10a), 135.8 (d, J(F-C) = 9.5 Hz, C-14), 157.6 (C-4a), 161.4 (C-11), 162.7 (d, J(F-C-16) = 263.6 Hz, C-16), 166.0 (C-12).
3.1.1.8. ({[(E/Z)-11-dibenz[b,e]oxepin-11(6H)-ylidene]amino}oxy)[5-fluoro-2-(trifluoromethyl)-phenyl]methanone (h) (E/Z isomers mixture in ratio 1:2.8 estimated by 1H-NMR)
FT-IR(solid in ATR, ν cm−1): 518 (w), 591 (m), 630 (w), 680 (w), 757 (s), 835 (m), 885 (m), 932 (m), 984 (m), 1031 (s), 1076 (s), 1115 (vs), 1153 (vs), 1196 (s), 1257 (s), 1305 (vs), 1440 (w), 1480 (w), 1597 (m), 1759 (vs), 2875 (w), 3078 (w)
19F-NMR (CDCl3): δ-59.25 (3F, bs, CF3); δ-106.49 (1F, bs, F-14).
1H-NMR(CDCl3): δ 5.19 (s, H-6 M), 5.25 (s, H-6 m), 6.90 (dd, J = 1.4, 8.2 Hz, H-4 M), 6.94 (td, J = 8.2, 1.4, H-2 m), 6.97 (dd, J = 1.4, 8.2 Hz, H-4 m), 7.02 (td, J = 8.2, 1.4 Hz, H-2 M), 7.23–7.49 (6H, m, H-3, H-7, H-8, H-9, H-16, H-18), 7.54 (dd, J = 1.8, 8.0 Hz, H-1 m), 7.66 (m, H-10 m), 7.72 (dd, J(F-H-15) = 5.1 Hz, J(H-15-H-16) = 8.8 Hz, H-15 M), 7.76(dd, J(F-H-15) = 5.1 Hz, J(H-15–H-16) = 8.8 Hz, H-15 m), 7.86 (dd, J = 1.8, 8.0 Hz, H-1 M).
13C-NMR (CDCl3, ppm): δ 70.4 (C-6 m), 70.5 (C-6 M), 117.7 (d, J(C-F) = 24.9 Hz, C-18 M), 117.7 (d, J(C-F)= 24.9 Hz, C-18 m), 118.4 (d, J(C-16-F) = 22.0 Hz, C-16 M), 118.4 (d, J(C-16-F) = 22.0 Hz, C-16 m), 119.5 (C-1a), 120.6, 120.9, 121.5, 127.1, 127.7, 127.9, 128.4, 129.6, 129.5 (dq, J(F-C-14) = 3.8 Hz, J(F3-C-14) = 5.1 Hz, C-14), 130.4 m, 130.6 M, 130.7 m, 130.8 M, 132.7, 132.7 m, 132.9 M, 134.1 (C-10a m), 134.2 (C-10a M), 155.8 (C-4a m), 157.6 (C-4a M), 163.9 (d, J(F-C-17) = 241.6 Hz, C-17), 165.1 (C-11), 165.9 (C-12).