The Mechanism of (R,R) ZX-5 on Increasing NO Release
Abstract
:1. Introduction
2. Materials and Methods
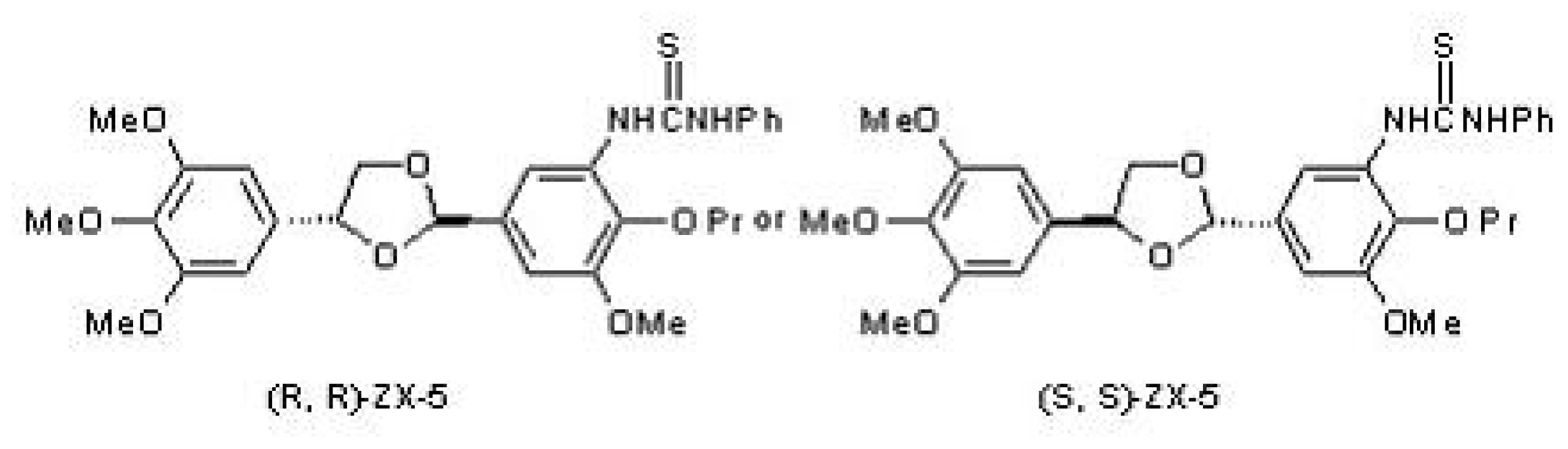
2.1. The Structure of (R,R) ZX-5
2.2. Cell Culture
2.3. Measurement of NO Release
2.4. Reverse-Transcriptase Polymerase Chain Reaction Analysis of eNOS and iNOS Expression
2.5. Western Blot Analysis
2.6. Endothelial NO Synthase (eNOS) Activity Assay
2.7. Statistical Analysis
3. Results
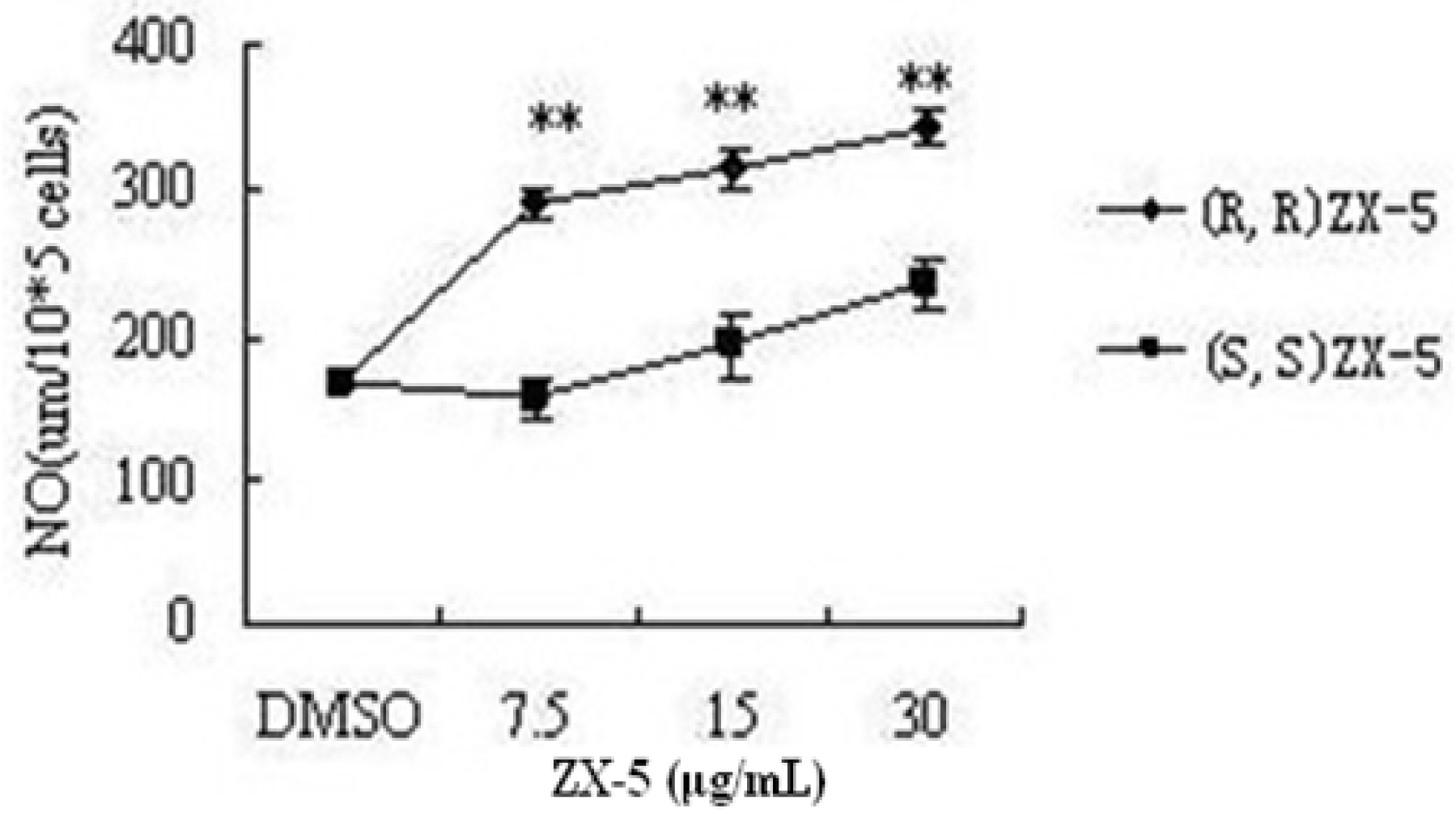
3.1. (R,R) ZX-5 Treatment Increases NO Production in HUVECs
3.2. (R,R) ZX-5 Up-Regulates iNOS mRNA in a Dose-Dependent Manner
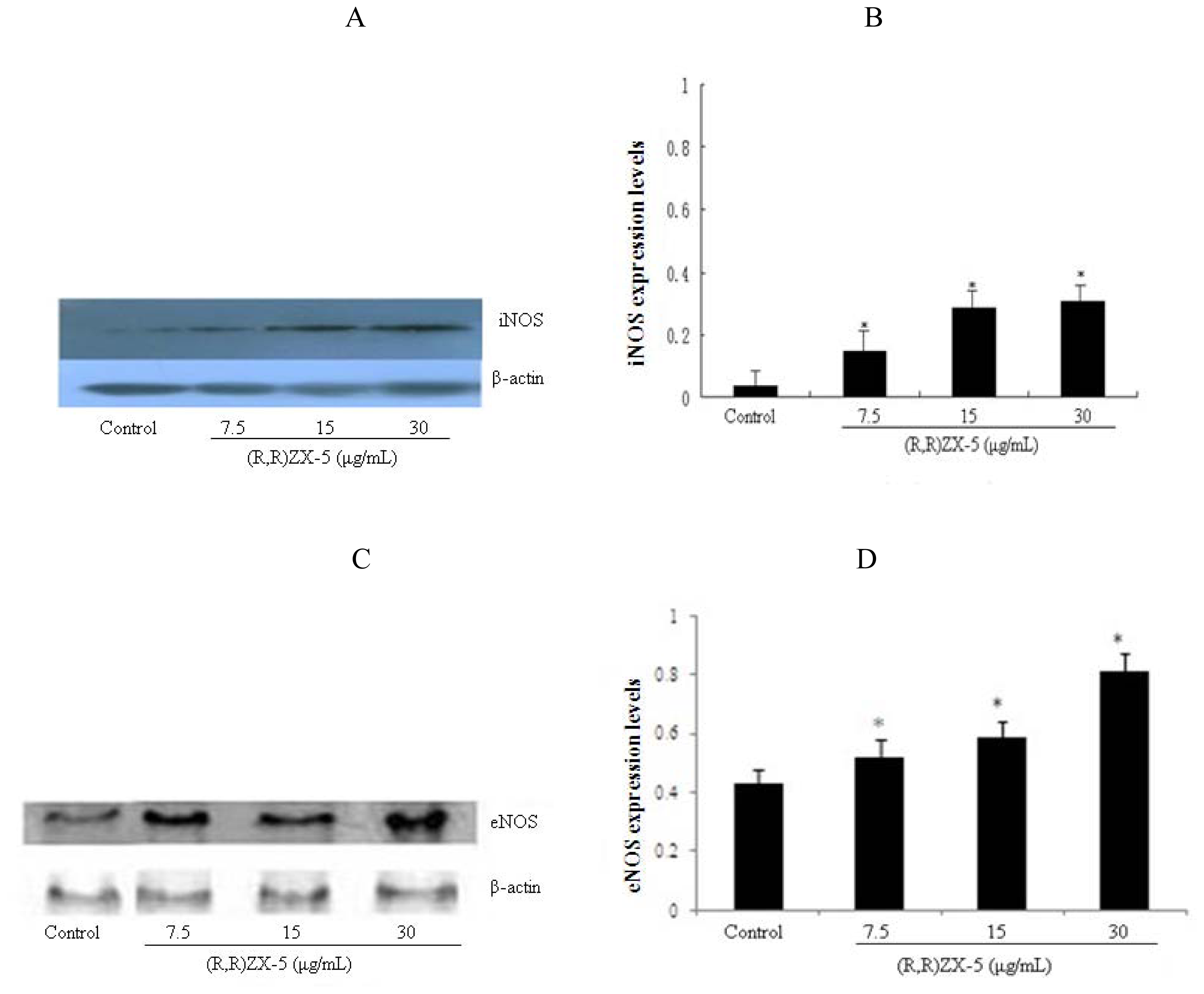
3.3. (R,R) ZX-5 Treatment Leads to Increased iNOS and eNOS Protein Expression
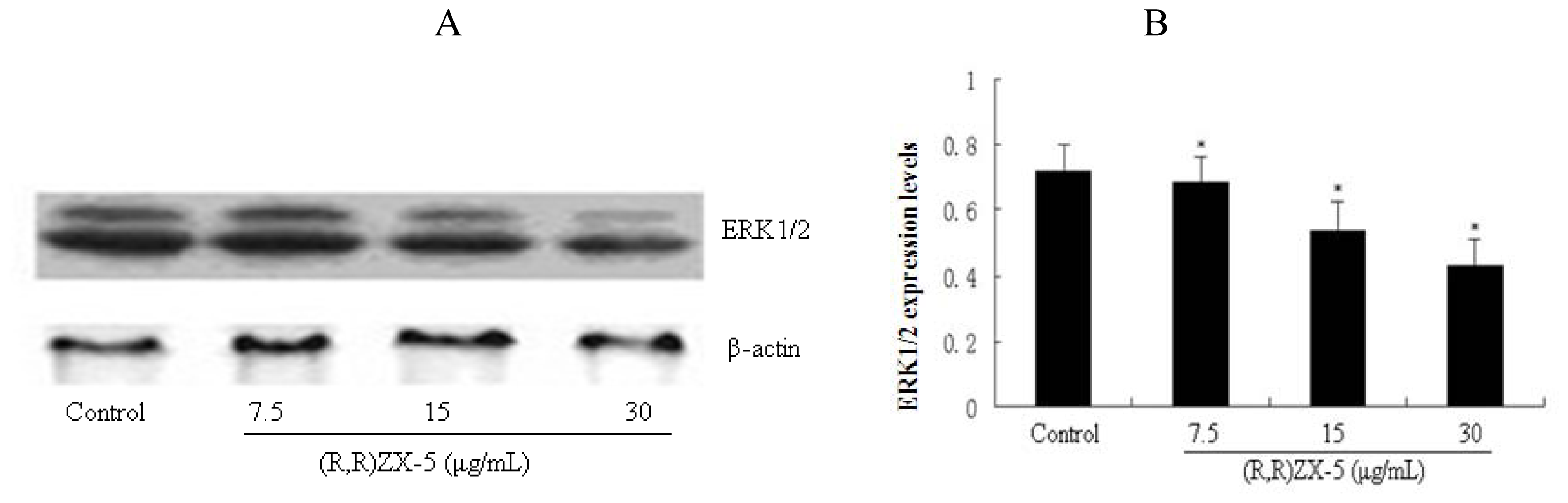
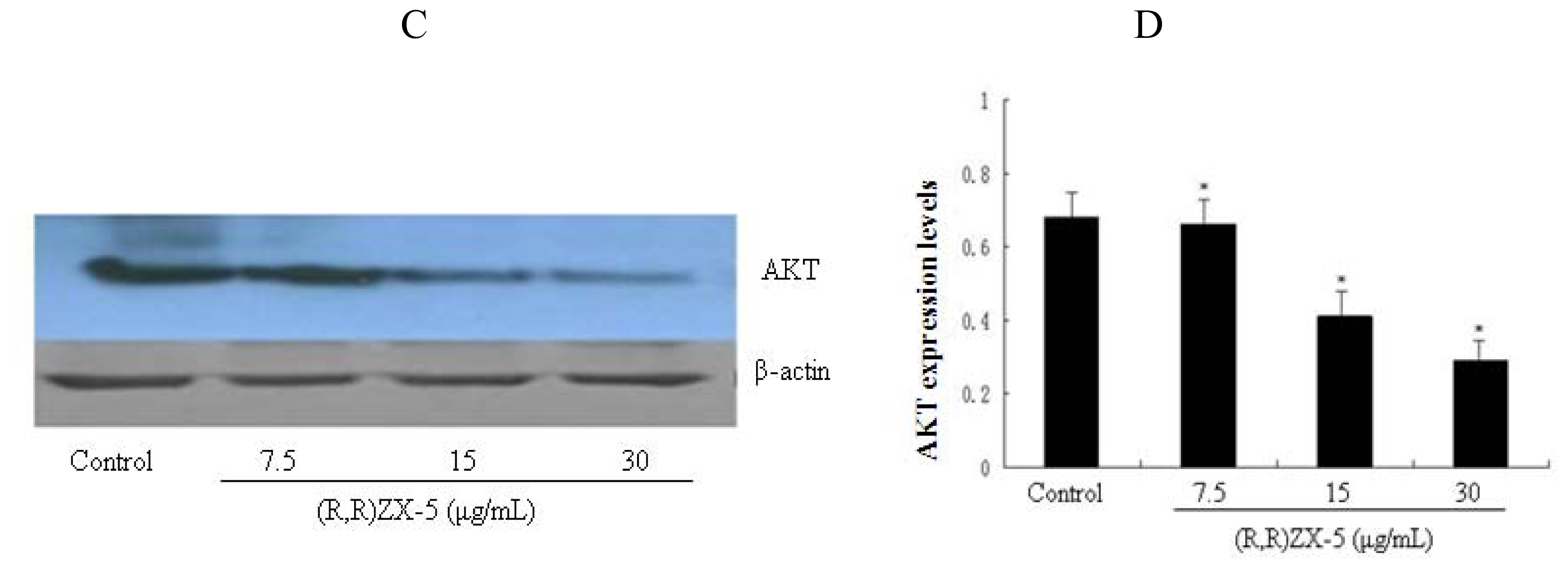
3.4. (R,R) ZX-5 Treatment Inhibits ERK and AKT Expression
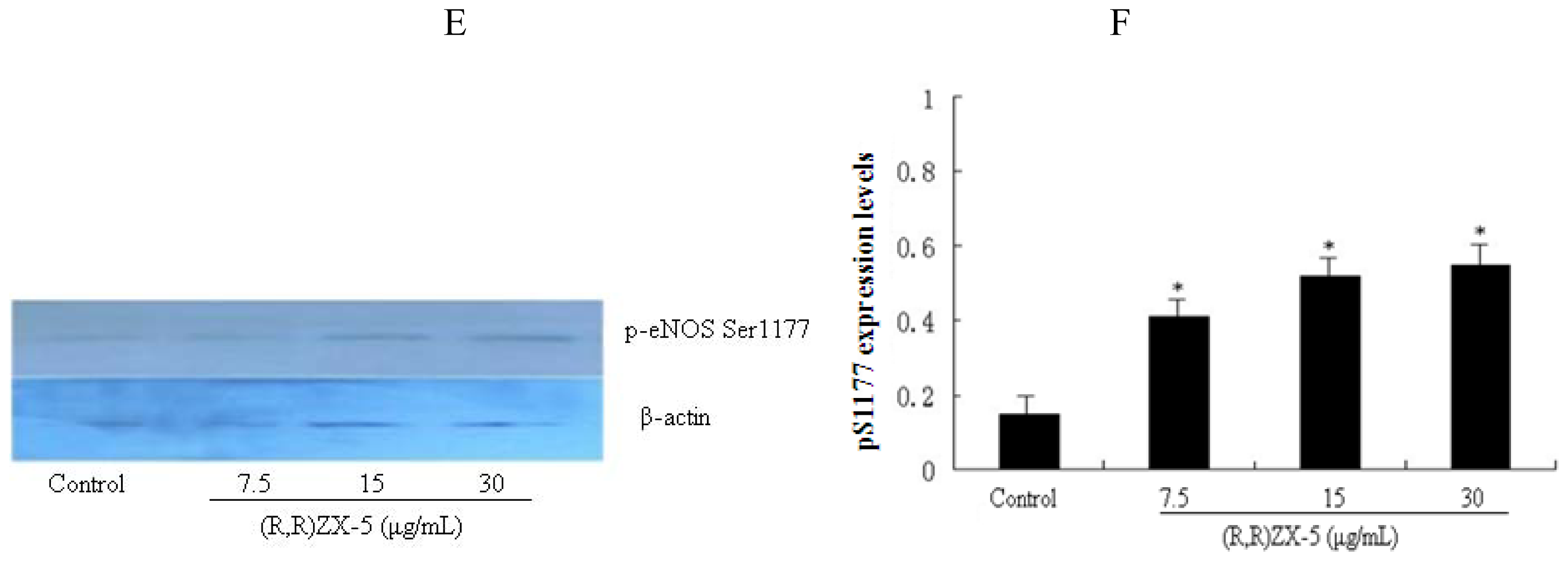
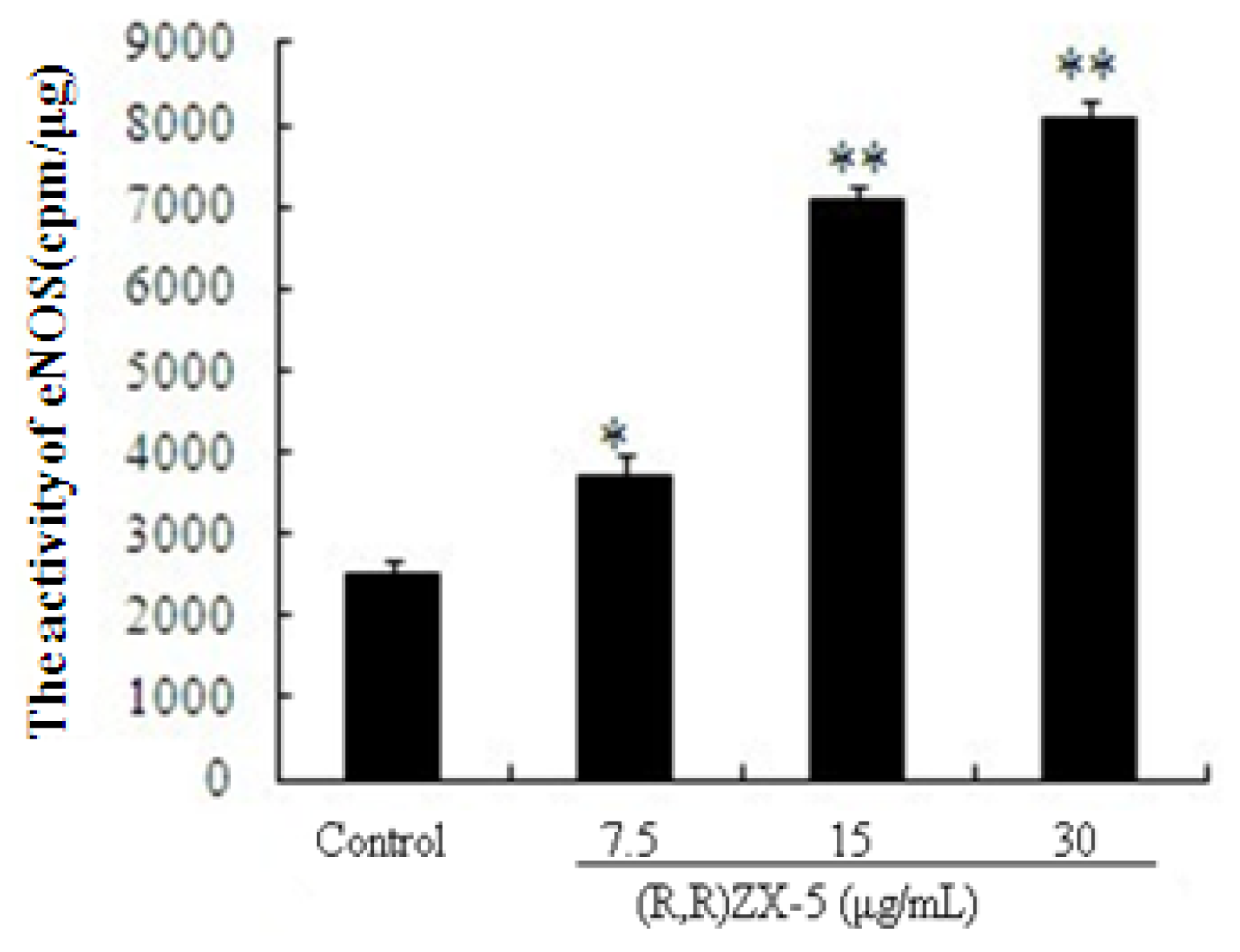
3.5. (R,R) ZX-5 Enhances eNOS Activity in HUVECs
4. Discussion
Acknowledgments
References
- van Leeuwen, R; Klaver, CC; Vingerling, JR; Hofman, A; de Jong, PT. Epidemiology of age-related maculopathy: A review. Eur. J. Epidemiol 2003, 18, 845–854. [Google Scholar]
- Klein, R; Peto, T; Bird, A; Vannewkirk, MR. The epidemiology of age-related macular degeneration. Am. J. Ophthalmol 2004, 137, 486–495. [Google Scholar]
- Chiou, GCY. Effects of nitric oxide on eye diseases and their treatment. J. Ocul. Pharmacol. Ther 2001, 17, 189–190. [Google Scholar]
- Xuan, B; Xu, XR; Chiou, GCY; Zhang, YH; Peng, SX. Relationship between nitric oxide production and choroidal blood flow. J. Ocul. Pharmacol. Ther 2003, 19, 247–253. [Google Scholar]
- Knowles, RG; Salter, M. Titheradge, MA, Ed.; Measurement of NOS activity by conversion of radiolabeled arginine to citrulline using ion-exchange separation. In Nitric Oxide Protocols. Humana: Totowa; Humana Press: Totowa, NJ, USA, 1998; pp. 67–73. [Google Scholar]
- Albrecht, EW; Stegeman, CA; Heeringa, P; Henning, RH; van Goor, H. Protective role of endothelial nitric oxide synthase. J. Pathol 2003, 199, 8–17. [Google Scholar]
- Cale, JM; Bird, IM. Inhibition of MEK/ERK signalling alters endothelial nitric oxide synthase activity in an agonist dependent manner. Biochem. J 2006, 398, 279–288. [Google Scholar]
- Mineo, C; Yuhanna, IS; Quon, MJ; Shaul, PW. High density lipoprotein-induced endothelial nitric-oxide synthase activation is mediated by Akt and MAP kinases. J. Biol. Chem 2003, 278, 9142–9149. [Google Scholar]
- Goetze, S; Bungenstock, A; Czupalla, C; Eilers, F; Stawowy, P; Kintscher, U; Spencer-Hansch, C; Graf, K; Nurnberg, B; Law, RE; Fleck, E; Grafe, M. Leptin induces endothelial cell migration through Akt, which is inhibited by PPARγ-ligands. Hypertension 2002, 40, 748–754. [Google Scholar]
- Wyatt, AW; Steinert, JR; Wheeler-Jones, CP; Morgan, AJ; Sugden, D; Pearson, JD; Sobrevia, L; Mann, GE. Early activation of the p42/p44MAPK pathway mediates adenosine-induced nitric oxide production in human endothelial cells: A novel calcium-insensitive mechanism. FASEB J 2002, 16, 1584–1594. [Google Scholar]
- Wei, Z; Al-Mehdi, AB; Fisher, AB. Signaling pathway for nitric oxide generation with simulated ischemia in flow-adapted endothelial cells. Am. J. Physiol. Heart. Circ. Physiol 2001, 281, H2226–H2232. [Google Scholar]
- Cha, MS; Lee, MJ; Je, GH; Kwak, JY. Endogenous production of nitric oxide by vascular endothelial growth factor down-regulates proliferation of choriocarcinoma cells. Biochem. Biophys. Res. Commun 2001, 282, 1061–1066. [Google Scholar]
- Kou, R; Igarashi, J; Michel, T. Lysophosphatidic acid and receptor-mediated activation of endothelial nitric-oxide synthase. Biochemistry 2002, 41, 4982–4988. [Google Scholar]
- Igarashi, J; Michel, T. Sphingosine 1-phosphate and isoform-specific activation of phosphoinositide 3-kinase β: Evidence for divergence and convergence of receptor-regulated endothelial nitric-oxide synthase signaling pathways. J. Biol. Chem 2001, 276, 36281–36288. [Google Scholar]
- Lorenz, M; Wessler, S; Follmann, E; Michaelis, W; Dusterhoft, T; Baumann, G; Stangl, K; Stangl, V. A constituent of green tea, epigallocatechin-3-gallate, activates endothelial nitric oxide synthase by a phosphatidylinositol-3-OH-kinase-, cAMP-dependent protein kinase and Akt-dependent pathway and leads to endothelial-dependent vasorelaxation. J. Biol. Chem 2004, 279, 6190–6195. [Google Scholar]
- Schmidt, K; Gibraeil, HD; Mayer, B. Lack of involvement of extracellular signal-regulated kinase (ERK) in the agonist-induced endothelial nitric oxide synthesis. Biochem. Pharmacol 2002, 63, 1137–1142. [Google Scholar]
- Bernier, SG; Haldar, S; Michel, T. Bradykinin-regulated interactions of the mitogen-activated protein kinase pathway with the endothelial nitric-oxide synthase. J. Biol. Chem 2000, 275, 30707–30715. [Google Scholar]
- Korkmaz, Y; Bloch, W; Steinritz, D; Baumann, MA; Addicks, K; Schneider, K; Raab, WH-M. Bradykinin mediates phosphorylation of eNOS in odontoblasts. J. Dent. Res 2006, 85, 536–541. [Google Scholar]
- Bernier, SG; Haldar, S; Michel, T. Bradykinin-regulated interactions of the mitogen-activated protein kinase pathway with the endothelial nitric-oxide synthase. J. Biol. Chem 2000, 275, 30707–30715. [Google Scholar]
- Smith, LE; Kopchick, JJ; Chen, W; Knapp, J; Kinose, F; Daley, D; Foley, E; Smith, RG; Schaeffer, JM. Essential role of growth hormone in ischemia-induced retinal neovascularization. Science 1997, 276, 1706–1709. [Google Scholar]
- Miller, JW. Vascular endothelial growth factor and ocular neovascularization. Am. J. Pathol 1997, 151, 13–23. [Google Scholar]
- Song, SJ; Chung, H; Yu, HG. Inhibitory effect of YC-1, 3-(5′-hydroxymethyl-2′-furyl)-1- benzylindazole, on experimental choroidal neovascularization in rat. Ophthalmic. Res 2008, 40, 35–40. [Google Scholar]
- Yang, XM; Wang, YS; Zhang, J; Li, Y; Xu, JF; Zhu, J; Zhao, W; Chu, DK; Wiedemann, P. Role of PI3K/Akt and MEK/ERK in mediating hypoxia-induced expression of HIF-1α and VEGF in laser-induced rat choroidal neovascularization. Investig. Ophthalmol. Vis. Sci 2009, 50, 1873–1879. [Google Scholar]

© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, H.-M.; Wei, J.; Pan, L.; Lin, H.; Wang, W.; Zhang, Y.; Shen, Z. The Mechanism of (R,R) ZX-5 on Increasing NO Release. Int. J. Mol. Sci. 2010, 11, 3323-3333. https://doi.org/10.3390/ijms11093323
Xu H-M, Wei J, Pan L, Lin H, Wang W, Zhang Y, Shen Z. The Mechanism of (R,R) ZX-5 on Increasing NO Release. International Journal of Molecular Sciences. 2010; 11(9):3323-3333. https://doi.org/10.3390/ijms11093323
Chicago/Turabian StyleXu, Han-Mei, Jin Wei, Li Pan, Hongying Lin, Weiqiang Wang, Yihua Zhang, and Zilong Shen. 2010. "The Mechanism of (R,R) ZX-5 on Increasing NO Release" International Journal of Molecular Sciences 11, no. 9: 3323-3333. https://doi.org/10.3390/ijms11093323
APA StyleXu, H.-M., Wei, J., Pan, L., Lin, H., Wang, W., Zhang, Y., & Shen, Z. (2010). The Mechanism of (R,R) ZX-5 on Increasing NO Release. International Journal of Molecular Sciences, 11(9), 3323-3333. https://doi.org/10.3390/ijms11093323



