Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review
Abstract
:1. Introduction
2. Recent Works on the Preparation of 1-D CeO2 Nanostructures
2.1. 1-D CeO2 Nanorod
2.2. 1-D CeO2 Nanowire/Nanofiber
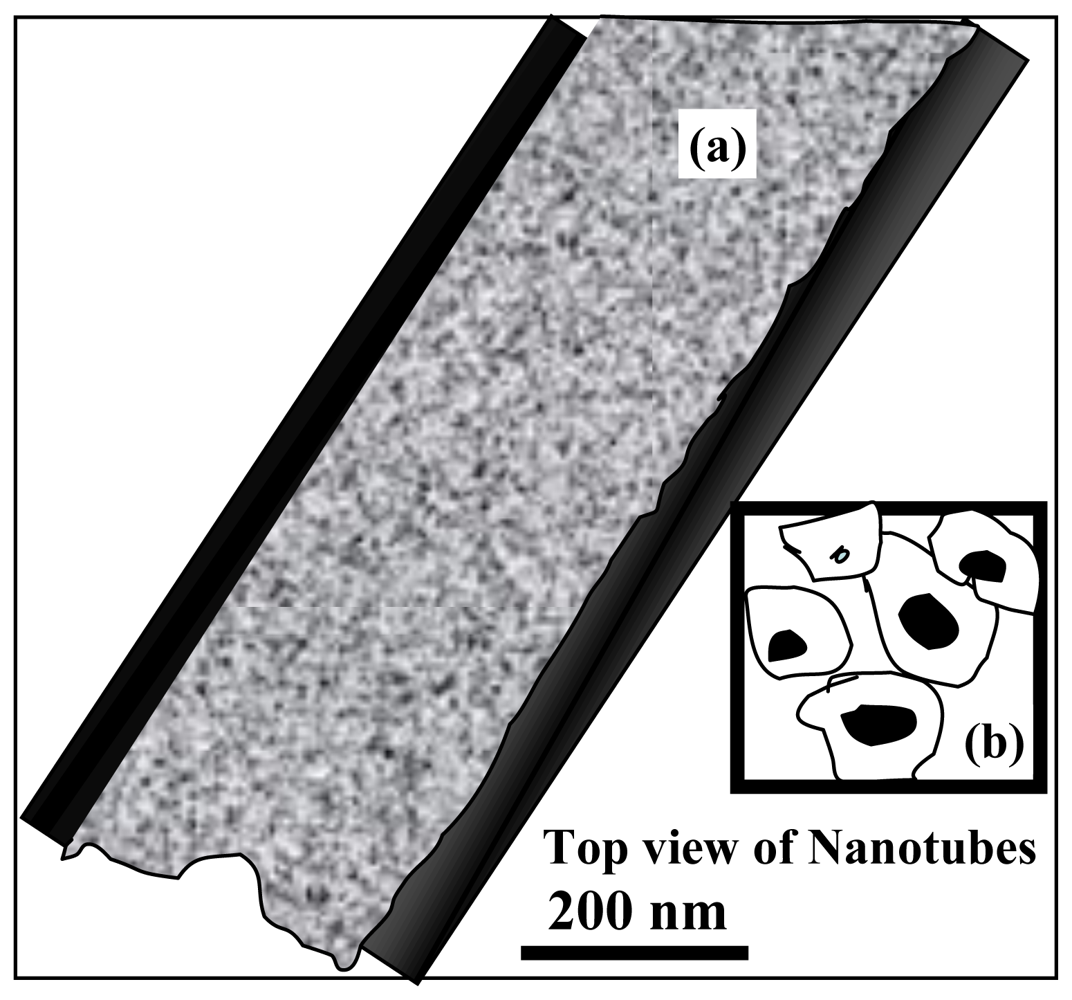
2.3. 1-D CeO2 Nanotube
2.4. Other Types of 1-D CeO2 Nanostructure
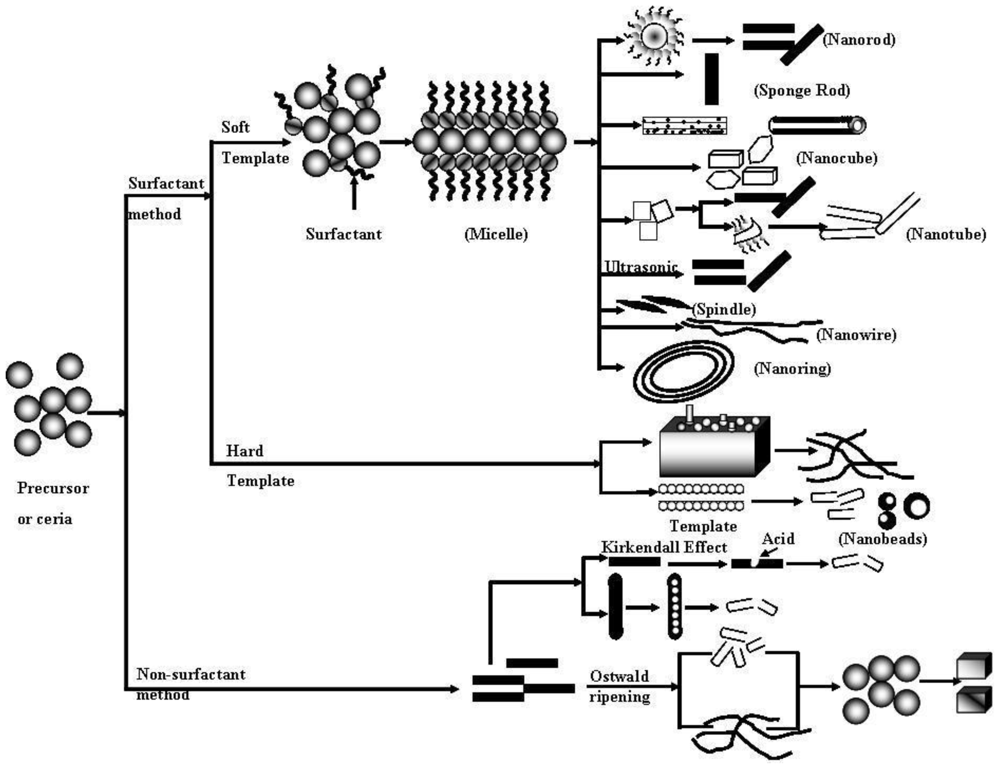
3. Formation Mechanism of 1-D CeO2 Nanostructures
3.1. Surfactant Assisted 1-D CeO2 Nanostructure Formation
3.2. Non-Surfactant Assisted 1-D CeO2 Nanostructure Formation
4. Applications of Ceria Nanostructure Materials
4.1. UV-Vis Absorption
4.2. UV-Vis Absorption Shift Phenomenon
4.3. Carbon-monoxide Oxidation Phenomenon
5. Conclusions
Acknowledgements
References
- Sun, C; Li, H; Zhang, H; Wang, Z; Chen, L. Controlled synthesis of CeO2 nanorods by a solvothermal method. Nanotechnology 2005, 16, 1454–1463. [Google Scholar]
- Ho, C; Yu, JC; Kwong, T; Mak, AC; Lai, S. Morphology-controllable synthesis of mesoporous CeO2 nano-and microstructures. Chem. Mater 2005, 17, 4514–4522. [Google Scholar]
- Tang, B; Zhuo, L; Ge, J; Wang, G; Shi, Z; Niu, J. A surfactant-free route to single-crystalline CeO2 nanowires. Chem. Commun 2005, 28, 3565–3567. [Google Scholar]
- Vantomme, A; Yuan, ZY; Du, G; Su, BL. Surfactant-assisted large-scale preparation of crystalline CeO2 nanorods. Langmuir 2005, 21, 1132–1135. [Google Scholar]
- Pan, C; Zhang, D; Shi, L. CTAB assisted hydrothermal synthesis, controlled conversion and CO oxidation properties of CeO2 nanoplates, nanotubes, and nanorods. J. Solid State Chem 2008, 181, 1298–1306. [Google Scholar]
- Zhou, K; Wang, X; Sun, X; Peng, Q; Li, Y. Enhanced catalytic activity of ceria nanorods from well-defined reactive crystal planes. J. Catal 2005, 229, 206–212. [Google Scholar]
- Huang, PX; Wu, F; Zhu, BL; Gao, XP; Zhu, HY; Yan, TY; Huang, WP; Wu, SH; Song, DY. CeO2 nanorods and gold nanocrystals supported on CeO2 nanorods as catalyst. J. Phys. Chem. B 2005, 109, 19169–19174. [Google Scholar]
- Mai, HX; Sun, LD; Zhang, YW; Si, R; Feng, W; Zhang, HP; Liu, HC; Yan, CH. Shape-selective synthesis and oxygen storage behavior of ceria nanopolyhedra, nanorods, and nanocubes. J. Phys. Chem. B 2005, 109, 24380–24385. [Google Scholar]
- Qi, RJ; Zhu, YJ; Cheng, GF; Huang, YH. Sonochemical synthesis of single-crystalline CeOHCO3 rods and their thermal conversion to CeO2 rods. Nanotechnology 2005, 16, 2502–2506. [Google Scholar]
- Zhang, DE; Ni, XM; Zheng, HG; Zhang, XJ; Song, JM. Fabrication of rod-like CeO2: Characterization, optical and electrochemical properties. Solid State Sci 2006, 8, 1290–1293. [Google Scholar]
- Zhang, D; Fu, H; Shi, L; Pan, C; Li, Q; Chu, Y; Yu, W. Synthesis of CeO2 nanorods via ultrasonication assisted by polyethylene glycol. Inorg. Chem 2007, 46, 2446–2451. [Google Scholar]
- Gao, F; Lu, Q; Komarneni, S. Fast synthesis of cerium oxide nanoparticles and nanorods. J. Nanosci. Nanotechnol 2006, 6, 3812–3819. [Google Scholar]
- Cui, Q; Dong, X; Wang, J; Li, M. Direct fabrication of cerium oxide hollow nanofibers by electrospinning. J. Rare Earths 2008, 26, 664–669. [Google Scholar]
- Gu, F; Wang, Z; Han, D; Shi, C; Guo, G. Reverse micelles directed synthesis of mesoporous ceria nanostructures. Mater. Sci. Eng. B 2007, 139, 62–68. [Google Scholar]
- La, RJ; Hu, ZA; Li, HL; Shang, XL; Yang, YY. Template synthesis of CeO2 ordered nanowire arrays. Mater. Sci. Eng. A 2004, 368, 145–148. [Google Scholar]
- Wu, GS; Xie, T; Yuan, XY; Cheng, BC; Zhang, LD. An improved sol-gel template synthetic route to large-scale CeO2 nanowires. Mater. Res. Bull 2004, 39, 1023–1028. [Google Scholar]
- Sun, C; Li, H; Wang, Z; Chen, L; Huang, X. Synthesis and characterization of polycrystalline CeO2 nanowires. Chem. Lett 2004, 33, 662–663. [Google Scholar]
- Yan, L; Xing, X; Yu, R; Deng, J; Chen, J; Liu, G. Facile alcohothermal synthesis of large-scale ceria nanowires with organic surfactant assistance. J. Phys. B: Condens. Matter 2007, 390, 59–64. [Google Scholar]
- Yada, M; Sakai, S; Torikai, T; Watari, T; Furuta, S; Katsuki, H. Cerium compound nanowires and nanorings templated by mixed organic molecules. Adv. Mater 2004, 16, 1222–1226. [Google Scholar]
- Yang, R; Guo, L. Synthesis of cubic fluorite CeO2 nanowires. J. Mater. Sci 2005, 40, 1305–1307. [Google Scholar]
- Bugayeva, N; Robinson, J. Synthesis of hydrated CeO2 nanowires and nanoneedles. Mater. Sci. Technol 2007, 23, 237–241. [Google Scholar]
- Fuentes, RO; Acuña, LM; Zimicz;, MG; Lamas, DG; Sacanell, JG; Gabriela Leyva, A; Baker, RT. Formation and structural properties of Ce–Zr mixed oxide nanotubes. Chem. Mater 2008, 20, 7356–7363. [Google Scholar]
- Yang, R; Guo, L. Synthesis of the nanotublar cubic fluorite CeO2. Chin. J. Inorg. Chem 2004, 20, 152–158. [Google Scholar]
- Chen, G; Sun, S; Sun, X; Fan, W; You, T. Formation of CeO2 nanotubes from Ce(OH)CO3 nanorods through kirkendall diffusion. Inorg. Chem 2009, 48, 1334–1338. [Google Scholar]
- Zhang, D; Fu, H; Shi, L; Fang, J; Li, Q. Carbon nanotube assisted synthesis of CeO2 nanotubes. J. Solid State Chem 2007, 180, 654–660. [Google Scholar]
- Fang, J; Cao, Z; Zhang, D; Shen, X; Ding, W; Shi, L. Preparation and CO conversion activity of ceria nanotubes by carbon nanotubes templating method. J. Rare Earths 2008, 26, 153–157. [Google Scholar]
- Li, Y; Ding, J; Chen, J; Xu, C; Wei, B; Liang, J; Wu, D. Preparation of ceria nanoparticles supported on carbon nanotubes. Mater. Res. Bull 2002, 37, 313–318. [Google Scholar]
- Wei, J; Ding, J; Zhang, X; Wu, D; Wang, Z; Luo, J; Wang, K. Coated double-walled carbon nanotubes with ceria nanoparticles. Mater. Lett 2005, 59, 322–325. [Google Scholar]
- Zhang, D; Shi, L; Fu, H; Fang, J. Ultrasonic-assisted preparation of carbon nanotube/cerium oxide composites. Carbon 2006, 44, 2849–2867. [Google Scholar]
- Fu, HX; Zhang, DS; Shi, LY; Fang, JH. Synthesis and characterization of cerium oxide nanotubes based on carbon nanotubes. Chem. J. Chin. Univ 2007, 28, 617–620. [Google Scholar]
- Zhang, D; Pan, C; Shi, L; Huang, L; Fang, J; Fu, H. A highly reactive catalyst for CO oxidation: CeO2 nanotubes synthesized using carbon nanotubes as removable templates. Microporous Mesoporous Mater 2009, 117, 193–200. [Google Scholar]
- Yu, KL; Ruan, GL; Ben, YH; Zou, JJ. Convenient synthesis of CeO2 nanotubes. Mater. Sci. Eng. B 2007, 139, 197–200. [Google Scholar]
- Miao, JJ; Wang, H; Li, YR; Zhu, JM; Zhu, JJ. Ultrasonic-induced synthesis of CeO2 nanotubes. J. Cryst. Growth 2005, 281, 525–529. [Google Scholar]
- Dos Santos, ML; Lima, RC; Riccardi, CS; Tranquilin, RL; Bueno, PR; Varela, JA; Longo, E. Preparation and characterization of ceria nanospheres by microwave-hydrothermal method. Mater. Lett 2008, 62, 4509–4511. [Google Scholar]
- Han, WQ; Wu, L; Zhu, Y. Formation and oxidation state of CeO2-x nanotubes. J. Am. Chem. Soc 2005, 127, 12814–12815. [Google Scholar]
- Tang, C; Bando, Y; Liu, B; Golberg, D. Cerium oxide nanotubes prepared from cerium hydroxide nanotubes. Adv. Mater 2005, 17, 3005–3009. [Google Scholar]
- Pan, C; Zhang, D; Shi, L; Fang, J. Template-free synthesis, controlled conversion, and CO oxidation properties of CeO2 nanorods, nanotubes, nanowires, and nanocubes. Eur. J. Inorg. Chem 2008, 15, 2429–2436. [Google Scholar]
- Chen, G; Xu, C; Song, X; Zhao, W; Ding, Y; Sun, S. Interface reaction route to two different kinds of CeO2 nanotubes. Inorg. Chem 2008, 47, 723–728. [Google Scholar]
- Zhou, K; Yang, Z; Yang, S. Highly reducible CeO2 nanotubes. Chem. Mater 2007, 19, 1215–1217. [Google Scholar]
- Wang, ZL; Feng, X. Polyhedral shapes of CeO2 nanoparticles. J. Phys. Chem. B 2003, 107, 13563–13566. [Google Scholar]
- Chen, YC; Chen, KB; Lee, CS; Lin, MC. Direct synthesis of Zr-doped ceria nanotubes. J. Phys. Chem. C 2009, 113, 5031–5034. [Google Scholar]
- Martin, P; Parker, SC; Sayle, DC; Watson, GW. Atomistic modeling of multilayered ceria nanotubes. Nano Lett 2007, 7, 543–546. [Google Scholar]
- Fu, Y; Wei, ZD; Ji, MB; Li, L; Shen, PK; Zhang, J. Morphology-controllable synthesis of CeO2 on a Pt electrode. Nanoscale Res. Lett 2008, 3, 431–434. [Google Scholar]
- Kuiry, SC; Patil, SD; Deshpande, S; Seal, S. Spontaneous self-assembly of cerium oxide nanoparticles to nanorods through supraaggregate formation. J. Phys. Chem. B 2005, 109, 6936–6939. [Google Scholar]
- Ge, M; Guo, C; Li, L; Zhang, B; Feng, Y; Wang, Y. Preparation of CeO2 novel sponge-like rods by emulsion liquid membrane system and its catalytic oxidation property. Mater. Lett 2009, 63, 1269–1271. [Google Scholar]
- Terribile, D; Trovarelli, A; Llorca, J; De Leitenburg, C; Dolcetti, G. The synthesis and characterization of mesoporous high-surface area ceria prepared using a hybrid organic/inorganic route. J. Catal 1998, 178, 299–308. [Google Scholar]
- Wang, H-C; Lu, C-H. Synthesis of cerium hydroxycarbonate powders via a hydrothermal technique. Mater. Res. Bull 2002, 37, 783–792. [Google Scholar]
- Zhang, D; Yan, T; Pan, C; Shi, L; Zhang, J. Carbon nanotube–assisted synthesis and high catalytic activity of CeO2 hollow nanobeads. Mater. Chem. Phys 2009, 113, 527–530. [Google Scholar]
- Zhang, D-E; Zhang, XJ; Ni, XM; Song, JM; Zheng, HG. Optical and electrochemical properties of CeO2 spindles. Chem. Phys. Chem 2006, 7, 2468–2470. [Google Scholar]
- Lu, X; Li, X; Chen, F; Ni, C; Chen, Z. Hydrothermal synthesis of prism-like mesocrystal CeO2. J. Alloys Compd 2009, 476, 958–962. [Google Scholar]
- Elidrissi, B; Addou, M; Regragui, M; Monty, C; Bougrine, A; Kachouane, A. Structural and optical properties of CeO2 thin films prepared by spray pyrolysis. Thin Solid Films 2000, 379, 23–27. [Google Scholar]
- Charitidis, C; Patsalas, P; Logothetidis, S. Optical and mechanical performance of nanostructured cerium oxides for applications in optical devices. J. Phys. Conf. Ser 2005, 10, 226–229. [Google Scholar]
- Masui, T; Fujiwara, K; Machida, K; Adachi, G. Characterization of cerium(IV) oxide ultrafine particles prepared using reversed micelles. Chem. Mater 1997, 9, 2197–2204. [Google Scholar]
- Tatar, B; Sam, ED; Kutlu, K; Ürgen, M. Synthesis and optical properties of CeO2 nanocrystalline films grown by pulsed electron beam. J. Mater. Sci 2008, 43, 5102–5108. [Google Scholar]
- Izu, N; Murayama, N; Shin, W; Matsubara, I; Kanzaki, S. Resistive oxygen sensors using cerium oxide thin films prepared by metal organic chemical vapor deposition and sputtering. Jpn. J. Appl. Phys.: Part 1 2004, 43, 6920–6924. [Google Scholar]
- Porqueras, I; Person, C; Corbella, C; Vives, M; Pinyol, A; Bertran, E. Characteristics of e-beam deposited electrochromic CeO2 thin films. Solid State Ionics 2003, 165, 131–137. [Google Scholar]
- Kanakaraju, S; Mohan, S; Sood, AK. Optical and structural properties of reactive ion beam sputter deposited CeO2 films. Thin Solid Films 1997, 305, 191–195. [Google Scholar]
- Guo, S; Arwin, H; Jacobsen, SN; Järrendahl, K; Helmersson, U. A spectroscopic ellipsometry study of cerium dioxide thin films grown on sapphire by rf magnetron sputtering. J. Appl. Phys 1995, 77, 5369–5376. [Google Scholar]
- Gallage, R; Matsuo, A; Watanabe, T; Matsushita, N; Yoshimura, M. Fabrication of transparent ceria films by spray deposition without post firing. J. Electroceram 2009, 22, 33–39. [Google Scholar]
- Özer, N. Optical properties and electrochromic characterization of sol–gel deposited ceria films. Sol. Energ. Mat. Sol. C 2001, 68, 391–400. [Google Scholar]
- Patsalas, P; Logothetidis, S; Metaxa, C. Optical performance of nanocrystalline transparent ceria films. Appl. Phys. Lett 2002, 81, 466–468. [Google Scholar]
- Zhang, Y; Cheng, T; Hu, Q; Fang, Z; Han, K. Study of the preparation and properties of CeO2 single/multiwall hollow microspheres. J. Mater. Res. Soc 2007, 22, 1472–1478. [Google Scholar]
- Cui, MY; He, JX; Lu, NP; Zheng, YY; Dong, WJ; Tang, WH; Chen, BY; Li, CR. Morphology and size control of cerium carbonate hydroxide and ceria micro/nanostructures by hydrothermal technology. Mater. Chem. Phys 2010, 121, 314–319. [Google Scholar]
- Sun, Z; Zhang, H; An, G; Yang, G; Liu, Z. Supercritical CO2-facilitating large-scale synthesis of CeO2 nanowires and their application for solvent-free selective hydrogenation of nitroarenes. J. Mater. Chem 2010, 20, 1947–1952. [Google Scholar]
- Guo, Z; Jian, F; Du, F. A simple method to controlled synthesis of CeO2 hollow microspheres. Scr. Mater 2009, 61, 48–51. [Google Scholar]
- Deshmukh, SS; Zhang, M; Kovalchuk, VI; D’Itri, JL. Effect of SO2 on CO and C3H6 oxidation over CeO2 and Ce0.75Zr0.25O2. Appl. Catal. B: Environ 2003, 45, 135–145. [Google Scholar]
- Chen, G; Xu, C; Song, X; Xu, S; Ding, Y; Sun, S. Template-free synthesis of single-crystalline- like CeO2 hollow nanocubes. Cryst. Growth Des 2008, 8, 4449–4453. [Google Scholar]
- Fornasiero, P; Dimonte, R; Rao, GR; Kaspar, J; Meriani, S; Trovarelli, A; Graziani, M. Rh-Loaded CeO2-ZrO2 solid-solutions as highly efficient oxygen exchangers: Dependence of the reduction behavior and the oxygen storage capacity on the structural-properties. J. Catal 1995, 151, 168–177. [Google Scholar]
- Sundar, RS; Deevi, S. CO oxidation activity of Cu-CeO nano-composite catalysts prepared by laser vaporization and controlled condensation. J. Nanopart. Res 2006, 8, 497–509. [Google Scholar]
- Sun, C; Li, H; Chen, L. Study of flowerlike CeO2 microspheres used as catalyst supports for CO oxidation reaction. J. Phys. Chem. Solids 2007, 68, 1785–1790. [Google Scholar]
- Guzman, J; Carrettin, S; Corma, A. Spectroscopic evidence for the supply of reactive oxygen during co oxidation catalyzed by gold supported on nanocrystalline CeO2. J. Am. Chem. Soc 2005, 127, 3286–3287. [Google Scholar]

| References | Preparation Procedure | Sample | Band Gap a(eV) | |
|---|---|---|---|---|
| Ed | Ei | |||
| e[2] | Polyol | Polycrystalline CeO2 | 3.19 | N.A. |
| CeO2 nanospheres (80–100 nm), | 3.46 | |||
| Microrods (d WD several100 nm; d L 15 to 20 μm, d AR 25 to 33), | 3.62 | |||
| Spindle-like (d WD several 100 nm, d L 2 to 4 μm, d AR 4-8) | 3.36 | |||
| [49] | Hydrothermal | Spindle like (d WD 800 nm and d L 5 μm) | 3.55 | N.A. |
| [50] | Hydrothermal | CeO2 prism-like mesocrystal Bulk CeO2 | 3.02 3.19 | N.A. |
| [51] | Spray pyrolysis | CeO2 films (cerium chloride) (cerium nitrate) | 3.6 3.53 | N.A. |
| [52] | Electron beam evaporation; Ion beam assisted deposition | Nanostructured CeO2−x | 3.48 | 3.18 |
| e[11] | Ultrasonication | CeO2 nanorods (d AR 10 to 15:1, d L 50–150 nm) | 2.9 | 2.67 |
| [53] | Microemulsion | Ceria ultrafine nanostructure | 3.44 2.6 | 2.87 2.73 |
| [54] | Pulsed electron beam | CeO2 nanocrystalline films | N.A. | 2.58 |
| [56–58] | Physical vapor-deposited | CeO2 films | N.A. | 3.15–3.5 |
| [59] | Spray deposition | CeO2 films | N.A. | 3.06–3.08 |
| [60] | Sol-gel method | CeO2 films | N.A. | 3.03–3.07 |
| References | Sample | T50 [°C] | T100 [°C] | BET [m2g−1] | Remarks |
|---|---|---|---|---|---|
| [54] | CeO2/Al2O3 | 270 | N.A.d | 165 | Microemulsion method provides higher catalytical activity |
| Microemulsion | N.A. | N.A. | 73 | ||
| CeO2/Al2O3 | 320 | N.A. | 167 | ||
| Coprecipitation | N.A. | N.A. | 73 | ||
| [62] | CeO2/single multiwall | 210 | 230 | 44.9 | In the second and third run, provides 100% conversion at 240 °C. |
| [67] | CeO2 hollow | 265 | N.A. | N.A. | Similar conversion provided at the second run. |
| Commercial | >300 | N.A. | N.A. | ||
| [69] | Mesoporous CeO2 with | N.A. | 220 | N.A. | Higher content of the CuO may alter the surface to volume ratio of the catalyst and affect the gas transfer. |
| [70] | CuO | N.A. | N.A. | N.A. | |
| Bulk CeO2 | N.A. | 500 | 141 | ||
| Nano CeO2(NC) | 435 | N.A. | N.A. | ||
| 2%Cu-NC | 166 | N.A. | 107 | ||
| 10%Cu-NC | 148 | N.A. | 131 | ||
| 20%Cu-NC | 150 | N.A. | 118 | ||
| [26] | Bulk CeO2 | >300 | N.A. | 5.67 | Ceria nanotubes are more active than the ceria nanoparticles and bulk ceria due to large surface area. |
| CeO2 nanoparticle | 298 | N.A. | 30.33 | ||
| CeO2 nanotube | 205 | 275 | 83.15 | ||
| e[5] | Nanoplate | 215 | >300 | 37.2 | Crystal plane (100) greatly affects the oxidation. |
| Nanorod | 273 | >340 | 52.5 | ||
| Nanotube | 264 | >325 | 80.1 | ||
| e[2] | CeO2-nanoparticle | 295 | 380 | N.A. | BET surface area increases after the calcination at 400 °C and that may influence the conversion. |
| Spherical | 284 | 315 | 40.3 | ||
| Rods | 265 | 315 | 67.8 | ||
| Spindle | 250 | 300 | 67.4 | ||
| [6] | CeO2 nanorod | N.A. | 275 | 50.1 | N.A. |
| [45] | CeO2 nanoparticle | N.A. | 300 | 62.4 | |
| CeO2 sponge rod | 190 | 205 | N.A. | ||
| [70] | Au/CeO2 nanorod | N.A. | >220 | N.A. | Au-supported nanoparticle provides better conversion due to the thermal stability. |
| CeO2 nano particle | >220 | N.A. | N.A. | ||
| CeO2 nanorod | >220 | N.A. | N.A. | ||
| Au/CeO2 nano particle | N.A. | 160 | N.A. | ||
| [48] | Ceria nanobead | 240 | 300 | 87.5 | CNT templates in the CeO2 hollow nanobeads may be formed from CeO2 − xC. |
| Ceria nanoparticle | >300 | N.A. | 5.7 | ||
| f[37] | Nanoroda | 290 | N.A. | 128.2 | Possesses enough aging time to increase BET surface area and consequently affect oxidation process. |
| Nanorodb | 224 | N.A. | 115.9 | ||
| Nanoparticle | 305 | N.A. | 105.1 | ||
| Nanowire | 245 | N.A. | 79.8 | ||
| Nanotube | 223 | N.A. | 98.2 | ||
| Nanocubec | 315 | N.A. | 3.5 | ||
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, K.-S.; Chowdhury, S. Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review. Int. J. Mol. Sci. 2010, 11, 3226-3251. https://doi.org/10.3390/ijms11093226
Lin K-S, Chowdhury S. Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review. International Journal of Molecular Sciences. 2010; 11(9):3226-3251. https://doi.org/10.3390/ijms11093226
Chicago/Turabian StyleLin, Kuen-Song, and Sujan Chowdhury. 2010. "Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review" International Journal of Molecular Sciences 11, no. 9: 3226-3251. https://doi.org/10.3390/ijms11093226
APA StyleLin, K.-S., & Chowdhury, S. (2010). Synthesis, Characterization, and Application of 1-D Cerium Oxide Nanomaterials: A Review. International Journal of Molecular Sciences, 11(9), 3226-3251. https://doi.org/10.3390/ijms11093226




