Applications of Chitin and Its Derivatives in Biological Medicine
Abstract
:1. Introduction
2. Antioxidant Activity
3. Anti-inflammatory Effects
4. Antimicrobial Effects
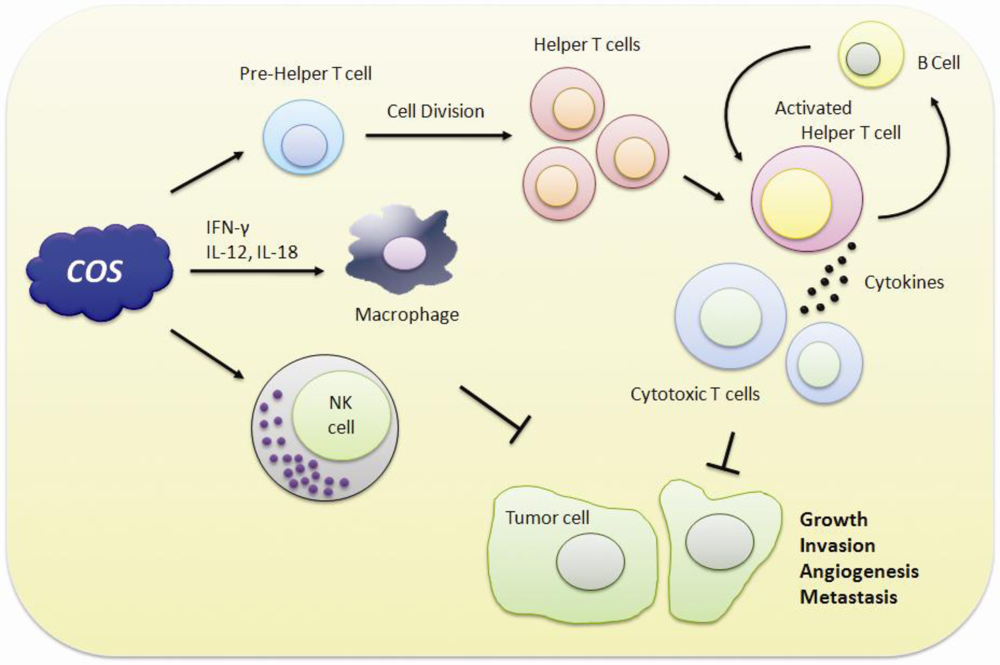
5. Immuno-Stimulating and Anticancer Effects
6. Application in Drug Delivery System
7. Conclusion
Acknowledgments
References
- Domard, A; Domard, M. Chitosan: Structureproperties relationship and biomedical applications. Polym. Biomater 2002, 9, 187–212. [Google Scholar]
- Lavall, R; Assis, O; Campana-Filho, S. [beta]-Chitin from the pens of Loligo sp.: Extraction and characterization. Bioresour. Technol 2007, 98, 2465–2472. [Google Scholar]
- Ngo, D; Kim, M; Kim, S. Chitin oligosaccharides inhibit oxidative stress in live cells. Carbohydr. Polym 2008, 74, 228–234. [Google Scholar]
- Abdou, E; Nagy, K; Elsabee, M. Extraction and characterization of chitin and chitosan from local sources. Bioresour. Technol 2008, 99, 1359–1367. [Google Scholar]
- Lee, D; Xia, W; Zhang, J. Enzymatic preparation of chitooligosaccharides by commercial lipase. Food Chem 2008, 111, 291–295. [Google Scholar]
- Lin, S; Lin, Y; Chen, H. Low molecular weight chitosan prepared with the aid of cellulase, lysozyme and chitinase: Characterisation and antibacterial activity. Food Chem 2009, 116, 47–53. [Google Scholar]
- Agnihotri, S; Mallikarjuna, N; Aminabhavi, T. Recent advances on chitosan-based micro-and nanoparticles in drug delivery. J. Controll. Release 2004, 100, 5–28. [Google Scholar]
- Kumar, M; Muzzarelli, R; Muzzarelli, C; Sashiwa, H; Domb, A. Chitosan chemistry and pharmaceutical perspectives. Chem. Rev 2004, 104, 6017–6084. [Google Scholar]
- Xia, W; Liu, P; Zhang, J; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll 2011, 25, 170–179. [Google Scholar]
- Knorr, D. Use of chitinous polymers in food: A challenge for food research and development. Food Technol. (USA) 1984, 38, 85–89. [Google Scholar]
- Kurita, K. Chemistry and application of chitin and chitosan. Polym. Degrad. Stabil 1998, 59, 117–120. [Google Scholar]
- Razdan, A; Pettersson, D. Effect of chitin and chitosan on nutrient digestibility and plasma lipid concentrations in broiler chickens. Brit. J. Nutr 1994, 72, 277–288. [Google Scholar]
- Ngo, D; Lee, S; Kim, M; Kim, S. Production of chitin oligosaccharides with different molecular weights and their antioxidant effect in RAW 264.7 cells. J. Funct. Foods 2009, 1, 188–198. [Google Scholar]
- Ribeiro, M; Espiga, A; Silva, D; Baptista, P; Henriques, J; Ferreira, C; Silva, J; Borges, J; Pires, E; Chaves, P. Development of a new chitosan hydrogel for wound dressing. Wound Repair Regen 2009, 17, 817–824. [Google Scholar]
- Lim, C; Yaacob, N; Ismail, Z; Halim, A. In vitro biocompatibility of chitosan porous skin regenerating templates (PSRTs) using primary human skin keratinocytes. Toxicol. in Vitro 2010, 24, 721–727. [Google Scholar]
- Li, Z; Yubao, L; Yi, Z; Lan, W; Jansen, J. In vitro and in vivo evaluation on the bioactivity of ZnO containing nano-hydroxyapatite/chitosan cement. J. Biomed. Mater. Res. A 2010, 93, 269–279. [Google Scholar]
- Kulikov, S; Chirkov, S; Il¡̄ina, A; Lopatin, S; Varlamov, V. Effect of the molecular weight of chitosan on its antiviral activity in plants. Appl. Biochem. Microbiol 2006, 42, 200–203. [Google Scholar]
- Wang, S; Chen, J. The protective effect of chitin and chitosan against Vibrio alginolyticus in white shrimp Litopenaeus vannamei. Fish Shellfish Immunol 2005, 19, 191–204. [Google Scholar]
- Rajesh Kumar, S; Ishaq Ahmed, V; Parameswaran, V; Sudhakaran, R; Sarath Babu, V; Sahul Hameed, A. Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in Asian sea bass (Lates calcarifer) to protect from Vibrio (Listonella) anguillarum. Fish Shellfish Immunol 2008, 25, 47–56. [Google Scholar]
- Arnaud, T; Neto, B; Diniz, F. Chitosan effect on dental enamel de-remineralization: An in vitro evaluation. J. Dentistry 2010, 38, 848–852. [Google Scholar]
- Khor, E; Lim, L. Implantable applications of chitin and chitosan. Biomaterials 2003, 24, 2339–2349. [Google Scholar]
- Mark, H; Bikales, N; Overberger, C; Menges, G. Encyclopedia of Polymer Science and Engineering; Wiley-Interscience: New York, NY, USA, 1986. [Google Scholar]
- Morganti, P; Morganti, G. Chitin nanofibrils for advanced cosmeceuticals. Clin. Dermatol 2008, 26, 334–340. [Google Scholar]
- Peniche Covas, C; Alvarez, L; Arguelles Monal, W. The adsorption of mercuric ions by chitosan. J. Appl. Polym. Sci 1992, 46, 1147–1150. [Google Scholar]
- Mckay, G; Blair, H; Gardner, J. Adsorption of dyes on chitin. I. Equilibrium studies. J. Appl. Polym. Sci 1982, 27, 3043–3057. [Google Scholar]
- Chatelet, C; Damour, O; Domard, A. Influence of the degree of acetylation on some biological properties of chitosan films. Biomaterials 2001, 22, 261–268. [Google Scholar]
- Kim, C; Choi, Y; Lee, S; Gin, Y; Son, Y. Development of chitosan dermal scaffold and its characterization. Key Eng. Mater 2007, 342, 181–184. [Google Scholar]
- Rahman, I; Biswas, S; Kode, A. Oxidant and antioxidant balance in the airways and airway diseases. Eur. J. Pharmacol 2006, 533, 222–239. [Google Scholar]
- Blagosklonny, M. Aging: ROS or TOR. Cell Cycle 2008, 7, 3344–3354. [Google Scholar]
- Maynard, S; Schurman, S; Harboe, C; de Souza-Pinto, N; Bohr, V. Base excision repair of oxidative DNA damage and association with cancer and aging. Carcinogenesis 2009, 30, 2–10. [Google Scholar]
- Mirshafiey, A; Mohsenzadegan, M. The role of reactive oxygen species in immunopathogenesis of rheumatoid arthritis. Iran J Allergy Asthma Immunol 2008, 7, 195–202. [Google Scholar]
- Pillai, S; Oresajo, C; Hayward, J. Ultraviolet radiation and skin aging: Roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation-a review. Int. J. Cosmet. Sci 2005, 27, 17–34. [Google Scholar]
- Leung, P; Chan, Y. Role of oxidative stress in pancreatic inflammation. Antioxid. Redox Signal 2009, 11, 135–166. [Google Scholar]
- Jeon, Y; Kim, S. Continuous production of chitooligosaccharides using a dual reactor system. Process Biochem 2000, 35, 623–632. [Google Scholar]
- Chen, A; Taguchi, T; Sakai, K; Kikuchi, K; Wang, M; Miwa, I. Antioxidant activities of chitobiose and chitotriose. Biol. Pharm. Bull 2003, 26, 1326–1330. [Google Scholar]
- Zeng, K; Deng, Y; Ming, J; Deng, L. Induction of disease resistance and ROS metabolism in navel oranges by chitosan. Sci. Horticulturae 2010, 128, 223–228. [Google Scholar]
- Mendis, E; Kim, M; Rajapakse, N; Kim, S. An in vitro cellular analysis of the radical scavenging efficacy of chitooligosaccharides. Life Sci 2007, 80, 2118–2127. [Google Scholar]
- Mori, T; Murakami, M; Okumura, M; Kadosawa, T; Uede, T; Fujinaga, T. Mechanism of macrophage activation by chitin derivatives. J. Vet. Med. Sci 2005, 67, 51–56. [Google Scholar]
- Liu, H; Li, W; Xu, G; Li, X; Bai, X; Wei, P; Yu, C; Du, Y. Chitosan oligosaccharides attenuate hydrogen peroxide-induced stress injury in human umbilical vein endothelial cells. Pharmacol. Res 2009, 59, 167–175. [Google Scholar]
- Xu, W; Huang, H; Lin, C; Jiang, Z. Chitooligosaccharides protect rat cortical neurons against copper induced damage by attenuating intracellular level of reactive oxygen species. Bioorg. Med. Chem. Lett 2010, 20, 3084–3088. [Google Scholar]
- Karadeniz, F; Artan, M; Kong, C; Kim, S. Chitooligosaccharides protect pancreatic [beta]-cells from hydrogen peroxide-induced deterioration. Carbohydr. Polym 2010, 82, 143–147. [Google Scholar]
- Xu, Q; Ma, P; Yu, W; Tan, C; Liu, H; Xiong, C; Qiao, Y; Du, Y. Chitooligosaccharides Protect Human Embryonic Hepatocytes Against Oxidative Stress Induced by Hydrogen Peroxide. Mar. Biotechnol 2010, 12, 292–298. [Google Scholar]
- Anraku, M; Kabashima, M; Namura, H; Maruyama, T; Otagiri, M; Gebicki, J; Furutani, N; Tomida, H. Antioxidant protection of human serum albumin by chitosan. Int. J. Biol. Macromol 2008, 43, 159–164. [Google Scholar]
- Rajapakse, N; Kim, M; Mendis, E; Kim, S. Inhibition of free radical-mediated oxidation of cellular biomolecules by carboxylated chitooligosaccharides. Bioorg. Med. Chem 2007, 15, 997–1003. [Google Scholar]
- Drayton, D; Liao, S; Mounzer, R; Ruddle, N. Lymphoid organ development: From ontogeny to neogenesis. Nat. Immunol 2006, 7, 344–353. [Google Scholar]
- Hu, J; Van den Steen, P; Sang, Q; Opdenakker, G. Matrix metalloproteinase inhibitors as therapy for inflammatory and vascular diseases. Nat. Rev. Drug Discov 2007, 6, 480–498. [Google Scholar]
- Jimi, E; Ghosh, S. Role of nuclear factor-kB in the immune system and bone. Immunol. Rev 2005, 208, 80–87. [Google Scholar]
- Epstein, F; Barnes, P; Karin, M. Nuclear factor-kB--a pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med 1997, 336, 1066–1071. [Google Scholar]
- Albert, S; Baldwin, J. Perspective series-NF-kB in defense and disease-The transcription factor NF-kB and human disease. J. Clin. Investig 2001, 107, 3–6. [Google Scholar]
- Macarthur, M; Hold, G; El-Omar, E. Inflammation and Cancer II. Role of chronic inflammation and cytokine gene polymorphisms in the pathogenesis of gastrointestinal malignancy. Am. J. Physiol.-Gastrointest. Liver Physiol 2004, 286, 515–520. [Google Scholar]
- Da Silva, C; Chalouni, C; Williams, A; Hartl, D; Lee, C; Elias, J. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J. Immunol 2009, 182, 3573–3582. [Google Scholar]
- Kim, M; You, H; You, M; Kim, N; Shim, B; Kim, H. Inhibitory Effect of water soluble chitosan on TNF α and IL 8 secretion from HMC 1. Cells 2004, 26, 401–409. [Google Scholar]
- Moon, J; Kim, H; Koo, H; Joo, Y; Nam, H; Park, Y; Kang, M. The antibacterial and immunostimulative effect of chitosan-oligosaccharides against infection by Staphylococcus aureus isolated from bovine mastitis. Appl. Microbiol. Biotechnol 2007, 75, 989–998. [Google Scholar]
- Yoon, H; Moon, M; Park, H; Im, S; Kim, Y. Chitosan oligosaccharide (COS) inhibits LPS-induced inflammatory effects in RAW 264.7 macrophage cells. Biochem. Biophys. Res. Commun 2007, 358, 954–959. [Google Scholar]
- Ueno, H; Mori, T; Fujinaga, T. Topical formulations and wound healing applications of chitosan. Adv. Drug Deliv. Rev 2001, 52, 105–115. [Google Scholar]
- Kim, M; Kim, S. Chitooligosaccharides inhibit activation and expression of matrix metalloproteinase-2 in human dermal fibroblasts. FEBS Lett 2006, 580, 2661–2666. [Google Scholar]
- Gorzelanny, C; Poppelmann, B; Strozyk, E; Moerschbacher, B; Schneider, S. Specific interaction between chitosan and matrix metalloprotease 2 decreases the invasive activity of human melanoma cells. Biomacromolecules 2007, 8, 3035–3040. [Google Scholar]
- Devlieghere, F; Vermeulen, A; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol 2004, 21, 703–714. [Google Scholar]
- Qin, C; Xiao, Q; Li, H; Fang, M; Liu, Y; Chen, X; Li, Q. Calorimetric studies of the action of chitosan-N-2-hydroxypropyl trimethyl ammonium chloride on the growth of microorganisms. Int. J. Biol. Macromol 2004, 34, 121–126. [Google Scholar]
- Chung, Y; Su, Y; Chen, C; Jia, G; Wang, H; Wu, J; Lin, J. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharmacol. Sinica 2004, 25, 932–936. [Google Scholar]
- Helander, I; Nurmiaho-Lassila, E; Ahvenainen, R; Rhoades, J; Roller, S. Chitosan disrupts the barrier properties of the outer membrane of Gram-negative bacteria. Int. J. Food Microbiol 2001, 71, 235–244. [Google Scholar]
- Je, J; Kim, S. Chitosan derivatives killed bacteria by disrupting the outer and inner membrane. J. Agric. Food Chem 2006, 54, 6629–6633. [Google Scholar]
- Liu, H; Du, Y; Wang, X; Sun, L. Chitosan kills bacteria through cell membrane damage. Int. J. Food Microbiol 2004, 95, 147–155. [Google Scholar]
- Wang, X; Du, Y; Liu, H. Preparation, characterization and antimicrobial activity of chitosan-Zn complex. Carbohydr. Polym 2004, 56, 21–26. [Google Scholar]
- Chen, C; Liau, W; Tsai, G. Antibacterial effects of N-sulfonated and N-sulfobenzoyl chitosan and application to oyster preservation. J. Food Protect 1998, 61, 1124–1128. [Google Scholar]
- Tsai, G; Su, W; Chen, H; Pan, C. Antimicrobial activity of shrimp chitin and chitosan from different treatments and applications of fish preservation. Fisher. Sci 2002, 68, 170–177. [Google Scholar]
- Jeon, Y; Park, P; Kim, S. Antimicrobial effect of chitooligosaccharides produced by bioreactor. Carbohydr. Polym 2001, 44, 71–76. [Google Scholar]
- Ilyina, A; Tikhonov, V; Albulov, A; Varlamov, V. Enzymic preparation of acid-free-water-soluble chitosan. J. Dentistry 2000, 35, 563–568. [Google Scholar]
- Jia, Z. Synthesis and antibacterial activities of quaternary ammonium salt of chitosan. Carbohydr. Res 2001, 333, 1–6. [Google Scholar]
- Suzuki, K; Mikami, T; Okawa, Y; Tokoro, A; Suzuki, S; Suzuki, M. Antitumor effect of hexa-N-acetylchitohexaose and chitohexaose. Carbohydr. Res 1986, 151, 403–408. [Google Scholar]
- Maeda, Y; Kimura, Y. Antitumor effects of various low-molecular-weight chitosans are due to increased natural killer activity of intestinal intraepithelial lymphocytes in sarcoma 180-bearing mice. J. Nutr 2004, 134, 945–950. [Google Scholar]
- Kobayashi, M; Watanabe, T; Suzuki, S; Suzuki, M. Effect of N-acetylchitohexaose against Candida albicans infection of tumor-bearing mice. Microbiol. Immunol 1990, 34, 413–426. [Google Scholar]
- Kimura, Y; Okuda, H. Prevention by chitosan of myelotoxicity, gastrointestinal toxicity and immunocompetent organic toxicity induced by 5-fluorouracil without loss of antitumor activity in mice. Cancer Sci 1999, 90, 765–774. [Google Scholar]
- Zhou, X; Liu, D; Liu, H; Yang, Q; Yao, K; Wang, X; Wang, L; Yang, X. Effect of low molecular weight chitosans on drug permeation through mouse skin: 1. Transdermal delivery of baicalin. J. Pharm. Sci 2010, 99, 2991–2998. [Google Scholar]
- Huang, R; Mendis, E; Rajapakse, N; Kim, S. Strong electronic charge as an important factor for anticancer activity of chitooligosaccharides (COS). Life Sci 2006, 78, 2399–2408. [Google Scholar]
- Shen, K; Chen, M; Chan, H; Jeng, J; Wang, Y. Inhibitory effects of chitooligosaccharides on tumor growth and metastasis. Food Chem. Toxicol 2009, 47, 1864–1871. [Google Scholar]
- Quan, H; Zhu, F; Han, X; Xu, Z; Zhao, Y; Miao, Z. Mechanism of anti-angiogenic activities of chitooligosaccharides may be through inhibiting heparanase activity. Med. Hypotheses 2009, 73, 205–206. [Google Scholar]
- Karagozlu, M; Kim, J; Karadeniz, F; Kong, C; Kim, S. Antiproliferative effect of aminoderivatized chitooligosaccharides on AGS human gastric cancer cells. Process Biochem 2010, 45, 1523–1528. [Google Scholar]
- Wu, H; Yao, Z; Bai, X; Du, Y; Ma, X. Chitooligosaccharides inhibit nitric oxide mediated migration of endothelial cells In vitro and tumor angiogenesis in vivo. Carbohydr. Polym 2010, 82, 927–932. [Google Scholar]
- Harish Prashanth, K; Tharanathan, R. Depolymerized products of chitosan as potent inhibitors of tumor-induced angiogenesis. Biochim. Biophys. Acta Gen. Subjects 2005, 1722, 22–29. [Google Scholar]
- Lee, J; Kang Decker, N; Chatterjee, S; Yao, J; Friedman, S; Shah, V. Mechanisms of nitric oxide interplay with Rho GTPase family members in modulation of actin membrane dynamics in pericytes and fibroblasts. Am. J. Pathol 2005, 166, 1861–1870. [Google Scholar]
- Cooke, J. NO and angiogenesis. Atheroscler. Suppl 2003, 4, 53–60. [Google Scholar]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci 2006, 31, 603–632. [Google Scholar]
- Di Martino, A; Sittinger, M; Risbud, M. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar]
- Janes, K; Fresneau, M; Marazuela, A; Fabra, A; Alonso, M. Chitosan nanoparticles as delivery systems for doxorubicin. J. Controll. Release 2001, 73, 255–267. [Google Scholar]
- De Campos, A; Sanchez, A; Alonso, M. Chitosan nanoparticles: A new vehicle for the improvement of the delivery of drugs to the ocular surface. Application to cyclosporin A. Int. J. Pharm 2001, 224, 159–168. [Google Scholar]
- Xu, Y; Du, Y. Effect of molecular structure of chitosan on protein delivery properties of chitosan nanoparticles. Int. J. Pharm 2003, 250, 215–226. [Google Scholar]
- Li, N; Zhuang, C; Wang, M; Sun, X; Nie, S; Pan, W. Liposome coated with low molecular weight chitosan and its potential use in ocular drug delivery. Int. J. Pharm 2009, 379, 131–138. [Google Scholar]
- Qi, L; Xu, Z. In vivo antitumor activity of chitosan nanoparticles. Bioorg. Med. Chem.Lett 2006, 16, 4243–4245. [Google Scholar]
- Qi, L; Xu, Z; Jiang, X; Li, Y; Wang, M. Cytotoxic activities of chitosan nanoparticles and copper-loaded nanoparticles. Bioorg. Med. Chem. Lett 2005, 15, 1397–1399. [Google Scholar]
- Moghimi, S; Hunter, A; Murray, J. Long-circulating and target-specific nanoparticles: Theory to practice. Pharmacol. Rev 2001, 53, 283–318. [Google Scholar]
- Kreuter, J. Peroral administration of nanoparticles. Adv. Drug Deliv. Rev 1991, 7, 71–86. [Google Scholar]
- Chouly, C; Pouliquen, D; Lucet, I; Jeune, J; Jallet, P. Development of superparamagnetic nanoparticles for MRI: Effect of particle size, charge and surface nature on biodistribution. J. Microencapsul 1996, 13, 245–255. [Google Scholar]
- Qi, L; Xu, Z; Jiang, X; Hu, C; Zou, X. Preparation and antibacterial activity of chitosan nanoparticles. Carbohydr. Res 2004, 339, 2693–2700. [Google Scholar]
- Qi, L; Xu, Z. Lead sorption from aqueous solutions on chitosan nanoparticles. Colloid. Surface Physicochem. Eng. Aspect 2004, 251, 183–190. [Google Scholar]
- Junping, W; Takayama, K; Nagai, T; Maitani, Y. Pharmacokinetics and antitumor effects of vincristine carried by microemulsions composed of PEG-lipid, oleic acid, vitamin E and cholesterol. Int. J. Pharm 2003, 251, 13–21. [Google Scholar]
- Yuan, F; Dellian, M; Fukumura, D; Leunig, M; Berk, D; Torchilin, V; Jain, R. Vascular permeability in a human tumor xenograft: Molecular size dependence and cutoff size. Cancer Res 1995, 55, 3752–3756. [Google Scholar]
- Williams, J; Lansdown, R; Sweitzer, R; Romanowski, M; LaBell, R; Ramaswami, R; Unger, E. Nanoparticle drug delivery system for intravenous delivery of topoisomerase inhibitors. J. Controll. Release 2003, 91, 167–172. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Park, B.K.; Kim, M.-M. Applications of Chitin and Its Derivatives in Biological Medicine. Int. J. Mol. Sci. 2010, 11, 5152-5164. https://doi.org/10.3390/ijms11125152
Park BK, Kim M-M. Applications of Chitin and Its Derivatives in Biological Medicine. International Journal of Molecular Sciences. 2010; 11(12):5152-5164. https://doi.org/10.3390/ijms11125152
Chicago/Turabian StylePark, Bae Keun, and Moon-Moo Kim. 2010. "Applications of Chitin and Its Derivatives in Biological Medicine" International Journal of Molecular Sciences 11, no. 12: 5152-5164. https://doi.org/10.3390/ijms11125152
APA StylePark, B. K., & Kim, M.-M. (2010). Applications of Chitin and Its Derivatives in Biological Medicine. International Journal of Molecular Sciences, 11(12), 5152-5164. https://doi.org/10.3390/ijms11125152



