Anaerobic Biodegradation Tests of Poly(lactic acid) under Mesophilic and Thermophilic Conditions Using a New Evaluation System for Methane Fermentation in Anaerobic Sludge
Abstract
:1. Introduction
2. Experimental Section
2.1. Plastic Sample
2.2. Apparatus for Anaerobic Biodegradation Test
2.3. Procedure of Anaerobic Degradation Test
2.4. Analysis of Evolved Biogas
2.5. Inorganic Carbon Measurement in Sludge
2.6. Calculation of Biodegradability
3. Results and Discussion
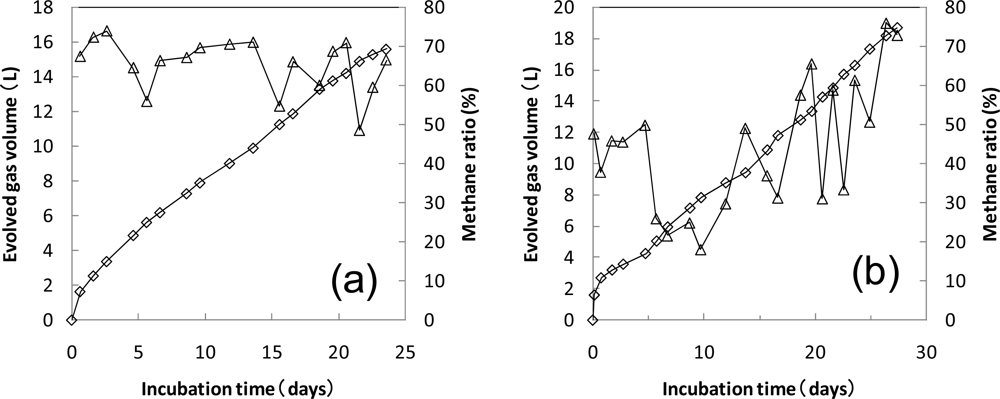
3.1. Preincubation of Sludge
3.2. Anaerobic Biodegradation Test of PLA by MODA-B
4. Conclusions
Acknowledgments
References and Notes
- Kunioka, M; Ninomiya, F; Funabashi, M. Biodegradation of poly(lactic acid) powder proposed as the reference test materials for the international standard of biodegradation evaluation methods. Polym. Degrad. Stab 2006, 91, 1919–1928. [Google Scholar]
- Funabashi, M; Ninomiya, F; Kunioka, M. Biodegradation of polycaprolactone powders proposed as reference test materials for international standard of biodegradation evaluation method. J. Polym. Environ 2007, 15, 7–17. [Google Scholar]
- Yagi, H; Ninomiya, F; Funabashi, M; Kunioka, M. Anaerobic biodegradation tests of poly (lactic acid) and polycaprolactone using new evaluation system for methane fermentation in anaerobic sludge. Polym. Degrad. Stab 2009, 94, 1397–1404. [Google Scholar]
- CRC Handbook of Chemistry and Physics, 75th ed; Lide, DR; Frederikse, HPR (Eds.) CRC Press: Boca Raton, FL, USA; pp. 1994–1995.
- Massardier-Nageotte, V; Pestre, C; Cruard-Pradet, T; Bayard, R. Aerobic and anaerobic biodegradability of polymer films and physico-chemical characterization. Polym. Degrad. Stab 2006, 91, 620–627. [Google Scholar]
- Abou-Zeid, DM; Müller, RJ; Deckwer, WD. Degradation of natural and synthetic polyesters under anaerobic conditions. J. Biotechnol 2001, 86, 113–126. [Google Scholar]
- Shin, PK; Kim, MH; Kim, JM. Biodegradability of degradable plastics exposed to anaerobic digested sludge and simulated landfill condition. J. Environ. Polym. Degrad 1997, 5, 33–39. [Google Scholar]
- Itävaara, M; Karjomaa, S; Selin, JF. Biodegradation of polylactide in aerobic and anaerobic thermophilic conditions. Chemosphere 2002, 46, 879–885. [Google Scholar]
- Bouallagui, H; Touhami, Y; Cheikh, RB; Hamdi, M. Bioreactor performance in anaerobic digestion of fruit and vegetable wastes. Process Biochem 2005, 40, 989–995. [Google Scholar]
- Demirel, B; Yenigün, O. Two-phase anaerobic digestion processes: A review. J. Chem. Technol. Biotechnol 2002, 77, 743–755. [Google Scholar]
- Patel, H; Madamwar, D. Single and multichamber fixed film anaerobic reactors for biomethanation of acidic petrochemical wastewater-systems performance. Process Biochem 2001, 36, 613–619. [Google Scholar]
 Temperature sensor,
Temperature sensor,  Gas sampling bag,
Gas sampling bag,  Pump (Gas phase in head space is exhausted from the bottom of the vessel by this pump),
Pump (Gas phase in head space is exhausted from the bottom of the vessel by this pump),  Surrounding envelope (there is a panel heater inside the black envelope),
Surrounding envelope (there is a panel heater inside the black envelope),  Test vessel,
Test vessel,  Panel heater.
Panel heater.
 Temperature sensor,
Temperature sensor,  Gas sampling bag,
Gas sampling bag,  Pump (Gas phase in head space is exhausted from the bottom of the vessel by this pump),
Pump (Gas phase in head space is exhausted from the bottom of the vessel by this pump),  Surrounding envelope (there is a panel heater inside the black envelope),
Surrounding envelope (there is a panel heater inside the black envelope),  Test vessel,
Test vessel,  Panel heater.
Panel heater.



| ISO 14853 | ISO 15985 | ASTM D5210 | ASTM D5526 | |
|---|---|---|---|---|
| Total solid concentration | 0.1-0.3% | >20% | >0.1% | 35, 45, 60% |
| Incubation temperature | 35 ± 2 °C | 52 ± 2 °C | 35 ± 2 °C | 35 ± 2 °C |
| Volume | 0.1-1 L | >750 mL | 100 mL | >800 g |
| pH | 6.8–7.2 | 7.5–8.5 | 7.5–8.5 | |
| Sample amount | 20–200 mg/L (as organic carbon) | 20 g/vessel | Sufficient carbon content sample | sufficient carbon content sample |
| Inoculum | domestic sewage or laboratory-grown anaerobic sludge | household waste | well-operated anaerobic sludge | household waste |
| Sludge in the tank of the Yamada Biomass Plant | 35 °C | 55 °C | |
|---|---|---|---|
| Total solid concentration (%) | 3.58 | 2.24 | 2.07 |
| Volatile solids (%) | 2.03 | 1.11 | 1.03 |
| Total organic carbon (%) | 0.98 | 0.38 | 0.37 |
| Total nitrogen (%) | 0.30 | 0.24 | 0.24 |
| C/N ratio | 3 | 2 | 2 |
| pH | 8.3 | 8.3 | 8.5 |
| Run | Dissolved CO2 amount (mg) in sludge (1.4 L) in the vessel | Excess dissolved CO2 amount (-blank) | ΣV1 (L) | Biodegradability2 (%) | Total methane (%) | pH |
|---|---|---|---|---|---|---|
| Incubation at 35 °C | ||||||
| 1 blank | 13,890 | 0.03 | 8.0 | |||
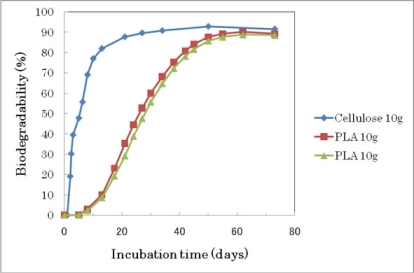
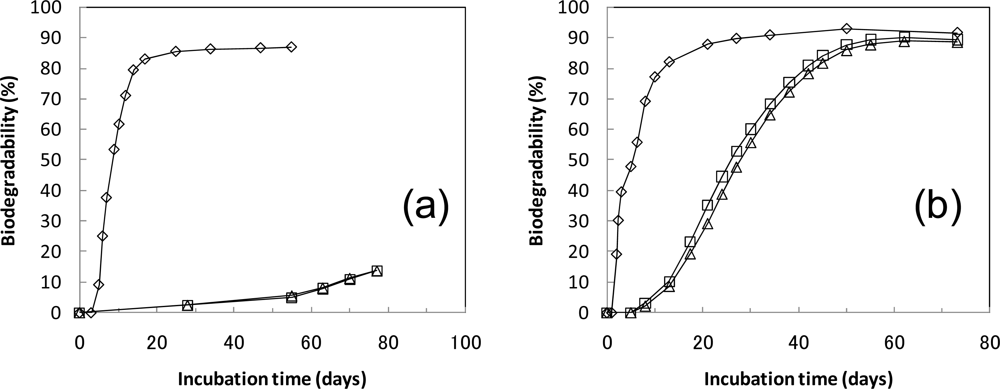
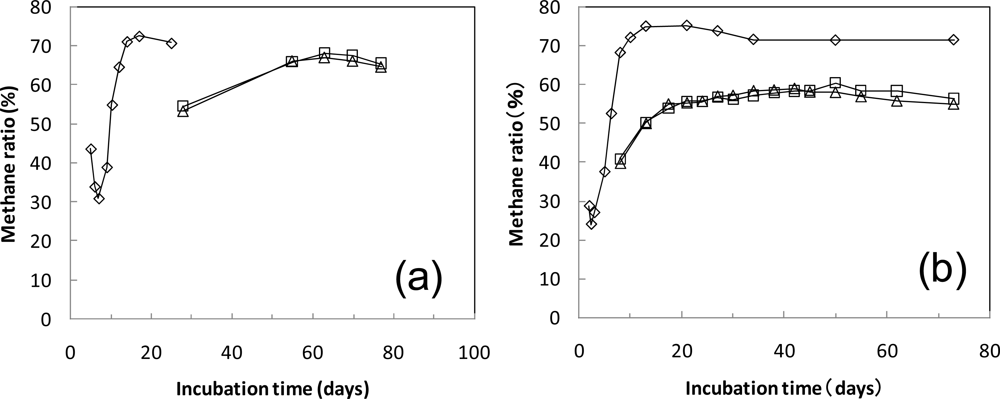
| 2 Cellulose 10 g | 14,000 | 110 mg (0.06 L) | 7.22 | 87 | 47 | 8.0 |
| 3 PLA 10 g | 15,290 | 1,400 mg (0.71 L) | 1.30 | 21* | 7.7 | |
| 4 PLA 10 g | 15,400 | 1,510 mg (0.77 L) | 1.30 | 22* | 7.8 | |
| Incubation at 55 °C | ||||||
| 5 blank | 14,390 | 0.79 | 8.5 | |||
| 6 Cellulose 10 g | 14,670 | 280 mg (0.14 L) | 8.39 | 93 | 50 | 8.4 |
| 7 PLA 10 g | 15,180 | 790 mg (0.40 L) | 9.10 | 94 | 55 | 8.3 |
| 8 PLA 10 g | 15,120 | 730 mg (0.37 L) | 9.02 | 92 | 56 | 8.2 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yagi, H.; Ninomiya, F.; Funabashi, M.; Kunioka, M. Anaerobic Biodegradation Tests of Poly(lactic acid) under Mesophilic and Thermophilic Conditions Using a New Evaluation System for Methane Fermentation in Anaerobic Sludge. Int. J. Mol. Sci. 2009, 10, 3824-3835. https://doi.org/10.3390/ijms10093824
Yagi H, Ninomiya F, Funabashi M, Kunioka M. Anaerobic Biodegradation Tests of Poly(lactic acid) under Mesophilic and Thermophilic Conditions Using a New Evaluation System for Methane Fermentation in Anaerobic Sludge. International Journal of Molecular Sciences. 2009; 10(9):3824-3835. https://doi.org/10.3390/ijms10093824
Chicago/Turabian StyleYagi, Hisaaki, Fumi Ninomiya, Masahiro Funabashi, and Masao Kunioka. 2009. "Anaerobic Biodegradation Tests of Poly(lactic acid) under Mesophilic and Thermophilic Conditions Using a New Evaluation System for Methane Fermentation in Anaerobic Sludge" International Journal of Molecular Sciences 10, no. 9: 3824-3835. https://doi.org/10.3390/ijms10093824
APA StyleYagi, H., Ninomiya, F., Funabashi, M., & Kunioka, M. (2009). Anaerobic Biodegradation Tests of Poly(lactic acid) under Mesophilic and Thermophilic Conditions Using a New Evaluation System for Methane Fermentation in Anaerobic Sludge. International Journal of Molecular Sciences, 10(9), 3824-3835. https://doi.org/10.3390/ijms10093824