Antioxidant Activity of a Red Lentil Extract and Its Fractions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Content of Total Phenolics and Condensed Tannins
2.2. UV Spectra
2.3. Total Antioxidant Activity
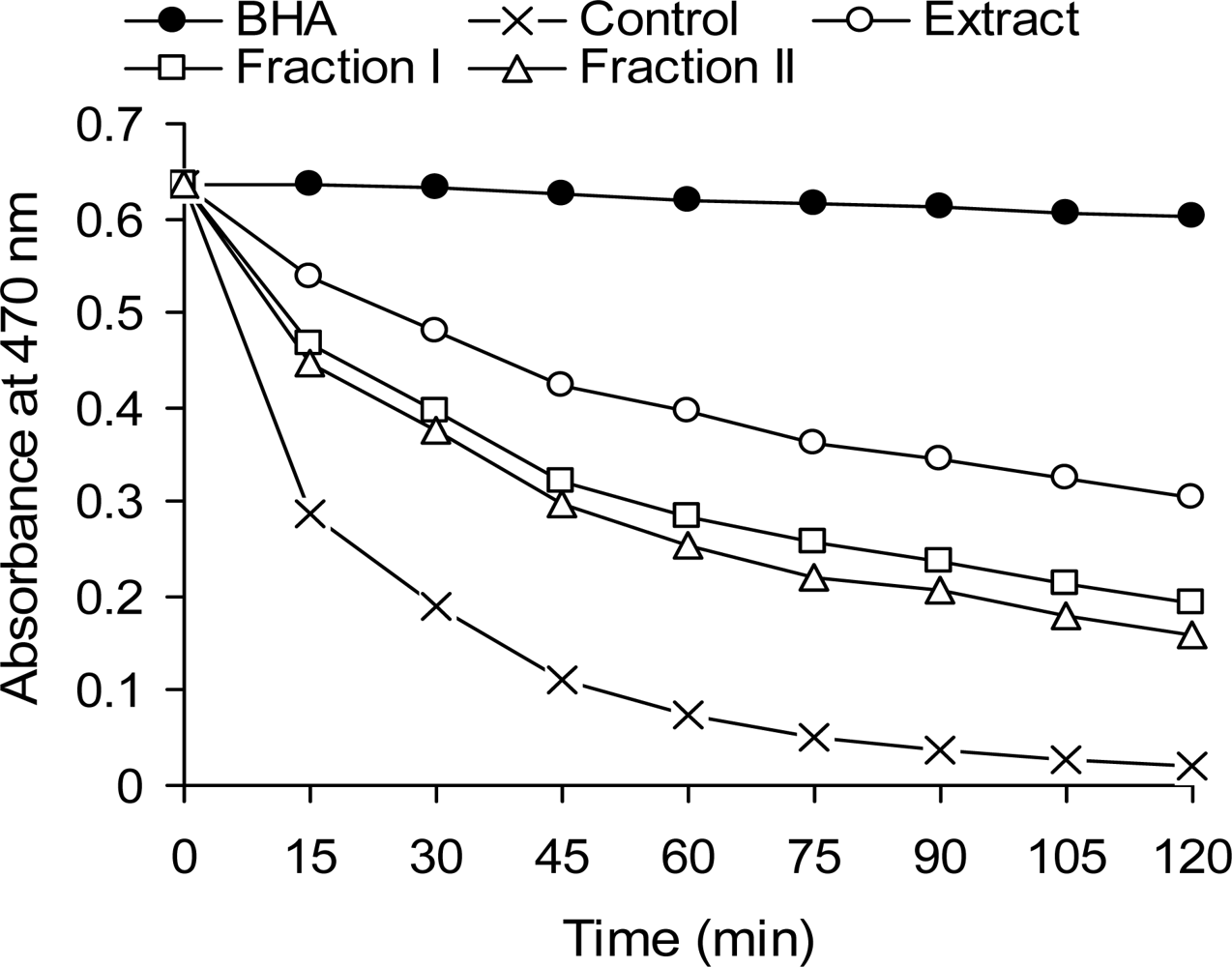
2.4. Antioxidant Activity in the β-Carotene-linoleate Model System
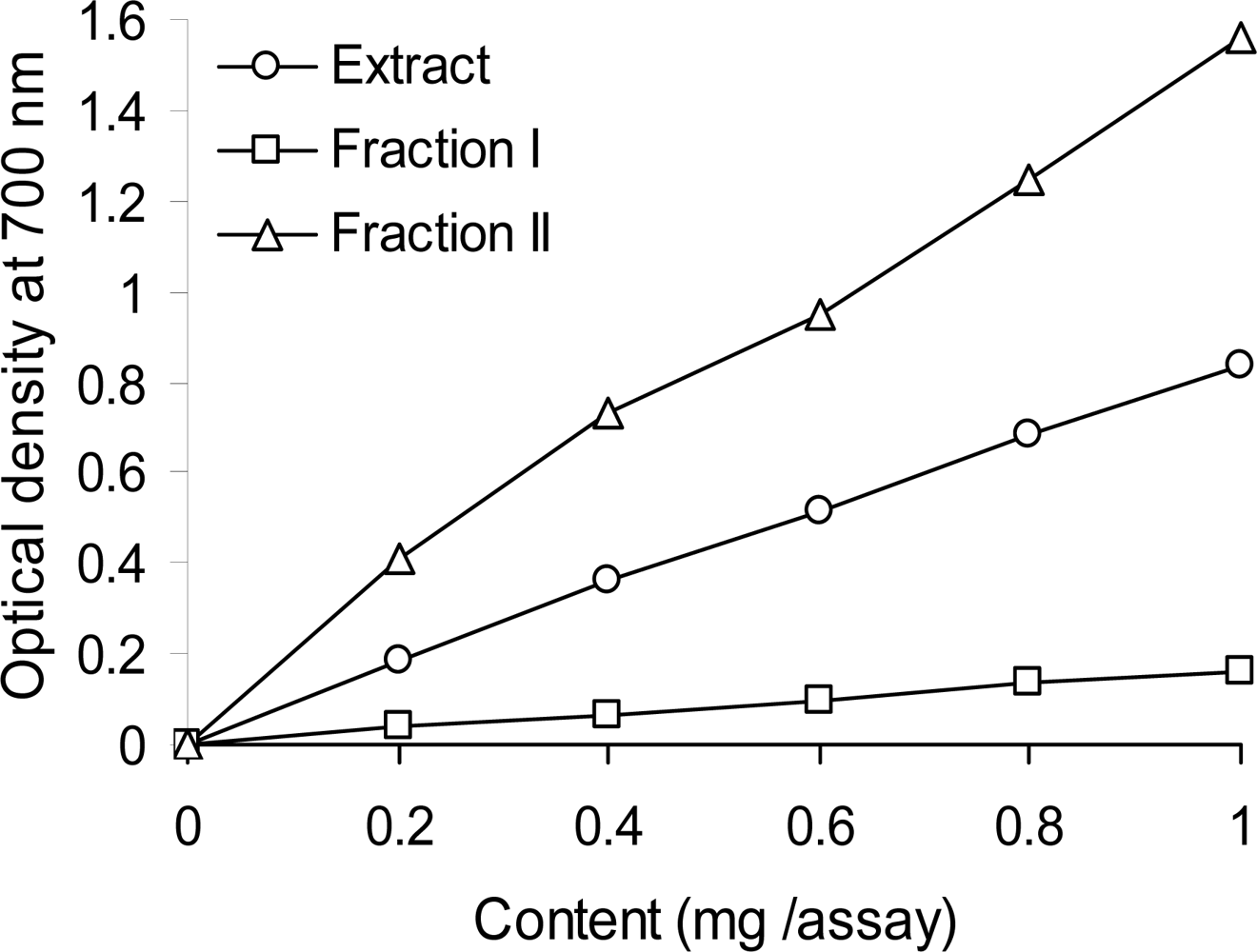
2.5. Reducing Power
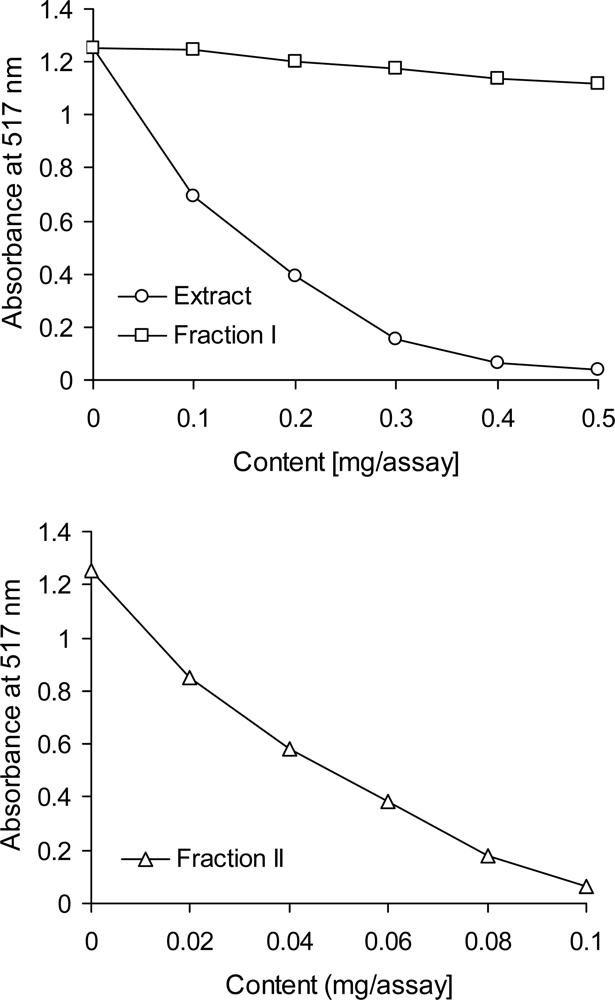
2.6. Scavenging of the DPPH Radical
2.7 Identification and Quantification of Phenolic Compounds
3. Experimental
3.1. Chemicals
3.2. Plant Material
3.3. Extract Preparation
3.4. Column Chromatography
3.5. Content of Total Phenolics
3.6. Condensed Tannins
3.7. UV Spectra
3.8. Total Antioxidant Activity (TAA)
3.9. Antioxidant Activity in a β-Carotene-linoleate Model System
3.10. Reducing Power
3.11. Scavenging of the DPPH Radical
3.12. HPLC-PAD Analysis
3.13. HPLC-ESI-MS Analysis
3.14. Identification and Quantification of Phenolic Compounds
4. Conclusions
References
- Halliwell, B; Gutteridge, JMC; Cross, CE. Free radicals, antioxidants, and human disease: where are we now? J. Lab. Clin. Med 1992, 119, 598–620. [Google Scholar]
- Willett, WC. Diet and health: What should we eat? Science 1994, 264, 532–537. [Google Scholar]
- Kris-Etherton, PM; Hecker, KD; Bonanome, A; Coval, SM; Binkoski, AE; Hilpert, KF. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med 2002, 113, 71S–88S. [Google Scholar]
- Kushi, LH; Meyer, KA; Jacobs, DR, Jr. Cereals, legumes, and chronic disease risk reduction: Evidence from epidemiological studies. Am. J. Clin. Nutr 1999, 70, 451S–458S. [Google Scholar]
- Amarowicz, R; Pegg, RB. Legumes as a source of natural antioxidants. Eur. J. Lipid Sci. Technol 2008, 110, 865–878. [Google Scholar]
- Madhujith, T; Amarowicz, R; Shahidi, F. Phenolic antioxidants in beans and their effects on inhibition of radical-induced DNA damage. J Am Oil Chem Soc 2004, 81, 691–696. [Google Scholar]
- Madhujith, T; Naczk, M; Shahidi, F. Antioxidant activity of common beans (Phaseolus vulgaris L.). J. Food Lipids 2004, 11, 220–233. [Google Scholar]
- Amarowicz, R; Raab, B. Antioxidative activity of leguminous seed extracts evaluated by chemiluminescence methods. Z. Naturforsch. C 1997, 52, 709–712. [Google Scholar]
- Amarowicz, R; Troszyńska, A. Antioxidant activity of extract of pea and its fractions of low molecular phenolics and tannins. Pol J Food Nutr Sci 2003, 12, 10–15. [Google Scholar]
- Amarowicz, R; Troszyńska, A. Antioxidant and antiradical activity of extracts of phenolic compounds from red bean. Czech J. Food Sci 2004, 22, 206–208. [Google Scholar]
- Amarowicz, R; Estrella, I; Hernández, T; Troszyńska, A. Antioxidant activity of extract of adzuki bean and its fractions. J. Food Lipids 2008, 15, 119–136. [Google Scholar]
- Amarowicz, R; Karamać, M; Kmita-Głażewska, H; Troszyńska, A; Kozłowska, H. Antioxidant activity of phenolic fractions of everlasting pea, faba bean and broad bean. J. Food Lipids 1996, 3, 199–211. [Google Scholar]
- Amarowicz, R; Troszyńska, A; Karamać, M; Kozłowska, H. Antioxidative properties of legume seed extracts. In Agri-Food Quality An Inter-Disciplinary Approach; Fenwick, GR, Hedley, C, Richards, RL, Khokhar, S, Eds.; The Royal Society of Chemistry: Cambridge, UK, 1996; pp. 376–379. [Google Scholar]
- Amarowicz, R; Troszyńska, A; Pegg, RB. Antioxidative and radical scavenging effects of phenolics from Vicia sativum. Fitoterapia 2008, 79, 121–122. [Google Scholar]
- Troszyńska, A; Kubicka, E. Superoxide scavenging activity of seed coat extracts from legume seeds. Pol. J. Food Nutr. Sci 2001, 10, 55–59. [Google Scholar]
- Chavan, UD; Amarowicz, R; Shahidi, F. Antioxidant activity of phenolic fractions of beach pea (Lathyrus maritimus L.). J. Food Lipids 1999, 6, 1–11. [Google Scholar]
- Xu, BJ; Chang, SKC. A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J. Food Sci 2007, 72, S159–S166. [Google Scholar]
- Xu, BJ; Yuan, SH; Chang, SKC. Comparative studies on the antioxidant activities of nine common food legumes against copper-induced human low-density lipoprotein oxidation in vitro. J. Food Sci 2007, 72, S522–S527. [Google Scholar]
- Xu, BJ; Yuan, SH; Chang, SKC. Comparative analyses of phenolic composition, antioxidant capacity, and color of cool season legumes and other selected food legumes. J. Food Sci 2007, 72, S167–S177. [Google Scholar]
- Heimler, D; Vignolini, P; Dini, MG; Romani, A. Rapid tests to assess the antioxidant activity of Phaseolus vulgaris L. dry beans. J. Agric. Food Chem 2005, 53, 3053–3056. [Google Scholar]
- Fernandez-Orozco, R; Zieliński, H; Piskuła, MK. Contribution of low-molecular-weight antioxidants to the antioxidant capacity of raw and processed lentil seeds. Nahrung 2003, 47, 291–299. [Google Scholar]
- Han, H; Baik, B-K. Antioxidant activity and phenolic content of lentils (Lens culinaris), chickpeas (Cicer arietinum L.), peas (Pisum sativum L.) and soybeans (Glycine max), and their quantitative changes during processing. Int. J. Food Sci. Technol 2008, 43, 1971–1978. [Google Scholar]
- Amarowicz, R; Piskuła, M; Honke, J; Rudnicka, B; Troszyńska, A; Kozłowska, H. Extraction of phenolic compounds from lentil seeds (Lens culinaris) with various solvents. Pol. J. Food Nutr. Sci 1995, 4, 53–62. [Google Scholar]
- Amarowicz, R; Karamać, M; Chavan, U. Influence of the extraction procedure on the antioxidative activity of lentil seed extracts in a β-carotene-linoleate model system. Grasas Aceites 2001, 52, 89–93. [Google Scholar]
- Amarowicz, R; Karamać, M; Shahidi, F. Antioxidant activity of phenolic fractions of lentil (Lens culinaris). J. Food Lipids 2003, 10, 1–10. [Google Scholar]
- Zieliński, H. Peroxyl radical-trapping capacity of germinated legume seeds. Nahrung 2002, 46, 100–104. [Google Scholar]
- Amarowicz, R; Estrella, I; Hernández, T; Robredo, S; Troszyńska, A; Kosińska, A; Pegg, RB. Free-radical scavenging capacity, antioxidant activity, and phenolic composition of green lentil (Lens culinaris). Food Chem 2009. [Google Scholar]
- Amarowicz, R. Tannins: the new natural antioxidants? Eur. J. Lipid Sci. Technol 2007, 109, 549–551. [Google Scholar]
- Alasalvar, C; Karamać, M; Amarowicz, R; Shahidi, F. Antioxidant and antiradical activities in extracts of hazelnut kernel (Corylus avellana L.) and hazelnut green leafy cover. J. Agric. Food Chem 2006, 54, 4826–4832. [Google Scholar]
- Amarowicz, R; Naczk, M; Shahidi, F. Antioxidant activity of crude tannins of canola and rapeseed hulls. J. Am. Oil Chem. Soc 2000a, 77, 957–961. [Google Scholar]
- Amarowicz, R; Naczk, M; Zadernowski, R; Shahidi, F. Antioxidant activity of condensed tannins of beach pea, canola hulls, evening primrose, and faba beans. J. Food Lipids 2000, 7, 195–205. [Google Scholar]
- Amarowicz, R; Pegg, RB; Rahimi-Moghaddam, P; Barl, B; Weil, JA. Free-radical scavenging capacity and antioxidant activity of selected plant species from the Canadian prairies. Food Chem 2004, 84, 551–562. [Google Scholar]
- Amarowicz, R; Raab, B; Karamać, M. Antioxidative activity of an ethanolic extract of evening primrose. Nahrung/Food 1999, 43, 216–217. [Google Scholar]
- Amarowicz, R; Troszyńska, A; Baryłko-Pikielna, N; Shahidi, F. Polyphenolics extracts from legume seeds: Correlations between total antioxidant activity, total phenolics content, tannins content and astringency. J. Food Lipids 2004, 11, 278–286. [Google Scholar]
- Amarowicz, R; Troszyńska, A; Shahidi, F. Antioxidant activity of almond seed extract and its fraction. J. Food Lipids 2005, 12, 344–358. [Google Scholar]
- Ariga, T; Hamano, M. Radical scavenging action and its mode in procyanidins B-1 and B-3 from azuki beans to peroxyl radicals. Agric. Biol. Chem 1990, 54, 2499–2504. [Google Scholar]
- Ariga, T; Koshiyama, I; Fukushima, D. Antioxidative properties of procyanidins B-1 and B-3 from azuki beans in aqueous systems. Agric. Biol. Chem 1988, 52, 2717–2722. [Google Scholar]
- Muir, AD. Antioxidative activity of condensed tannins. In Natural Antioxidants Chemistry, Health Effects, and Applications; Shahidi, F, Ed.; AOCS Press: Champaign, IL, USA, 1996; pp. 204–212. [Google Scholar]
- Amarowicz, R; Shahidi, F. A rapid chromatographic method for separation of individual catechins from green tea. Food Res. Int 1996, 29, 71–76. [Google Scholar]
- Naczk, M; Wanasundara, PKJPD; Shahidi, F. Facile spectrophotometric quantification method of sinapic acid in hexane-extracted and methanol–ammonia–water-treated mustard rapeseed meals. J. Agric. Food Chem 1992, 40, 444–448. [Google Scholar]
- Siriwardhana, SSKW; Shahidi, F. Antiradical activity of extracts of almond and its by-products. J. Am. Oil. Chem. Soc 2002, 79, 903–908. [Google Scholar]
- Dueñas, M; Hernández, T; Estrella, I. Assessment of in vitro antioxidant capacity of the seed coat and the cotyledon of legumes in relation to their phenolic contents. Food Chem 2006, 98, 95–103. [Google Scholar]
- Dueñas, M; Fernández, D; Hernández, T; Estrella, I; Muñoz, R. Bioactive phenolic compounds of cowpeas (Vigna sinensis L). Modifications by fermentation with natural microflora and with Lactobacillus plantarum ATCC 14917. J. Sci. Food Agric 2005, 85, 297–304. [Google Scholar]
- Strumeyer, DH; Malin, MJ. Condensed tannins in grain sorghum: Isolation, fractionation, and characterization. J. Agric. Food Chem 1975, 23, 909–914. [Google Scholar]
- Price, ML; van Scoyoc, S; Butler, LG. A critical evaluation of the vanillin reaction as an assay for tannin in sorghum grain. J. Agric. Food Chem 1978, 26, 1214–1218. [Google Scholar]
- Miller, HE. A simplified method for the evaluation of antioxidants. J. Am. Oil Chem. Soc 1971, 48, 91. [Google Scholar]
- Oyaizu, M. Studies on products of browning reaction–Antioxidative activities of products of browning reaction prepared from glucosamine. Jap. J. Nutr 1978, 44, 307–315. [Google Scholar]
- Amarowicz, R; Karamać, M; Weidner, S; Abe, S; Shahidi, F. Antioxidant activity of wheat caryopses and embryos extracts. J. Food Lipids 2002, 9, 201–210. [Google Scholar]
- Dueñas, M; Estrella, I; Hernández, T. Occurrence of phenolic compounds in the seed coat and the cotyledon of peas (Pisum sativum L.). Eur. Food Res. Technol 2004, 219, 116–123. [Google Scholar]
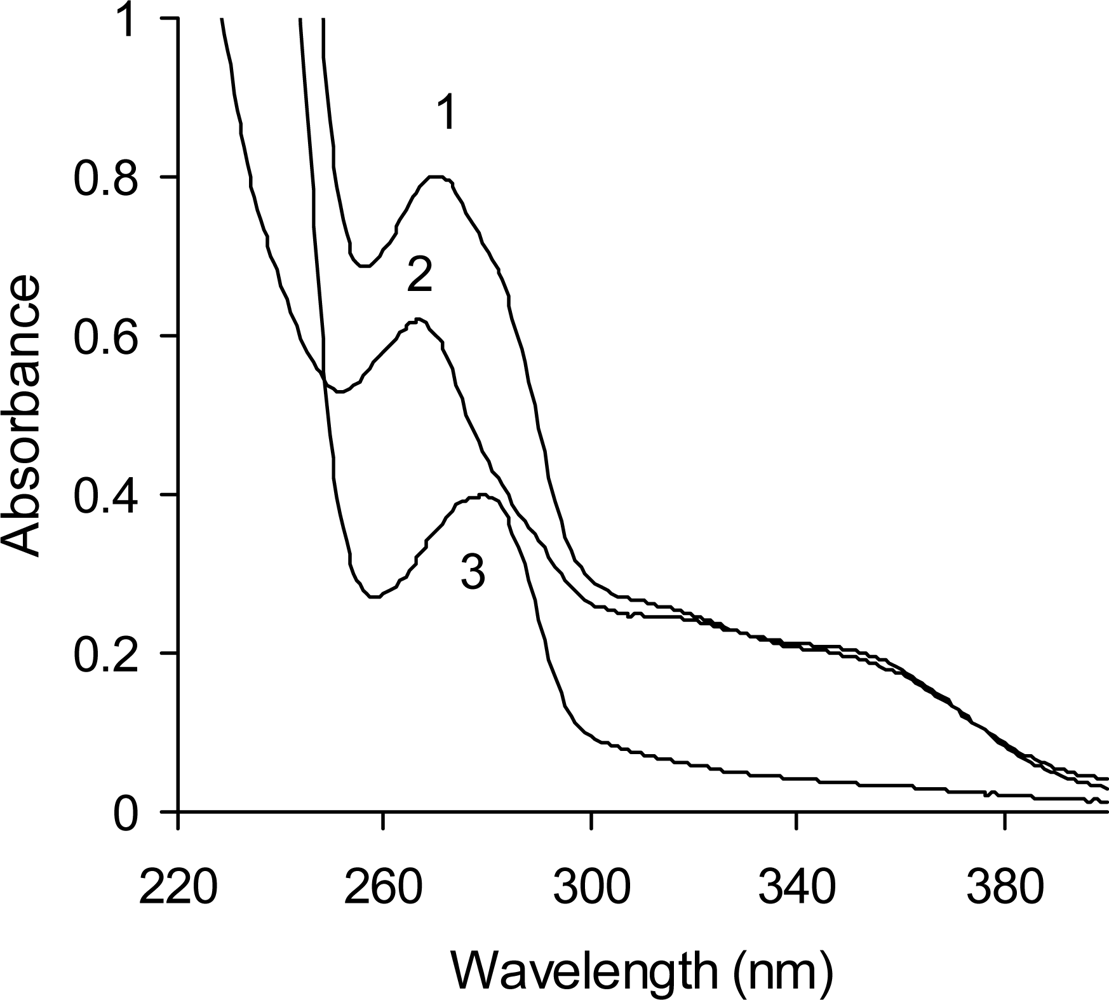
| Analyzed material | Total phenolics (mg/g) | Tannins (A500/g) | Total Antioxidant Activity (μmol Trolox® eq./mg) | λmax (nm) |
|---|---|---|---|---|
| Crude extract | 58 ± 2 | 70 ± 2 | 0.68 ± 0.03 | 271 |
| Fraction 1 | 12 ± 1 | 1.52 ± 0.03 | 0.33 ± 0.02 | 267 |
| Fraction 2 | 290 ± 8 | 129 ± 4 | 5.85 ± 0.20 | 280 |
| Compound number | λmax (nm) | [M-H]− (m/z) | Fragment Ions (m/z) | Compounds | Content (μg/g) |
|---|---|---|---|---|---|
| 1 | 302.5 | 153.1 | Gallic aldehyde | 13.45 ± 1.02 | |
| 2 | 278.8 | 577.1 | 289 | Procyanidin dimer (1) | 20.68 ± 1.23 |
| 3 | 276.4 | 593.1 | 289.1 | Prodelphinidin dimer (1) | 154.8 ± 4.14 |
| 4 | 282.3 | 881.3 | 577; 289 | Digallate procyanidin dimer | 83.29 ± 3.69 |
| 5 | 278.8 | 865 | 577.1; 289 | Procyanidin trimer (1) | 48.2 ±2.11 |
| 6 | 276.5 | 593.1 | 289 | Prodelphinidin dimer (2) | 6.8 ± 0.19 |
| 7 | 255,5 | 137.1 | p-Hydroxybenzoic acid | 73.46 ± 2.09 | |
| 8 | 278.8 | 451.1 | 289.1 | Catechin glucoside | 51.95 ±2.62 |
| 9 | 278.8 | 289 | Catechin | 36.02 ±1.36 | |
| 10 | 277.2 | 745.1 | 577; 169 | Procyanidin gallate | 32.78 ±1.23 |
| 11 | 278.8 | 577 | 289 | Procyanidin dimer (2) | 18.7 ±1.14 |
| 12 | 277 | 203.1 | Tryptophan | 122.4 ±3.7 | |
| 13 | 278.8 | 451.1 | 289 | Epicatechin glucoside | 6.65 ± 0.22 |
| 14 | 278.8 | 289 | Epicatechin | 98.21 ± 3.57 | |
| 15 | 278.8 | 865 | 577.1; 289 | Procyanidin trimer (2) | 39.3 ±1.19 |
| 16 | 309 | 163 | trans-p-Coumaric acid | 38.84 ±2.11 | |
| 17 | 256; 355.5 | 625.3 | 301 | Quercetin diglycoside | 287.84 ±14.3 |
| 18 | 322.9 | 193 | trans-Ferulic acid | 15.99 ±1.12 | |
| 19 | 278.8 | 577 | 289 | Procyanidin dimer (3) | 26.6 ±1.28 |
| 20 | 265; 347.8 | 447,1 | 285 | Kaempferol derivative | 37.56 ±1.96 |
| 21 | 235.7 | 223.2 | Sinapic acid | 0.06 ± 0.02 | |
| 22 | 256; | 505.4 | 301.1 | Quercetin hexose, acylated | 21.17 ±1.03 |
| 23 | 267sh, | 505.4 | 301.1 | Quercetin hexose, acylated | 0.27 ± 0.04 |
| 24 | 325, 335 | 430,1 | 269.1 | apigenin hexose | 14.45 ±1.08 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Amarowicz, R.; Estrella, I.; Hernández, T.; Dueñas, M.; Troszyńska, A.; Kosińska, A.; Pegg, R.B. Antioxidant Activity of a Red Lentil Extract and Its Fractions. Int. J. Mol. Sci. 2009, 10, 5513-5527. https://doi.org/10.3390/ijms10125513
Amarowicz R, Estrella I, Hernández T, Dueñas M, Troszyńska A, Kosińska A, Pegg RB. Antioxidant Activity of a Red Lentil Extract and Its Fractions. International Journal of Molecular Sciences. 2009; 10(12):5513-5527. https://doi.org/10.3390/ijms10125513
Chicago/Turabian StyleAmarowicz, Ryszard, Isabell Estrella, Teresa Hernández, Montserrat Dueñas, Agnieszka Troszyńska, Agnieszka Kosińska, and Ronald B. Pegg. 2009. "Antioxidant Activity of a Red Lentil Extract and Its Fractions" International Journal of Molecular Sciences 10, no. 12: 5513-5527. https://doi.org/10.3390/ijms10125513
APA StyleAmarowicz, R., Estrella, I., Hernández, T., Dueñas, M., Troszyńska, A., Kosińska, A., & Pegg, R. B. (2009). Antioxidant Activity of a Red Lentil Extract and Its Fractions. International Journal of Molecular Sciences, 10(12), 5513-5527. https://doi.org/10.3390/ijms10125513





