Functionality, in Vitro Digestibility and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Protein Concentrates
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Proximate Analysis
2.3. Crude Fibre Determination
2.4. Preparation of Protein Concentrates
2.5. Amino Acid Analysis
2.6. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.7. Protein Digestibility by Trypsin
2.8. Protein Solubility
2.9. Foaming Capacity (FC) and Foam Stability (FS)
2.10. Emulsifying Capacity
2.11. Water/Oil Absorption Capacity
2.12. Bulk Density
2.13. Differential Scanning Calorimetry (DSC)
2.14. Statistical Analysis
3. Results and Discussion
3.1. Proximate Chemical Composition
3.2. Protein content of Foxtail Millet Protein Concentrates
3.3. Amino Acid Analysis
3.4. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
3.5. In Vitro Trypsin Digestibility
3.6. Protein Solubility
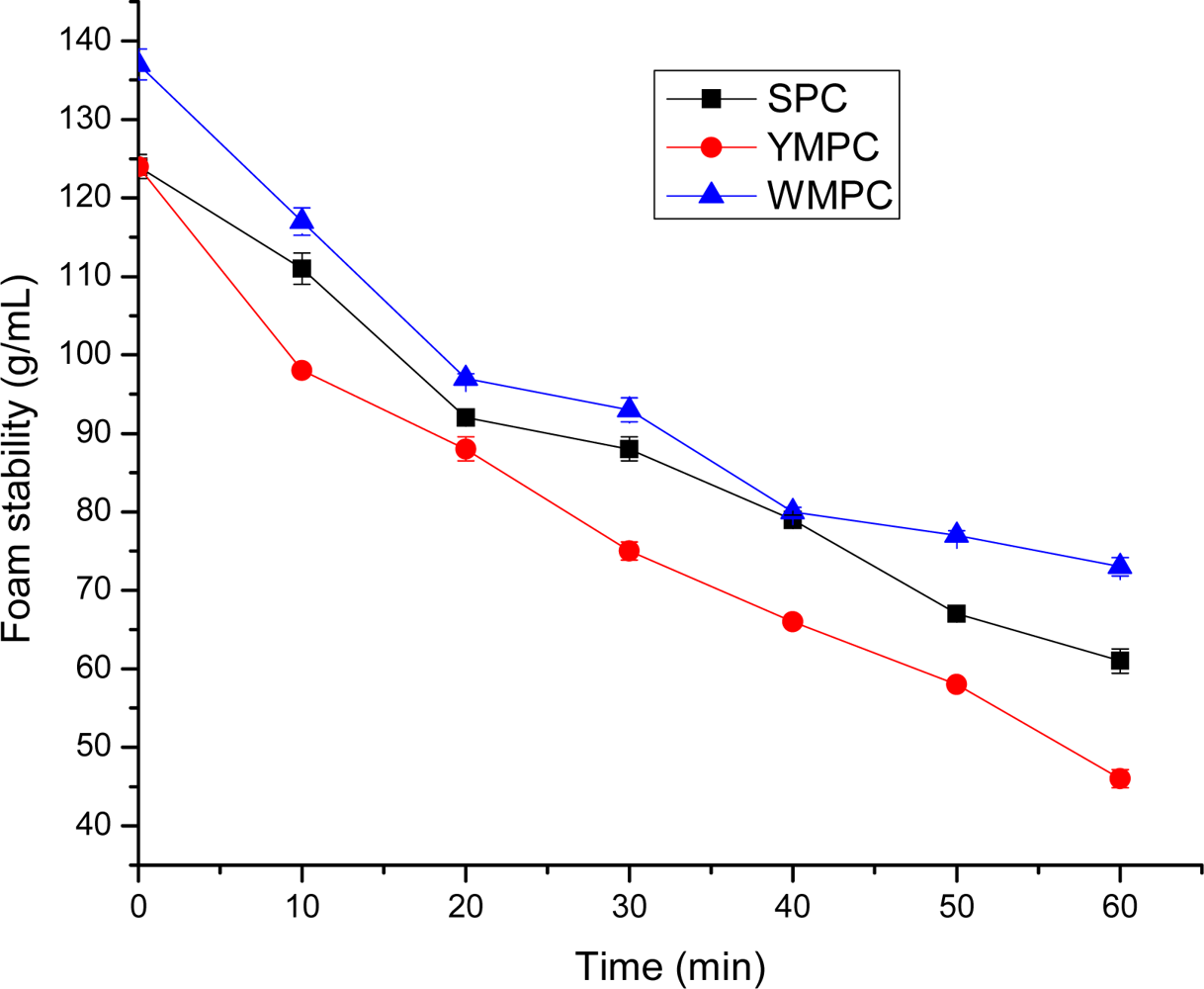
3.7. Foam Capacity and Foam Stability
3.8. Emulsifying Capacity
3.9. Water/Oil Absorption
3.10. Bulk Density
3.11. Differential Scanning Calorimetry (DSC)
4. Conclusions
Acknowledgments
References
- Venktesh, A; Prakash, V. Functional properties of total sunflower (Helianthus annuus L.) seed-effect of physical and chemical treatments. J. Agric. Food Chem 1993, 41, 218–223. [Google Scholar]
- Ahmenda, M; Prinyawiwatkul, W; Rao, R. Solubilized wheat protein isolate: Functional properties and potential food applications. J Agric Food Chem 1999, 47, 1340–1345. [Google Scholar]
- Wilding, P; Lillford, PJ; Regenstein, JM. Functional properties of proteins in foods. J Chem Technol Biotechnol 1984, 34, 182–189. [Google Scholar]
- Obilana, AB; Manyasa, E. Millets grain properties and utilization potential. In Pseudocereals and less common cereals; Belton, PS, Taylor, JRN, Eds.; Springer-Verlag: New York, NY, USA, 2002; pp. 177–217. [Google Scholar]
- Vithal, DS; Machewad, GM. Processing of foxtail millet for improved nutrient availability. J. Food Process Pres 2006, 30, 269–279. [Google Scholar]
- Usha, A; Sipriya, G; Chandra, TS. The effect of fermentation on the primary nutrients in foxtail millet (Setaria italica). Food Chem 1996, 56, 381–384. [Google Scholar]
- Gopalan, C; Ramasashtri, BV; Balasubramanian, SC. Nutritive Value of Indian Foods; National Institute of Nutrition, ICMR: Hyderabad, India, 1987; p. 60. [Google Scholar]
- Li, D. Inhibitory action of ‘food therapy No.1’ on the growth of Sarcoma 180 preliminary report [in Chinese]. J. Foxtail Millet 1986, 1, 1–4. [Google Scholar]
- Damodaran, S. Interfaces, protein films, and foams. Adv. Food Nutr. Res 1990, 34, 1–7. [Google Scholar]
- Kinsella, JE. Functional properties of proteins of proteolytic enzyme modified soy protein isolates. J. Agric. Food Chem 1976, 38, 651–656. [Google Scholar]
- McWatters, A; Holmes, M. Influence of moist heat on solubility and emulsification properties of soy and peanut flours. J. Food Sci 1979, 44, 774–776. [Google Scholar]
- Yatsumatsu, K; Toda, J; Wada, T; Misaki, M; Issi, K. Studies on the functional properties of food-grade soy bean products. Agric. Biol. Chem 1972a, 36, 537–541. [Google Scholar]
- Chandi, GK; Sogi, DS. Functional properties of rice bran protein concentrates. J. Food Eng 2007, 79, 592–597. [Google Scholar]
- Mustafa, AL; Al-Wessali, MS; Al-Basha, OM; Al-Amir, RH. Utilization of cowpea flour and protein isolate in bakery products. Cereal Food World 1986, 31, 756–758. [Google Scholar]
- Ceirwyn, JS. Analytical Chemistry of Foods; Blackie Academic: London, UK, 1995. [Google Scholar]
- Olayide, SL. Functionality of Africa Locus beans (Parkia biglobossa) protein isolate: Effect of pH, ionic strength and various protein concentration. Food Chem 2003, 86, 345–355. [Google Scholar]
- Moore, S. On the determination of cystine and cysteic acid. J. Biol. Chem 1963, 238, 235–237. [Google Scholar]
- Ravindran, G; Bryden, WL. Tryptophan determination in proteins and feedstuffs by ion exchange chromatography. Food Chem 2005, 89, 309–314. [Google Scholar]
- Laemmli, UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
- Elkhalil, EAJ; El Tinay, AH; Mohamed, BE; Elshseikh, EAE. Effect of malt pretreatment on phytic acid and in vitro protein digestibility of sorghum flour. Food chem 2001, 72, 29–32. [Google Scholar]
- Bera, MB; Mukherjee, RK. Solubility, emulsifying, and foaming properties of rice bran protein concentrates. J. Food Sci 1989, 54, 142–145. [Google Scholar]
- Bernardi Don, LS; Pilosof, AMR; Bartholomai, GB. Enzymatic modification of soy protein concentrates by fungal and bacterial proteases. J. Am. oil Chem. Soc 1991, 68, 102–105. [Google Scholar]
- Yasumatsu, K; Sawada, K; Moritaka, S; Misaki, M; Toda, J; Wada, T; Ishi, K. Whipping and emulsifying properties of soy beans products. J. Agric. Boil. Chem 1972b, 36, 719–727. [Google Scholar]
- Sudharshana, L; Monteiro, PV; Ramachandra, G. Studies of kodo millet (Paspalumscro biculatum). J. Sci. Food Agric 1988, 42, 315–323. [Google Scholar]
- Oshodi, AA; Ogungbenle, HN. Chemical composition, nutritionally valuable minerals and functional properties of benniseed, pearl millet and quinoa flours. Int. J. Food Sci. Nutr 1999, 50, 325–331. [Google Scholar]
- Amado, R; Arrigoni, E. Nutritive and functional properties of wheat germ. Int. Food Ingred 1992, l4, 30–34. [Google Scholar]
- Monteiro, PV; Sudharshana, L; Ramachandra, G. Japanese barnyard millet (Echinochloa frumentacea): protein content, quality and SDS-PAGE of protein fractions. J. Sci. Food Agric 1988, 43, 17–25. [Google Scholar]
- Van Der Plancken, I; Van Remoortere, M; Van Loey, A; Hendrickx, ME. Heat induced changes in the susceptibility of egg white proteins to enzymatic hydrolysis: A kinetic study. J. Agric. Food Chem 2003, 51, 3819–3823. [Google Scholar]
- Zahra, S; Ahmed, GM; Yassen, AAE; El-Moniem, Abd. Comparative studies on protein fractions and amino acid composition from sorghum and pearl millet. Mol. Nutr. Food Res 1996, 40, 305–309. [Google Scholar]
- Damodaran, S. Structure-function relationship of food proteins. In Protein Functionality in Food Systems; Hettiarachchy, NS, Zieger, GR, Eds.; Marcel Dekker: New York, NY, USA, 1994; pp. 190–225. [Google Scholar]
- Singh, RU; Shrikantan, LN; Malleshi, G. The functional properties of popped, flaked, extruded and roller-dried foxtail millet (Setaria italica). Int. J. Food Sci. Technol 2004, 39, 907–915. [Google Scholar]
- Petruccelli, S; Anon, MC. Soy protein isolates components and their interactions. J. Agric. Food Chem 1994, 43, 1762–1767. [Google Scholar]
- Kanu, PJ; Zhu, K; Huming, Z; Haifeng, Q; Kanu, JB; Kexue, Z. Semame Protein 11: Functional properties of sesame (Sesamum indicum L). Protein isolate as influenced by pH, temperature, time, and ratio of flour to water during its production. Asian J. Biochem 2007, 2, 289–301. [Google Scholar]
- Tang, S; Hettiarachchy, NS; Horax, R; Eswaranandam, S. Physicochemical properties and functionality of rice bran protein hydrolyzate prepared from heat-stabilized defatted rice bran with the aid of enzymes. J. Food Sci 2003, 68, 152–157. [Google Scholar]
- Barbut, S. Determining Water and fat holding. In Methods of Testing Protein Functionality; Hall, GM, Ed.; Blackie Academic and Professional: New York, NY, USA, 1999; pp. 186–225. [Google Scholar]
- Kruse, JP; Bagger, C; Schwenke, KD. Reheological properties of modified lupin proteins. Naung/Food 2001, 45, 412–415. [Google Scholar]
- Wang, JC; Kinsella, JE. Functional properties of novel proteins, Alfalfa leaf protein. J. Food Sci 1976, 41, 286–289. [Google Scholar]
- Ju, ZY; Hettiarachchy, NS; Rath, N. Extraction, denaturation and hydrophobic properties of rice flour proteins. J. Food Sci 2001, 66, 229–232. [Google Scholar]
- Wang, M; Hettiarachchy, NS; Qi, M; Burks, W; Siebenmorgen, TJ. Preparation and functional properties of rice bran protein isolate. J. Agric. Food Chem 1999, 47, 411–416. [Google Scholar]


| Sample | Protein | Moisture | Fat | Ash | Crude fiber | Carbohydrate |
|---|---|---|---|---|---|---|
| WMF | 11.50 ± 1.08 | 10.45 ± 0.14 | 2.38 ± 0.03 | 0.47 ± 0.03 | 1.88 ± 0.01 | 73.33 ± 0.03 |
| YMF | 11.41 ± 0.15 | 10.22 ± 0.13 | 2.90 ± 0.35 | 0.68 ± 0.04 | 1.92 ± 0.02 | 73.00 ± 0.14 |
| DWMF | 11.92 ± 0.30 | 12.23 ± 0.04 | 0.41 ± 0.15 | 0.44 ± 0.04 | 1.95 ± 0.02 | 72.92 ± 0.01 |
| DYMF | 11.39 ± 0.38 | 12.09 ± 0.10 | 0.66 ± 0.17 | 0.91 ± 0.03 | 2.02 ± 0.10 | 72.91 ± 0.08 |
| Essential Amino Acids | DWMF | DYMF | WMPC | YMPC | SPC | FAO/WHO/UNUa | |
|---|---|---|---|---|---|---|---|
| Child | Adult | ||||||
| Isoleucine (Ile) | 4.58 | 4.59 | 3.82 | 3.91 | 5.18 | 3.0 | 3.0 |
| Leucine (Leu) | 13.14 | 13.60 | 8.58 | 8.71 | 8.13 | 6.0 | 5.9 |
| Lysine (lys) | 3.43 | 3.85 | 5.17 | 6.07 | 6.74 | 4.8 | 4.5 |
| Methionine (Met) | 2.72 | 3.06 | 2.66 | 2.42 | 1.99 | ||
| Met + Cys | 3.06 | 3.50 | 3.79 | 3.15 | 2.49 | 2.3b | 1.6b |
| Phenylalanine (Phe) | 7.73 | 6.27 | 5.21 | 5.34 | 5.54 | ||
| Phe + Tyr | 10.68 | 8.71 | 9.33 | 9.00 | 8.79 | 4.1c | 3.8c |
| Threonine (Thr) | 2.76 | 3.68 | 4.28 | 4.55 | 3.57 | 2.5 | 2.3 |
| Valine (Val) | 5.58 | 5.81 | 5.74 | 5.79 | 5.57 | 2.9 | 3.9 |
| Histidine (His) | 2.06 | 2.11 | 3.01 | 2.80 | 2.54 | 1.6 | 1.5 |
| Tryptophan (Trp) | 1.14 | 1.39 | 1.51 | 1.53 | 0.02 | 0.66 | 0.6 |
| Nonessential Amino Acids | |||||||
| Alanine (Ala) | 10.89 | 9.30 | 6.15 | 6.35 | 4.29 | ||
| Arginine (Arg) | 4.91 | 4.78 | 8.63 | 7.86 | 7.83 | ||
| Aspartic acid (Asp)d | 6.51 | 7.71 | 8.49 | 8.97 | 11.27 | ||
| Cysteine (Cys)e | 0.34 | 0.45 | 1.23 | 0.74 | 0.50 | ||
| Glutamic Acid (Glu)f | 23.77 | 22.00 | 14.97 | 14.08 | 21.04 | ||
| Glycine (Gly) | 2.22 | 2.91 | 5.06 | 5.21 | 4.23 | ||
| Serine (Ser) | 5.17 | 4.56 | 5.14 | 5.19 | 2.54 | ||
| Tyrosine (Tyr) | 2.94 | 2.44 | 4.11 | 3.66 | 3.35 | ||
| Proline (Pro) | 5.10 | 5.54 | 5.38 | 6.49 | 3.58 | ||
| SPC* | YMPC** | WMPC*** | |
|---|---|---|---|
| In vitro digestibility (%) | 84 ± 1.15b | 78 ± 0.58a | 81 ± 1.53ab |
| Emulsifying capacity (%) | 53 ± 1.2a | 65 ± 1.50b | 74 ± 1.00c |
| Foam capacity (g/mL) | 124 ± 1.53a | 124 ± 1.00a | 137 ± 2.00b |
| Water holding capacity (g/g) | 5 ± 0.07a | 6 ± 0.06b | 7 ± 0.15c |
| Oil holding capacity (g/g) | 6 ± 0.22ab | 5 ± 0.09a | 6 ± 0.61b |
| Bulk density (g/mL) | 0.15 ± 0.03a | 0.17 ± 0.01ab | 0.22 ± 0.02b |
| DSC Measure a (°C) | |||||
|---|---|---|---|---|---|
| Protein sourceb | To | Tp | Te | ΔH J/g | Ar. mJ |
| WMPC | 84.06 | 86.79 | 88.29 | 0.10 | 0.15 |
| YMPC | 87.55 | 88.98 | 88.98 | 0.01 | 0.03 |
| SPC | 86.25 | 88.31 | 88.84 | 0.01 | 0.03 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mohamed, T.K.; Zhu, K.; Issoufou, A.; Fatmata, T.; Zhou, H. Functionality, in Vitro Digestibility and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Protein Concentrates. Int. J. Mol. Sci. 2009, 10, 5224-5238. https://doi.org/10.3390/ijms10125224
Mohamed TK, Zhu K, Issoufou A, Fatmata T, Zhou H. Functionality, in Vitro Digestibility and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Protein Concentrates. International Journal of Molecular Sciences. 2009; 10(12):5224-5238. https://doi.org/10.3390/ijms10125224
Chicago/Turabian StyleMohamed, Tabita Kamara, Kexue Zhu, Amadou Issoufou, Tarawalie Fatmata, and Huiming Zhou. 2009. "Functionality, in Vitro Digestibility and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Protein Concentrates" International Journal of Molecular Sciences 10, no. 12: 5224-5238. https://doi.org/10.3390/ijms10125224
APA StyleMohamed, T. K., Zhu, K., Issoufou, A., Fatmata, T., & Zhou, H. (2009). Functionality, in Vitro Digestibility and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Protein Concentrates. International Journal of Molecular Sciences, 10(12), 5224-5238. https://doi.org/10.3390/ijms10125224




