Structure-Properties Relationship of Dimeric Surfactants from Butyl Glucosides
Abstract
:Introduction
Experimental
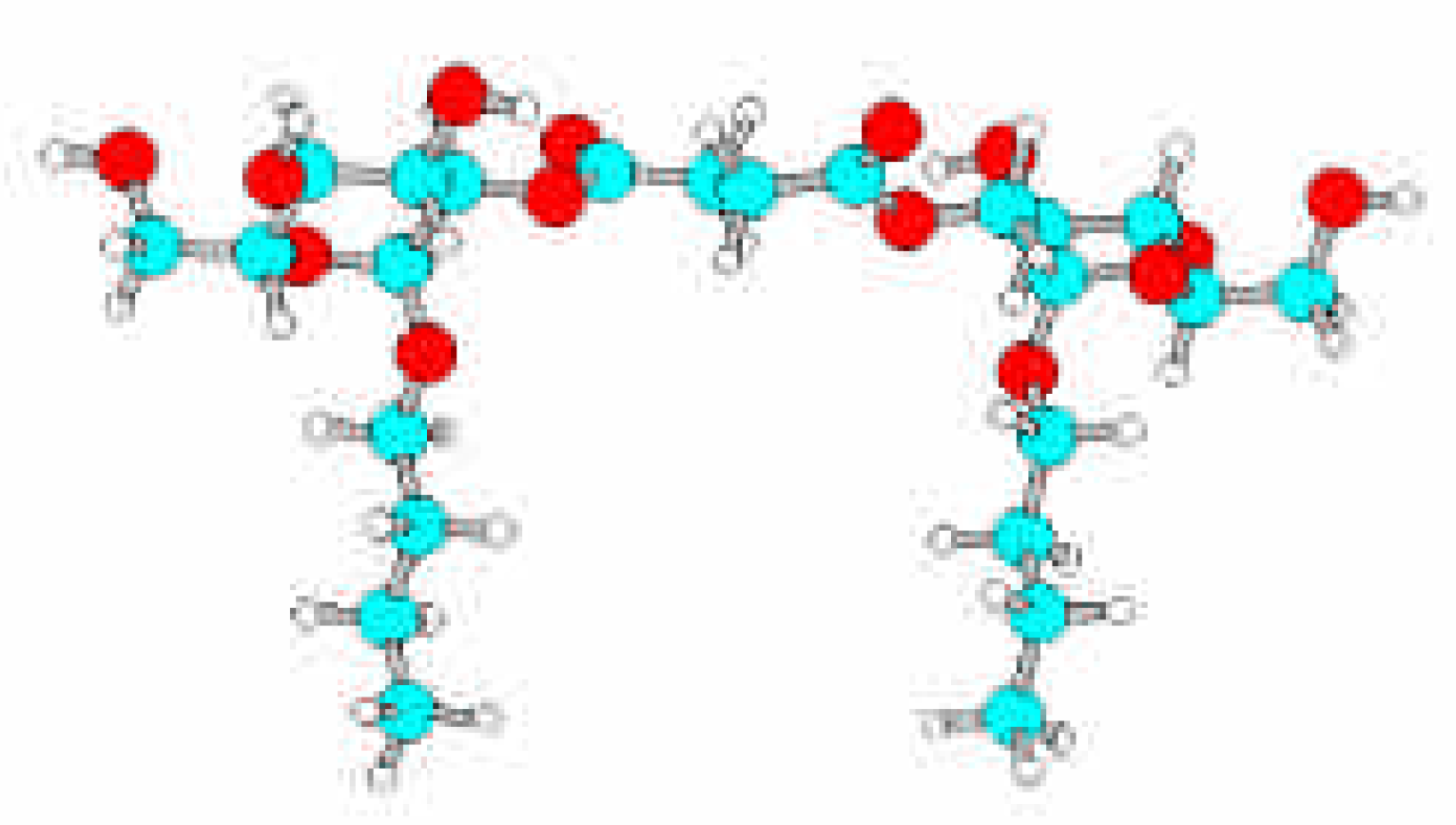
Results and Discussion
Acknowledgements
References and Notes
- Zana, R.; Benrraou, M.; Rueff, R. Langmuir 1991, 7, 1072.
- Menger, F. M.; Littau, C. A. J. Am. Chem. Soc. 1991, 113, 1451. [CrossRef]
- Castro, M. J. L.; Kovensky, J.; Fernández Cirelli, A. Tetrahedron Lett. 1997, 38, 3995.
- Castro, M. J. L.; Kovensky, J.; Fernández Cirelli, A. Tetrahedron 1999, 55, 12711.
- Möbius, D.; Miller, R. Drops and Bubbles in Interfacial Research; Elsevier Science, 1998; pp. 279–326. [Google Scholar]
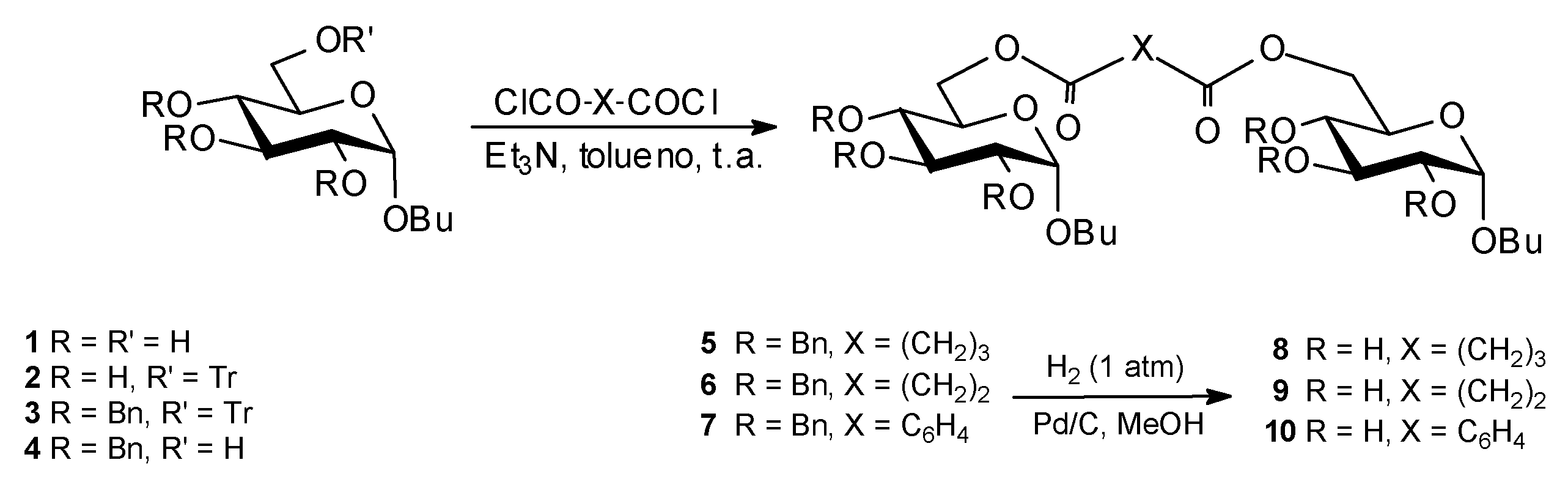
Share and Cite
Castro, M.J.L.; Kovensky, J.; Cirelli, A.F. Structure-Properties Relationship of Dimeric Surfactants from Butyl Glucosides. Molecules 2000, 5, 608-609. https://doi.org/10.3390/50300608
Castro MJL, Kovensky J, Cirelli AF. Structure-Properties Relationship of Dimeric Surfactants from Butyl Glucosides. Molecules. 2000; 5(3):608-609. https://doi.org/10.3390/50300608
Chicago/Turabian StyleCastro, Mariano J. L., José Kovensky, and Alicia Fernández Cirelli. 2000. "Structure-Properties Relationship of Dimeric Surfactants from Butyl Glucosides" Molecules 5, no. 3: 608-609. https://doi.org/10.3390/50300608
APA StyleCastro, M. J. L., Kovensky, J., & Cirelli, A. F. (2000). Structure-Properties Relationship of Dimeric Surfactants from Butyl Glucosides. Molecules, 5(3), 608-609. https://doi.org/10.3390/50300608



