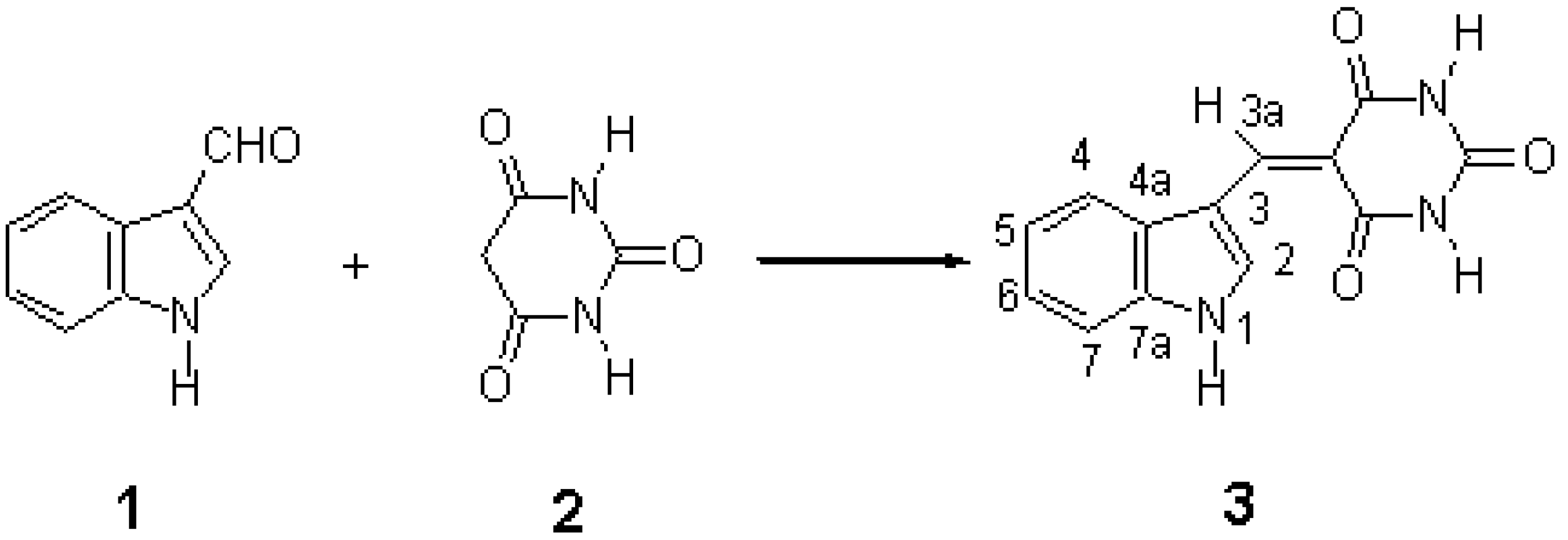
5-(Indol-3-yl)barbituric acid
Supplementary materials
Supplementary File 1Supplementary File 2References
- Jones, G. Org. React. 1967, 15, 203.
- Tanaka, K.; Chen, X.; Yoneda, F. Teteraheron 1988, 44, 3241.
- Sample availability: available from the authors and MDPI.
© 2000 MDPI. All rights reserved. Molecules website www.mdpi.org/molecules/
Share and Cite
Asiri, A.M. 5-(Indol-3-yl)barbituric acid. Molecules 2000, 5, M183. https://doi.org/10.3390/M183
Asiri AM. 5-(Indol-3-yl)barbituric acid. Molecules. 2000; 5(12):M183. https://doi.org/10.3390/M183
Chicago/Turabian StyleAsiri, Abdullah Mohamed. 2000. "5-(Indol-3-yl)barbituric acid" Molecules 5, no. 12: M183. https://doi.org/10.3390/M183
APA StyleAsiri, A. M. (2000). 5-(Indol-3-yl)barbituric acid. Molecules, 5(12), M183. https://doi.org/10.3390/M183



