Synthetic Strategies and Biological Diversity of Biflavonoids: Current Status and Perspective
Abstract
1. Introduction
2. Synthetic Methods of Biflavonoids
2.1. Chemical Synthesis
2.1.1. Direct Coupling Reactions
2.1.2. Precursor Cyclization Reaction
2.2. Emerging Synthetic Methods
2.2.1. Oxidative Coupling
2.2.2. Electrocatalytic Synthesis
2.3. Biosynthetic Strategies
3. Biological Activity of Biflavonoids
3.1. Anti-Tumor Activity
3.2. Anti-Alzheimer’s Disease
3.3. Anti-Inflammatory Activity
3.4. Antimicrobial Activity
4. Challenges and Perspectives
4.1. Chemical Synthesis
4.2. Biological Activities
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Baker, W.; Simmonds, W.H.C. 258. Derivatives of 5 : 6 : 4′- and 5 : 8 : 4′-trihydroxyflavones, and a note on the structure of ginkgetin. J. Chem. Soc. 1940, 1370–1374. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Li, Q.Y.; Yan, L.L.; Shi, Y. Structural characterization and identification of biflavones in Selaginella tamariscina by liquid chromatography-diode-array detection/electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 2173–2186. [Google Scholar] [CrossRef]
- Lee, T.H.; Ham, S.L.; Lee, D.Y.; Lee, J.R.; Kim, J.; Kim, C.S. Structure Revision of Balsamisides A-D and Establishment of an Empirical Rule for Distinguishing Four Classes of Biflavonoids. J. Nat. Prod. 2022, 85, 2461–2467. [Google Scholar] [CrossRef]
- Sundaram, R.; Muthu, K.; Shanthi, P.; Sachdanandam, P. Antioxidant and antihyperlipidemic activities of catechol derivatives and biflavonoid isolated from Semecarpus anacardium seeds. Toxicol. Mech. Methods 2022, 32, 123–131. [Google Scholar] [CrossRef]
- Liu, Y.; Ye, J.; Fan, Z.; Wu, X.; Zhang, Y.; Yang, R.; Jiang, B.; Wang, Y.; Wu, M.; Zhou, J.; et al. Ginkgetin Alleviates Inflammation and Senescence by Targeting STING. Adv. Sci. 2025, 12, e2407222. [Google Scholar] [CrossRef]
- Dai, X.H.; Zhu, J.M.; Wang, G.Y.; Ren, Y.H.; Liu, H.L.; Wang, Y. Gymnosperm-specific CYP90Js enable biflavonoid biosynthesis and microbial production of amentoflavone. Nat. Commun. 2025, 16, 7792. [Google Scholar] [CrossRef]
- Huang, W.; Xu, S.; Lin, R.; Xiong, X.; Song, J.; Liu, Y.; Li, J. Enzymatic Synthesis of Biflavonoid Glycosides with Enhanced Antitumor Activity Using Glycosyltransferase and Sucrose Synthase. J. Agric. Food Chem. 2025, 73, 4807–4819. [Google Scholar] [CrossRef]
- Sun, Y.L.; Guo, Y.; Wang, X.Y.; Zhang, Y.S.; Jin, X.Q.; Zheng, J.; Chen, Y.; Cheng, X.; Guo, Y.H.; Li, X.M.; et al. Elimination of gut microbiota hinders the therapeutic effect of amentoflavone on respiratory syncytial virus-induced lung inflammation injury by regulating innate immunity. Phytomedicine 2025, 145, 157033. [Google Scholar] [CrossRef]
- Nakazawa, K.; Ito, M. Synthesis of ginkgetin. Tetrahedron Lett. 1962, 3, 317–319. [Google Scholar] [CrossRef]
- Sagrera, G.; Bertucci, A.; Vazquez, A.; Seoane, G. Synthesis and antifungal activities of natural and synthetic biflavonoids. Bioorganic Med. Chem. 2011, 19, 3060–3073. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.C.; Ueng, T.; Chen, C.Y.; Chang, P.W. Synthesis of Biflavonyl Ethers. J. Chin. Chem. Soc. 1970, 17, 251–254. [Google Scholar] [CrossRef]
- Che, H.; Park, B.K.; Lim, H.; Kim, H.P.; Chang, H.W.; Jeong, J.H.; Park, H. Synthesis of biflavones having a 6-O-7″ linkage and effects on cyclooxygenase-2 and inducible nitric oxide synthase. Bioorganic Med. Chem. Lett. 2009, 19, 74–76. [Google Scholar] [CrossRef]
- Muller, D.; Fleury, J.P. A new strategy for the synthesis of biflavonoids via arylboronic acids. Tetrahedron Lett. 1991, 32, 2229–2232. [Google Scholar] [CrossRef]
- Ramos, I.T.L.; Silva, R.J.M.; Silva, T.M.S.; Camara, C.A. Palladium-catalyzed coupling reactions in flavonoids: A retrospective of recent synthetic approaches. Synth. Commun. 2021, 51, 3520–3545. [Google Scholar] [CrossRef]
- Zembower, D.; Zhang, H. Total Synthesis of Robustaflavone, a Potential Anti-Hepatitis B Agent. J. Org. Chem. 1998, 63, 9300–9305. [Google Scholar] [CrossRef]
- Park, H.I. Total Synthesis of Amentoflavone. Med. Chem. 2015, 5, 467–469. [Google Scholar] [CrossRef]
- Lim, H.; Kim, S.B.; Park, H.; Chang, H.W.; Kim, H.P. New anti-inflammatory synthetic biflavonoid with C-C (6-6″) linkage: Differential effects on cyclooxygenase-2 and inducible nitric oxide synthase. Arch. Pharmacal Res. 2009, 32, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Nakano, H.; Hoshino, Y.; Matsuyama, H.; Kohari, Y. Efficient synthesis of biflavones having a ring-A ring of two flavone units using Suzuki cross-coupling reactions. Heterocycles 2010, 81, 1871–1879. [Google Scholar] [CrossRef] [PubMed]
- Sum, T.J.; Sum, T.H.; Galloway, W.R.J.D.; Twigg, D.G.; Ciardiello, J.J.; Spring, D.R. Synthesis of structurally diverse biflavonoids. Tetrahedron 2018, 74, 5089–5101. [Google Scholar] [CrossRef]
- Chen, J.; Chang, H.W.; Kim, H.P.; Park, H. Synthesis of phospholipase A2 inhibitory biflavonoids. Bioorganic Med. Chem. Lett. 2006, 16, 2373–2375. [Google Scholar] [CrossRef]
- Moon, T.C.; Quan, Z.; Kim, J.; Kim, H.P.; Kudo, I.; Murakami, M.; Park, H.; Chang, H.W. Inhibitory effect of synthetic C–C biflavones on various phospholipase A2s activity. Bioorganic Med. Chem. 2007, 15, 7138–7143. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Ribeiro, D.; Silva, V.L.M.; Fernandes, E. Synthetic flavonoid dimers: Synthesis strategies and biological activities. Eur. J. Med. Chem. 2025, 291, 117669. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Yang, K.; Jia, X.; Gao, X.; Yu, P. Total Synthesis of I3,II8-Biapigenin and Ridiculuflavone A. Org. Chem. Front. 2017, 4, 578–586. [Google Scholar] [CrossRef]
- Lu, K.; Li, M.; Huang, Y.; Sun, Y.; Gong, Z.; Wei, Q.; Zhao, X.; Zhang, Y.; Yu, P. Total synthesis of wikstrol A and wikstrol B. Org. Biomol. Chem. 2019, 17, 8206–8213. [Google Scholar] [CrossRef]
- Pathe, G.K.; Konduru, N.K.; Parveen, I.; Ahmed, N. Anti-proliferative activities of flavone–estradiol Stille-coupling adducts and of indanone-based compounds obtained by SnCl4/Zn-catalysed McMurry cross-coupling reactions. RSC Adv. 2015, 5, 83512–83521. [Google Scholar] [CrossRef]
- Fregoso-López, D.; Miranda, L.D. Visible-Light Mediated Radical Alkylation of Flavones: A Modular Access to Nonsymmetrical 3,3″-Biflavones. Org. Lett. 2022, 24, 8615–8620. [Google Scholar] [CrossRef]
- Cardullo, N.; Muccilli, V.; Tringali, C. Laccase-mediated synthesis of bioactive natural products and their analogues. RSC Chem. Biol. 2022, 3, 614–647. [Google Scholar] [CrossRef]
- Molyneux, R.J.; Waiss, A.C.; Haddon, W.F. Oxidative coupling of apigenin. Tetrahedron 1970, 26, 1409–1416. [Google Scholar] [CrossRef]
- Nanjan, P.; Nambiar, J.; Nair, B.G.; Banerji, A. Synthesis and discovery of (I-3,II-3)-biacacetin as a novel non-zinc binding inhibitor of MMP-2 and MMP-9. Bioorganic Med. Chem. 2015, 23, 3781–3787. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Lim, S.H.M.; Lin, J.; Wu, J.; Tang, H.; Zhao, F.; Liu, F.; Sun, C.; Shi, X.; Kuang, Y.; et al. Oxygen mediated oxidative couplings of flavones in alkaline water. Nat. Commun. 2022, 13, 6424. [Google Scholar] [CrossRef]
- Nadirov, R.K.; Nadirov, K.S.; Esimova, A.M.; Nadirova, Z.K. Electrochemical Synthesis of Amino Derivatives of Biflavonoids. Chem. Nat. Compd. 2014, 50, 735–736. [Google Scholar] [CrossRef]
- Jung, Y.J.; Lee, E.H.; Lee, C.G.; Rhee, K.J.; Jung, W.S.; Choi, Y.; Pan, C.H.; Kang, K. AKR1B10-inhibitory Selaginella tamariscina extract and amentoflavone decrease the growth of A549 human lung cancer cells in vitro and in vivo. J. Ethnopharmacol. 2017, 202, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Zhen, P.; Li, D.; Liu, X.; Ding, X.; Liu, H. Amentoflavone promotes ferroptosis by regulating reactive oxygen species (ROS)/5′AMP-activated protein kinase (AMPK)/mammalian target of rapamycin (mTOR) to inhibit the malignant progression of endometrial carcinoma cells. Bioengineered 2022, 13, 13269–13279. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Lin, Z.; Shi, P.; Chen, B.; Wang, G.; Huang, J.; Sui, Y.; Liu, Q.; Li, S.; Lin, X. Delicaflavone induces ROS-mediated apoptosis and inhibits PI3K/AKT/mTOR and Ras/MEK/Erk signaling pathways in colorectal cancer cells. Biochem. Pharmacol. 2020, 171, 113680. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.; Matlin, A.J.; Lowell, A.M.; Moore, M.J. The Biflavonoid Isoginkgetin Is a General Inhibitor of Pre-mRNA Splicing. J. Biol. Chem. 2008, 283, 33147–33154. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, S.; Wang, J.; Zhang, P.; Lu, T.; Zhang, L. Hinokiflavone Inhibits Growth of Esophageal Squamous Cancer By Inducing Apoptosis via Regulation of the PI3K/AKT/mTOR Signaling Pathway. Front. Oncol. 2022, 12, 833719. [Google Scholar] [CrossRef]
- Windsor, P.K.; Plassmeyer, S.P.; Mattock, D.S.; Bradfield, J.C.; Han, B.H. Biflavonoid-Induced Disruption of Hydrogen Bonds Leads to Amyloid-b Disaggregation. Int. J. Mol. Sci. 2021, 22, 2888. [Google Scholar] [CrossRef]
- Sirimangkalakitti, N.; Juliawaty, L.D.; Hakim, E.H.; Waliana, I.; Kinoshita, K. Naturally occurring biflavonoids with amyloid β aggregation inhibitory activity for development of anti-Alzheimer agents. Bioorganic Med. Chem. Lett. 2019, 29, 1994–1997. [Google Scholar] [CrossRef]
- Gutmann, H.; Bruggisser, R.; Schaffner, W.; Bogman, K.; Botomino, A.; Drewe, J. Transport of amentoflavone across the blood-brain barrier in vitro. Planta Medica 2002, 68, 804–807. [Google Scholar] [CrossRef]
- Ella, F.A.; Tchamgoue, J.; Ambamba, B.D.A.; Mountessou, B.Y.G.; Essouman, F.M.; Essola, N.N.; Wilhelm, A.; Ngondi, J.L.; Njayou, F.N.; Kouam, S.F. Anti-Alzheimer potential of biflavonoids from Allanblackia floribunda: Multi-target inhibition of monoamine oxidase, β-secretase, and glycogen synthase kinase-3β. Fitoterapia 2026, 188, 106967. [Google Scholar] [CrossRef]
- Ayoub, I.M.; Korinek, M.; Hwang, T.L.; Chen, B.H.; Chang, F.R.; El-Shazly, M.; Singab, A.N.B. Probing the Antiallergic and Anti-inflammatory Activity of Biflavonoids and Dihydroflavonols from Dietes bicolor. J. Nat. Prod. 2018, 81, 243–253. [Google Scholar] [CrossRef]
- Shim, S.Y.; Lee, S.G.; Lee, M. Biflavonoids Isolated from Selaginella tamariscina and Their Anti-Inflammatory Activities via ERK 1/2 Signaling. Molecules 2018, 23, 926. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Zhang, Y.; Shan, G.Y.; Cheng, J.Y.; Wan, H.; Zhang, Y.X.; Li, H.J. Hinokiflavone exerts dual regulation on apoptosis and pyroptosis via the SIX4/Stat3/Akt pathway to alleviate APAP-induced liver injury. Life Sci. 2024, 354, 122968. [Google Scholar] [CrossRef] [PubMed]
- Gil, B.; Sanz, M.J.; Terencio, M.C.; Gunasegaran, R.; Payá, M.; Alcaraz, M.J. Morelloflavone, a novel biflavonoid inhibitor of human secretory phospholipase A2 with anti-inflammatory activity. Biochem. Pharmacol. 1997, 53, 733–740. [Google Scholar] [CrossRef]
- Park, H.; Kim, Y.H.; Chang, H.W.; Kim, H.P. Anti-inflammatory activity of the synthetic C-C biflavonoids. J. Pharm. Pharmacol. 2006, 58, 1661–1667. [Google Scholar] [CrossRef] [PubMed]
- Owona, B.A.; Mary, A.; Messi, A.N.; Ravichandran, K.A.; Mbing, J.N.; Pegnyemb, E.; Moundipa, P.F.; Heneka, M.T. Biflavonoid Methylchamaejasmin and Khaya grandifoliola Extract Inhibit NLRP3 Inflammasome in THP-1 Cell Model of Neuroinflammation. Mol. Neurobiol. 2025, 62, 1605–1619. [Google Scholar] [CrossRef]
- de Almeida, M.M.A.; Souza, C.D.S.; Dourado, N.S.; da Silva, A.B.; Ferreira, R.S.; David, J.M.; David, J.P.; Costa, M.F.D.; da Silva, V.D.A.; Butt, A.M.; et al. Phytoestrogen Agathisflavone Ameliorates Neuroinflammation-Induced by LPS and IL-1β and Protects Neurons in Cocultures of Glia/Neurons. Biomolecules 2020, 10, 562. [Google Scholar] [CrossRef]
- Bai, Y.; Wang, W.; Shi, M.; Wei, X.; Zhou, X.; Li, B.; Zhang, J. Novel Antibiofilm Inhibitor Ginkgetin as an Antibacterial Synergist against Escherichia coli. Int. J. Mol. Sci. 2022, 23, 8809. [Google Scholar] [CrossRef]
- Linden, M.; Brinckmann, C.; Feuereisen, M.M.; Review; Schieber, A. Effects of structural differences on the antibacterial activity of biflavonoids from fruits of the Brazilian peppertree (Schinus terebinthifolius Raddi). Food Res. Int. 2020, 133, 109134. [Google Scholar] [CrossRef]
- Cane, H.P.C.A.; Saidi, N.; Yahya, M.; Darusman, D.; Musman, M. Macrophylloflavone: A New Biflavonoid from Garcinia macrophylla Mart. (Clusiaceae) for Antibacterial, Antioxidant, and Anti-Type 2 Diabetes Mellitus Activities. Sci. World J. 2020, 2020, 2983129. [Google Scholar] [CrossRef]
- Miki, K.; Nagai, T.; Suzuki, K.; Tsujimura, R.; Koyama, K.; Kinoshita, K.; Furuhata, K.; Yamada, H.; Takahashi, K. Anti-influenza virus activity of biflavonoids. Bioorganic Med. Chem. Lett. 2007, 17, 772–775. [Google Scholar] [CrossRef]
- Lee, J.; Choi, Y.; Woo, E.R.; Lee, D.G. Isocryptomerin, a novel membrane-active antifungal compound from Selaginella tamariscina. Biochem. Biophys. Res. Commun. 2009, 379, 676–680. [Google Scholar] [CrossRef]
- Menezes, J.; Campos, V.R. Natural biflavonoids as potential therapeutic agents against microbial diseases. Sci. Total Environ. 2021, 769, 145168. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Yang, F.; Huang, X. Proceedings of Chemistry, Pharmacology, Pharmacokinetics and Synthesis of Biflavonoids. Molecules 2021, 26, 6088. [Google Scholar] [CrossRef]
- Khodzhaieva, R.S.; Gladkov, E.S.; Kyrychenko, A.; Roshal, A.D. Progress and Achievements in Glycosylation of Flavonoids. Front. Chem. 2021, 9, 637994. [Google Scholar] [CrossRef]
- Lu, S.M.; Chen, C.; Liu, C.; Liu, R.; Chen, J.H.; Zhang, Z.; Yang, Z. Regioselective Hydroxylation of Flavonoids by Transition-Metal-Catalyzed C-H Bond Oxidation. Org. Lett. 2023, 25, 2264–2269. [Google Scholar] [CrossRef]
- Lin, D.; Jiang, S.; Zhang, A.; Wu, T.; Qian, Y.; Shao, Q. Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis. Nat. Prod. Bioprospecting 2022, 12, 8. [Google Scholar] [CrossRef]
- Rahman, M.; Riaz, M.; Desai, U.R. Synthesis of biologically relevant biflavanoids—A review. Chem. Biodivers. 2007, 4, 2495–2527. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Patel, N.D.; Xu, G.; Xu, X.; Savoie, J.; Ma, S.; Hao, M.H.; Keshipeddy, S.; Capacci, A.G.; Wei, X.; et al. Efficient chiral monophosphorus ligands for asymmetric Suzuki-Miyaura coupling reactions. Org. Lett. 2012, 14, 2258–2261. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Capacci, A.G.; Wei, X.; Li, W.; White, A.; Patel, N.D.; Savoie, J.; Gao, J.J.; Rodriguez, S.; Qu, B.; et al. A general and special catalyst for Suzuki-Miyaura coupling processes. Angew. Chem. Int. Ed. 2010, 49, 5879–5883. [Google Scholar] [CrossRef]
- Vavříková, E.; Vacek, J.; Valentová, K.; Marhol, P.; Ulrichová, J.; Kuzma, M.; Křen, V. Chemo-enzymatic synthesis of silybin and 2,3-dehydrosilybin dimers. Molecules 2014, 19, 4115–4134. [Google Scholar] [CrossRef] [PubMed]

















| Name (Source) | Activity | Mechanism/Pathway | Type [Ref] |
|---|---|---|---|
 Amentoflavone Amentoflavone(Selaginella) | AKR1B10 inhibitor (IC50 = 1.54 μM) | Blocking doxorubicin metabolism reverses resistance | Lung cancer [32] |
| Treatment concentration: 50–100 μM | 1. ROS/AMPK/mTOR pathway 2. Downregulating SLC7A11 GPX4 | Endometrial carcinoma [33] | |
 Delicaflavone Delicaflavone(Selaginella) | Colorectal Cancer Cells, MTT Assay HT29 (IC50 = 18.8 μM) | 1. In Vitro: PI3K/AKT/mTOR pathway, apoptosis 2. Ras/MEK/Erk signaling pathway 3. In Vivo: upregulating Caspase-3/9 and downregulating CD34/Ki67 | Colorectal cancer [34] |
 Isoginkgetin Isoginkgetin(Ginkgo biloba) | pre-mRNA splicing (IC50 = 30 μM) | Inhibiting pre-mRNA splicing | Anti-tumor activity [35] |
 Hinokiflavone Hinokiflavone(Platycladus orientalis) | 50 mg/kg (in vivo) | 1. SIX4/Akt/Stat3 pathway 2. PI3K/AKT/mTOR and 3. Downregulating MMP2/9 | Esophageal squamous cell carcinoma [36] |
 Amentoflavone-7-O-β-D-glucoside Amentoflavone-7-O-β-D-glucoside | Human prostate cancer PC-3 cells (IC50 = 57.7μM) Amentoflavone(IC50 = 102.2 μM) | Downregulating PDE6A, PDE10A | Human prostate cancer [7] |
| Name (Source) | Activity | Mechanism/Pathway | Ref |
|---|---|---|---|
 Amentoflavone (Selaginella) Amentoflavone (Selaginella) | Depolymerize Aβ42 fibrils (EC50 = 0.70 ± 0.16 μM) Inhibit Aβ40 aggegation (IC50 = 4.8 ± 0.1 μM) | 1. Binding to the Aβ42 N-terminal pocket (4–14) via π-π stacking 2. Hydroxyl groups form hydrogen bonds with the backbone of Aβ peptides 3. Disrupting the β-sheet structure that maintains fibril stability | [37,38,39] |
 Sequiaflavone Sequiaflavone (Selaginella) | Depolymerize Aβ42 fibrils (EC50 = 1.89 ± 0.74 μM) Inhibit Aβ40 aggregation (IC50 = 4.9 ± 0.1 μM) | ||
 Bilobetin (Selaginella) Bilobetin (Selaginella) | Depolymerize Aβ42 fibrils (EC50 = 2.75 ± 1.72 μM) Inhibit Aβ40 aggegation (IC50 = 4.7 ± 0.7 μM) | ||
 podocarpusflavone A podocarpusflavone A(Podocarpus neriifolius) | Inhibit Aβ40 aggegation (IC50 = 4.9 ± 0.2 μM) | ||
 Sotetsuflavone (Cycas revoluta) Sotetsuflavone (Cycas revoluta) | Inhibit Aβ40 aggregation (IC50 > 50 μM) | ||
 (2S,3S)-volkensiflavone-7-O-β-glucopyranoside (Allanblackia floribunda) (2S,3S)-volkensiflavone-7-O-β-glucopyranoside (Allanblackia floribunda) | Inhibit MAO-A (IC50 = 35.85 ± 0.03 μM) Inhibit BACE-1 (IC50 = 2.48 ± 0.11 μM) Inhibit GSK-3β (IC50 = 9.39 ± 0.06 μM) | 1. Neurotransmitter metabolic imbalance pathway (mediated by MAO-A) 2. β-amyloid (Aβ) deposition pathway (mediated by BACE-1) 3. Tau hyperphosphorylation pathway (mediated by GSK-3β) | [40] |
 (2R,3S)-volkensiflavone-7-O-β-D-acetylglucopyranoside (Allanblackia floribunda) (2R,3S)-volkensiflavone-7-O-β-D-acetylglucopyranoside (Allanblackia floribunda) | Inhibit MAO-A (IC50 = 25.54 ± 0.05 μM) Inhibit BACE-1 (IC50 = 2.50 ± 0.17 μM) Inhibit GSK-3β (IC50 = 7.17 ± 0.09 μM) |
| Name (Source) | Activity | Mechanism/Pathway | Refs |
|---|---|---|---|
 Robustaflavone Robustaflavone(Selaginella) | Inhibit superoxide anion (IC50 = 1.01 μM) Inhibit elastase (IC50 = 0.45 μM) | 1. Suppress superoxide anion generation 2. Inhibit elastase release | [41] |
 Amentoflavone Amentoflavone (Selaginella) | Inhibit superoxide anion (IC50 = 1.01 μM) Inhibit elastase (IC50 = 0.75 μM) | 1. Inhibit superoxide 2. Inhibit elastase | [41] |
 Hinokiflavone Hinokiflavone (Platycladus orientalis) | Range from 1 to 10 μM | 1. ERK 1/2/NF-κB signaling pathway 2. Downregulate the iNOS and COX-2 3. regulate the SIX4/Akt/Stat3 pathway | [42,43] |
 Morelloflavone (Garcinia dulcis) Morelloflavone (Garcinia dulcis) | II and III sPLA2 inhibitor (IC50 = 0.9 μM) | sPLA2 inhibition and ROS scavenging | [44] |
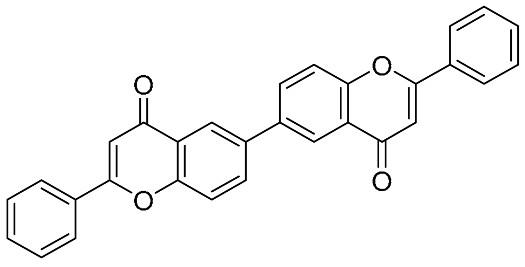 6,6″- linked biflavonoid 6,6″- linked biflavonoid(synthetic biflavonoids) | COX-2 inhibitor (IC50 = 3.7 μM) | Directly inhibit COX-2 and synergistically inhibit sPLA2-IIA | [45] |
 7,7″-di-O-methylchamaejasmin 7,7″-di-O-methylchamaejasmin(Khaya grandifoliola) | Concentrations of 10, 20 μM and 40 μM | 1. Block the formation of new ASC specks 2. Downregulate the gene expression of NFκB, NLRP3, and Caspase-1 3. Inhibit NO production and the secretion of IL-1β and IL-18 | [46] |
 Agathisflavone Agathisflavone(Agathis palmerstonii) | Concentrations of 0.1 μM and 1 μM | 1. NF-κB signaling pathway 2. Downregulate TNF-α, IL-1β, CCL2, CCL5, NOS2 3. Upregulate IL-10 4. Reduce NO 5. Block M1 polarization of microglia 6. Inhibit microglial proliferation (reduce Iba-1+/BrdU+ cells | [47] |
| Name (Source) | Activity | Mechanism/Pathway | Type [Refs] |
|---|---|---|---|
 Ginkgetin (Ginkgo biloba) Ginkgetin (Ginkgo biloba) | Minimum biofilm inhibitory concentration: 6.25 µM (inhibition rate > 20%) | 1. Reduce the production of AI-2 2. Downregulate the transcription of key genes (csgA, csgD, flhC, flhD, fliC, luxS, lsrB, lsrK, lsrR) 3. Inhibit EPS production | Antibacterial activity [48] |
 Tetrahydroamentoflavone Tetrahydroamentoflavone(Semecarpus prainii) | 1. MIC = 0.063 mg/mL 2. MBC = 0.063–0.125 mg/mL | Inhibit Gram-positive bacteria such as Bacillus subtilis and staphylococcus carnosus | Antibacterial activity [49] |
 Macrophylloflavone Macrophylloflavone(Garcinia macrophylla) | 1. The inhibition zone for Escherichia coli ranges from 16.65 ± 0.43 mm to 20.29 ± 0.28 mm 2. For staphylococcus aureus, it ranges from 15.54 ± 0.39 mm to 23.16 ± 0.32 mm | Antibacterial activity [50] | |
 Ginkgetin derivatives Ginkgetin derivatives(Ginkgo biloba) | 1. H1N1 inhibitor (IC50 = 5.50 μg/mL) 2. H3N2 inhibitor (IC50 = 0.82 μg/mL) | 1. Target influenza virus sialidase to block viral release and spread 2. Conjugation with sialic acid enables dual-site synergistic inhibition and reduces cytotoxicity | Antiviral activity [51] |
 Amentoflavone Amentoflavone(Selaginella) | 1. RSV inhibitor (IC50 = 5.50 μg/mL) 2. 120 mg/kg/d | Inhibit RSV replication | Antiviral activity [52] |
 Isocryptomerin Isocryptomerin(Selaginella tamariscina) | MIC = 18.11 µM against Candida albicans, Geotrichumcandidum, Saccharomyces cerevisiae | Disruption of fungal cell membranes | Antifungal activity [53] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhang, Y.; Chai, Y.; Wu, J.; Wang, S.; Shi, J.; Wei, M.; Liu, R.; Zhuang, C. Synthetic Strategies and Biological Diversity of Biflavonoids: Current Status and Perspective. Molecules 2026, 31, 925. https://doi.org/10.3390/molecules31060925
Zhang Y, Chai Y, Wu J, Wang S, Shi J, Wei M, Liu R, Zhuang C. Synthetic Strategies and Biological Diversity of Biflavonoids: Current Status and Perspective. Molecules. 2026; 31(6):925. https://doi.org/10.3390/molecules31060925
Chicago/Turabian StyleZhang, Yu, Yue Chai, Jiabin Wu, Shuyu Wang, Jinxin Shi, Minzhen Wei, Runhui Liu, and Chunlin Zhuang. 2026. "Synthetic Strategies and Biological Diversity of Biflavonoids: Current Status and Perspective" Molecules 31, no. 6: 925. https://doi.org/10.3390/molecules31060925
APA StyleZhang, Y., Chai, Y., Wu, J., Wang, S., Shi, J., Wei, M., Liu, R., & Zhuang, C. (2026). Synthetic Strategies and Biological Diversity of Biflavonoids: Current Status and Perspective. Molecules, 31(6), 925. https://doi.org/10.3390/molecules31060925






