The Molecular Network of Neutrophil Extracellular Traps in Hepatocellular Carcinoma: Biogenesis, Function, and Therapeutic Implications
Abstract
1. Introduction
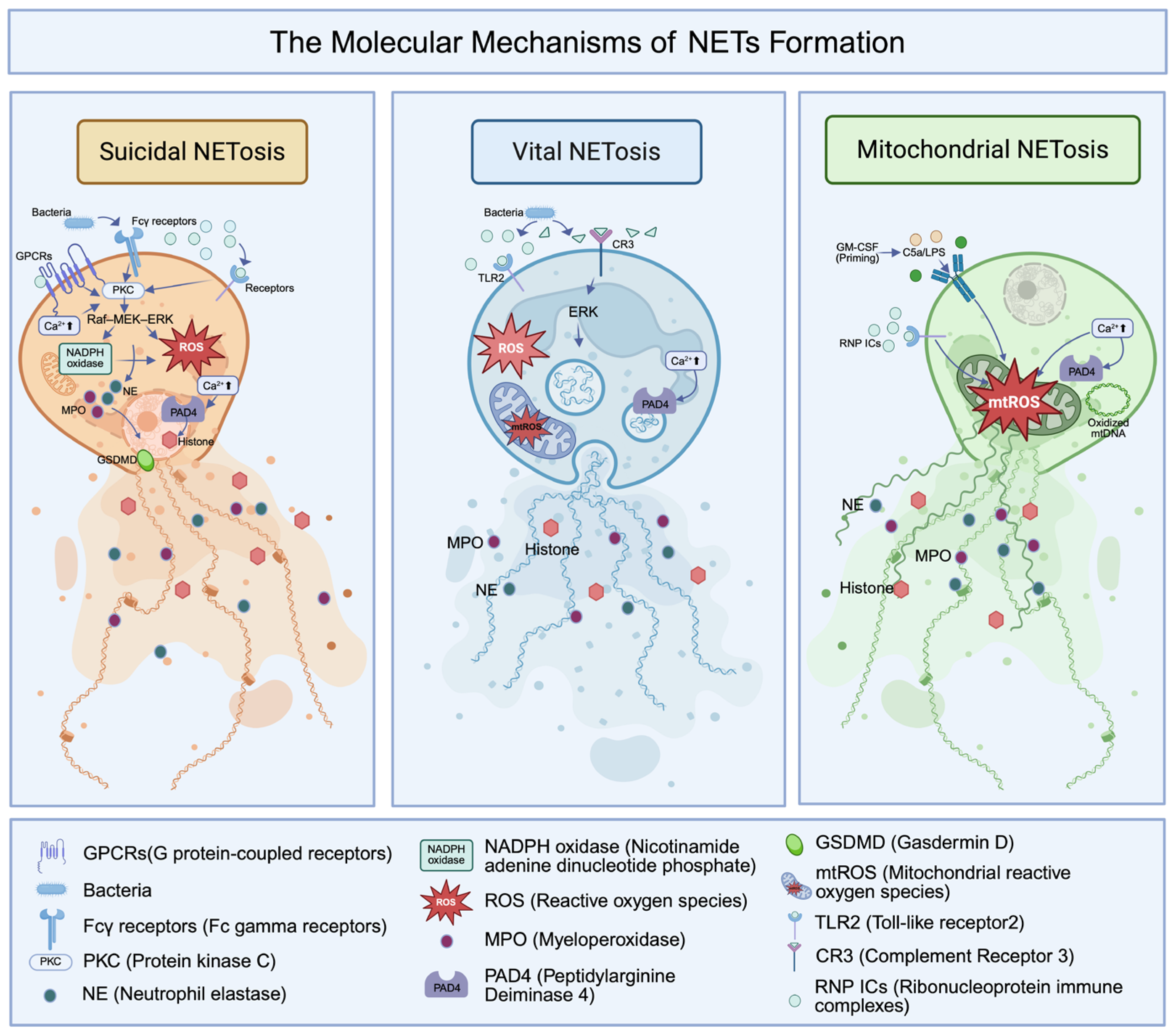
2. The Molecular Mechanisms of NETs Formation
2.1. Suicidal NETosis: The NADPH Oxidase-Dependent Pathway
2.2. Vital NETosis: Rapid Release Without Cell Death
2.3. Mitochondrial NETosis: ROS-Dependent Extrusion of mtDNA
3. The Origins of NETs in the HCC Microenvironment
3.1. HCC Cells Trigger NETosis
3.2. Stromal Cells in the HCC Microenvironment Trigger NETosis
3.3. Extracellular Matrix (ECM) in the HCC Microenvironment Trigger NETosis
3.4. Immunological Crosstalk: Immune Cells and NETs in HCC
4. The Roles of NETs in HCC Progression
4.1. NETs Drive the Development of HCC
4.2. NETs Drive the Process of HCC Metastasis
4.3. NETs Drive Immune Escape and Postoperative Recurrence of HCC
4.4. NETs Drive the Occurrence of HCC Related Systemic Complications
5. Clinical Translation: NETs as Biomarkers and Therapeutic Targets
5.1. Diagnostic and Prognostic Utility: From Liquid Biopsies to Multi-Omics Signatures
| Main Detection Category | Purpose of Detection | Detected Component/Method | References |
|---|---|---|---|
| Serum/Plasma Circulating NETs Markers | Prognostic Assessment and Risk Stratification | Preoperative serum MPO–DNA levels correlate with shorter RFS and OS in HCC patients. | [105] |
| Serum/Plasma Circulating NETs Markers | Prediction of Extrahepatic Metastasis | In HBV-related HCC, circulating NETs levels significantly correlate with and can predict extrahepatic metastasis. | [15] |
| Tissue NETs Markers | Prognostic Assessment and Risk Stratification | Expression of CitH3 in tumor tissue serves as an independent prognostic factor for postoperative recurrence and OS. | [105,124] |
| Specific Protein Expression Levels | Key Regulatory Molecules as Markers | Significantly decreased serum levels of the secreted protein PRSS35 in HCC patients are associated with poor prognosis. | [115] |
| Specific Protein Expression Levels | Key Regulatory Molecules as Markers | High expression of the methyltransferase METTL5 in tumor tissue correlates with adverse prognosis and TNM stage in HCC. | [117] |
| NET-Related Gene Expression Signatures | Prognostic Assessment and Risk Stratification | Risk scoring models based on transcriptomic data of NET-related genes (NRGs) effectively predict OS in HCC patients and serve as independent prognostic factors. | [119,120,122] |
| NET-Related Gene Expression Signatures | Prognostic Assessment and Risk Stratification | Prognostic models integrating NETs and m6A methylation-related long non-coding RNAs accurately stratify HCC patients into different risk groups. | [123] |
| NET-Related Gene Expression Signatures | Prognostic Assessment and Risk Stratification | Prognostic signatures based on NETosis-correlated long non-coding RNAs can distinguish different risk subgroups in HCC. | [121] |
| Radiomics Biomarkers | Prediction of Immunotherapy Response | CT image-based radiomics features of NETs can predict the response of HCC patients to PD-1 inhibitor immunotherapy (objective response rate). | [23] |
5.2. Immune-Targeted Therapy: Strategies Against NETs
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HCC | Hepatocellular carcinoma |
| TME | Tumor microenvironment |
| NETs | Neutrophil extracellular traps |
| ICI | Immune checkpoint inhibitor |
| TAN | Tumor-associated neutrophil |
| NE | Neutrophil elastase |
| MPO | Myeloperoxidase |
| TMCO6 | Transmembrane and coiled-coil domains 6 |
| NASH | Non-alcoholic steatohepatitis |
| Tregs | Regulatory T-cells |
| Ox-mtDNA | Oxidized mitochondrial DNA |
| CitH3 | Citrullinated histone H3 |
| mtDNA | Mitochondrial DNA |
| GPCR | G protein-coupled receptor |
| PMA | Phorbol 12-myristate 13-acetate |
| PKC | Protein kinase C |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NOX | NADPH oxidase complex |
| PAD4 | Peptidylarginine Deiminase 4 |
| ROS | Reactive oxygen species |
| GSDMD | Gasdermin D |
| TLR | Toll-like receptor |
| CR3 | Complement Receptor 3 |
| ECM | Extracellular matrix |
| PMN | Polymorphonuclear cell |
| C5a | Complement factor 5a |
| LPS | Lipopolysaccharide |
| GM-CSF | Granulocyte/macrophage colony-stimulating factor |
| mtROS | Mitochondrial reactive oxygen species |
| RNP ICs | Ribonucleoprotein immune complexes |
| CGD | Chronic granulomatous disease |
| SK3 | Small conductance potassium channel 3 |
| cGAS | Cyclic GMP-AMP synthase |
| IL-8/CXCL8 | Interleukin-8 |
| CXCR | C-X-C chemokine receptor |
| CXCL | Chemokine (C-X-C motif) ligand |
| ACOT12 | Acetyl-CoA thioesterase 12 |
| ACSS1/2 | Acyl-CoA synthetase short-chain family member 1/2 |
| HBV | Hepatitis B Virus |
| DAMP | Damage-associated molecular pattern |
| COX2 | Cyclooxygenase 2 |
| HSC | Hepatic stellate cell |
| CAF | Tumor-associated fibroblast |
| TEC | Tumor endothelial cell |
| MSC | Mesenchymal stem cell |
| EC | Endothelial cell |
| TF | Tissue factor |
| G-MDSC | Granulocyte-myeloid derived suppressor cell |
| Col1 | Type I collagen |
| DDR1 | Discoidin domain receptor 1 |
| NLR | Neutrophil-lymphocyte ratio |
| TAM | Tumor-associated macrophage |
| EV | Extracellular vesicle |
| FGL2 | Fibrinogen-like 2 |
| A2AR | Adenosine receptor 2a |
| DEN | Diethylnitrosamine |
| HFD | High-fat diet |
| NASH | Nonalcoholic steatohepatitis |
| MASH | Metabolic dysfunction–associated steatohepatitis |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| ALD | Alcoholic liver disease |
| EMT | Epithelial–mesenchymal transition |
| PAMP | Pathogen-associated molecular pattern |
| PGE2 | Prostaglandin E2 |
| CTC | Circulating tumor cell |
| TIDE | Tumor Immune Dysfunction and Exclusion |
| I/R | Ischemia–reperfusion |
| RFS | Recurrence-free survival |
| OS | Overall survival |
| PVT | Portal vein thrombosis |
| cfDNA | Cell-free DNA |
| HMWK | High-molecular-weight kininogen |
| MELD | Model for end-stage liver disease |
| LDH | Lactate dehydrogenase |
| ccf-mtDNA | Circulating free mitochondrial DNA |
| NRG | NET-related gene |
| R-NETs | NET-related radiomic signature |
| FMT | Fecal microbiota transplantation |
| PPARα | Peroxisome proliferator-activated receptor alpha |
| DNase I | Deoxyribonuclease I |
| GSK3a | Glycogen synthase kinase 3 alpha |
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Rumgay, H.; Ferlay, J.; de Martel, C.; Georges, D.; Ibrahim, A.S.; Zheng, R.; Wei, W.; Lemmens, V.; Soerjomataram, I. Global, regional and national burden of primary liver cancer by subtype. Eur. J. Cancer 2022, 161, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Vogel, A.; Meyer, T.; Sapisochin, G.; Salem, R.; Saborowski, A. Hepatocellular carcinoma. Lancet 2022, 400, 1345–1362. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Yang, X.R.; Chung, W.Y.; Dennison, A.R.; Zhou, J. Targeted therapy for hepatocellular carcinoma. Signal Transduct. Target. Ther. 2020, 5, 146. [Google Scholar] [CrossRef]
- Ye, X.; Fang, X.; Li, F.; Jin, D. Targeting TIME in advanced hepatocellular carcinoma: Mechanisms of drug resistance and treatment strategies. Crit. Rev. Oncol. Hematol. 2025, 211, 104735. [Google Scholar] [CrossRef]
- Bloom, M.; Podder, S.; Dang, H.; Lin, D. Advances in Immunotherapy in Hepatocellular Carcinoma. Int. J. Mol. Sci. 2025, 26, 1936. [Google Scholar] [CrossRef]
- Shen, M.; Hu, P.; Donskov, F.; Wang, G.; Liu, Q.; Du, J. Tumor-associated neutrophils as a new prognostic factor in cancer: A systematic review and meta-analysis. PLoS ONE 2014, 9, e98259. [Google Scholar] [CrossRef]
- Uribe-Querol, E.; Rosales, C. Neutrophils in Cancer: Two Sides of the Same Coin. J. Immunol. Res. 2015, 2015, 983698. [Google Scholar] [CrossRef]
- Shen, K.Y.; Zhu, Y.; Xie, S.Z.; Qin, L.X. Immunosuppressive tumor microenvironment and immunotherapy of hepatocellular carcinoma: Current status and prospectives. J. Hematol. Oncol. 2024, 17, 25. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Steinberg, B.E.; Grinstein, S. Unconventional roles of the NADPH oxidase: Signaling, ion homeostasis, and cell death. Sci. STKE 2007, 2007, pe11. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Miao, C.; Zhang, H. Targeting neutrophil extracellular traps in cancer progression and metastasis. Theranostics 2025, 15, 5846–5869. [Google Scholar] [CrossRef] [PubMed]
- Masucci, M.T.; Minopoli, M.; Del Vecchio, S.; Carriero, M.V. The Emerging Role of Neutrophil Extracellular Traps (NETs) in Tumor Progression and Metastasis. Front. Immunol. 2020, 11, 1749. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Wysocki, R.W.; Amoozgar, Z.; Maiorino, L.; Fein, M.R.; Jorns, J.; Schott, A.F.; Kinugasa-Katayama, Y.; Lee, Y.; Won, N.H.; et al. Cancer cells induce metastasis-supporting neutrophil extracellular DNA traps. Sci. Transl. Med. 2016, 8, 361ra138. [Google Scholar] [CrossRef]
- Zhan, X.; Wu, R.; Kong, X.H.; You, Y.; He, K.; Sun, X.Y.; Huang, Y.; Chen, W.X.; Duan, L. Elevated neutrophil extracellular traps by HBV-mediated S100A9-TLR4/RAGE-ROS cascade facilitate the growth and metastasis of hepatocellular carcinoma. Cancer Commun. 2023, 43, 225–245. [Google Scholar] [CrossRef]
- Yang, L.Y.; Luo, Q.; Lu, L.; Zhu, W.W.; Sun, H.T.; Wei, R.; Lin, Z.F.; Wang, X.Y.; Wang, C.Q.; Lu, M.; et al. Increased neutrophil extracellular traps promote metastasis potential of hepatocellular carcinoma via provoking tumorous inflammatory response. J. Hematol. Oncol. 2020, 13, 3. [Google Scholar] [CrossRef]
- Seyhan, D.; Allaire, M.; Fu, Y.; Conti, F.; Wang, X.W.; Gao, B.; Lafdil, F. Immune microenvironment in hepatocellular carcinoma: From pathogenesis to immunotherapy. Cell Mol. Immunol. 2025, 22, 1132–1158. [Google Scholar] [CrossRef]
- Song, M.; Zhang, C.; Cheng, S.; Ouyang, D.; Ping, Y.; Yang, J.; Zhang, Y.; Tang, Y.; Chen, H.; Wang, Q.J.; et al. DNA of Neutrophil Extracellular Traps Binds TMCO6 to Impair CD8+ T-cell Immunity in Hepatocellular Carcinoma. Cancer Res. 2024, 84, 1613–1629. [Google Scholar] [CrossRef]
- Velliou, R.I.; Mitroulis, I.; Chatzigeorgiou, A. Neutrophil extracellular traps contribute to the development of hepatocellular carcinoma in NASH by promoting Treg differentiation. Hepatobiliary Surg. Nutr. 2022, 11, 415–418. [Google Scholar] [CrossRef]
- Yang, L.Y.; Shen, X.T.; Sun, H.T.; Zhu, W.W.; Zhang, J.B.; Lu, L. Neutrophil extracellular traps in hepatocellular carcinoma are enriched in oxidized mitochondrial DNA which is highly pro-inflammatory and pro-metastatic. J. Cancer 2022, 13, 1261–1271. [Google Scholar] [CrossRef]
- Zhu, W.; Fan, C.; Dong, S.; Li, X.; Chen, H.; Zhou, W. Neutrophil extracellular traps regulating tumorimmunity in hepatocellular carcinoma. Front. Immunol. 2023, 14, 1253964. [Google Scholar] [CrossRef] [PubMed]
- Masuda, S.; Nakazawa, D.; Shida, H.; Miyoshi, A.; Kusunoki, Y.; Tomaru, U.; Ishizu, A. NETosis markers: Quest for specific, objective, and quantitative markers. Clin. Chim. Acta 2016, 459, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Lai, Q.; Zhou, Y.; He, J.; Song, Y.; Liao, M.; Sun, J.; Li, M.; Zhang, M.; Liang, W.; et al. Noninvasive evaluation of neutrophil extracellular traps signature predicts clinical outcomes and immunotherapy response in hepatocellular carcinoma. Front. Immunol. 2023, 14, 1134521. [Google Scholar] [CrossRef] [PubMed]
- Sue, T.; Ichikawa, T.; Hattori, S.; Otani, H.; Fujimura, S.; Higuchi, T.; Okumura, N.; Higuchi, Y. Quantitative evaluation of citrullinated fibrinogen for detection of neutrophil extracellular traps. Immunol. Res. 2024, 72, 409–417. [Google Scholar] [CrossRef]
- Liu, K.; Sun, E.; Lei, M.; Li, L.; Gao, J.; Nian, X.; Wang, L. BCG-induced formation of neutrophil extracellular traps play an important role in bladder cancer treatment. Clin. Immunol. 2019, 201, 4–14. [Google Scholar] [CrossRef]
- Schedel, F.; Mayer-Hain, S.; Pappelbaum, K.I.; Metze, D.; Stock, M.; Goerge, T.; Loser, K.; Sunderkötter, C.; Luger, T.A.; Weishaupt, C. Evidence and impact of neutrophil extracellular traps in malignant melanoma. Pigment Cell Melanoma Res. 2020, 33, 63–73. [Google Scholar] [CrossRef]
- Kalafati, L.; Kourtzelis, I.; Schulte-Schrepping, J.; Li, X.; Hatzioannou, A.; Grinenko, T.; Hagag, E.; Sinha, A.; Has, C.; Dietz, S.; et al. Innate Immune Training of Granulopoiesis Promotes Anti-tumor Activity. Cell 2020, 183, 771–785.e12. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Abed, U.; Goosmann, C.; Hurwitz, R.; Schulze, I.; Wahn, V.; Weinrauch, Y.; Brinkmann, V.; Zychlinsky, A. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 2007, 176, 231–241. [Google Scholar] [CrossRef]
- Yipp, B.G.; Kubes, P. NETosis: How vital is it? Blood 2013, 122, 2784–2794. [Google Scholar] [CrossRef]
- Pilsczek, F.H.; Salina, D.; Poon, K.K.; Fahey, C.; Yipp, B.G.; Sibley, C.D.; Robbins, S.M.; Green, F.H.; Surette, M.G.; Sugai, M.; et al. A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus. J. Immunol. 2010, 185, 7413–7425. [Google Scholar] [CrossRef]
- Yipp, B.G.; Petri, B.; Salina, D.; Jenne, C.N.; Scott, B.N.; Zbytnuik, L.D.; Pittman, K.; Asaduzzaman, M.; Wu, K.; Meijndert, H.C.; et al. Infection-induced NETosis is a dynamic process involving neutrophil multitasking in vivo. Nat. Med. 2012, 18, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, S.; Mihalache, C.; Kozlowski, E.; Schmid, I.; Simon, H.U. Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps. Cell Death Differ. 2009, 16, 1438–1444. [Google Scholar] [CrossRef] [PubMed]
- Sil, P.; Hayes, C.P.; Reaves, B.J.; Breen, P.; Quinn, S.; Sokolove, J.; Rada, B. P2Y6 Receptor Antagonist MRS2578 Inhibits Neutrophil Activation and Aggregated Neutrophil Extracellular Trap Formation Induced by Gout-Associated Monosodium Urate Crystals. J. Immunol. 2017, 198, 428–442. [Google Scholar] [CrossRef] [PubMed]
- Behnen, M.; Leschczyk, C.; Möller, S.; Batel, T.; Klinger, M.; Solbach, W.; Laskay, T. Immobilized immune complexes induce neutrophil extracellular trap release by human neutrophil granulocytes via FcγRIIIB and Mac-1. J. Immunol. 2014, 193, 1954–1965. [Google Scholar] [CrossRef]
- Hoppenbrouwers, T.; Autar, A.S.A.; Sultan, A.R.; Abraham, T.E.; van Cappellen, W.A.; Houtsmuller, A.B.; van Wamel, W.J.B.; van Beusekom, H.M.M.; van Neck, J.W.; de Maat, M.P.M. In vitro induction of NETosis: Comprehensive live imaging comparison and systematic review. PLoS ONE 2017, 12, e0176472. [Google Scholar] [CrossRef]
- Hakkim, A.; Fuchs, T.A.; Martinez, N.E.; Hess, S.; Prinz, H.; Zychlinsky, A.; Waldmann, H. Activation of the Raf-MEK-ERK pathway is required for neutrophil extracellular trap formation. Nat. Chem. Biol. 2011, 7, 75–77. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef]
- DeSouza-Vieira, T.; Guimarães-Costa, A.; Rochael, N.C.; Lira, M.N.; Nascimento, M.T.; Lima-Gomez, P.S.; Mariante, R.M.; Persechini, P.M.; Saraiva, E.M. Neutrophil extracellular traps release induced by Leishmania: Role of PI3Kγ, ERK, PI3Kσ, PKC, and [Ca2+]. J. Leukoc. Biol. 2016, 100, 801–810. [Google Scholar] [CrossRef]
- Mansouri, P.; Mansouri, P.; Behmard, E.; Najafipour, S.; Kouhpayeh, S.A.; Farjadfar, A. Peptidylarginine deiminase (PAD): A promising target for chronic diseases treatment. Int. J. Biol. Macromol. 2024, 278, 134576. [Google Scholar] [CrossRef]
- Wang, Y.; Li, M.; Stadler, S.; Correll, S.; Li, P.; Wang, D.; Hayama, R.; Leonelli, L.; Han, H.; Grigoryev, S.A.; et al. Histone hypercitrullination mediates chromatin decondensation and neutrophil extracellular trap formation. J. Cell Biol. 2009, 184, 205–213. [Google Scholar] [CrossRef]
- Kenny, E.F.; Herzig, A.; Krüger, R.; Muth, A.; Mondal, S.; Thompson, P.R.; Brinkmann, V.; Bernuth, H.V.; Zychlinsky, A. Diverse stimuli engage different neutrophil extracellular trap pathways. eLife 2017, 6, e24437. [Google Scholar] [CrossRef] [PubMed]
- Papayannopoulos, V.; Metzler, K.D.; Hakkim, A.; Zychlinsky, A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 2010, 191, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, M.; Lindberg, M.R.; Kennett, M.J.; Xiong, N.; Wang, Y. PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. J. Exp. Med. 2010, 207, 1853–1862. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Monteleone, M.; Boucher, D.; Sollberger, G.; Ramnath, D.; Condon, N.D.; von Pein, J.B.; Broz, P.; Sweet, M.J.; Schroder, K. Noncanonical inflammasome signaling elicits gasdermin D-dependent neutrophil extracellular traps. Sci. Immunol. 2018, 3, eaar6676. [Google Scholar] [CrossRef]
- Thiam, H.R.; Wong, S.L.; Qiu, R.; Kittisopikul, M.; Vahabikashi, A.; Goldman, A.E.; Goldman, R.D.; Wagner, D.D.; Waterman, C.M. NETosis proceeds by cytoskeleton and endomembrane disassembly and PAD4-mediated chromatin decondensation and nuclear envelope rupture. Proc. Natl. Acad. Sci. USA 2020, 117, 7326–7337. [Google Scholar] [CrossRef]
- Urban, C.; Zychlinsky, A. Netting bacteria in sepsis. Nat. Med. 2007, 13, 403–404. [Google Scholar] [CrossRef]
- Byrd, A.S.; O’Brien, X.M.; Johnson, C.M.; Lavigne, L.M.; Reichner, J.S. An extracellular matrix-based mechanism of rapid neutrophil extracellular trap formation in response to Candida albicans. J. Immunol. 2013, 190, 4136–4148. [Google Scholar] [CrossRef]
- Clark, S.R.; Ma, A.C.; Tavener, S.A.; McDonald, B.; Goodarzi, Z.; Kelly, M.M.; Patel, K.D.; Chakrabarti, S.; McAvoy, E.; Sinclair, G.D.; et al. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nat. Med. 2007, 13, 463–469. [Google Scholar] [CrossRef]
- Heyworth, P.G.; Cross, A.R.; Curnutte, J.T. Chronic granulomatous disease. Curr. Opin. Immunol. 2003, 15, 578–584. [Google Scholar] [CrossRef]
- Lood, C.; Blanco, L.P.; Purmalek, M.M.; Carmona-Rivera, C.; De Ravin, S.S.; Smith, C.K.; Malech, H.L.; Ledbetter, J.A.; Elkon, K.B.; Kaplan, M.J. Neutrophil extracellular traps enriched in oxidized mitochondrial DNA are interferogenic and contribute to lupus-like disease. Nat. Med. 2016, 22, 146–153. [Google Scholar] [CrossRef]
- Douda, D.N.; Khan, M.A.; Grasemann, H.; Palaniyar, N. SK3 channel and mitochondrial ROS mediate NADPH oxidase-independent NETosis induced by calcium influx. Proc. Natl. Acad. Sci. USA 2015, 112, 2817–2822. [Google Scholar] [CrossRef]
- Azzouz, D.; Palaniyar, N. How Do ROS Induce NETosis? Oxidative DNA Damage, DNA Repair, and Chromatin Decondensation. Biomolecules 2024, 14, 1307. [Google Scholar] [CrossRef] [PubMed]
- Wise, A.D.; TenBarge, E.G.; Mendonça, J.D.C.; Mennen, E.C.; McDaniel, S.R.; Reber, C.P.; Holder, B.E.; Bunch, M.L.; Belevska, E.; Marshall, M.G.; et al. Mitochondria sense bacterial lactate and drive release of neutrophil extracellular traps. Cell Host Microbe 2025, 33, 341–357.e9. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Yan, X.; Zhang, H. Mitochondrial DNA-activated cGAS-STING pathway in cancer: Mechanisms and therapeutic implications. Biochim. Biophys. Acta Rev. Cancer 2025, 1880, 189249. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Zhao, Y.; Shen, M.; Wu, L.; Yang, H.; Yao, Y.; Yang, Q.; Du, J.; Liu, L.; Li, Y.; Bai, Y. Stromal cells in the tumor microenvironment: Accomplices of tumor progression? Cell Death Dis. 2023, 14, 587. [Google Scholar] [CrossRef]
- James, P.; Kaushal, D.; Beaumont Wilson, R. NETosis in Surgery: Pathophysiology, Prevention, and Treatment. Ann. Surg. 2024, 279, 765–780. [Google Scholar] [CrossRef]
- Kwong, T.T.; Xiong, Z.; Zhang, Y.; Wu, H.; Cao, J.; Pak-Chun Wong, P.; Liu, X.; Wang, J.; Wong, C.H.; Man-Kit Tse, G.; et al. Overcoming immunotherapy resistance in hepatocellular carcinoma by targeting myeloid IL-8/CXCR2 signaling. Mol. Ther. 2025, 33, 1659–1673. [Google Scholar] [CrossRef]
- de Andrea, C.E.; Ochoa, M.C.; Villalba-Esparza, M.; Teijeira, Á.; Schalper, K.A.; Abengozar-Muela, M.; Eguren-Santamaría, I.; Sainz, C.; Sánchez-Gregorio, S.; Garasa, S.; et al. Heterogenous presence of neutrophil extracellular traps in human solid tumours is partially dependent on IL-8. J. Pathol. 2021, 255, 190–201. [Google Scholar] [CrossRef]
- Guan, X.; Lu, Y.; Zhu, H.; Yu, S.; Zhao, W.; Chi, X.; Xie, C.; Yin, Z. The Crosstalk Between Cancer Cells and Neutrophils Enhances Hepatocellular Carcinoma Metastasis via Neutrophil Extracellular Traps-Associated Cathepsin G Component: A Potential Therapeutic Target. J. Hepatocell. Carcinoma 2021, 8, 451–465. [Google Scholar] [CrossRef]
- Pan, J.J.; Xie, S.Z.; Zheng, X.; Xu, J.F.; Xu, H.; Yin, R.Q.; Luo, Y.L.; Shen, L.; Chen, Z.R.; Chen, Y.R.; et al. Acetyl-CoA metabolic accumulation promotes hepatocellular carcinoma metastasis via enhancing CXCL1-dependent infiltration of tumor-associated neutrophils. Cancer Lett. 2024, 592, 216903. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Gu, Z.; Yuan, X.; Jin, R.; Li, J. Prognostic Signature of NETs-Related Genes in Hepatocellular Carcinoma Based on Bulk and Single-Cell Transcriptomics. J. Hepatocell. Carcinoma 2025, 12, 2351–2367. [Google Scholar] [CrossRef] [PubMed]
- Hidaka, H.; Ishiko, T.; Ishikawa, S.; Ikeda, O.; Mita, S.; Iwamura, T.; Chijiiwa, K.; Ogawa, M. Constitutive IL-8 expression in cancer cells is associated with mutation of p53. J. Exp. Clin. Cancer Res. 2005, 24, 127–133. [Google Scholar] [PubMed]
- Lei, M.M.L.; Leung, C.O.N.; Leung, R.W.H.; Wu, X.Q.; Chung, K.P.S.; Gu, C.Y.J.; Chan, M.S.M.; Chau, W.K.; Mu, Q.H.; Ng, K.Y.; et al. Wild-type KRAS activation drives evasion of interferon-mediated immunity and resistance to immunotherapy in hepatocellular carcinoma. Nat. Commun. 2025, 16, 9913. [Google Scholar] [CrossRef]
- Zhou, K.; Luo, X.; Wang, Y.; Cao, D.; Sun, G. MicroRNA-30a suppresses tumor progression by blocking Ras/Raf/MEK/ERK signaling pathway in hepatocellular carcinoma. Biomed. Pharmacother. 2017, 93, 1025–1032. [Google Scholar] [CrossRef]
- Qiu, Y.; Wang, Y.; Chai, Z.; Ni, D.; Li, X.; Pu, J.; Chen, J.; Zhang, J.; Lu, S.; Lv, C.; et al. Targeting RAS phosphorylation in cancer therapy: Mechanisms and modulators. Acta Pharm. Sin. B 2021, 11, 3433–3446. [Google Scholar] [CrossRef]
- Pan, B.; Zhang, Z.; Ye, D.; Zhang, X.; Yao, Y.; Luo, Y.; Hong, H.; Cai, X.; Chen, Y.; Tang, N. PPARα suppresses growth of hepatocellular carcinoma in a high-fat diet context by reducing neutrophil extracellular trap release. JHEP Rep. 2025, 7, 101228. [Google Scholar] [CrossRef]
- Yang, J.D.; Nakamura, I.; Roberts, L.R. The tumor microenvironment in hepatocellular carcinoma: Current status and therapeutic targets. Semin. Cancer Biol. 2011, 21, 35–43. [Google Scholar] [CrossRef]
- Xue, J.; Zhang, J.; Chen, G.; Chen, L.; Lu, X. Advances in Tumor Microenvironment and Immunotherapeutic Strategies for Hepatocellular Carcinoma. Oncol. Res. 2025, 33, 2309–2329. [Google Scholar] [CrossRef]
- Zou, Z.; Lin, Z.; Wu, C.; Tan, J.; Zhang, J.; Peng, Y.; Zhang, K.; Li, J.; Wu, M.; Zhang, Y. Micro-Engineered Organoid-on-a-Chip Based on Mesenchymal Stromal Cells to Predict Immunotherapy Responses of HCC Patients. Adv. Sci. 2023, 10, e2302640. [Google Scholar] [CrossRef]
- Liu, J.; Chen, S.; Wang, W.; Ning, B.F.; Chen, F.; Shen, W.; Ding, J.; Chen, W.; Xie, W.F.; Zhang, X. Cancer-associated fibroblasts promote hepatocellular carcinoma metastasis through chemokine-activated hedgehog and TGF-β pathways. Cancer Lett. 2016, 379, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ren, H.; Dai, B.; Li, J.; Shang, L.; Huang, J.; Shi, X. Hepatocellular carcinoma-derived exosomal miRNA-21 contributes to tumor progression by converting hepatocyte stellate cells to cancer-associated fibroblasts. J. Exp. Clin. Cancer Res. 2018, 37, 324. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wang, Y.; Xiong, Q.; He, J.; Wang, H.; Islam, M.; Zhou, X.; Kim, A.; Zhang, H.; Huang, H.; et al. Neutrophil extracellular traps promote MASH fibrosis by metabolic reprogramming of HSC. Hepatology 2025, 81, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Blanco, L.P.; Wang, X.; Carlucci, P.M.; Torres-Ruiz, J.J.; Romo-Tena, J.; Sun, H.W.; Hafner, M.; Kaplan, M.J. RNA Externalized by Neutrophil Extracellular Traps Promotes Inflammatory Pathways in Endothelial Cells. Arthritis Rheumatol. 2021, 73, 2282–2292. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, C.; Yu, M.; Zhao, X.; Du, J.; Li, Y.; Jing, H.; Dong, Z.; Kou, J.; Bi, Y.; et al. Neutrophil extracellular traps induced by activated platelets contribute to procoagulant activity in patients with colorectal cancer. Thromb. Res. 2019, 180, 87–97. [Google Scholar] [CrossRef]
- Li, J.C.; Zou, X.M.; Yang, S.F.; Jin, J.Q.; Zhu, L.; Li, C.J.; Yang, H.; Zhang, A.G.; Zhao, T.Q.; Chen, C.Y. Neutrophil extracellular traps participate in the development of cancer-associated thrombosis in patients with gastric cancer. World J. Gastroenterol. 2022, 28, 3132–3149. [Google Scholar] [CrossRef]
- Peng, J.; Pan, F.; Xu, Y.; Yan, Y.; Gao, M.; Zang, H.; Lin, G.; Cheng, L.; Zhou, Y. Mesenchymal stem cell transplantation alleviated TBI-induced lung injury by inhibiting PAD4-dependent NET formation. J. Adv. Res. 2025. [Google Scholar] [CrossRef]
- Pedrazza, L.; Cunha, A.A.; Luft, C.; Nunes, N.K.; Schimitz, F.; Gassen, R.B.; Breda, R.V.; Donadio, M.V.; de Souza Wyse, A.T.; Pitrez, P.M.C.; et al. Mesenchymal stem cells improves survival in LPS-induced acute lung injury acting through inhibition of NETs formation. J. Cell Physiol. 2017, 232, 3552–3564. [Google Scholar] [CrossRef]
- Ye, Y.; Ye, Y.; Tian, M.; Zhao, Y.; Guo, Z.; Jin, C.; Duan, S.; Zheng, Y. Mesenchymal Stem Cell-Derived Exosomes in Anti-NET Therapy: Mechanisms, Challenges, and Future Perspectives. Int. J. Nanomed. 2025, 20, 14481–14497. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhang, X.; Zhang, L.; Li, W.; Wu, H.; Yuan, X.; Mao, F.; Wang, M.; Zhu, W.; Qian, H.; et al. The IL-6-STAT3 axis mediates a reciprocal crosstalk between cancer-derived mesenchymal stem cells and neutrophils to synergistically prompt gastric cancer progression. Cell Death Dis. 2014, 5, e1295. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Y.; Dong, K.; Sun, Z.; Zhao, D.; Wang, W.; Yu, G.; Liu, W.; Xu, G.; Han, Z.; et al. Programming of the development of tumor-promoting neutrophils by mesenchymal stromal cells. Cell Physiol. Biochem. 2014, 33, 1802–1814. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Weaver, V.M.; Werb, Z. The extracellular matrix: A dynamic niche in cancer progression. J. Cell Biol. 2012, 196, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Friedman, S.L. Fibrosis-dependent mechanisms of hepatocarcinogenesis. Hepatology 2012, 56, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.T.; Xie, S.Z.; Zheng, X.; Zou, T.T.; Hu, B.Y.; Xu, J.; Liu, L.; Xu, Y.F.; Wang, X.F.; Wang, H.; et al. Cirrhotic-extracellular matrix attenuates aPD-1 treatment response by initiating immunosuppressive neutrophil extracellular traps formation in hepatocellular carcinoma. Exp. Hematol. Oncol. 2024, 13, 20. [Google Scholar] [CrossRef]
- Wang, Z.; Cheng, Z.X.; Abrams, S.T.; Lin, Z.Q.; Yates, E.D.; Yu, Q.; Yu, W.P.; Chen, P.S.; Toh, C.H.; Wang, G.Z. Extracellular histones stimulate collagen expression in vitro and promote liver fibrogenesis in a mouse model via the TLR4-MyD88 signaling pathway. World J. Gastroenterol. 2020, 26, 7513–7527. [Google Scholar] [CrossRef]
- Chen, D.; Jin, Z.; Chu, H.; Wu, Y.; Bian, Y.; Yuan, T.; Lv, H.; Xia, Q.; Wang, L.; Chu, Q.; et al. DNASE1L3-expressing dendritic cells promote CD8+ T cell function and anti-PD-(L)1 therapy efficacy by degrading neutrophil extracellular traps. Cancer Cell 2025, 43, 1758–1775.e8. [Google Scholar] [CrossRef]
- Kaltenmeier, C.; Yazdani, H.O.; Morder, K.; Geller, D.A.; Simmons, R.L.; Tohme, S. Neutrophil Extracellular Traps Promote T Cell Exhaustion in the Tumor Microenvironment. Front. Immunol. 2021, 12, 785222. [Google Scholar] [CrossRef]
- Teijeira, Á.; Garasa, S.; Gato, M.; Alfaro, C.; Migueliz, I.; Cirella, A.; de Andrea, C.; Ochoa, M.C.; Otano, I.; Etxeberria, I.; et al. CXCR1 and CXCR2 Chemokine Receptor Agonists Produced by Tumors Induce Neutrophil Extracellular Traps that Interfere with Immune Cytotoxicity. Immunity 2020, 52, 856–871.e8. [Google Scholar] [CrossRef]
- Fridlender, Z.G.; Sun, J.; Kim, S.; Kapoor, V.; Cheng, G.; Ling, L.; Worthen, G.S.; Albelda, S.M. Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 2009, 16, 183–194. [Google Scholar] [CrossRef]
- Tan, X.; Wang, Y.; Yu, Y.; Zheng, R.; Li, J.; Chen, S.; Xie, Q.; Guo, S.; Zhang, C.; Deng, X.; et al. Neutrophil-to-lymphocyte ratio predicts a poor prognosis for penile cancer with an immunosuppressive tumor microenvironment. Front. Immunol. 2025, 16, 1568825. [Google Scholar] [CrossRef]
- Sun, X.; Gui, Y.; Yang, T.; Chen, L.; Zhang, Y.; Yan, L.; Chen, W.; Wang, B. PD-L1+ neutrophils induced NETs in malignant ascites is a potential biomarker in HCC. Cancer Immunol. Immunother. 2024, 73, 254. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Wu, R.; Kong, X.; Wang, X.; Duan, Y.; Cao, S.; Yu, S.; Zhao, Y.; Li, S.; Zhou, J.; et al. Neutrophil-macrophage crosstalk via NETs-IL-17/VEGF/S100A9 axis promotes hepatocellular carcinoma progression. J. Exp. Clin. Cancer Res. 2025, 45, 27. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Guan, B.; Liu, Y.; Gu, Q.; Chen, W.; Xie, B.; Zhou, M.; Xiang, J.; Zhao, S.; Zhao, Q.; et al. Fibrinogen-like 2 in tumor-associated macrophage-derived extracellular vesicles shapes an immunosuppressive microenvironment in colorectal liver metastases by promoting tumor stemness and neutrophil extracellular traps formation. Cancer Lett. 2025, 618, 217642. [Google Scholar] [CrossRef] [PubMed]
- Lei, Q.; Zhen, S.; Zhang, L.; Zhao, Q.; Yang, L.; Zhang, Y. A2AR-mediated CXCL5 upregulation on macrophages promotes NSCLC progression via NETosis. Cancer Immunol. Immunother. 2024, 73, 108. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, X.; Chen, S.; Wang, J.; Yu, S.; Li, Y.; Xu, M.; Aboubacar, H.; Li, J.; Shan, T.; et al. Gut-derived lipopolysaccharide promotes alcoholic hepatosteatosis and subsequent hepatocellular carcinoma by stimulating neutrophil extracellular traps through toll-like receptor 4. Clin. Mol. Hepatol. 2022, 28, 522–539. [Google Scholar] [CrossRef]
- van der Windt, D.J.; Sud, V.; Zhang, H.; Varley, P.R.; Goswami, J.; Yazdani, H.O.; Tohme, S.; Loughran, P.; O’Doherty, R.M.; Minervini, M.I.; et al. Neutrophil extracellular traps promote inflammation and development of hepatocellular carcinoma in nonalcoholic steatohepatitis. Hepatology 2018, 68, 1347–1360. [Google Scholar] [CrossRef]
- Sardana, S.; Singh, K.P.; Saminathan, M.; Vineetha, S.; Panda, S.; Dinesh, M.; Maity, M.; Varshney, R.; Sulabh, S.; Sahoo, M.; et al. Effect of inhibition of Toll-like receptor 3 signaling on pathogenesis of rabies virus in mouse model. Acta Trop. 2022, 234, 106589. [Google Scholar] [CrossRef]
- Cools-Lartigue, J.; Spicer, J.; McDonald, B.; Gowing, S.; Chow, S.; Giannias, B.; Bourdeau, F.; Kubes, P.; Ferri, L. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J. Clin. Investig. 2013, 123, 3446–3458. [Google Scholar] [CrossRef]
- Jiang, Z.Z.; Peng, Z.P.; Liu, X.C.; Guo, H.F.; Zhou, M.M.; Jiang, D.; Ning, W.R.; Huang, Y.F.; Zheng, L.; Wu, Y. Neutrophil extracellular traps induce tumor metastasis through dual effects on cancer and endothelial cells. Oncoimmunology 2022, 11, 2052418. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Shang, H.; Qiu, J.; Zhang, R.; Xiong, Y.; Wang, T.; Wang, F.; Wu, A.; Lin, X.; et al. In Situ Assembly of Transformable Monopeptide on Activated Neutrophils Attenuates NETs-Induced Hepatocellular Carcinoma Metastasis by Disrupting NE Nuclear Translocation. Adv. Sci. 2026, 13, e17415. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, C.; Dong, B.; Zhang, Z.; Li, X.; Huang, S.; Tang, D.; Jing, X.; Yu, S.; Zheng, T.; et al. Neutrophil extracellular traps promote immune escape in hepatocellular carcinoma by up-regulating CD73 through Notch2. Cancer Lett. 2024, 598, 217098. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, H.; Wang, Y.; Brown, Z.J.; Xia, Y.; Huang, Z.; Shen, C.; Hu, Z.; Beane, J.; Ansa-Addo, E.A.; et al. Regulatory T-cell and neutrophil extracellular trap interaction contributes to carcinogenesis in non-alcoholic steatohepatitis. J. Hepatol. 2021, 75, 1271–1283. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Yang, L.; Shen, X.; Pan, J.; Chen, Y.; Chen, J.; Wang, H.; Meng, J.; Chen, Z.; Xie, S.; et al. Targeting Gsk3a reverses immune evasion to enhance immunotherapy in hepatocellular carcinoma. J. Immunother. Cancer 2024, 12, e009642. [Google Scholar] [CrossRef] [PubMed]
- Tohme, S.; Yazdani, H.O.; Al-Khafaji, A.B.; Chidi, A.P.; Loughran, P.; Mowen, K.; Wang, Y.; Simmons, R.L.; Huang, H.; Tsung, A. Neutrophil Extracellular Traps Promote the Development and Progression of Liver Metastases after Surgical Stress. Cancer Res. 2016, 76, 1367–1380. [Google Scholar] [CrossRef]
- Kaltenmeier, C.T.; Yazdani, H.; van der Windt, D.; Molinari, M.; Geller, D.; Tsung, A.; Tohme, S. Neutrophil extracellular traps as a novel biomarker to predict recurrence-free and overall survival in patients with primary hepatic malignancies. HPB 2021, 23, 309–320. [Google Scholar] [CrossRef]
- Cheng, Y.; Gong, Y.; Chen, X.; Zhang, Q.; Zhang, X.; He, Y.; Pan, L.; Ni, B.; Yang, F.; Xu, Y.; et al. Injectable adhesive hemostatic gel with tumor acidity neutralizer and neutrophil extracellular traps lyase for enhancing adoptive NK cell therapy prevents post-resection recurrence of hepatocellular carcinoma. Biomaterials 2022, 284, 121506. [Google Scholar] [CrossRef]
- Gong, Y.; Cheng, Y.; Zeng, F.; Liu, X.; Yang, Y.; Zhang, F.; Wen, C.; Yang, F.; Li, H.; He, Y.; et al. A self-gelling hemostatic powder boosting radiotherapy-elicited NK cell immunity to combat postoperative hepatocellular carcinoma relapse. Biomaterials 2025, 317, 123068. [Google Scholar] [CrossRef]
- Seo, J.D.; Gu, J.Y.; Jung, H.S.; Kim, Y.J.; Kim, H.K. Contact System Activation and Neutrophil Extracellular Trap Markers: Risk Factors for Portal Vein Thrombosis in Patients With Hepatocellular Carcinoma. Clin. Appl. Thromb. Hemost. 2019, 25, 1076029618825310. [Google Scholar] [CrossRef]
- Han, F.; Guo, J.; Mu, M.; Bian, K.; Cui, Z.; Duan, Q.; Ma, J.; Jin, L.; Liu, W.; Chen, F. Mechanism of ozone alleviation of malignant ascites in hepatocellular carcinoma through the inhibition of neutrophil extracellular traps. PNAS Nexus 2023, 2, pgad280. [Google Scholar] [CrossRef]
- McGlynn, K.A.; Petrick, J.L.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma. Hepatology 2021, 73, 4–13. [Google Scholar] [CrossRef]
- Li, N.; Zheng, X.; Chen, M.; Huang, L.; Chen, L.; Huo, R.; Li, X.; Huang, Y.; Sun, M.; Mai, S.; et al. Deficient DNASE1L3 facilitates neutrophil extracellular traps-induced invasion via cyclic GMP-AMP synthase and the non-canonical NF-κB pathway in diabetic hepatocellular carcinoma. Clin. Transl. Immunol. 2022, 11, e1386. [Google Scholar] [CrossRef] [PubMed]
- Dunn, G.P.; Old, L.J.; Schreiber, R.D. The three Es of cancer immunoediting. Annu. Rev. Immunol. 2004, 22, 329–360. [Google Scholar] [CrossRef] [PubMed]
- Asafo-Agyei, K.O.; Samant, H. Hepatocellular Carcinoma. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2025. [Google Scholar]
- Han, M.; Liu, Y.; Cao, Y.; Zhang, Y.; Yan, Y.; Deng, S.; Yuan, X.; Xing, H.; Huang, Y.; Zhu, L. The Imbalance of Homeostasis in Neutrophil Extracellular Traps is Associated with Portal Vein Thrombosis in Patients with Decompensated Cirrhosis. J. Clin. Transl. Hepatol. 2024, 12, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhou, Y.; Zhou, Z.; Zhang, P.; Yan, R.; Sun, L.; Ma, W.; Zhang, T.; Shen, S.; Liu, H.; et al. Secreted protease PRSS35 suppresses hepatocellular carcinoma by disabling CXCL2-mediated neutrophil extracellular traps. Nat. Commun. 2023, 14, 1513. [Google Scholar] [CrossRef]
- Chu, Y.D.; Chen, W.T.; Lin, W.R.; Lai, M.W.; Yeh, C.T. Mitochondrial echoes in the bloodstream: Decoding ccf-mtDNA for the early detection and prognosis of hepatocellular carcinoma. Cell Biosci. 2025, 15, 118. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, Y.; Zhu, Y.; Zhang, W.; Wang, B.; Du, X.; Dai, Q.; Zhang, F.; Fang, Z. The m6A methyltransferase METTL5 promotes neutrophil extracellular trap network release to regulate hepatocellular carcinoma progression. Cancer Med. 2024, 13, e7165. [Google Scholar] [CrossRef]
- Yuan, D.; Zhang, F.; Lv, P.; Zhu, J.; Zhang, H.; Zhang, Z. A reliable prognostic model for hepatocellular carcinoma using neutrophil extracellular traps and immune related genes. Sci. Rep. 2025, 15, 19390. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Li, Y.; Qi, X.; Fan, X. A signature based on neutrophil extracellular trap-related genes for the assessment of prognosis, immunoinfiltration, mutation and therapeutic response in hepatocellular carcinoma. J. Gene Med. 2024, 26, e3588. [Google Scholar] [CrossRef]
- Yuan, Z.; Yang, X.; Hu, Z.; Gao, Y.; Yan, P.; Zheng, F.; Guo, Y.; Wang, X.; Zhou, J. Characterization of a predictive signature for tumor microenvironment and immunotherapy response in hepatocellular carcinoma involving neutrophil extracellular traps. Heliyon 2024, 10, e30827. [Google Scholar] [CrossRef]
- Ji, Z.; Zhang, C.; Yuan, J.; He, Q.; Zhang, X.; Yang, D.; Xu, N.; Chu, J. Predicting the immunity landscape and prognosis with an NCLs signature in liver hepatocellular carcinoma. PLoS ONE 2024, 19, e0298775. [Google Scholar] [CrossRef]
- Xu, Q.; Ying, H.; Xie, C.; Lin, R.; Huang, Y.; Zhu, R.; Liao, Y.; Zeng, Y.; Yu, F. Characterization of neutrophil extracellular traps related gene pair for predicting prognosis in hepatocellular carcinoma. J. Gene Med. 2023, 25, e3551. [Google Scholar] [CrossRef]
- Zhan, T.; Wang, W.; Guan, X.; Bao, W.; Lu, N.; Zhang, J. Construction of an m6A- and neutrophil extracellular traps-related lncRNA model to predict hepatocellular carcinoma prognosis and immune landscape. Front. Immunol. 2023, 14, 1231543. [Google Scholar] [CrossRef]
- Chen, J.; Fang, Y.; Tang, Z.; Dong, E.; Gao, J.; Zhu, G.; Kwangwari, P.; Feng, S.; Qu, W.; Wu, X.; et al. Predictive value of neutrophil-to-lymphocyte ratio in recurrent HCC after repeat hepatectomy or salvage liver transplantation. Hepatol. Int. 2025, 19, 856–865. [Google Scholar] [CrossRef]
- Xie, S.Z.; Yang, L.Y.; Wei, R.; Shen, X.T.; Pan, J.J.; Yu, S.Z.; Zhang, C.; Xu, H.; Xu, J.F.; Zheng, X.; et al. Targeting SPP1-orchestrated neutrophil extracellular traps-dominant pre-metastatic niche reduced HCC lung metastasis. Exp. Hematol. Oncol. 2024, 13, 111. [Google Scholar] [CrossRef]
- Yi, N.; Zhang, L.; Huang, X.; Ma, J.; Gao, J. Lenvatinib-activated NDUFA4L2/IL33/PADI4 pathway induces neutrophil extracellular traps that inhibit cuproptosis in hepatocellular carcinoma. Cell Oncol. 2025, 48, 487–504. [Google Scholar] [CrossRef]
- Deng, Z.; Mei, S.; Ouyang, Z.; Wang, R.; Wang, L.; Zou, B.; Dai, J.; Mao, K.; Li, Q.; Guo, Q.; et al. Dysregulation of gut microbiota stimulates NETs-driven HCC intrahepatic metastasis: Therapeutic implications of healthy faecal microbiota transplantation. Gut Microbes 2025, 17, 2476561. [Google Scholar] [CrossRef]
- Yin, Y.; Dai, H.; Sun, X.; Xi, Z.; Zhang, J.; Pan, Y.; Huang, Y.; Ma, X.; Xia, Q.; He, K. HRG inhibits liver cancer lung metastasis by suppressing neutrophil extracellular trap formation. Clin. Transl. Med. 2023, 13, e1283. [Google Scholar] [CrossRef] [PubMed]




| Mechanism Category | Key Molecules/Pathways | HCC Models Used | Reference |
|---|---|---|---|
| Promoting Tumor Initiation and Development | S100A9/TLR4/RAGE-ROS axis | HBV-stable HCC cell lines (e.g., HepG2.2.15), HBV-related HCC patient samples | [15] |
| LPS/TLR4-ROS axis | Ethanol-induced fatty liver and HCC model (ethanol/diethylnitrosamine (DEN) + a 4% Lieber-DeCarli liquid alcohol diet) in mice | [95] | |
| cGAS-STING-NF-κB-NLRP3-GSDMD axis, ox-mtDNA | HCC patient samples, High-fat diet (HFD) + DEN induced HCC mouse model | [67] | |
| Free fatty acids direct stimulation | NASH patient samples, STAM mouse model of nonalcoholic steatohepatitis (NASH)-HCC (Streptozotocin + HFD) | [96] | |
| TLR3/COX-2/PGE2 pathway, HSC activation | Western diet/carbon tetrachloride-induced metabolic dysfunction–associated steatohepatitis (MASH) fibrosis model | [73] | |
| Metabolic reprogramming, Naive CD4+ T-cell entiation into Tregs | STAM mouse model, choline-deficient + HFD + DEN mouse model | [97] | |
| Promoting Tumor Metastasis | VEGF, CD31, EMT markers, MMP2/MMP9 | HBV-stable HCC cell lines (e.g., HepG2.2.15), C57BL/6 mice were injected with LPS and HCC cell line H22 | [15] |
| β2 integrins, ICAM-1, MMP9 | Murine Lewis lung carcinoma cell subline H59, A549 human lung carcinoma cell line | [98] | |
| TLR4/9-COX2, inflammatory response | Immunocompetent C57BL/6 mouse lung metastasis model, Human HCC tissue samples | [16] | |
| EMT (E-cadherin ↓, Vimentin ↑) | HBV-related HCC cell line (HepG2, HepG2.2.15) and mouse lung metastasis model | [15] | |
| EMT, E-cadherin ↓, N-Cadherin ↑, Snail/Slug ↑ | Human HCC tissue samples, peripheral blood neutrophils from healthy donors + HCC tumor supernatants | [99] | |
| ox-mtDNA, IL-8, IL-6 | Peripheral blood neutrophils from HCC patients, HepG2 cells and mouse lung metastasis model | [20] | |
| NE, EMT, ECM | The Hepa1-6/luc cell line | [100] | |
| NET-related cathepsin G (cG), E-cadherin ↓ | Neutrophils from HCC patients and healthy donors, Lung metastasis model in nude mice | [60] | |
| Promoting Immune Evasion | NETs → Notch2 → NF-κB → CD73 ↑ → Treg infiltration | Mouse HCC model (hydrodynamic transfection), HCC organoids, clinical HCC samples | [101] |
| NETs → TLR4 → CD4+ T-cell → Treg → TCR | NASH-HCC mouse models, clinical NASH-HCC patient sample | [102] | |
| NETs-DNA → TMCO6 (on CD8+ T-cells) → Inhibition of TCR/NF-κB → CD8+ T cell exhaustion | Wild-type and TMCO6−/− mouse HCC models, clinical HCC samples | [18] | |
| Liver cirrhosis ECM/Col1 → DDR1 → NF-κB → CXCL8 ↑ → Neutrophil recruitment/NETs barrier | Liver cirrhosis mouse models (DMN-induced), subcutaneous/orthotopic models, humanized mouse models | [84] | |
| Gsk3a → LRG1 ↑ → Neutrophil recruitment and PD-L1 + NETs → CTL inhibition | C57BL/6 subcutaneous tumor models, immunodeficient NPG mouse models, co-culture systems | [103] | |
| Driving Postoperative Recurrence | Liver ischemia–reperfusion (I/R) → NETs ↑ → HMGB1 release → TLR9 activation | Mouse liver I/R model combined with MC38 colon cancer metastasis model, clinical mCRC patient samples | [104] |
| Pre/postoperative serum MPO–DNA, Cit-H3 levels ↑ → Predicts shorter RFS and OS | Cohort of HCC patients undergoing hepatectomy | [105] | |
| Postoperative local DNase I → Degrades NETs → Breaks physical barrier → Enhances NK/CD8+ T-cell immunity → Inhibits recurrence | Mouse liver resection recurrence models, co-culture experiments | [106] | |
| Radiotherapy (RT) → Induces NETs → Combined with autophagy/CD73/NETs inhibitors → Enhances RT-induced NK cell immunity → Prevents recurrence | Hepa1-6 mouse HCC model | [107] | |
| Promoting Systemic Complications | PVT: NETs markers ↑ (cfDNA, histone-DNA)/DNase activity ↓ → Hypercoagulability | Cohort of decompensated cirrhosis patients (with/without HCC) | [107] |
| PVT: NETs → Contact system activation (Factor XIIa ↑, HMWK ↓) → Hypercoagulability | HCC patient cohort (with/without PVT) | [108] | |
| Malignant Ascites: NETs markers (MPO–DNA, CitH3) ↑ in ascitic fluid vs. benign ascites | HCC patients with malignant ascites vs. benign ascites patient cohort | [91] | |
| Malignant Ascites: NETs release pro-inflammatory factors (TNF-α, IL-6, IFN-γ), VEGF, MMP-9 → Promotes ascites formation | H22 cell-bearing mouse malignant ascites model | [109] |
| Therapeutic Mechanism | Target | Compound/Method | References |
|---|---|---|---|
| Inhibition of Neutrophil Chemotaxis and Recruitment | Chemokine receptor CXCR2 | SB225002 | [61,125] |
| Inhibition of Key Enzymes for NETs Formation | PAD4 | GSK484 | [99,126] |
| Inhibition of Key Enzymes for NETs Formation | NE | Peptide nanomaterial (FTP-NPs) | [100] |
| Metabolic Modulation to Reduce NETs Generation | Mitochondrial oxidative stress | Metformin | [20] |
| Metabolic Modulation to Reduce NETs Generation | Peroxisome proliferator-activated receptor alpha (PPARα) | Bezafibrate/Fenofibrate | [67] |
| Gut Microbiota Modulation to Indirectly Inhibit NETs | Gut microbiota ecology | Fecal microbiota transplantation (FMT) from healthy donors | [127] |
| Neutralization of NETs-Inducing Factors | Interleukin-33 (IL-33), Mitochondrial complex I subunit NDUFA4L2 | IL-33 neutralizing antibody, Gene knockdown (shRNA/siRNA) | [126] |
| Direct Degradation of NETs Structure | DNA backbone of NETs | Deoxyribonuclease I (DNase I) | [16,20,60,99,101,106,107,125,126] |
| Blockade of Pro-Metastatic Signaling Upon NETs Internalization | TLR4/9—COX2 signaling pathway | Aspirin/Hydroxychloroquine (HCQ) | [16] |
| Supplementation/Activation of Endogenous NETs-Inhibitory Proteins | Secreted protease PRSS35 | PRSS35 overexpression | [115] |
| Supplementation/Activation of Endogenous NETs-Inhibitory Proteins | Histidine-rich glycoprotein (HRG) | HRG overexpression | [128] |
| Postoperative Adjuvant Clearance of Local NETs | NETs and acidic microenvironment at the surgical margin | Injectable hydrogel/powder loaded with DNase I | [106,107] |
| Sensitization to Immune Checkpoint Inhibitors | Collagen receptor DDR1 (in combination with anti-PD-1) | Nilotinib + Anti-PD-1 antibody | [84] |
| Sensitization to Immune Checkpoint Inhibitors | NETs-mediated immunosuppressive microenvironment (in combination with anti-PD-1) | DNase I + Anti-PD-1 antibody | [101] |
| Sensitization to Immune Checkpoint Inhibitors | Glycogen synthase kinase 3 alpha (GSK3a) | SB216763 + Anti-PD-1 antibody | [103] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, C.; Lu, J.; Tian, Y.; Lu, S.; Wang, W.; Jiang, J.; Zheng, X.; Yan, S. The Molecular Network of Neutrophil Extracellular Traps in Hepatocellular Carcinoma: Biogenesis, Function, and Therapeutic Implications. Molecules 2026, 31, 749. https://doi.org/10.3390/molecules31040749
Liu C, Lu J, Tian Y, Lu S, Wang W, Jiang J, Zheng X, Yan S. The Molecular Network of Neutrophil Extracellular Traps in Hepatocellular Carcinoma: Biogenesis, Function, and Therapeutic Implications. Molecules. 2026; 31(4):749. https://doi.org/10.3390/molecules31040749
Chicago/Turabian StyleLiu, Chang, Jienan Lu, Yang Tian, Sinan Lu, Weili Wang, Jun Jiang, Xiang Zheng, and Sheng Yan. 2026. "The Molecular Network of Neutrophil Extracellular Traps in Hepatocellular Carcinoma: Biogenesis, Function, and Therapeutic Implications" Molecules 31, no. 4: 749. https://doi.org/10.3390/molecules31040749
APA StyleLiu, C., Lu, J., Tian, Y., Lu, S., Wang, W., Jiang, J., Zheng, X., & Yan, S. (2026). The Molecular Network of Neutrophil Extracellular Traps in Hepatocellular Carcinoma: Biogenesis, Function, and Therapeutic Implications. Molecules, 31(4), 749. https://doi.org/10.3390/molecules31040749










