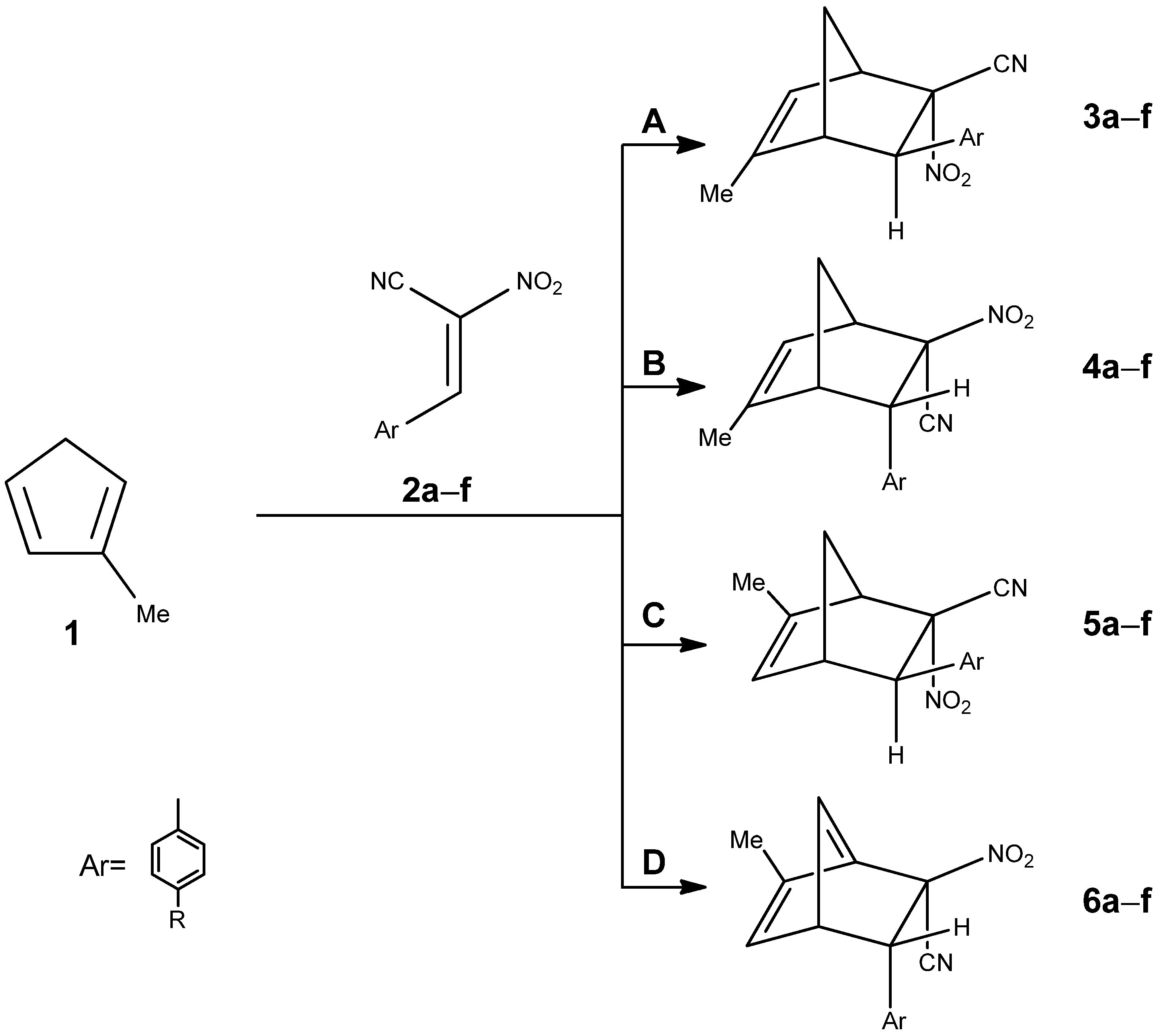
2.1. Energetic Profiles and Solvent Effects on the Reaction Mechanism
The first step involved the examination of all four possible reaction pathways. To this end, DFT calculations were performed using the ωB97X-D functional and the 6-311G(d) basis set, in combination with the PCM solvation model.
Calculations were performed for solvents with varying polarity, and it was shown that the reaction course is strongly dependent on solvent polarity. A distinct reactivity pattern was observed in non-polar (e.g., toluene) versus polar solvents (e.g., methanol, nitromethane, acetonitrile, water), with acetone exhibiting intermediate behavior consistent with its polar aprotic nature. Among the solvents investigated, toluene and acetone stand out due to their inability to act as hydrogen bond donors and their relatively low dielectric constants. This sets them apart from more polar, protic solvents such as water or methanol, which are capable of stronger solvation of charged or zwitterionic species. The findings have been collected and are detailed in the subsequent sections of the manuscript.
2.1.1. Stepwise Processes with the Heterocyclic Intermediate
Regardless of the solvent used, the reaction pathways leading to norbornene derivatives bearing a nitro group in the endo position (pathways A and C,
Figure 6) proceed via a two-step mechanism with a heterocyclic intermediate.
The first reaction phase is realized via the formation of a pre-reaction complex (MC), accompanied by a slight increase in Gibbs free energy (ca. 3 kcal/mol). Subsequently, the formation of the transition state (TS1) is associated with a more significant rise in Gibbs free energy, reaching approximately 16–18 kcal/mol (
Table 2 and
Table 3).
In nitromethane, the reaction proceeds through the formation of a weakly stabilized pre-reactive complex (MC), which is enthalpically favored (ΔH = −8.4 kcal·mol−1) but entropically penalized, resulting in a slightly positive free energy (ΔG = +3.1 kcal·mol−1). The first transition state (TS1) constitutes a barrier of ΔG≠ = 13.4 kcal·mol−1 relative to MC (ΔG = +16.5 kcal·mol−1 in absolute terms), indicating that the initial bond reorganization is kinetically accessible under ambient conditions. Passage through TS1 leads to the formation of a stabilized intermediate (INT, ΔG = −3.8 kcal·mol−1), which lies significantly lower in energy than both MC and TS1.
The subsequent progression toward products requires surmounting a second barrier, with TS2 located at ΔG = +11.4 kcal·mol−1. Relative to INT, this corresponds to ΔG≠ = 15.2 kcal·mol−1, a value higher than the preceding step, suggesting that the second transition state is kinetically more demanding. As a result, the intermediate is expected to be kinetically persistent and may accumulate to measurable concentrations before conversion to the final products. Ultimately, the reaction affords a strongly stabilized product state (PR, ΔG = −10.1 kcal·mol−1), confirming that the overall transformation is exergonic.
Taken together, these results demonstrate that in nitromethane the reaction proceeds via a two-step mechanism in which the first barrier (TS1) initiates intermediate formation, while the second barrier (TS2) controls the overall rate.
The calculated thermodynamic and kinetic parameters reveal that the investigated transformation proceeds exothermically in all studied solvents, with reaction enthalpies ranging from −18.7 to −26.7 kcal·mol−1 and Gibbs free energies from −2.4 to −10.3 kcal·mol−1. The reaction is therefore thermodynamically favorable, although the degree of stabilization of the products varies with the solvent. In particular, acetone appears to diminish the driving force, yielding the least negative ΔG (–2.4 kcal·mol−1), whereas toluene and the more polar solvents (methanol, acetonitrile, nitromethane, water) provide a significantly larger stabilization (ΔG ≈ −10 kcal·mol−1).
The kinetic profile is dominated by the first transition state (TS1), which consistently constitutes the highest energy barrier across all solvents (ΔG≠ ≈ 16–18 kcal·mol−1). The second transition state (TS2) is lower in free energy (ΔG≠ ≈ 11–16 kcal·mol−1), confirming that the first step is rate-determining. Intermediate species (INT) are enthalpically well-stabilized (ΔH ≈ −20 kcal·mol−1), but their Gibbs free energies are close to zero or slightly positive in polar media due to the strongly unfavorable entropic contributions (ΔS ≈ −55 cal·mol−1·K−1). This substantial entropy loss suggests a pronounced ordering effect during complexation and solvation, consistent with the formation of tightly bound intermediates.
Overall, solvent polarity exerts only a minor effect on the kinetic barriers but has a more pronounced influence on the relative thermodynamic stability of the products. These results indicate that the transformation is under kinetic control with a rate-determining first step, while product distribution and thermodynamic stability are modulated by solvent effects.
The reaction pathway in methanol was examined for both mechanistic alternatives (pathway A and pathway C). In both cases, the formation of the pre-reactive complex (MC) is only marginally stabilized (ΔG = +3.1 kcal·mol−1 for A and +3.3 kcal·mol−1 for C), reflecting a small entropic penalty associated with complexation. The subsequent energy profiles, however, diverge significantly.
For pathway A, the first transition state (TS1) appears at ΔG = +16.5 kcal·mol−1, corresponding to a relative barrier of 13.4 kcal·mol−1 with respect to MC. This step leads to a stabilized intermediate (INT, ΔG = −3.8 kcal·mol−1), from which the second transition state (TS2) requires an additional 15.3 kcal·mol−1 to be surmounted. The overall process culminates in an exergonic product state (PR, ΔG = −10.1 kcal·mol−1). Thus, pathway A involves moderate and balanced barriers in both steps, making it kinetically feasible and consistent with a mechanism where the intermediate may accumulate before progressing to products.
In contrast, pathway C is associated with higher barriers. TS1 is located at ΔG = +18.7 kcal·mol−1, giving a relative barrier of 15.4 kcal·mol−1, while TS2 rises to ΔG = +16.4 kcal·mol−1, corresponding to a barrier of 20.7 kcal·mol−1 above the intermediate (ΔG = −4.3 kcal·mol−1). Although the final products are similarly stabilized (ΔG = −9.2 kcal·mol−1), the kinetic demands of pathway C are substantially greater.
These results clearly indicate that pathway A is kinetically favored in methanol, with significantly lower activation barriers relative to pathway C. While both mechanisms are thermodynamically viable, the high second barrier in pathway C renders it unlikely to contribute meaningfully under standard conditions, suggesting that the observed reactivity is dominated by pathway A.
2.1.2. Single Step Mechanisms
The energy profile of pathway D in nitromethane reveals a simple scenario without a discrete intermediate. The pre-reactive complex (MC) is only weakly stabilized, with ΔG = +2.9 kcal·mol
−1, consistent with the entropic cost of association (
Table 4). The single transition state (TS) is located at ΔG = +19.6 kcal·mol
−1, corresponding to a relative activation barrier of 16.7 kcal·mol
−1 with respect to MC. This makes the process moderately demanding from a kinetic perspective.
The reaction products (PR) are substantially stabilized (ΔG = −8.7 kcal·mol
−1), confirming that the transformation is exergonic overall. Compared with the more complex mechanisms (pathways A and C), pathway D (
Figure 7) is thermodynamically similar (final product stabilization of ca. −9 kcal·mol
−1) but kinetically less favorable. Specifically, the single-step barrier in pathway D (ΔG
≠ = 19.4 kcal·mol
−1) exceeds the initial and secondary barriers of pathway A (16.5 and 15.2 kcal·mol
−1, respectively) and approaches the higher barriers characteristic of pathway C.
2.1.3. Stepwise Zwitterionic Mechanisms
In contrast to pathways A, C, and D, the reaction proceeding along pathway B exhibits completely different qualitative behavior. In nonpolar solvents (acetone and toluene), the reaction follows a mechanism analogous to pathway D—it is one-step process proceeding directly through the pre-reactive complex (MC) and the transition state to form the final product, nitronorbornene.
However, in more polar solvents, the reaction pathway changes markedly. After the formation of the molecular complex (MC), the system passes through a first transition state (TS1), leading to the formation of a zwitterionic intermediate (ZW). In the subsequent steps, this intermediate evolves through a second transition state (TS2) to yield the final product, nitronorbornene.
This represents one of the few well-documented examples of a reaction proceeding via such a zwitterionic mechanism [
32]. Therefore, we focused on a detailed characterization of the zwitterionic intermediate and the corresponding energy profile of this reaction pathway.
In nitromethane, pathway B (
Figure 8) proceeds through a sequence involving the pre-reactive complex (MC), a first transition state, a zwitterionic intermediate (ZW), a second transition state, and the final products. The MC is weakly stabilized (ΔG = +3.0 kcal·mol
−1), while the first transition state lies at ΔG = +18.2 kcal·mol
−1, giving an activation barrier of 15.0 kcal·mol
−1 relative to MC (
Table 5).
From this transition state, the reaction does not proceed directly to the products but instead forms a zwitterionic intermediate at ΔG = +13.7 kcal·mol−1. This species reflects charge separation stabilized by solvation in nitromethane and constitutes a shallow minimum on the potential energy surface. Progression from ZW to the products requires crossing a second transition state (TS2, ΔG = +15.2 kcal·mol−1), only slightly higher in energy than ZW itself. The final products are thermodynamically stabilized (ΔG = −9.1 kcal·mol−1), confirming a strongly exergonic outcome.
The presence of a zwitterionic intermediate in methanol contrasts with its absence in less polar solvents (toluene, acetone) and indicates that solvent polarity is crucial in stabilizing charge-separated species. In methanol, Pathway B therefore operates as a two-step process with a ZW intermediate, although the overall barrier is comparable to a single-step mechanism.
To gain deeper insight into the polar nature of the reaction, we analyzed the critical structures along the reaction pathway. The evaluation of the global electron density transfer (GEDT) (
Table 6) between the interacting fragments allows for a quantitative description of charge flow and provides additional evidence for the polar character of the process.
GEDT values of 0.43 e (TS1) and 0.79 e (TS2) indicate a highly polar process; the markedly larger GEDT for TS2 reflects pronounced charge separation and a stronger zwitterionic character, implying significant electrostatic stabilization and potential solvent sensitivity.
To assess the extent of asynchronicity in the examined reactions, all stationary structures were subjected to detailed analysis. The principal descriptors—interatomic distances between the reactive centers (r), the bond-formation progress parameter (l), and the asymmetry index (Δl)—are compiled in
Table 6.
Based on the analysis of the C1–C6 and C4–C7 bond lengths along the reaction pathway (
Table 6,
Figure 9 and
Figure 10), it can be clearly concluded that the studied cycloaddition proceeds in an asynchronous, two-step manner. In the initial complex (MC), both distances exceed 3.2 Å, indicating the absence of any significant chemical interaction. In the first transition state (TS1), the C1–C6 distance decreases substantially to 2.05 Å, while C4–C7 remains long (2.98 Å), suggesting that the first bond starts to form earlier. In the zwitterionic intermediate (ZW), the C1–C6 bond is already fully formed (1.61 Å), whereas C4–C7 is still elongated (3.24 Å), supporting the presence of a stepwise mechanism involving a charge-separated intermediate. In the second transition state (TS2), the C4–C7 distance begins to contract, ultimately leading to the formation of the final product, where both bond lengths (≈1.57–1.58 Å) correspond to typical σ bonds within the newly formed ring. These results clearly demonstrate that the cycloaddition does not proceed via a single, concerted process, but rather through two distinct stages characterized by sequential C–C bond formation. The plot shows (
Figure 9) the variation of key interatomic distances along the reaction coordinate. A clear parabolic trend can be observed, nearly symmetrical for both forming bonds. The Cartesian coordinates of the key structures along pathway B are provided in the
Supplementary Materials.
The progress indices (Δl) for the forming bonds differ for most TSs, indicating asynchronous bond formation In TS1, lC1–C6 = 0.70, while lC4−C7 = 0.12. This disparity indicates that the C1–C6 bond forms significantly earlier than the C4–C7 bond, suggesting pronounced asynchronicity. Similarly, in TS2, lC1−C6 = 0.97, while lC4−C7 = 0.00, showing that the C1–C6 bond is almost completely formed, whereas the C4–C7 bond has not yet begun to develop. Higher Δl values indicate greater asynchronicity.
2.3. Reaction Mechanism Elucidated by Bonding Evolution Theory (BET)
To gain detailed insight into the reaction mechanism and the evolution of chemical bonds within the zwitterionic system, we performed a Bonding Evolution Theory (BET) analysis [
34]. This approach allows for a precise characterization of bond formation and cleavage along the reaction pathway, providing a clear picture of electron density redistribution during the transformation. By applying BET, we aimed to identify key stages in the reaction, understand the sequence of bond-making and bond-breaking events, and correlate these changes with the overall stability and reactivity of the zwitterionic species.
The analysis of the ELF valence basin population changes along the IRC of the 42CA reaction between 1 and 2c shows that this reaction can be topologically characterized by 10 distinct phases of bond evolution for the reaction, clearly demonstrating a highly asynchronous and polar cycloaddition mechanism (
Figure 14,
Table 9).
At the early stages of the process (points 1–3), the disynaptic basins V(C1, C2) and V(C3, C4) increase in population, which corresponds to the initial strengthening of the σ-framework during reactant approach. Subsequently, both basins undergo a marked decrease, reflecting the rupture of the original C–C bonds. Subsequently, monosynaptic basins appear at C1 and C6, which then merge into a disynaptic basin V(C1–C6), corresponding to the formation of the C–C bond in the zwitterionic structure (ZWB). In the following steps, monosynaptic basins at C4 and C7 are formed first, which subsequently evolve into the disynaptic basin V(C4–C7), leading to the formation of the nitronorbornene structure. The process is polar and highly asynchronous, proceeding through a zwitterionic intermediate.
The basins associated with the aromatic ring (V(C10–C15)) remain nearly constant (2.7–2.8 e), confirming that the phenyl substituent does not undergo significant electronic reorganization and mainly serves as an electron-withdrawing stabilizer.
Throughout the reaction coordinate, a systematic redistribution of electron density occurs within the π-system and the nitro group. The evolution of V(C7, N2) and V(C9, N1) basins clearly indicates charge polarization toward the nitron fragment, supporting the zwitterionic character of the intermediate.
Phase I The key event in this stage is the merging of two disynaptic basins V(C6, C7) and V′(C6, C7), initially integrating 1.80 e and 1.70 e, respectively. As a result, a single, larger basin is formed with a population of 3.49 e.
Phase II At this stage, the disynaptic basins V(C1, C2) and V′(C1, C2) merge, increasing their populations from 1.75 e and 1.53 e to 3.24 e. The auxiliary basins V′(C1, C2) vanish, indicating the formation of stronger and more localized bonding interactions.
Phase III At this stage, the disynaptic basins V(C2, C3) and V′(C2, C3) merge, increasing their populations from 1.69 e and 1.56 e to 3.11 e. The auxiliary basins V′(C3, C4) vanish. The increase in V(C7, N2) (from 2.36 e to 2.65 e) suggests a change in the bonding regime in this region of the structure.
Phase IV A two newly formed monosynaptic basins with a population of 0.34 e and 0.09 e appear, indicating that atoms C1 and C6 begin to exhibit a certain degree of non-bonding electron density. This correlates with a slight decrease in V(C1, C2) (from 3.11 e to 2.77 e) and V(C1, C5) (from 2.01 e to 1.99 e), reflecting a partial weakening of bonding around C1.
Phase V The monosynaptic basins V(C1) and V(C6), formed in the previous phase, disappear, and a new disynaptic basin V(C1, C6) is formed from their merging electron densities. Additionally, a new monosynaptic basin with a total electron population of 0.39 e appears. A noticeable decrease in V(C6, C7) (from 3.44 e to 3.01 e) suggests that part of the C6–C7 bonding electron density has been redistributed into the new basins V(C6), V(C7), and V(C1, C6).
Phase VI A second monosynaptic basin V’(C7) with a population of 0.47 e appears, accompanied by an increase in V(C1, C6) (from 0.62 e to 0.99 e) and a further decrease in V(C6, C7) (from 3.01 e to 2.39 e). Atom C7 seems to exhibit two independent regions of non-bonding electron density, and the C6–C7 bond continues to weaken, favoring the formation of a new C1–C6 interaction.
Phase VII The V′(C7) basin disappears completely, indicating the loss of one of the previously separated regions of electron localization around atom C7. Simultaneously, the population of the V(C7) basin increases significantly, suggesting that the electron density formerly associated with V′(C7) has been transferred to V(C7).
Phase VIII A new monosynaptic basin V(C4) (0.10 e) appears, indicating a partial localization of electron density at the C4 atom. This newly formed basin reflects a transient accumulation of electronic charge that precedes the formation of the C4–C7 bond. The emergence of V(C4) thus marks an intermediate stage of the electronic reorganization, where the weakening of the former C3–C4 interaction facilitates the redistribution of electron density toward the forthcoming bonding region between C4 and C7.
Phase IX The disynaptic basin splits into two distinct basins, V(C2, C3) and V′(C2, C3), whose populations decrease from 3.10 e to 1.56 e and 1.58 e, respectively. This division indicates a redistribution of the electron density within the C2–C3 bonding region, suggesting a partial weakening or polarization of this bond.
Phase X A new disynaptic basin V(C4, C7) with a population of 1.11 e appears, marking the formation of a new C–C bond between atoms C4 and C7. This process is accompanied by a redistribution of electron density from the previously existing monosynaptic basins V(C4) and V(C7), which disappear at this stage. The vanishing of these monosynaptic basins indicates that the non-bonding electron density localized on C4 and C7 has been fully incorporated into the new bonding region, confirming the establishment of a stable σ-type C–C.
2.4. Evaluation of Substituent Effects on the Mechanistic Pathways
To verify whether the reaction mechanism established for the parent system can be considered general, we extended our study to include a substituent effect analysis. For this purpose, a series of conjugated nitroalkenes bearing para-substituted phenyl rings were investigated. The substituents were selected to cover a broad range of electronic effects as defined by their Hammett σ parameters, encompassing both electron-donating (-OMe, -Me) and electron-withdrawing (-F, -Cl, -NO2) groups.
The analysis of the corresponding potential energy surfaces revealed that the overall mechanistic pattern remains preserved across the entire substituent series. In all cases, the reaction proceeds via formation of a pre-reactive molecular complex, followed by a single or double transition-state pathway, depending on the specific reaction channel (A–D). However, quantitative differences were observed in the relative energies of the critical points, indicating that the electronic nature of the substituent subtly modulates the activation barriers and the stability of intermediates.
The relative thermodynamic parameters calculated for the stationary points along path A of the cycloaddition reaction of
1 with
2a–
f (
Table 10). The substituent effect on the reaction profile is clearly visible in the first step, where the Gibbs free energy barriers (ΔG
≠ for MC → TS1) decrease from 15.4 kcal·mol
−1 for the electron-donating OMe group to 11.2 kcal·mol
−1 for the strongly electron-withdrawing NO
2 group. This trend indicates that electron-accepting substituents facilitate the initial cycloaddition step by stabilizing the transition state. In contrast, the second-step barriers (INT → TS2) remain nearly constant (~15 kcal·mol
−1) across all derivatives, suggesting a less pronounced substituent influence. The relative stability of the products (PR) follows the order NO
2 > Cl > H ≈ F > Me > OMe, in agreement with the electron-withdrawing strength of the substituents. Entropic contributions are similar for all systems (ΔS ≈ −50 to −55 cal·mol
−1·K
−1), reflecting comparable structural reorganization upon cycloaddition.
In contrast to path A, this route (path B) proceeds through a zwitterionic intermediate (ZW) stabilized to various extents depending on the substituent at the phenyl ring. The first activation barrier (MC → TS) gradually decreases with increasing electron-withdrawing strength of the substituent, from 17.3 kcal·mol−1 for OMe to only 13.2 kcal·mol−1 for NO2, indicating that electrophilic activation of the dienophile favors the polar transition state. The zwitterionic intermediates are notably stabilized in the presence of electron-accepting substituents (ΔG(ZW) = 15.6 for Ome and 10.6 kcal·mol−1 for NO2), supporting a polar, two-step mechanism. The relative Gibbs free energies of the products follow the same trend as in path A, decreasing in the order OMe < Me < F < H < Cl < NO2. The NO2-substituted system exhibits the lowest energy barriers and the most stable product, confirming that path B is strongly favored for electron-deficient derivatives. Entropic effects remain similar across the series (ΔS ≈ −50 cal·mol−1·K−1), reflecting comparable degrees of structural ordering during the reaction progress.
Similarly to path A, this mechanism proceeds via a two-step process involving an intermediate (INT) (path C). The Gibbs free energy barriers of the first step (MC → TS1) range from 16.9 kcal·mol−1 for the OMe derivative to 13.2 kcal·mol−1 for the NO2-substituted system, showing a clear substituent effect consistent with the electron-withdrawing strength at the phenyl ring. Electron-accepting groups significantly lower both activation barriers and stabilize the intermediate and final product. The second transition state (INT → TS2) shows slightly lower and more uniform barriers (ΔG≠ ≈ 17–14 kcal·mol−1), suggesting a relatively similar energy demand for the ring closure step across all derivatives. The product stability follows the same trend as in paths A and B, increasing with the electron-withdrawing character of the substituent (OMe < Me < F < H < Cl < NO2). Overall, the NO2-substituted system exhibits the lowest free energies throughout the entire reaction profile, indicating a highly favorable reaction pathway. Entropic variations (ΔS ≈ −50 cal·mol−1·K−1) remain small, confirming that the reaction proceeds with comparable degrees of molecular organization for all substituents.
Unlike paths A–C, this mechanism proceeds through a transition state without the formation of an intermediate (path D). The Gibbs free energy barriers (MC → TS) systematically decrease with increasing electron-withdrawing ability of the substituent, from 18.6 kcal·mol−1 for the OMe derivative to 13.8 kcal·mol−1 for the NO2-substituted system. This trend clearly indicates that electron-accepting groups promote a more facile, concerted cycloaddition process. The relative stability of the products again follows the order OMe < Me < F < H < Cl < NO2, in line with their electronic nature. Among all derivatives, the NO2-substituted system exhibits both the lowest activation barrier and the most stable product, suggesting that the D path is thermodynamically and kinetically favored under polar conditions for electron-deficient substrates. Entropic changes remain consistent across the series (ΔS ≈ −50 cal·mol−1·K−1), implying similar degrees of molecular organization in the transition states.
Comparison of the four examined reaction pathways (A–D) indicates that the substituent at the phenyl ring has a pronounced influence on both the mechanism and energetics of the cycloaddition. For all cases, the product stability increases with the electron-withdrawing strength of the substituent (OMe < Me < F < H < Cl < NO2). Overall, the NO2-substituted system exhibits the lowest Gibbs free energies and the smallest barriers across all pathways, confirming that electron-deficient derivatives react more readily via a polar, nearly concerted mechanism. Electron-withdrawing groups were found to lower the activation energy, thereby facilitating the cycloaddition process, while electron-donating substituents produced the opposite effect. These observations are consistent with the general trend predicted by the Hammett correlation, confirming that the reaction is governed primarily by electronic rather than steric factors. Consequently, the investigated mechanism can be regarded as general and robust with respect to substituent-induced perturbations in the electronic structure of the nitroalkene component.