Changes in the Fatty Acid Composition and Antioxidant Properties in Mono-Protein Commercial Dry Dog Foods During Storage
Abstract
1. Introduction
2. Results
3. Discussion
3.1. Chemical Composition and Nutrient Stability
3.2. Antioxidant Stability and Its Role in Dog Food Preservation
3.3. Changes in the Fatty Acid Composition During Storage
4. Material and Methods
4.1. Material
4.2. Chemical Composition
4.3. Calculation of Metabolizable Energy
4.4. Fatty Acid Content
4.5. Index of Atherogenicity (IA)
4.6. Index of Thrombogenicity (IT)
4.7. Index of Hypocholesterolemic/Hypercholesterolemic Acids (h/H)
4.8. Hypocholesterolemic Acids (DFA) and Hypercholesterolemic Acids (OFA)
4.9. Determination of Total Phenolic Compound Content
4.10. Determination of Antioxidant Properties
4.10.1. ABTS Method
4.10.2. DPPH Method
4.10.3. FRAP Method
4.11. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) |
| AFR | adverse food reaction |
| AI | atherogenic index |
| ANOVA | two-way analysis of variance |
| BF3 | boron trifluoride |
| BHA | butylated hydroxyanisole |
| BHT | butylated hydroxytoluene |
| CA | crude ash |
| CF | crude fiber |
| CGA | chlorogenic acid equivalents |
| CP | crude protein |
| DE | digestible energy |
| DFA | hypocholesterolemic acids |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ED | energy digestibility |
| EE | ether extract |
| FA | fatty acids |
| FRAP | ferric reducing antioxidant power |
| GAE | gallic acid equivalents |
| GC-FID | flame ionization detector |
| GE | gross energy |
| h/H | hypocholesterolemic/hypercholesterolemic acids |
| HSD | honestly significant difference |
| ME | metabolizable energy |
| MUFA | monounsaturated fatty acids |
| NFE | nitrogen-free extracts |
| OFA | hypercholesterolemic acids |
| ORAC | oxygen radical absorbance capacity |
| OTC | over-the-counter |
| PCA | principal component analysis |
| PUFA | polyunsaturated fatty acids |
| ROS | reactive oxygen species |
| RSA | radical scavenging activity |
| SFA | saturated fatty acids |
| TI | thrombogenic index |
| TPTZ | 2,4,6-tris(2-pyridyl)-s-triazine |
| UFA | unsaturated fatty acids |
References
- FEDIAF. Facts & Figures. The European Pet Food Industry. 2024. Available online: https://europeanpetfood.org/wp-content/uploads/2024/06/FEDIAF-Facts-Figures-2022_Online100.pdf (accessed on 1 May 2024).
- FEDIAF. Nutritional Guidelines from Complete and Complementary Pet Food for Cats and Dogs. 2024. Available online: https://europeanpetfood.org/self-regulation/nutritional-guidelines/ (accessed on 1 May 2024).
- FEDIAF. Code of Good Labelling Practice for Pet Food. 2019. Available online: https://europeanpetfood.org/self-regulation/labelling/ (accessed on 1 May 2024).
- Regulation (EC) No 767/2009 of the European Parliament and of the Council of 13 July 2009 on the Placing on the Market and Use of Feed, Amending European Parliament and Council Regulation (EC) No 1831/2003 and Repealing Council Directive 79/373/EEC, Commission Directive 80/511/EEC, Council Directives 82/471/EEC, 83/228/EEC, 93/74/EEC, 93/113/EC and 96/25/EC and Commission Decision 2004/217/EC. Available online: https://eur-lex.europa.eu/eli/reg/2009/767/oj/eng (accessed on 1 May 2024).
- Regulation (EC) No 1831/2003 of the European Parliament and of the Council of 22 September 2003 on Additives for Use in Animal Nutrition. Available online: https://eur-lex.europa.eu/eli/reg/2003/1831/2021-03-27 (accessed on 1 May 2024).
- Fossati, L.A.; Larsen, J.A.; Villaverde, C.; Fascetti, A.J. Determination of mammalian DNA in commercial canine diets with uncommon and limited ingredients. Vet. Med. Sci. 2019, 5, 30–38. [Google Scholar] [CrossRef]
- Olatunde, G.A.; Atungulu, G.G. Emerging pet food drying and storage strategies to maintain safety. FFSA J 2018, 2018, 45–61. [Google Scholar]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 1541–4337. [Google Scholar] [CrossRef]
- Turek, J.J.; Watkins, B.A.; Schoenlein, I.A.; Allen, K.G.D.; Hayek, M.G.; Aldrich, C.G. Oxidized lipid depresses canine growth, immune function and bone formation. J. Nutr. Biochem. 2003, 4, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Glodde, F.; Günal, M.; Kinsel, M.E.; AbuGhazaleh, A. Effects of natural antioxidants on the stability of omega-3 fatty acids in dog food. J. Vet. Res. 2018, 62, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Morelli, G.; Stefanutti, D.; Ricci, R. A Survey among dog and cat owners on pet food storage and preservation in the households. Animals 2021, 11, 273. [Google Scholar] [CrossRef] [PubMed]
- Usuga, A.; Rojano, B.A.; Duque, J.C.; Mesa, C.; Restrepo, O.; Gomez, L.M.; Restrepo, G. Dry food affects the oxidative/antioxidant profile of dogs. Vet. Med. Sci. 2023, 9, 687–697. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Lipid oxidation and improving the oxidative stability. Chem. Soc. Rev. 2010, 39, 4067–4079. [Google Scholar] [CrossRef]
- Hołda, K.; Głogowski, R. Selected quality properties of lipid fraction and oxidative stability of dry dog foods under typical storage conditions. J. Therm. Anal. Calorim. 2016, 126, 91–96. [Google Scholar] [CrossRef]
- Błaszczyk, A.; Augustyniak, A.; Skolimowski, J. Ethoxyquin: An antioxidant used in animal feed. Int. J. Food Sci. 2013, 2013, 585931. [Google Scholar] [CrossRef]
- Corsato Alvarenga, I.; Keller, L.C.; Waldy, C.; Aldrich, C.G. Extrusion processing modifications of a dog kibble at large scale alter levels of starch available to animal enzymatic digestion. Foods 2021, 10, 2526. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP); Bampidis, V.; Azimonti, G.; Bastos, M.L.; Christensen, H.; Dusemund, B.; Fašmon Durjava, M.; Kouba, M.; López-Alonso, M.; López Puente, S.; et al. Safety and efficacy of a feed additive consisting of butylated hydroxytoluene (BHT) for all animal species (Lanxess Deutschland GmbH). EFSA J. 2022, 20, e07286. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP); Bampidis, V.; Azimonti, G.; Bastos, M.L.; Christensen, H.; Dusemund, B.; Kouba, M.; Kos Durjava, M.; López-Alonso, M.; López Puente, S.; et al. Scientific opinion on the safety of butylated hydroxy anisole (BHA) for all animal species. EFSA J. 2019, 17, 5913. [Google Scholar]
- Hegab, D.; Mohammed, A.; Metwally, M.; Ghoneim, M.; Abou-Hadeed, A. Ethoxyquin and butylated hydroxyl toluene induced hepatotoxic effect via apoptosis, oxidative stress in rats: Tissue injury-related CYP1A1 gene expression. Zagazig Vet. J. 2021, 49, 456–470. [Google Scholar] [CrossRef]
- Bunghez, F.; Socaciu, C.; Catunescu, G.M. Antioxidants used in pet feed. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Agric. 2012, 69, 1843–5386. [Google Scholar] [CrossRef]
- Park, D.H.; Kothari, D.; Niu, K.M.; Han, S.G.; Yoon, J.E.; Lee, H.G.; Kim, S.K. Effect of fermented medicinal plants as dietary additives on food preference and fecal microbial quality in dogs. Animals 2019, 9, 690. [Google Scholar] [CrossRef]
- Casazza, A.A.; Pettinato, M.; Perego, P. Polyphenols from apple skins: A study on microwave-assisted extraction optimization and exhausted solid characterization. Sep. Purif. Technol. 2020, 240, 116640. [Google Scholar] [CrossRef]
- Doseděl, M.; Jirkovský, E.; Macáková, K.; Krčmová, L.K.; Javorská, L.; Pourová, J.; Mercolini, L.; Remião, F.; Nováková, L.; Mladěnka, P. Vitamin C-sources, physiological role, kinetics, deficiency, use, toxicity, and determination. Nutrients 2021, 13, 615. [Google Scholar] [CrossRef]
- Trela, A.; Szymańska, R. Less widespread plant oils as a good source of vitamin E. Food Chem. 2019, 296, 160–166. [Google Scholar] [CrossRef]
- Milgram, N.W.; Head, E.; Muggenburg, B.; Holowachuk, D.; Murphey, H.; Estrada, J.; Ikeda-Douglas, C.; Zicker, S.; Cotman, C. Landmark discrimination learning in the dog: Effects of age, an antioxidant fortified food, and cognitive strategy. Neurosci. Biobehav. Rev. 2002, 26, 679–695. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Wannenmacher, J.; Cotterchio, C.; Schlumberger, M.; Reuber, V.; Gastl, M.; Becker, T. Technological influence on sensory stability and antioxidant activity of beers measured by ORAC and FRAP. J. Sci. Food. Agric. 2019, 99, 6628–6637. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Dunmire, K.M.; Truelock, C.N.; Paulk, C.B.; Aldrich, G.; Li, Y. Antioxidant performances of corn gluten meal and DDGS protein hydrolysates in food, pet food, and feed systems. J. Agric. Food. Res. 2020, 2, 100030. [Google Scholar] [CrossRef]
- Dogu-Baykut, E.; Gunes, G.; Decker, E.A. Impact of shortwave ultraviolet (UV-C) radiation on the antioxidant activity of thyme (Thymus vulgaris L.). Food Chem. 2014, 157, 167–173. [Google Scholar] [CrossRef]
- Schlieck, T.M.M.; Petrolli, T.G.; Bissacotti, B.F.; Copetti, P.M.; Bottari, N.B.; Morsch, V.M.; da Silva, A.S. Addition of a blend of essential oils (cloves, rosemary and oregano) and vitamin E to replace conventional chemical antioxidants in dog feed: Effects on food quality and health of beagles. Arch. Anim. Nutr. 2021, 75, 389–403. [Google Scholar] [CrossRef]
- Vhangani, L.N.; Van Wyk, J. Antioxidant activity of Maillard reaction products (MRPs) in a lipid-rich model system. Food Chem. 2016, 208, 301–308. [Google Scholar] [CrossRef]
- Nooshkam, M.; Varidi, M. Antioxidant and antibrowning properties of Maillard reaction products in food and biological systems. Vitam. Horm. 2024, 125, 367–399. [Google Scholar]
- Bolchini, S.; Morozova, K.; Ferrentino, G. Assessing antioxidant properties of Maillard reaction products: Methods and potential applications as food preservatives. Eur. Food Res. Technol. 2025, 251, 1–21. [Google Scholar] [CrossRef]
- Rodrigues, R.B.A.; Zafalon, R.V.A.; Rentas, M.F.; Risolia, L.W.; Macedo, H.T.; Perini, M.P.; Silva, A.M.G.D.; Marchi, P.H.; Balieiro, J.C.C.; Mendes, W.S.; et al. The supplementation of docosahexaenoic acid-concentrated fish oil enhances cognitive function in puppies. Animals 2023, 13, 2938. [Google Scholar] [CrossRef]
- Tynes, V.V.; Landsberg, G.M. Nutritional management of behavior and brain disorders in dogs and cats. Vet. Clin. Small Anim. Pract. 2021, 51, 711–727. [Google Scholar] [CrossRef]
- Ahlstrøm, Ø.; Krogdahl, Å.; Vhile, S.G.; Skrede, A. Fatty acid composition in commercial dog foods. J. Nutr. 2004, 134, 2145S–2147S. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.A.; Stockman, J.; Li, X.; Wang, S.C. Fatty acid analysis and stability of selected edible oils used in homemade pet diets. J. Vet. Intern. Med. 2025, 39, e70119. [Google Scholar] [CrossRef] [PubMed]
- Kara, K. Effect of stocking conditions on fatty acid composition and oxidation capacities of different class and type dog food. Ital. J. Anim. Sci. 2021, 20, 1042–1053. [Google Scholar] [CrossRef]
- Esterbauer, H. Cytotoxicity and genotoxicity of lipid-oxidation products. Am. J. Clin. Nutr. 1993, 57, 779S–785S. [Google Scholar] [CrossRef]
- Phung, A.S.; Bannenberg, G.; Vigor, C.; Reversat, G.; Oger, C.; Roumain, M.; Galano, J.-M.; Durand, T.; Muccioli, G.G.; Ismail, A. Chemical compositional changes in over-oxidized fish oils. Foods 2020, 9, 1501. [Google Scholar] [CrossRef]
- Sullivan, J.C.; Budge, S.M.; St-Onge, M. Modeling the primary oxidation in commercial fish oil preparations. Lipids 2011, 46, 87–93. [Google Scholar] [CrossRef]
- Liu, K.; Liu, Y.; Chen, F. Effect of storage temperature on lipid oxidation and changes in nutrient contents in peanuts. Food Sci. Nutr. 2019, 7, 2280–2290. [Google Scholar] [CrossRef]
- Macotpet, A.; Suksawat, F.; Sukon, P.; Pimpakdee, K.; Pattarapanwichien, E.; Tangrassameeprasert, R.; Boonsiri, P. Oxidative stress in cancer-bearing dogs assessed by measuring serum malondialdehyde. BMC Vet. Res. 2013, 9, 101. [Google Scholar] [CrossRef]
- Barrouin-Melo, S.M.; Anturaniemi, J.; Sankari, S.; Griinari, M.; Atroshi, F.; Ounjaijean, S.; Hielm-Björkman, A.K. Evaluating oxidative stress, serological- and haematological status of dogs suffering from osteoarthritis, after supplementing their diet with fish or corn oil. Lipids Health Dis. 2016, 15, 139. [Google Scholar] [CrossRef]
- Sechi, S.; Carta, S.; Correddu, F.; Di Cerbo, A.; Nudda, A.; Cocco, R. Effects of commercially available antioxidant-enriched fish- and chicken-based diets on biochemical parameters and blood fatty acid profile of old dogs. Animals 2022, 12, 1326. [Google Scholar] [CrossRef]
- Costa-Santos, K.; Damasceno, K.; Portela, R.D. Lipid and metabolic profiles in female dogs with mammary carcinoma receiving dietary fish oil supplementation. BMC Vet. Res. 2019, 15, 401. [Google Scholar] [CrossRef] [PubMed]
- Wright-Rodgers, A.S.; Waldron, M.K.; Bigley, K.E.; Lees, G.E.; Bauer, J.E. Dietary fatty acids alter plasma lipids and lipoprotein distributions in dogs during gestation, lactation, and the perinatal period. J. Nutr. 2005, 135, 2230–2235. [Google Scholar] [CrossRef]
- Moussavi Javardi, M.S.; Madani, Z.; Movahedi, A.; Karandish, M.; Abbasi, B. The correlation between dietary fat quality indices and lipid profile with atherogenic index of plasma in obese and non-obese volunteers: A cross-sectional descriptive-analytic case-control study. Lipids Health Dis. 2020, 19, 213. [Google Scholar] [CrossRef]
- ISO Method 6498; Animal Feeding Stuffs–Guidelines for Sample Preparation. International Organization for Standardization: Geneva, Switzerland, 2012. Available online: https://www.iso.org/standard/52285.html (accessed on 1 May 2024).
- Commission Regulation (EC) No 152/2009 of 27 January 2009 Laying Down the Methods of Sampling and Analysis for the Official Control of Feed. Available online: https://eur-lex.europa.eu/eli/reg/2009/152/oj/eng (accessed on 1 May 2024).
- AOAC. Official Methods of Analysis of the AOAC, 22nd ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2023; Available online: https://www.aoac.org/official-methods-of-analysis/ (accessed on 1 April 2024).
- Ulbricht, T.; Southgate, D. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- Swain, T.; Hillis, W.E. The phenolic constituents of Prunus domestica L. The quantity of analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Miliauskas, G.; Yenkutonis, P.R.; Van Beek, T.A. Screening of radical scavenging activity of some medicinal and aromatic plants extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- TIBCO. Statistica. 2023. Available online: https://docs.tibco.com/pub/stat/14.1.0/doc/pdf/TIB_stat_14.1.0_installation.pdf (accessed on 1 April 2024).





















| Dog Food | Dry Matter (g/100 g of Fresh Matter) | Crude Protein | Nitrogen-Free Extracts | Crude Ash | Crude Fiber | Metabolizable Energy |
|---|---|---|---|---|---|---|
| A | 94.17 abc | 29.68 b | 38.45 c | 6.91 a | 5.62 c | 398.2 a |
| B | 94.56 bc | 41.41 e | 27.19 a | 8.36 a | 4.19 ab | 382.6 a |
| C | 93.76 abc | 26.67 a | 47.04 d | 6.02 a | 6.09 c | 384.9 a |
| D | 91.42 a | 39.87 e | 30.04 ab | 7.80 a | 4.17 ab | 383.4 a |
| E | 92.19 ab | 34.44 c | 37.05 bc | 6.96 a | 5.21 bc | 384.8 a |
| F | 95.44 c | 36.47 d | 26.94 a | 9.45 a | 3.78 a | 388.4 a |
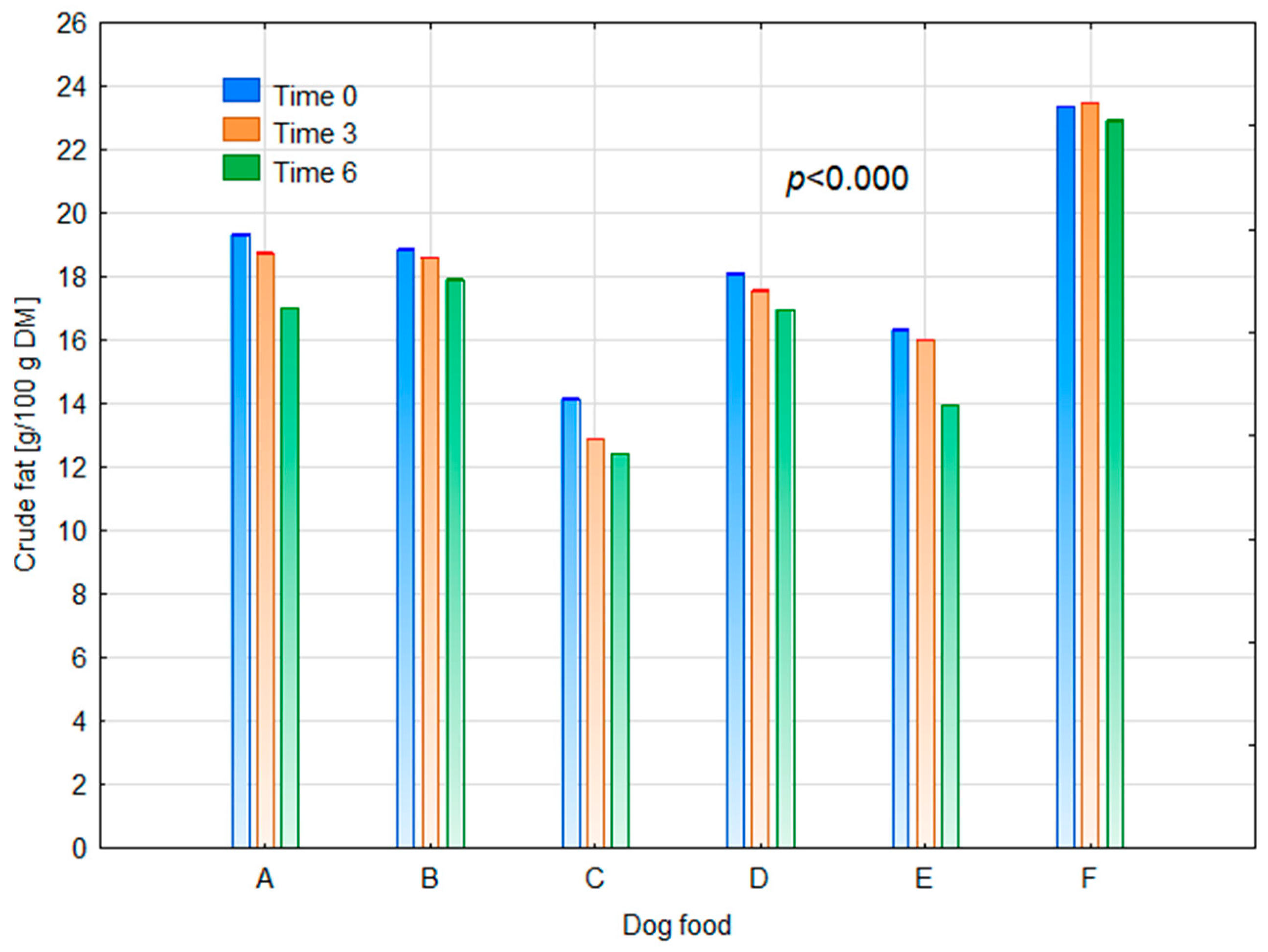
| Dog Food | Dry Matter (g/100 g of Fresh Matter) | Crude Fat (g/100 g of DM) |
|---|---|---|
| A | 94.17 abc | 19.35 e |
| B | 94.56 bc | 18.86 d |
| C | 93.76 abc | 14.17 a |
| D | 91.42 a | 18.12 c |
| E | 92.19 ab | 16.34 b |
| F | 95.44 c | 23.37 f |
| Time After Opening (Months) | ||
| 0 | 93.59 b | 18.37 c |
| 3 | 92.89 a | 17.88 b |
| 6 | 95.04 c | 16.87 a |
| Dog Food | C4:0 | C6:0 | C8:0 | C10:0 | C11:0 | C12:0 | C13:0 | C14:0 | C15:0 | C16:0 | C17:0 | C18:0 | C20:0 | C21:0 | C22:0 | C23:0 | C24:0 | SFA |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | 0.00 | 0.01 | 0.03 | 0.02 ab | 0.01 | 0.19 bc | 0.01 | 1.01 a | 0.15 a | 12.66 a | 0.31 bc | 5.97 d | 0.38 e | 0.00 | 0.12 | 0.22 b | 0.07 a | 21.14 a |
| B | 0.00 | 0.02 | 0.01 | 0.03 ab | 0.00 | 0.44 d | 0.05 | 1.82 c | 0.23 b | 23.96 e | 0.40 e | 7.89 e | 0.11 a | 0.00 | 0.14 | 0.32 c | 0.02 a | 35.45 d |
| C | 0.00 | 0.00 | 0.00 | 0.49 c | 0.01 | 25.47 e | 0.04 | 6.93 f | 0.21 b | 17.99 d | 0.17 a | 3.97 b | 0.12 a | 0.01 | 0.04 | 0.05 a | 0.03 a | 55.54 f |
| D | 0.00 | 0.00 | 0.05 | 0.01 a | 0.01 | 0.23 c | 0.02 | 4.08 e | 0.29 c | 14.62 b | 0.27 b | 3.69 a | 0.28 c | 0.00 | 0.00 | 0.27 bc | 3.56 d | 27.37 c |
| E | 0.01 | 0.01 | 0.01 | 0.04 ab | 0.01 | 0.15 ab | 0.01 | 1.58 b | 0.12 a | 24.12 f | 0.37 de | 13.30 f | 0.18 b | 0.00 | 0.00 | 0.29 bc | 0.25 b | 40.45 e |
| F | 0.00 | 0.01 | 0.02 | 0.06 b | 0.01 | 0.10 a | 0.01 | 2.42 d | 0.23 b | 14.76 c | 0.33 cd | 4.73 c | 0.31 d | 0.01 | 0.00 | 0.24 b | 1.75 c | 25.04 b |
| Time After Opening (Months) | ||||||||||||||||||
| 0 | 0.01 | 0.01 | 0.03 | 0.08 a | 0.01 | 2.91 a | 0.05 | 3.26 c | 0.32 b | 20.72 c | 0.42 b | 7.61 b | 0.19 a | 0.01 | 0.05 | 0.26 b | 0.62 a | 36.57 c |
| 3 | 0.00 | 0.01 | 0.02 | 0.12 b | 0.01 | 4.98 b | 0.01 | 2.79 a | 0.14 a | 16.75 b | 0.24 a | 6.13 a | 0.25 b | 0.02 | 0.04 | 0.25 b | 1.09 b | 32.83 a |
| 6 | 0.00 | 0.01 | 0.02 | 0.13 b | 0.01 | 5.40 c | 0.02 | 2.87 b | 0.16 a | 16.57 a | 0.27 a | 6.03 a | 0.26 b | 0.01 | 0.05 | 0.19 a | 1.13 c | 33.10 b |
| Dog Food | C14:1 (n-9) | C15:1 | C16:1 (n-9) | C17:1 (n-9) | C18:1 (n-9 cis) | C20:1 (n-9) | C22:1 (n-9) | C24:1 | C18:2 (n-6 trans) | C18:2 (n-6 cis) | C20:2 (n-9) | C22:2 (n-6) | C18:3 (n-3) | C18:3 (n-6) | C20:3 (n-3) | C20:3 (n-6) | C20:4 (n-6) | C20:5 (n-3) | C22:6 (n-3) | Unidentified |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | 0.05 b | 0.04 | 1.39 a | 0.18 ab | 53.79 f | 4.67 f | 0.05 | 0.04 | 0.07 | 16.92 d | 0.06 | 0.04 ab | 0.94 d | 0.16 | 0.02 | 0.22 c | 0.01 a | 0.03 b | 0.07 ab | 0.11 a |
| B | 0.17 d | 0.00 | 3.97 f | 0.23 c | 37.95 b | 1.66 c | 0.01 | 0.08 | 0.04 | 19.71 f | 0.03 | 0.01 a | 0.30 b | 0.05 | 0.07 | 0.09 b | 0.01 a | 0.01 a | 0.01 a | 0.16 ab |
| C | 0.09 c | 0.05 | 2.71 c | 0.17 a | 20.68 a | 2.99 d | 0.03 | 0.02 | 0.00 | 17.19 e | 0.01 | 0.06 b | 0.24 a | 0.01 | 0.02 | 0.03 a | 0.02 a | 0.04 b | 0.02 a | 0.10 a |
| D | 0.02 a | 0.14 | 3.45 e | 0.16 a | 41.37 d | 1.52 b | 6.11 | 0.24 | 0.06 | 7.37 a | 1.56 | 0.23 c | 5.51 f | 0.01 | 0.00 | 0.24 c | 0.09 c | 0.06 c | 3.50 d | 0.48 c |
| E | 0.02 a | 0.05 | 2.48 b | 0.21 bc | 39.66 c | 1.19 a | 0.05 | 0.02 | 0.03 | 13.79 c | 0.42 | 0.19 c | 0.78 c | 0.03 | 0.11 | 0.01 a | 0.07 b | 0.01 a | 0.11 b | 0.32 bc |
| F | 0.07 c | 0.09 | 3.07 d | 0.16 a | 45.19 e | 3.77 e | 2.28 | 0.19 | 0.06 | 12.91 b | 1.07 | 0.46 d | 3.43 e | 0.06 | 0.00 | 0.08 b | 0.25 d | 0.01 a | 0.97 c | 0.85 d |
| Time After Opening (Months) | ||||||||||||||||||||
| 0 | 0.11 c | 0.06 | 3.05 c | 0.21 b | 40.19 c | 2.45 a | 1.10 | 0.07 | 0.03 | 13.20 a | 0.38 | 0.10 a | 1.43 a | 0.04 | 0.04 | 0.09 a | 0.05 a | 0.03 a | 0.50 a | 0.32 ab |
| 3 | 0.06 b | 0.07 | 2.71 a | 0.15 a | 39.84 b | 2.70 b | 1.64 | 0.11 | 0.04 | 15.42 c | 0.58 | 0.16 b | 2.08 b | 0.05 | 0.04 | 0.14 c | 0.08 b | 0.03 a | 0.88 b | 0.41 b |
| 6 | 0.05 a | 0.06 | 2.78 b | 0.20 b | 39.28 a | 2.74 b | 1.78 | 0.12 | 0.05 | 15.32 b | 0.62 | 0.24 c | 2.09 b | 0.06 | 0.03 | 0.12 b | 0.09 c | 0.03 a | 0.97 c | 0.28 a |
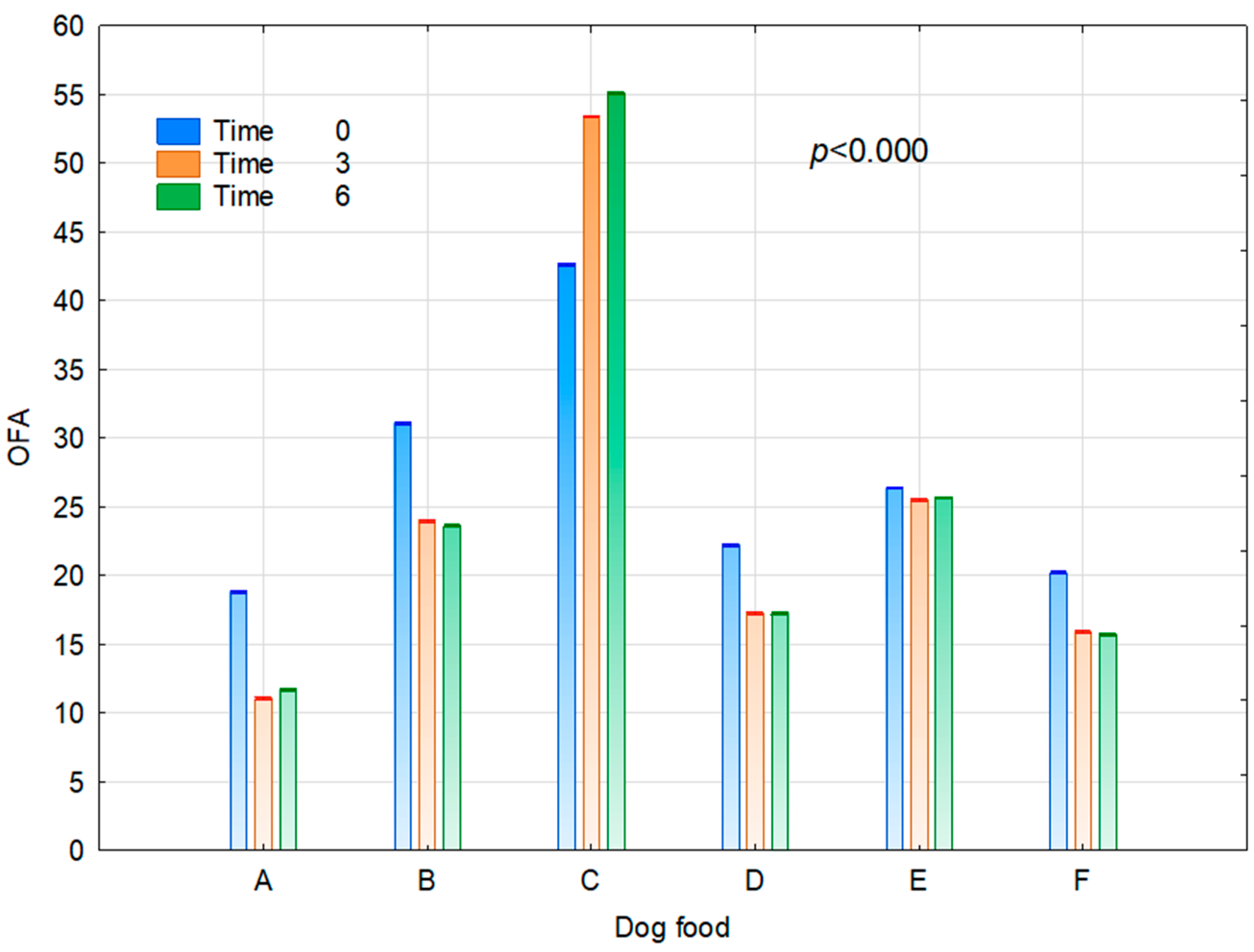
| Dog Food | MUFA | PUFA | n-3 | n-6 | n-6/n-3 | h/H | DFA | OFA | AI | TI |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 90.30 a | 27.78 a | 1.06 c | 17.38 d | 16.39 b | 5.55 f | 84.73 f | 13.85 a | 0.22 a | 3.75 b |
| B | 66.05 a | 30.49 a | 0.40 b | 19.89 e | 50.15 c | 2.26 c | 72.28 b | 26.23 e | 0.50 d | 2.29 a |
| C | 40.09 a | 26.44 a | 0.32 a | 17.25 d | 58.91 d | 0.78 a | 48.34 a | 50.39 f | 1.63 f | 2.28 a |
| D | 80.15 a | 26.19 a | 9.07 e | 7.77 a | 0.93 a | 2.91 d | 75.83 d | 18.93 c | 0.43 c | 29.3 f |
| E | 65.50 a | 23.28 a | 1.01 c | 13.93 c | 13.78 b | 2.10 b | 72.53 c | 25.85 d | 0.52 e | 4.47 c |
| F | 82.15 a | 28.45 a | 4.41 d | 13.35 b | 3.15 a | 3.62 e | 78.84 e | 17.28 b | 0.33 b | 14.19 d |
| Time After Opening (Months) | ||||||||||
| 0 | 70.81 a | 23.58 a | 1.99 a | 13.41 a | 18.11 a | 2.27 a | 70.72 a | 26.90 c | 0.61 b | 7.25 a |
| 3 | 70.86 a | 28.79 a | 3.02 b | 15.73 c | 29.53 c | 3.21 c | 72.89 c | 24.52 a | 0.59 a | 10.27 b |
| 6 | 70.44 a | 28.95 a | 3.12 c | 15.65 b | 24.01 b | 3.13 b | 72.65 b | 24.84 b | 0.61 b | 10.55 c |
| Item | MUFA | PUFA | n-6 | n-3 | n-6/n-3 | h/H | DFA | OFA | AI |
|---|---|---|---|---|---|---|---|---|---|
| PUFA | 0.76 p < 0.000 | 1.00 p = --- | - | - | - | - | - | - | - |
| n-6 | −0.17 p = 0.330 | 0.20 p = 0.240 | 1.00 p = --- | - | - | - | - | - | - |
| n-3 | 0.25 p = 0.146 | 0.03 p = 0.870 | −0.81 p < 0.000 | 1.00 p = --- | - | - | - | - | - |
| n-6/n-3 | −0.44 p = 0.007 | 0.10 p = 0.559 | 0.74 p < 0.000 | −0.63 p < 0.000 | 1.00 p = --- | - | - | - | - |
| h/H | 0.49 p = 0.002 | 0.13 p = 0.443 | 0.04 p = 0.832 | 0.19 p = 0.281 | −0.49 p = 0.002 | 1.00 p = --- | - | - | - |
| DEFA | 0.55 p = 0.001 | 0.09 p = 0.622 | -0.16 p = 0.364 | 0.32 p = 0.059 | −0.69 p < 0.000 | 0.84 p < 0.000 | 1.00 p = --- | - | - |
| OFA | −0.55 p < 0.000 | −0.08 p = 0.627 | 0.26 p = 0.134 | −0.43 p = 0.009 | 0.75 p < 0.000 | −0.82 p < 0.000 | −0.99 p < 0.000 | 1.00 p = --- | - |
| AI | −0.53 p = 0.001 | −0.07 p = 0.683 | 0.19 p = 0.277 | −0.32 p = 0.055 | 0.70 p < 0.000 | −0.74 p < 0.000 | −0.98 p < 0.000 | 0.98 p < 0.000 | 1.00 p = --- |
| TI | 0.23 p = 0.171 | 0.02 p = 0.901 | −0.82 p < 0.000 | 1.00 p < 0.000 | −0.62 p < 0.000 | 0.16 p = 0.366 | 0.29 p = 0.081 | −0.41 p = 0.014 | −0.30 p = 0.073 |
| Correlation | Regression Equations | r2 |
|---|---|---|
| MUFA vs. PUFA | PUFA = 9.427 + 0.250·MUFA | 0.59 |
| MUFA vs. n-6/n-3 | n-6/n-3 = 49.272 − 0.359·MUFA | 0.19 |
| MUFA vs. h/H | h/H = 0.938 + 0.027·MUFA | 0.24 |
| MUFA vs. DFA | DFA = 56.456 + 0.221·MUFA | 0.30 |
| MUFA vs. OFA | OFA = 42.024 − 0.235·MUFA | 0.31 |
| MUFA vs. AI | AI = 1.231 − 0.009·MUFA | 0.28 |
| n-6 vs. n-3 | n-3 = 12.207 − 0.636·n-6 | 0.66 |
| n-6 vs. n-6/n-3 | n-6/n-3 = −39.460 + 4.243·n-6 | 0.55 |
| n-6 vs. TI | TI = 38.925 − 1.981·n-6 | 0.67 |
| n-3 vs. n-6/n-3 | n-6/n-3 = 36.236 − 4.555·n-3 | 0.39 |
| n-3 vs. OFA | OFA = 29.790 − 1.612·n-3 | 0.18 |
| n-3 vs. TI | TI = 1.001 + 3.078·n-3 | 1.00 |
| n-6/n-3 vs. h/H | h/H = 3.666 − 0.033·n-6/n-3 | 0.24 |
| n-6/n-3 vs. DFA | DFA = 80.281 − 0.343·n-6/n-3 | 0.48 |
| n-6/n-3 vs. OFA | OFA = 16.184 + 0.387·n-6/n-3 | 0.56 |
| n-6/n-3 vs. AI | AI = 0.265 + 0.014·n-6/n-3 | 0.49 |
| n-6/n-3 vs. TI | TI = 15.611 − 0.262·n-6/n-3 | 0.38 |
| h/H vs. DFA | DFA = 54.412 + 6.161·h/H | 0.71 |
| h/H vs. OFA | OFA = 43.495 − 6.301·h/H | 0.68 |
| h/H vs. AI | AI = 1.247 − 0.224·h/H | 0.55 |
| DFA vs. OFA | OFA = 10.44 − 1.041·DFA | 0.98 |
| DFA vs. AI | AI = 3.544 − 0.041·DFA | 0.97 |
| OFA vs. AI | AI = −0.378 + 0.039·OFA | 0.96 |
| OFA vs. TI | TI = 17.783 − 0.332·OFA | 0.17 |
| Dog Food | Polyphenols (mg CAE/100 g DM) | Polyphenols (mg GAE/100 g DM) | DPPH (µM Trolox/1 g DM) | FRAP (µM Trolox/1 g DM) | ABTS (µM Trolox/1 g DM) | RSA ABTS (%) |
|---|---|---|---|---|---|---|
| A | 276.10 b | 137.20 b | 6.25 b | 29.76 d | 15.77 bc | 53.74 e |
| B | 196.30 a | 97.60 a | 4.57 a | 23.99 a | 10.31 a | 46.65 a |
| C | 359.90 d | 178.90 d | 7.67 cd | 32.78 e | 15.21 b | 51.83 b |
| D | 350.00 d | 17400 d | 6.70 bc | 26.19 b | 17.38 d | 53.72 d |
| E | 293.40 c | 145.80 c | 7.94 d | 27.93 c | 17.00 cd | 53.53 c |
| F | 1049.60 e | 521.70 e | 1202 e | 38.30 f | 22.58 e | 60.15 f |
| Time After Opening (Months) | ||||||
| 0 | 382.30 b | 190.00 b | 9.73 c | 47.40 c | 16.29 b | 45.57 a |
| 3 | 361.00 a | 179.40 a | 5.78 a | 18.48 a | 14.09 a | 53.51 b |
| 6 | 519.40 c | 258.20 c | 7.07 b | 23.59 b | 18.74 c | 60.73 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kępińska-Pacelik, J.; Biel, W.; Witkowicz, R.; Micek, P.; Piątkowska, E.; Patla, A. Changes in the Fatty Acid Composition and Antioxidant Properties in Mono-Protein Commercial Dry Dog Foods During Storage. Molecules 2025, 30, 3524. https://doi.org/10.3390/molecules30173524
Kępińska-Pacelik J, Biel W, Witkowicz R, Micek P, Piątkowska E, Patla A. Changes in the Fatty Acid Composition and Antioxidant Properties in Mono-Protein Commercial Dry Dog Foods During Storage. Molecules. 2025; 30(17):3524. https://doi.org/10.3390/molecules30173524
Chicago/Turabian StyleKępińska-Pacelik, Jagoda, Wioletta Biel, Robert Witkowicz, Piotr Micek, Ewa Piątkowska, and Aleksandra Patla. 2025. "Changes in the Fatty Acid Composition and Antioxidant Properties in Mono-Protein Commercial Dry Dog Foods During Storage" Molecules 30, no. 17: 3524. https://doi.org/10.3390/molecules30173524
APA StyleKępińska-Pacelik, J., Biel, W., Witkowicz, R., Micek, P., Piątkowska, E., & Patla, A. (2025). Changes in the Fatty Acid Composition and Antioxidant Properties in Mono-Protein Commercial Dry Dog Foods During Storage. Molecules, 30(17), 3524. https://doi.org/10.3390/molecules30173524









