A Hydrophilic Copper–Viologen Hybrid Exhibiting High Degradation Efficiency on Commercial Dye in Maritime Accidents
Abstract
1. Introduction
2. Results and Discussion
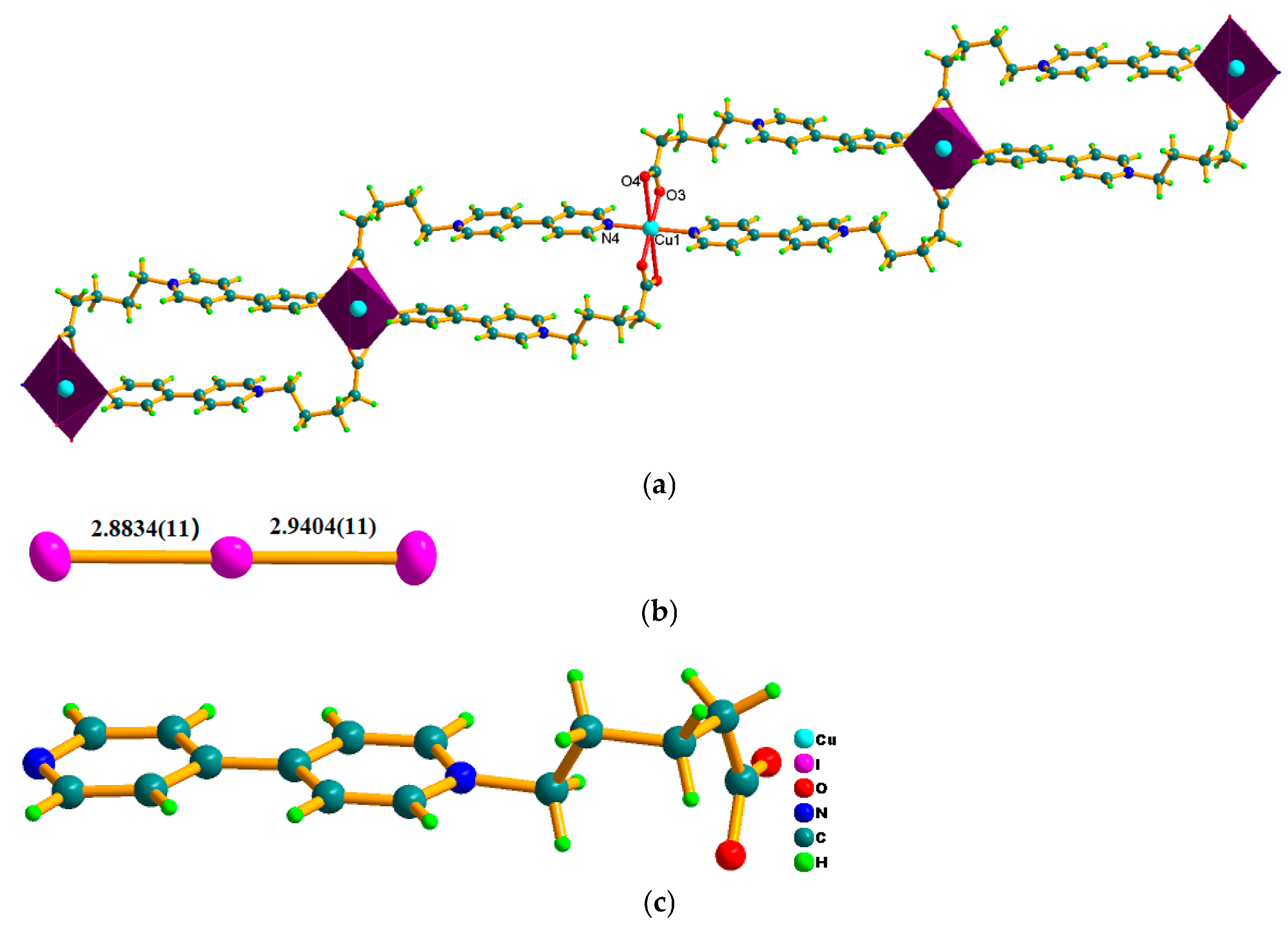
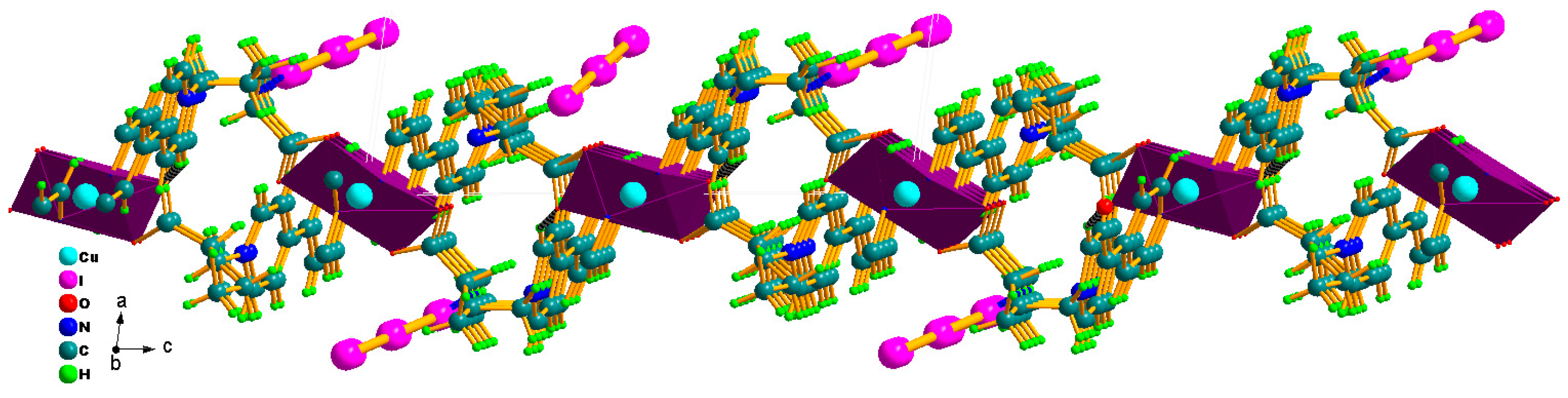
2.1. Structural Characterization of Photocatalyst
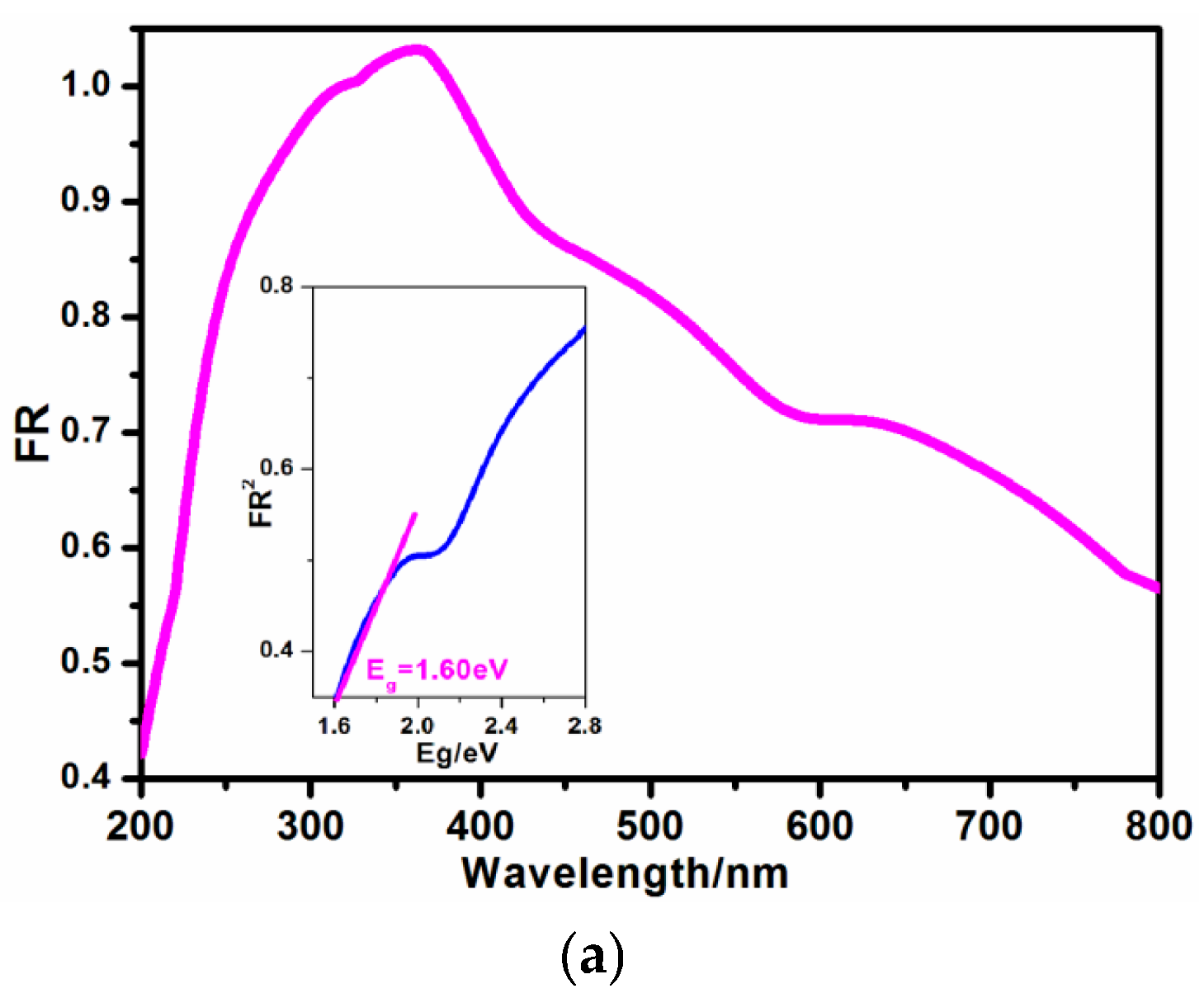
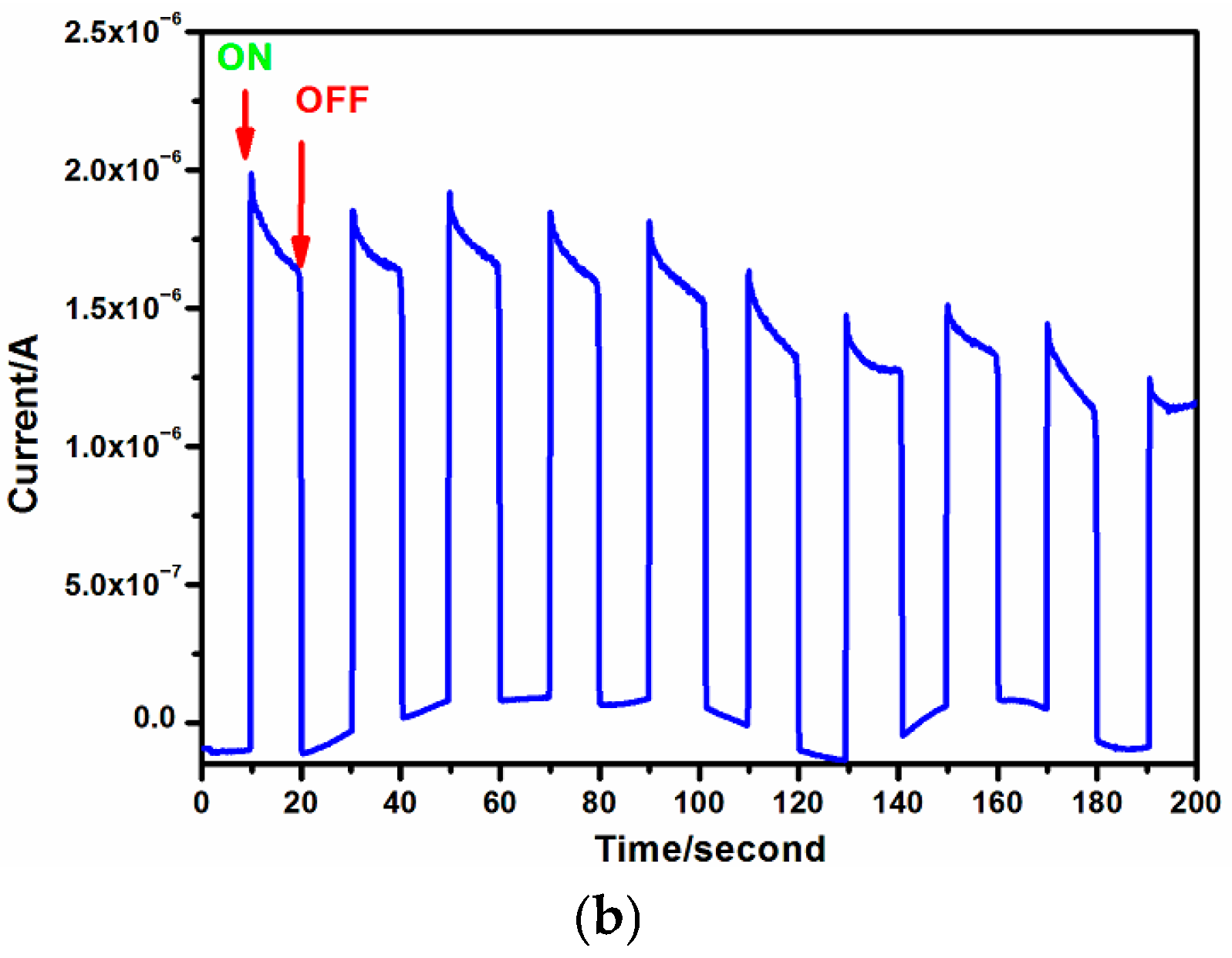
2.2. Visible Absorption Behavior and Transient Photocurrent Response
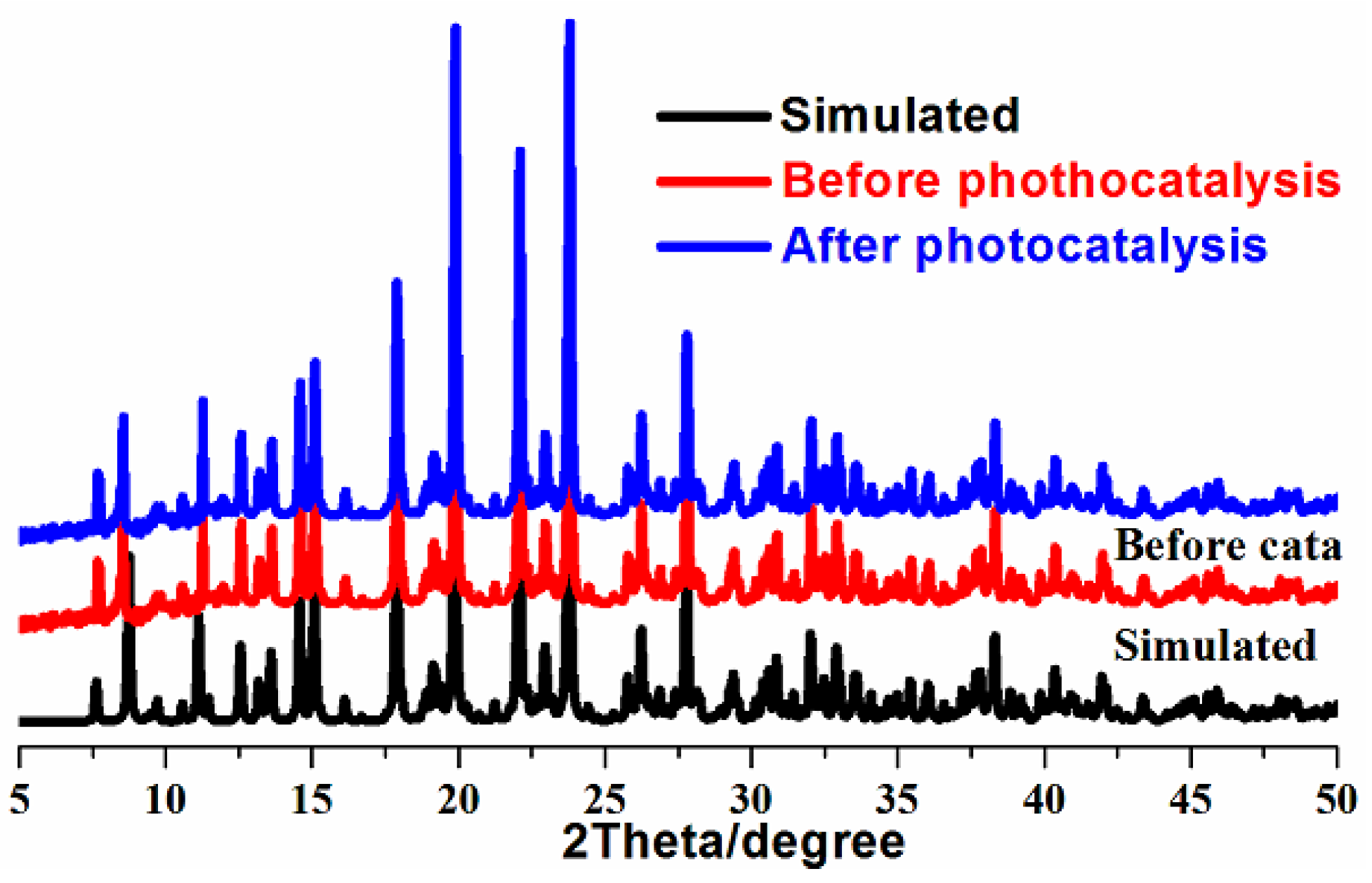
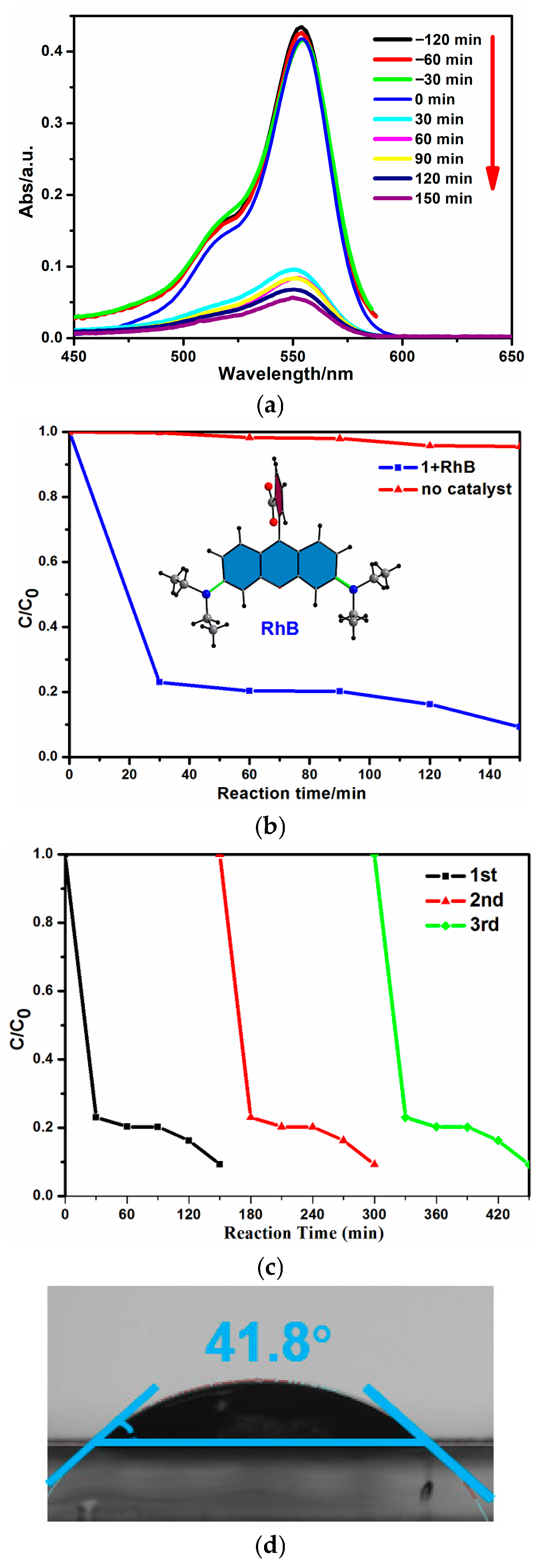
2.3. Photocatalytic Degradation of RhB in Seawater
3. Experiment
3.1. Materials and Methods
3.2. Synthesis of Photocatalyst {[Cu(BPA)2]·2I3}n (1)
3.3. X-Ray Crystallography
3.4. Photocurrent Measurements
3.5. Photodegradation of RhB
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Shabbir, S.; Faheem, M.; Ali, N.; Kerr, P.G.; Wu, Y. Evaluating role of immobilized periphyton in bioremediation of azo dye amaranth. Bioresour. Technol. 2017, 225, 395–401. [Google Scholar] [CrossRef]
- Wang, X.P.; Shang, H.Y.; Liu, X.D.; Bai, Y.J. Fuzzy assessment methodology for emergency response capability in hazardous materials transportation. Inter. J. Innov. Comput. Info. Contr. 2008, 4, 2689–2696. [Google Scholar]
- Maruthapandi, M.; Luong, J.H.T.; Gedanken, A. Kinetic, isotherm and mechanism studies of organic dye adsorption on poly(4,4′-oxybisbenzenamine) and copolymer of poly(4,4′-oxybisbenzenamine-pyrrole) macro-nanoparticles synthesized by multifunctional carbon dots. New J. Chem. 2019, 43, 1926–1935. [Google Scholar] [CrossRef]
- Maryanti, S.A.; Suciati, S.; Wahyuni, E.S.; Santoso, S.; Wiyasa, I.W.A. Rhodamine B triggers ovarian toxicity through oxidative stress, decreases in the number of follicles, 17B-estradiol level, and thickness of endometrium. Cukurova. Med. J. 2014, 39, 451–457. [Google Scholar]
- Robi, S.D.; Jian, Z.; Md, M.; Benjamin, J.C.; Yue, Z.B.; Hareem, K.; Nitu, S.; Ali, Z.; Farjana, H.; Torben, D.; et al. Two dimensional PbMoO4: A photocatalytic material derived from a naturally non-layered crystal. Nano Energ. 2018, 49, 237–246. [Google Scholar]
- Horzum, N.; Hilal, M.E.; Isık, T. Enhanced bactericidal and photocatalytic activities of ZnO nanostructures by changing the cooling route. New J. Chem. 2018, 42, 11831–11838. [Google Scholar] [CrossRef]
- Sudha, D.; Sivakumar, P. Review on the photocatalytic activity of various composite catalysts. Chem. Eng. Process. 2015, 97, 112–133. [Google Scholar] [CrossRef]
- Meng, X.C.; Zhang, Z.S. Bismuth-based photocatalytic semiconductors: Introduction, challenges and possible approaches. J. Mol. Catal. A Chem. 2016, 423, 533–549. [Google Scholar] [CrossRef]
- Samsudin, M.F.R.; Sufian, S.; Hameed, B.H. Epigrammatic progress and perspective on the photocatalytic properties of BiVO4-based photocatalyst in photocatalytic water treatment technology: A review. J. Mol. Liq. 2018, 268, 438–459. [Google Scholar] [CrossRef]
- Zhang, N.; Yang, M.Q.; Liu, S.Q.; Sun, Y.G.; Xu, Y.J. Waltzing with the Versatile Platform of Graphene to Synthesize Composite Photocatalysts. Chem. Rev. 2015, 115, 10307–10377. [Google Scholar] [CrossRef]
- Mamba, G.; Mishra, A.K. Graphitic carbon nitride (g-C3N4) nanocomposites: A new and exciting generation of visible light driven photocatalysts for environmental pollution remediation. Appl. Catal. B 2016, 198, 347–377. [Google Scholar] [CrossRef]
- Zhao, L.M.; Zhang, W.T.; Song, K.Y.; Wu, Q.Q.; Li, Y.; Li, H.H.; Chen, Z.R. Lead-carboxylate/polyiodide hybrids constructing from halogen bonding and asymmetric viologen: Structures, visible-light-driven photocatalytic properties and enhanced photocurrent responses. CrystEngComm 2018, 20, 2245–2252. [Google Scholar] [CrossRef]
- Wang, D.H.; Lin, X.Y.; Wang, Y.K.; Zhang, W.T.; Song, K.Y.; Lin, H.; Li, H.H.; Chen, Z.R. A new iodiplumbate-based hybird constructed from asymmetric viologen and polyiodides: Structure, properties and photocatalytic aactivity for the degradation of organic dye. Chin. J. Struct. Chem. 2017, 36, 2000–2006. [Google Scholar]
- Luo, J.; Li, W.; Yin, R.; Liu, Q.; Xin, X.; Yang, L.; He, K.; Ma, D.; Lv, S.; Xing, D. Photocatalyst degradation of perfluorooctanoic acid in water: Mechanisms, approaches, and perspectives. Sep. Purif. Technol. 2024, 338, 126503. [Google Scholar] [CrossRef]
- Jiang, Y.; Qu, F.; Tian, L.; Yang, X.; Zou, Z.; Lin, Z. Self-assembled g-C3N4 nanoarchitectures with boosted photocatalytic solar-to-hydrogen efficiency. Appl. Surf. Sci. 2019, 487, 59–67. [Google Scholar] [CrossRef]
- Li, H.H.; Zeng, X.H.; Wu, H.Y.; Jie, X.; Zheng, S.T.; Chen, Z.R. Incorporating guest molecules into honeycomb structures constructed from uranium(VI)-polycarboxylates: Structural diversities and photocatalytic activities for the degradation of organic dye. Cryst. Growth Des. 2015, 15, 10–13. [Google Scholar] [CrossRef]
- Kumari, P.; Panda, T. Role of N-rich coordination environment in metal−organic frameworks for enhanced photocatalytic dye degradation. Cryst. Growth Des. 2024, 24, 4493–4500. [Google Scholar] [CrossRef]
- Wang, Q.; Cai, C.; Wang, M.; Guo, Q.; Wang, B.; Luo, W.; Wang, Y.; Zhang, C.; Zhou, L.; Zhang, D.; et al. Efficient photocatalytic degradation of malachite green in seawater by the hybrid of zinc-oxide nanorods grown on three-dimensional (3D) reduced graphene oxide(RGO)/Ni foam. Materials 2018, 11, 1004. [Google Scholar] [CrossRef]
- Chen, P.; Hu, X.; Qi, Y.; Wang, X.; Li, Z.; Zhao, L.; Liu, S.; Cui, C. Rapid degradation of azo dyes by melt-spun Mg-Zn-Ca metallic glass in artificial seawater. Metals 2017, 7, 485–491. [Google Scholar] [CrossRef]
- Makita, M.; Harata, A. Photocatalytic decolorization of rhodamine B dye as a model of dissolved organic compounds: Influence of dissolved inorganic chloride salts in seawater of the sea of Japan. Chem. Eng. Process. 2008, 47, 859–863. [Google Scholar] [CrossRef]
- Zhuang, X.Q.; Wu, Q.Q.; Huang, X.H.; Li, H.H.; Lin, T.J.; Gao, Y.L. A three-component hybrid templated by asymmetric viologen exhibiting visible-light-driven photocatalytic degradation on dye pollutant in maritime accident seawater. Catalysts 2021, 11, 640. [Google Scholar] [CrossRef]
- Chen, H.Y.; Zahraa, O.; Bouchy, M. Inhibition of the adsorption and photocatalytic degradation of an organic contaminant in an aqueous suspension of TiO2 by inorganic ions. J. Photochem. Photobiol. A 1997, 108, 37–44. [Google Scholar] [CrossRef]
- Li, Z.H.; Li, M.; Xu, T.Y.; Zhao, B.T. A viologen-derived luminescent material exhibiting photochromism, photocontrolled luminescence and selective detection of Cr2O72− in aqueous solution. Spectrochim. Acta A 2024, 306, 123579. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.; Pu, S.; Xiao, Q.; Liu, G.; Cui, S. Synthesis and photochromism of a novel water-soluble diarylethene with glucosyltriazolyl groups. Tetra. Lett. 2013, 54, 474–477. [Google Scholar] [CrossRef]
- Patel, E.N.; Arthur, R.B.; Nicholas, A.D.; Reinheimer, E.W.; Omary, M.A.; Brichacek, M.; Patterson, H.H. Synthesis, structure and photophysical properties of a 2D network with gold dicyanide donors coordinated to aza[5]helicene viologen acceptors. Dalton Trans. 2019, 48, 10288–10297. [Google Scholar] [CrossRef]
- Karmakar, S.; Panda, B.; Sahoo, B.; Routray, K.L.; Varma, S.; Behera, D. A Study on optical and dielectric properties of Ni-ZnO nanocomposite. Mater. Sci. Semicond. Process. 2018, 88, 198–206. [Google Scholar] [CrossRef]
- Zhou, X.J.; Chen, C.; Ren, C.X.; Sun, J.K.; Zhang, J. Tunable solid-state photoluminescence based on proton-triggered structural transformation of 4,4′-bipyridinium derivative. J. Mater. Chem. C 2013, 1, 744–750. [Google Scholar] [CrossRef]
- Wang, C.; Wang, S.J.; Kong, F.G. Calixarene-protected titanium-oxo clusters and their photocurrent responses and photocatalytic performances. Inorg. Chem. 2021, 60, 5034–5041. [Google Scholar] [CrossRef]
- Alhasan, H.S.; Omran, A.R.; Mahmud, A.A.; Mady, A.H.; Thalji, M.R. Toxic congo red dye photodegradation employing green synthesis of zinc oxide nanoparticles using gum arabic. Water 2024, 16, 2202. [Google Scholar] [CrossRef]
- El-Sabban, H.A.; Mady, A.H.; Diab, M.A.; Zoubi, W.A.; Kang, J.H.; Ko, Y.G. Novel environmentally-friendly 0D/3D TiO2@FeMoO4 binary composite with boosted photocatalytic performance: Structural analysis and Z-scheme interpretations. J. Alloys Compd. 2025, 1010, 177265. [Google Scholar] [CrossRef]
- Li, J.; Yang, F.; Zhou, Q.; Ren, R.; Wu, L.; Lv, Y. A regularly combined magnetic 3D hierarchical Fe3O4/BiOBr heterostructure: Fabrication, visible-light photocatalytic activity and degradation mechanism. J. Colloid Interface Sci. 2019, 546, 139–151. [Google Scholar] [CrossRef]
- Wendlandt, W.M.; Hecht, H.G. Reflectance Spectroscopy; Interscience: New York, NY, USA, 1966. [Google Scholar]
- Sheldrick, G.M. SHELXS-97: Program for Crystal Structure Solution; Göttingen University: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. SHELXL-97: Program for the Refinement of Crystal Structures; Göttingen University: Göttingen, Germany, 1997. [Google Scholar]
- Lin, X.Y.; Zhao, L.M.; Wang, D.H.; Wang, Y.K.; Li, M.; Li, H.H.; Chen, Z.R. Structural diversities of squarate-based complexes: Photocurrent responses and thermochromic behaviours enchanced by viologens. Inorg. Chem. Front. 2018, 5, 189–199. [Google Scholar] [CrossRef]
- Bailey, R.A.; Clark, H.M.; Ferris, J.P.; Krause, S.; Strong, R.L. 10-The environmental chemistry of some important elements. In Chemistry of the Environment; Academic Press: San Diego, CA, USA, 2002; pp. 347–414. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Y.; Hu, C.; Huang, X.; Li, H.; Lou, T.; Zhuang, X. A Hydrophilic Copper–Viologen Hybrid Exhibiting High Degradation Efficiency on Commercial Dye in Maritime Accidents. Molecules 2025, 30, 3525. https://doi.org/10.3390/molecules30173525
Gao Y, Hu C, Huang X, Li H, Lou T, Zhuang X. A Hydrophilic Copper–Viologen Hybrid Exhibiting High Degradation Efficiency on Commercial Dye in Maritime Accidents. Molecules. 2025; 30(17):3525. https://doi.org/10.3390/molecules30173525
Chicago/Turabian StyleGao, Yali, Chaojian Hu, Xihe Huang, Haohong Li, Tong Lou, and Xueqiang Zhuang. 2025. "A Hydrophilic Copper–Viologen Hybrid Exhibiting High Degradation Efficiency on Commercial Dye in Maritime Accidents" Molecules 30, no. 17: 3525. https://doi.org/10.3390/molecules30173525
APA StyleGao, Y., Hu, C., Huang, X., Li, H., Lou, T., & Zhuang, X. (2025). A Hydrophilic Copper–Viologen Hybrid Exhibiting High Degradation Efficiency on Commercial Dye in Maritime Accidents. Molecules, 30(17), 3525. https://doi.org/10.3390/molecules30173525





