Methods and Applications of Lanthanide/Transition Metal Ion-Doped Luminescent Materials
Abstract
1. Introduction
- (1).
- Photoinduced excitation (most common)
- (2).
- Electro-excitation
- (3).
- Cathodic ray excitation
- (4).
- Thermal excitation (thermoluminescence)
- (5).
- Chemical excitation (chemiluminescence)
- (1).
- 4f electronic configuration: The 4f electron number of rare earth ions varies from 0 (La3+) to 14 (Lu3+), resulting in a complex and diverse energy level structure. The angular quantum number l of the 4f orbital is 3, and the magnetic quantum number m can range from −3 to +3, with a total of seven orbitals and a maximum capacity of 14 electrons.
- (2).
- Energy level splitting: The 4f energy level of a free rare earth ion splits into multiple energy levels (such as 2S + 1L-J) due to spin–orbit coupling. In crystals, the crystal field further causes energy level splitting, but due to the shielding of 4f electrons, the degree of splitting is relatively small (usually several hundred wavenumbers).
- (1).
- f-f transition: Mechanism: 4f electrons transition within the same shell (Δl = 0). Electric dipole transitions are originally forbidden, but due to the symmetry breaking of the crystal field or mixing with configurations of opposite parity, the transition is allowed. Characteristics: The spectrum is sharp and linear, with a high color purity. Low transition probability and long excited state lifetime (in milliseconds) are called metastable states. The emission wavelength is determined by the rare earth ions themselves and is less affected by the external environment.
- (2).
- f-d transition: Mechanism: 4f electrons transition to 5d orbitals (Δl = 1), allowing for electric dipole transitions. It is commonly found in low-valence rare earth ions (such as Ce2+, Eu2+, and Yb2+). Characteristics: The spectrum exhibits broadband, high intensity, and a short fluorescence lifetime (nanosecond level). The emission wavelength is significantly affected by the crystal field.
- (3).
- Charge transfer band (CTS): Electrons migrate from the molecular orbitals of ligands (such as O2−) to the 4f orbitals of rare earth ions, forming a broad band absorption. It is commonly used to enhance the light absorption efficiency of rare earth ions.
- (1).
- LED lighting and display
- (2).
- Imaging and visualization
- (3).
- Security and anti-counterfeiting measures
2. Technology and Methods of Lanthanide/Transition Metal Ion-Doped Luminescent Materials
2.1. Preparation of Luminescent Materials by the High-Temperature Solid-State Method
2.2. Preparation of Luminescent Materials by the Sol-Gel Method
2.3. Hydrothermal Synthesis of Transition Metal- and Lanthanide-Doped Luminescent Materials
2.4. Brief Summary
- Low-temperature nanoparticle synthesis
- 2.
- Development of Multi-sulfur Oxide Matrix
- 3.
- Optimization of fluoride stability
3. Application of Lanthanide/Transition Metal Ion-Doped Luminescent Materials
3.1. Application of Luminescent Materials in Solar Cells
3.1.1. Crystal Silicon Solar Cell
3.1.2. Perovskite Solar Cells
3.2. Application of Luminescent Materials in Night Vision Devices
3.3. Biological Imaging and Biosensing
3.3.1. Luminescent Transition Metal Complexes as Probes for Cell Viability
3.3.2. Luminescent Transition Metal Complexes as Probes for Bacterial Cells
3.3.3. Luminescent Transition Metal Complexes as Probes for Microenvironment
3.4. Non-Contact Temperature Measurement
3.4.1. Reading Based on the Luminous Intensity Ratio
3.4.2. Temperature Sensing Through Luminescence Lifetime
3.4.3. Optical Temperature Measurement Based on Temperature-Induced Spectroscopy
3.5. Anti-Counterfeiting Applications
4. Conclusions and Outlook
4.1. Advantages of Rare Earth and Transition Metal-Doped Luminescent Materials
4.1.1. Excellent Optical Performance
4.1.2. Near-Infrared Luminescence Characteristics
4.1.3. High Conversion Efficiency and Long Lifespan
4.1.4. Multiple Application Scenarios
4.2. Challenges Faced by Rare Earth and Transition Metal-Doped Luminescent Materials
4.2.1. High Preparation Cost
4.2.2. Shortage of Resources
4.2.3. Toxicity of Elements
4.2.4. Color Saturation Is Not as Good as Direct Luminescence
4.3. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Tiwari, A.; Dhoble, S.J. Tunable lanthanide/transition metal ion-doped novel phosphors for possible application in w-LEDs: A review. Luminescence 2020, 35, 4–33. [Google Scholar] [CrossRef]
- Liu, J.; Kaczmarek, A.M.; Deun, R.V. Advances in tailoring luminescent rare-earth mixed inorganic materials. Chem. Soc. Rev. 2018, 47, 7225–7238. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, T.; Chen, G. Recent progress in lanthanide ions doped inorganic metal halide perovskites. J. Rare Earths 2024, 42, 237–250. [Google Scholar] [CrossRef]
- Ansari, A.A.; Parchur, A.K.; Nazeeruddin, M.K.; Tavakoli, M.M. Luminescent lanthanide nanocomposites in thermometry: Chemistry of dopant ions and host matrices. Coord. Chem. Rev. 2021, 444, 214040. [Google Scholar] [CrossRef]
- Forde, A.; Inerbaev, T.; Hobbie, E.K.; Kilin, D.S. Excited State Dynamics of a CsPbBr3 Nanocrystal Terminated with Binary Ligands: Sparse Density of States with Giant Spin-Orbit Coupling Suppresses Carrier Cooling. J. Am. Chem. Soc. 2019, 141, 4388–4397. [Google Scholar] [CrossRef] [PubMed]
- Erol, E.; Vahedigharehchopogh, N.; Kıbrıslı, O.; Ersundu, M.Ç.; Ersundu, A.E. Recent progress in lanthanide-doped luminescent glasses for solid-state lighting applications—A review. J. Phys. Condens. Matter 2021, 33, 483001. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Jiang, Y.; Sun, T.; Zhao, J.; Huang, B.; Peng, D.; Wang, F. Mechanically Excited Multicolor Luminescence in Lanthanide Ions. Adv. Mater. 2018, 31, 1807062. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Han, S.; Li, C.; Tu, D.; Chen, X. Lanthanide-Doped Inorganic Nanoprobes for Luminescent Assays of Biomarkers. Acc. Mater. Res. 2023, 4, 193–204. [Google Scholar] [CrossRef]
- Hou, Z.; Li, G.; Lian, H.; Lin, J. One-dimensional luminescent materials derived from the electrospinning process: Preparation, characteristics and application. J. Mater. Chem. 2012, 22, 5254–5276. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, H.; Qian, J.; Li, Z.; Hu, X.; Guo, Y.; Zhou, L. Rare Earth Ion Doped Luminescent Materials: A Review of Up/Down Conversion Luminescent Mechanism, Synthesis, and Anti-Counterfeiting Application. Photonics 2023, 10, 1014. [Google Scholar] [CrossRef]
- Zhang, C.; Lin, J. Defect-related luminescent materials: Synthesis, emission properties and applications. Chem. Soc. Rev. 2012, 41, 7938–7961. [Google Scholar] [CrossRef]
- Xu, Y.; Wu, M.; Liu, Y.; Feng, X.Z.; Yin, X.B.; He, X.W.; Zhang, Y.K. Nitrogen-Doped Carbon Dots: A Facile and General Preparation Method, Photoluminescence Investigation, and Imaging Applications. Chem.-A Eur. J. 2013, 19, 2276–2283. [Google Scholar] [CrossRef]
- Zeng, S.; Yi, Z.; Lu, W.; Qian, C.; Wang, H.; Rao, L.; Hao, J. Simultaneous realization of phase/size manipulation, upconversion luminescence enhancement, and blood vessel imaging in multifunctional nanoprobes through transition metal Mn2+ doping. Adv. Funct. Mater. 2014, 24, 4051–4059. [Google Scholar] [CrossRef]
- Bazhukova, I.N.; Pustovarov, V.A.; Myshkina, A.V.; Ulitko, M.V. Luminescent Nanomaterials Doped with Rare Earth Ions and Prospects for Their Biomedical Applications (A Review). Opt. Spectrosc. 2020, 128, 2050–2068. [Google Scholar] [CrossRef]
- Zheng, B.; Fan, J.; Chen, B.; Qin, X.; Wang, J.; Wang, F.; Liu, X. Rare-Earth Doping in Nanostructured Inorganic Materials. Chem. Rev. 2022, 122, 5519–5603. [Google Scholar] [CrossRef]
- Liu, D.; Dang, P.; Zhang, G.; Lian, H.; Li, G.; Lin, J. Near-infrared emitting metal halide materials: Luminescence design and applications. InfoMat 2024, 6, e12542. [Google Scholar] [CrossRef]
- Zhao, F.; Song, Z.; Liu, Q. Advances in Fe3+-activated luminescent materials for near-infrared light sources. Prog. Solid State Chem. 2024, 74, 100456. [Google Scholar] [CrossRef]
- Lin, J.; Zhou, L.; Ren, L.; Shen, Y.; Chen, Y.; Fu, J.; Xu, S. Broadband near-infrared emitting Sr3Sc4O9: Cr3+ phosphors: Luminescence properties application in light-emitting diodes. J. Alloys Compd. 2022, 908, 164582. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, L.; Shi, L.; Zhang, D. Near-Infrared Lanthanide Luminescence for Functional Materials. Prog. Chem. 2011, 23, 153–164. [Google Scholar]
- Li, G.; Lin, J. Recent progress in low-voltage cathodoluminescent materials: Synthesis, improvement and emission properties. Chem. Soc. Rev. 2014, 43, 7099–7131. [Google Scholar] [CrossRef]
- Zhou, N.; Wang, D.; Bao, Y.; Zhu, R.; Yang, P.; Song, L. A Review of Perovskite Nanocrystal Applications in Luminescent Solar Concentrators. Adv. Opt. Mater. 2023, 11, 2202681. [Google Scholar] [CrossRef]
- Li, L.; Chen, J.; Zhang, Q.; Yang, Z.; Sun, Y.; Zou, G. Methane dry reforming over activated carbon supported Ni-catalysts prepared by solid phase synthesis. J. Clean. Prod. 2020, 274, 122256. [Google Scholar] [CrossRef]
- Zhao, J.; Hu, C.; Fang, J.; Sun, R.; Lu, J.; Su, S.; Teng, B. High-temperature solid-phase synthesis of eulyite-type Ba3Yb(PO4)(3) as a single host for narrow-band Tb3+ green emission. Journal of Materials Science. Mater. Electron. 2023, 34, 918. [Google Scholar] [CrossRef]
- Raj, C.R.; Suresh, S.; Bhavsar, R.R.; Singh, V.K. Recent developments in thermo-physical property enhancement and applications of solid solid phase change materials A review. J. Therm. Anal. Calorim. 2020, 139, 3023–3049. [Google Scholar] [CrossRef]
- Misevicius, M.; Pinkas, J.; Balevicius, V. Solid-state synthesis and luminescence of europium doped and co-doped with dysprosium SrAl4O7. J. Alloys Compd. 2020, 820, 153110. [Google Scholar] [CrossRef]
- Song, Y.; Guo, N.; Li, J.; Ouyang, R.; Miao, Y.; Shao, B. Photoluminescence and temperature sensing of lanthanide Eu3+ and transition metal Mn4+ dual-doped antimoniate phosphor through site-beneficial occupation. Ceram. Int. 2020, 46, 22164–22170. [Google Scholar] [CrossRef]
- Pavani, K.; Kumar, J.S.; Moorthy, L.R. Photoluminescence properties of Tb3+ and Eu3+ ions co-doped SrMg2La2W2O12 phosphors for solid state lighting applications. J. Alloys Compd. 2014, 586, 722–729. [Google Scholar] [CrossRef]
- Tang, Q.; Guo, N.; Xin, Y.; Li, W.; Shao, B.; Ouyang, R. Luminous tuning in Eu3+/Mn4+ co-doped double perovskite structure by designing the site-occupancy strategy for solid-state lighting and optical temperature sensing. Mater. Res. Bull. 2022, 149, 111704. [Google Scholar] [CrossRef]
- Zhang, N.N.; Yang, Y.G.; Yan, X.Y.; Wang, M.Q.; Zhang, Y.Y.; Zhang, H.D.; Wang, J.Y. Preparation and photoluminescence properties of novel orange-red La3Ga5SiO14: Sm3+ phosphors. Ceram. Int. 2023, 49, 16080–16088. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, C.; Zheng, W.; Zhang, Q.; Bao, Z.; Kong, L.; Li, L. Large-Scale Synthesis of Highly Luminescent Perovskite Nanocrystals by Template-Assisted Solid-State Reaction at 800 °C. Chem. Mater. 2020, 32, 308–314. [Google Scholar] [CrossRef]
- Wang, T.C.; He, Z.L.; Luo, J.B.; Peng, Q.P.; Wei, J.H.; Chen, K.L.; Kuang, D.B. Organic–Inorganic Hybrid Rare Earth Halide Glasses for Tunable Multicolor X-ray Scintillation. Angew. Chem. Int. Ed. 2025, 64, e202504658. [Google Scholar] [CrossRef]
- Zhang, G.; Wang, D.; Lou, B.; Ma, C.G.; Meijerink, A.; Wang, Y. Efficient broadband near-infrared emission from lead-free halide double perovskite single crystal. Angew. Chem. Int. Ed. 2022, 61, e202207454. [Google Scholar] [CrossRef]
- Zhang, Q.; Zheng, W.; Wan, Q.; Liu, M.; Feng, X.; Kong, L.; Li, L. Confined synthesis of stable and uniform CsPbBr3 nanocrystals with high quantum yield up to 90% by high temperature solid-state reaction. Adv. Opt. Mater. 2021, 9, 2002130. [Google Scholar]
- Yang, X.; Shao, Z.; Ru, H. Preparation and characterisation of Sr2CeO4: Eu3+ rare earth luminescent material by high temperature mechano-chemical method. J. Mater. Sci. Technol. 2016, 32, 1066–1070. [Google Scholar] [CrossRef]
- Duan, J.; Bai, C.; Sun, J.; Wang, D.; Wang, Y. Highly Efficient Novel Garnet-Structured Yellow Emitting Phosphor for High Power Laser-Driven Lighting. Laser Photonics Rev. 2025, 19, 2401371. [Google Scholar] [CrossRef]
- Cao, J.; Liu, R.; Guo, H.; Tian, S.; Zhang, K.; Ren, X.; Liang, G. High-temperature solid-phase synthesis of lithium iron phosphate using polyethylene glycol grafted carbon nanotubes as the carbon source for rate-type lithium-ion batteries. J. Electroanal. Chem. 2022, 907, 116049. [Google Scholar] [CrossRef]
- Al-Qahtani, S.D.; Alzahrani, S.O.; Snari, R.M. Preparation of photoluminescent and photochromic smart glass window using sol-gel technique and lanthanides-activated aluminate phosphor. Ceram. Int. 2022, 48, 17489–17498. [Google Scholar] [CrossRef]
- Fan, N.; Du, Q.; Guo, R.; Luo, L.; Wang, L. Sol-Gel Synthesis and Photoluminescence Properties of a Far-Red Emitting Phosphor BaLaMgTaO 6:Mn4+ for Plant Growth LEDs. Materials 2023, 16, 4029. [Google Scholar] [CrossRef] [PubMed]
- Tanwar, V.; Singh, S.; Gupta, I.; Kumar, P.; Kumar, H.; Mari, B. Singh DPreparation luminescence characterization of Eu(III)-activated Forsterite for optoelectronic applications. J. Mol. Struct. 2021, 1250, 131802. [Google Scholar] [CrossRef]
- Gökçe, S.; Keskin, İ.Ç.; Katı, M.İ.; Kibar, R.; Çetin, A.; Arslanlar, Y.T. Kinetic parameters and luminescence properties of rare earth (Tb, Nd) doped and transition metal (Mn) doped/co-doped YAlO3 prepared via sol-gel method. J. Lumin. 2023, 261, 119883. [Google Scholar] [CrossRef]
- Vijayaprasath, G.; Murugan, R.; Ravi, G.; Mahalingam, T.; Hayakawa, Y. Characterization of dilute magnetic semiconducting transition metal doped ZnO thin films by sol–gel spin coating method. Appl. Surf. Sci. 2014, 313, 870–876. [Google Scholar] [CrossRef]
- El Ghoul, J.; El Mir, L. Sol–gel synthesis and luminescence of undoped and Mn-doped zinc orthosilicate phosphor nanocomposites. J. Lumin. 2014, 148, 82–88. [Google Scholar] [CrossRef]
- Talane, T.E.; Mbule, P.S.; Noto, L.L.; Shingange, K.; Mhlongo, G.H.; Mothudi, B.M.; Dhlamini, M.S. Sol-gel preparation and characterization of Er3+ doped TiO2 luminescent nanoparticles. Mater. Res. Bull. 2018, 108, 234–241. [Google Scholar] [CrossRef]
- Szpikowska-Sroka, B.; Pawlik, N.; Goryczka, T.; Pisarski, W.A. Technological aspects for Tb3+-doped luminescent sol–gel nanomaterials. Ceram. Int. 2015, 41, 11670–11679. [Google Scholar] [CrossRef]
- Lauria, A.; Villa, I.; Fasoli, M.; Niederberger, M.; Vedda, A. Multifunctional role of rare earth doping in optical materials: Nonaqueous sol–gel synthesis of stabilized cubic HfO2 luminescent nanoparticles. Acs Nano 2013, 7, 7041–7052. [Google Scholar] [CrossRef]
- Feinle, A.; Lavoie-Cardinal, F.; Akbarzadeh, J.; Peterlik, H.; Adlung, M.; Wickleder, C.; Husing, N. Novel Sol–Gel Precursors for Thin Mesoporous Eu3+-Doped Silica Coatings as Efficient Luminescent Materials. Chem. Mater. 2012, 24, 3674–3683. [Google Scholar] [CrossRef]
- De La Rosa, A.S.; Cortés-Hernández, D.A.; Escorcia-García, J.; Lopez-Herrera, H.U. White-light luminescence from Tm3+-doped borosilicate glass-ceramics synthesized by the sol-gel route. Ceram. Int. 2023, 49, 23985–23995. [Google Scholar] [CrossRef]
- Keskin, I.Ç.; Türemiş, M.; Katı, M.I.; Gültekin, S.; Arslanlar, Y.T.; Çetin, A.; Kibar, R. Detailed luminescence (RL, PL, CL, TL) behaviors of Tb3+ and Dy3+ doped LiMgPO4 synthesized by sol-gel method. J. Lumin. 2020, 225, 117276. [Google Scholar] [CrossRef]
- Gahlaut, U.P.S.; Kumar, V.; Pandey, R.K.; Goswami, Y.C. Highly luminescent ultra small Cu doped ZnO nanostructures grown by ultrasonicated sol–gel route. Optik 2016, 127, 4292–4295. [Google Scholar] [CrossRef]
- Cai, J.; Pan, H.; Wang, Y. Luminescence properties of red-emitting Ca2 Al2 SiO7: Eu 3+ nanoparticles prepared by sol-gel method. Rare Met. 2011, 30, 374–380. [Google Scholar] [CrossRef]
- Boukerika, A.; Guerbous, L. Annealing effects on structural and luminescence properties of red Eu3+-doped Y2O3 nanophosphors prepared by sol–gel method. J. Lumin. 2014, 145, 148–153. [Google Scholar] [CrossRef]
- Parayil, R.T.; Gupta, S.K.; Mohapatra, M. A review on defect engineered NIR persistent luminescence through transition metal ion (Cr, Mn, Fe and Ni) doping: Wider perspective covering synthesis, characterization, fundamentals and applications. Coord. Chem. Rev. 2025, 522, 216200. [Google Scholar] [CrossRef]
- Masjedi-Arani, M.; Salavati-Niasari, M. Metal (Mn, Co, Ni and Cu) doped ZnO-Zn2SnO4-SnO2 nanocomposites: Green sol-gel synthesis, characterization and photocatalytic activity. J. Mol. Liq. 2017, 248, 197–204. [Google Scholar] [CrossRef]
- Sung Lim, C.; Aleksandrovsky, A.; Atuchin, V.; Molokeev, M.; Oreshonkov, A. Microwave-Employed Sol-Gel Synthesis of Scheelite-Type Microcrystalline AgGd(MoO4)2:Yb3+/Ho3+ Upconversion Yellow Phosphors and Their Spectroscopic Properties. Crystals 2020, 256, 278–294. [Google Scholar] [CrossRef]
- Prerna Arya, S.; Sharma, A.; Singh, B.; Tomar, A.; Singh, S.; Sharma, R. Morphological and Optical Characterization of Sol-Gel Synthesized Ni-Doped ZnO Nanoparticles. Integr. Ferroelectr. 2020, 205, 1–13. [Google Scholar] [CrossRef]
- Jovanovi, D. Lanthanide-doped orthovanadate phosphors: Syntheses, structures, and photoluminescence properties. Spectrosc. Lanthan. Doped Oxide Mater. 2020, 211, 198–213. [Google Scholar]
- Shen, S.; Chen, J.; Wang, X.; Zhao, L.; Guo, L. Microwave-assisted hydrothermal synthesis of transition-metal doped ZnIn2S4 and its photocatalytic activity for hydrogen evolution under visible light. J. Power Sources 2011, 196, 10112–10119. [Google Scholar] [CrossRef]
- Sadetskaya, A.V.; Bobrysheva, N.P.; Osmolowsky, M.G.; Osmolovskaya, O.M.; Voznesenskiy, M.A. Correlative experimental and theoretical characterization of transition metal doped hydroxyapatite nanoparticles fabricated by hydrothermal method. Mater. Charact. 2021, 173, 110911. [Google Scholar] [CrossRef]
- Kaynar, Ü.H.; Çoban, M.B.; Madkhli, A.Y.; Ayvacikli, M.; Can, N. Phase transition and luminescence characteristics of dysprosium doped strontium stannate phosphor synthesized using hydrothermal method. Ceram. Int. 2023, 49, 11641–11646. [Google Scholar] [CrossRef]
- Hu, Y.; Liang, X.; Wang, Y.; Liu, E.; Hu, X.; Fan, J. Enhancement of the red upconversion luminescence in NaYF4: Yb3+, Er3+ nanoparticles by the transition metal ions doping. Ceram. Int. 2015, 41, 14545–14553. [Google Scholar] [CrossRef]
- Huang, W.; Lu, C.; Jiang, C.; Jin, J.; Ding, M.; Ni, Y.; Xu, Z. Rare earth doped LiYbF4 phosphors with controlled morphologies: Hydrothermal synthesis and luminescent properties. Mater. Res. Bull. 2012, 47, 1310–1315. [Google Scholar] [CrossRef]
- Chavan, A.B.; Gawande, A.B.; Gaikwad, V.B.; Jain, G.H.; Deore, M.K. Hydrothermal synthesis and luminescence properties of Dy3+ doped SrMoO4 nano-phosphor. J. Lumin. 2021, 234, 117996. [Google Scholar] [CrossRef]
- Reszczyńska, J.; Grzyb, T.; Wei, Z.; Klein, M.; Kowalska, E.; Ohtani, B.; Zaleska-Medynska, A. Photocatalytic activity and luminescence properties of RE3+–TiO2 nanocrystals prepared by sol–gel and hydrothermal methods. Appl. Catal. B Environ. 2016, 181, 825–837. [Google Scholar] [CrossRef]
- Zhou, M.; Han, D.; Liu, X.; Ma, C.; Wang, H.; Tang, Y.; Yang, J. Enhanced visible light photocatalytic activity of alkaline earth metal ions-doped CdSe/rGO photocatalysts synthesized by hydrothermal method. Appl. Catal. B Environ. 2015, 172, 174–184. [Google Scholar] [CrossRef]
- Macedo, W.C.; Junior, A.G.B.; de Oliveira Rocha, K.; de Souza Albas, A.E.; Pires, A.M.; Teixeira, S.R.; Longo, E. Photoluminescence of Eu3+-doped CaZrO3 red-emitting phosphors synthesized via microwave-assisted hydrothermal method. Mater. Today Commun. 2020, 24, 100966. [Google Scholar] [CrossRef]
- Zhao, W.; Wei, Z.; Zhang, L.; Wu, X.; Wang, X.; Jiang, J. Optical magnetic properties of Co Ni co-doped ZnSnanorods prepared by hydrothermal method. J. Alloys Compd. 2017, 698, 754–760. [Google Scholar] [CrossRef]
- Ming, J.; Chen, Y.; Miao, H.; Fan, Y.; Wang, S.; Chen, Z.; Zhang, F. High-brightness transition metal-sensitized lanthanide near-infrared luminescent nanoparticles. Nat. Photonics 2024, 18, 1254–1262. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, Y.J.; Hao, X.; Peng, P.; Shi, J.Y.; Peng, F.; Sun, R.C. Hydrothermal synthesis and applications of advanced carbonaceous materials from biomass: A review. Adv. Compos. Hybrid Mater. 2020, 3, 267–284. [Google Scholar] [CrossRef]
- Huang, P.; Zheng, W.; Gong, Z.; You, W.; Wei, J.; Chen, X. Rare earth ion–and transition metal ion–doped inorganic luminescent nanocrystals: From fundamentals to biodetection. Mater. Today Nano 2019, 5, 100031. [Google Scholar] [CrossRef]
- Xian, Y.; Li, K. Hydrothermal Synthesis of High-Yield Red Fluorescent Carbon Dots with Ultra-Narrow Emission by Controlled O/N Elements. Adv. Mater. 2022, 34, e2201031. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Bai, X.; Yang, D.; Chen, X.; Jing, P.; Qu, S.; Song, H. Doping lanthanide into perovskite nanocrystals: Highly improved and expanded optical properties. Nano Lett. 2017, 17, 8005–8011. [Google Scholar] [CrossRef]
- Milstein, T.J.; Kroupa, D.M.; Gamelin, D.R. Picosecond quantum cutting generates photoluminescence quantum yields over 100% in ytterbium-doped CsPbCl3 nanocrystals. Nano Lett. 2018, 18, 3792–3799. [Google Scholar] [CrossRef] [PubMed]
- De la Mora, M.B.; Amelines-Sarria, O.; Monroy, B.M.; Hernández-Pérez, C.D.; Lugo, J.E. Materials for downconversion in solar cells: Perspectives and challenges. Sol. Energy Mater. Sol. Cells 2017, 165, 59–71. [Google Scholar] [CrossRef]
- Sun, T.; Chen, X.; Jin, L.; Li, H.W.; Chen, B.; Fan, B.; Wang, F. Broadband Ce(III)-Sensitized Quantum Cutting in Core–Shell Nanoparticles: Mechanistic Investigation and Photovoltaic Application. J. Phys. Chem. Lett. 2017, 8, 5099–5104. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Chen, Z.; Leung, M.H.M.; Zheng, B.; Wang, L.; An, M.; Yuan, Z. Photo-Induced Synthesis of Ytterbium and Manganese-Doped CsPbCl3 Nanocrystals for Visible to Near–Infrared Photoluminescence with Negative Thermal Quenching. Adv. Sci. 2025, 12, 2408927. [Google Scholar] [CrossRef]
- Zang, Y.; Lin, W.; Zheng, Y.; Su, C.; Wu, P.; Chang, Q. Acoustic radiation torque of a Bessel vortex wave on a viscoelastic spherical shell nearby an impedance boundary. J. Sound Vib. 2021, 509, 116261. [Google Scholar] [CrossRef]
- Jin, J.; Li, H.; Chen, C.; Zhang, B.; Bi, W.; Song, Z.; Dai, Q. Improving Efficiency and Light Stability of Perovskite Solar Cells by Incorporating YVO4:Eu3+, Bi3+Nanophosphor into the Mesoporous TiO2 Layer. ACS Appl. Energy Mater. 2018, 1, 2096–2102. [Google Scholar] [CrossRef]
- Zhou, D.; Sun, R.; Xu, W.; Ding, N.; Li, D.; Chen, X.; Song, H. Impact of host composition, codoping, or tridoping on quantum-cutting emission of ytterbium in halide perovskite quantum dots and solar cell applications. Nano Lett. 2019, 19, 6904–6913. [Google Scholar] [CrossRef]
- Ding, N.; Xu, W.; Zhou, D.; Ji, Y.; Wang, Y.; Sun, R.; Song, H. Extremely efficient quantum-cutting Cr3+, Ce3+, Yb3+ tridoped perovskite quantum dots for highly enhancing the ultraviolet response of Silicon photodetectors with external quantum efficiency exceeding 70%. Nano Energy 2020, 78, 105278. [Google Scholar] [CrossRef]
- Cai, T.; Wang, J.; Li, W.; Hills-Kimball, K.; Yang, H.; Nagaoka, Y.; Chen, O. Mn2+/Yb3+ codoped CsPbCl3 perovskite nanocrystals with triple-wavelength emission for luminescent solar concentrators. Adv. Sci. 2020, 7, 2001317. [Google Scholar] [CrossRef]
- Wang, Y.; Ding, N.; Zhou, D.; Xu, W.; Sun, R.; Li, W.; Song, H. ZnS-coated Yb3+-doped perovskite quantum dots: A stable and efficient quantum cutting photon energy converter for silicon-based electronics. Chem. Eng. J. 2024, 487, 150347. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, Y.; Ren, Z.; Wang, W.; Tu, D.; Li, G. Simultaneous Luminescence Color Tuning and Spectral Broadening of Cr3+ Near-Infrared Emission for Night Vision Application. ACS Appl. Opt. Mater. 2023, 1, 1088–1096. [Google Scholar] [CrossRef]
- Yu, H.; Chen, J.; Mi, R.; Yang, J.; Liu, Y.G. Broadband near-infrared emission of K3ScF:Cr3+ phosphors for night vision imaging system sources. Chem. Eng. J. 2021, 417, 129271. [Google Scholar] [CrossRef]
- Das, S.; Sharma, S.K.; Manam, J. Near infrared emitting Cr3+ doped Zn3Ga2Ge2O10 long persistent phosphor for night vision surveillance and anti-counterfeit applications. Ceram. Int. 2022, 48, 824–831. [Google Scholar] [CrossRef]
- Zhou, Z.; Wang, X.; Yi, X.; Ming, H.; Ma, Z.; Peng, M. Rechargeable and sunlight-activated Sr3Y2Ge3O12:Bi3+ UV–Visible-NIRpersistent luminescence material for night-vision signage optical information storage. Chem. Eng. J. 2021, 421, 127820. [Google Scholar] [CrossRef]
- Jiménez-Hernández, M.E.; Orellana, G.; Montero, F.; Portolés, M.T. A Ruthenium Probe for Cell Viability Measurement Using Flow Cytometry, Confocal Microscopy and Time-Resolved Luminescence. Photochem. Photobiol. 2000, 72, 28–34. [Google Scholar] [CrossRef]
- Manikandamathavan, V.M.; Duraipandy, N.; Kiran, M.S.; Vaidyanathan, V.G.; Nair, B.U. A New Platinum(II) Complex for Bioimaging Applications. RSC Adv. 2015, 5, 24877–24885. [Google Scholar] [CrossRef]
- Chen, M.; Wu, Y.; Liu, Y.; Yang, H.; Zhao, Q.; Li, F. A Phosphorescent Iridium(III) Solvent Complex for Multiplex Assays of Cell Death. Biomaterials 2014, 35, 8748–8755. [Google Scholar] [CrossRef]
- Ouyang, C.; Li, Y.; Rees, T.W.; Liao, X.; Jia, J.; Chen, Y.; Chao, H. Supramolecular Assembly of An Organoplatinum (II) Complex with Ratiometric Dual Emission for Two-Photon Bioimaging. Angew. Chem. 2021, 133, 4196–4203. [Google Scholar] [CrossRef]
- Li, B.; Wang, Y.; Chan, M.H.-Y.; Pan, M.; Li, Y.; Yam, V.W.-W. Supramolecular Assembly of Organoplatinum(II) Complexes for Subcellular Distribution and Cell Viability Monitoring with Differentiated Imaging. Angew. Chem. Int. Ed. 2022, 61, e202210703. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, W.; Chung, J.; Yin, J.; Yoon, J. Recent Progress in Fluorescent Probes for Bacteria. Chem. Soc. Rev. 2021, 50, 7725–7744. [Google Scholar] [CrossRef]
- Gautam, A.; Gupta, A.; Prasad, P.; Sasmal, P.K. AIE-Active Cyclometalated Iridium(III) Complexes for the Detection of Lipopolysaccharides and Wash-Free Imaging of Bacteria. Dalton Trans. 2023, 52, 7843–7853. [Google Scholar] [CrossRef]
- Xu, L.; Deng, P.; Song, W.; Liu, M.; Wang, M.; Yu, Y.; Wang, F. An AIE-Active Cyclometalated Iridium(III) Photosensitizer for Selective Discrimination, Imaging, and Synergistic Elimination of Gram-Positive Bacteria. ACS Mater. Lett. 2023, 5, 162–171. [Google Scholar] [CrossRef]
- Gupta, A.; Prasad, P.; Gupta, S.; Sasmal, P.K. Simultaneous Ultrasensitive Detection and Elimination of Drug-Resistant Bacteria by Cyclometalated Iridium(III) Complexes. ACS Appl. Mater. Interfaces 2020, 12, 35967–35976. [Google Scholar] [CrossRef]
- Jian, Y.; Jin, Z.; Qi, S.; Da, X.; Wang, Z.; Wang, X.; Zhou, Q. An Alkynyl-Dangling Ru(II) Polypyridine Complex for Targeted Antimicrobial Photodynamic Therapy. Chem. Eur. J. 2022, 28, e202103359. [Google Scholar] [CrossRef]
- Yang, Z.; Cao, J.; He, Y.; Yang, J.H.; Kim, T.; Peng, X.; Kim, J.S. Macro-/Micro-Environment-Sensitive Chemosensing and Biological Imaging. Chem. Soc. Rev. 2014, 43, 4563–4601. [Google Scholar] [CrossRef]
- Yin, J.; Huang, L.; Wu, L.; Li, J.; James, T.D.; Lin, W. Small Molecule Based Fluorescent Chemosensors for Imaging the Microenvironment within Specific Cellular Regions. Chem. Soc. Rev. 2021, 50, 12098–12150. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, L.; Dai, T.; Qin, Z.; Lu, H.; Zhang, L.; Zhou, F. Liquid−Liquid Phase Separation in Human Health and Diseases. Signal Transduct. Target. Ther. 2021, 6, 290. [Google Scholar] [CrossRef]
- Yan, Z.; Xue, J.; Zhou, M.; Wang, J.; Zhang, Y.; Wang, Y.; Qiao, J.; He, Y.; Li, P.; Zhang, S. Dynamic Monitoring of Phase-Separated Biomolecular Condensates by Photoluminescence Lifetime Imaging. Anal. Chem. 2021, 93, 2988–2995. [Google Scholar] [CrossRef]
- Wang, W.-J.; Mu, X.; Tan, C.-P.; Wang, Y.-J.; Zhang, Y.; Li, G.; Mao, Z.-W. Induction and Monitoring of DNA Phase Separation in Living Cells by a Light-Switching Ruthenium Complex. J. Am. Chem. Soc. 2021, 143, 11370–11381. [Google Scholar] [CrossRef]
- Zhou, S.; Duan, C.; Yin, M.; Liu, X.; Han, S.; Zhang, S.; Li, X. Optical thermometry based on cooperation of temperature-induced shift of charge transfer band edge and thermal coupling. Opt. Express 2018, 26, 27339–27345. [Google Scholar] [CrossRef]
- Wang, X.; Wolfbeis, O.S.; Meier, R.J. Luminescent probes and sensors for temperature. Chem. Soc. Rev. 2013, 42, 7834–7869. [Google Scholar] [CrossRef]
- Jaque, D.; Vetrone, F. Luminescence nanothermometry. Nanoscale 2012, 4, 4301–4326. [Google Scholar] [CrossRef]
- Xu, W.; Song, Q.; Zheng, L.; Zhang, Z.; Cao, W. Optical temperature sensing based on the near-infrared emissions from Nd 3+/Yb 3+ codoped CaWO4. Opt. Lett. 2014, 39, 4635–4638. [Google Scholar] [CrossRef]
- Zheng, S.; Chen, W.; Tan, D.; Zhou, J.; Guo, Q.; Jiang, W.; Qiu, J. Lanthanide-doped NaGdF4 core–shell nanoparticles for non-contact self-referencing temperature sensors. Nanoscale 2014, 6, 5675–5679. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhao, H.; Li, Y.; Zheng, L.; Zhang, Z.; Cao, W. Optical temperature sensing through the upconversion luminescence from Ho3+/Yb3+ codoped CaWO4. Sens. Actuators B Chem. 2013, 188, 1096–1100. [Google Scholar] [CrossRef]
- Zhou, S.; Jiang, G.; Wei, X.; Duan, C.; Chen, Y.; Yin, M. Pr3+-Doped β-NaYF4 for temperature sensing with fluorescence intensity ratio technique. J. Nanosci. Nanotechnol. 2014, 14, 3739–3742. [Google Scholar] [CrossRef]
- Wawrzynczyk, D.; Bednarkiewicz, A.; Nyk, M.; Strek, W.; Samoc, M. Neodymium (III) doped fluoride nanoparticles as non-contact optical temperature sensors. Nanoscale 2012, 4, 6959–6961. [Google Scholar] [CrossRef]
- Zheng, K.; Liu, Z.; Lv, C.; Qin, W. Temperature sensor based on the UV upconversion luminescence of Gd 3+ in Yb 3+–Tm 3+–Gd 3+ codoped NaLuF 4 microcrystals. J. Mater. Chem. C 2013, 1, 5502–5507. [Google Scholar] [CrossRef]
- Boruc, Z.; Kaczkan, M.; Fetlinski, B.; Turczynski, S.; Malinowski, M. Blue emissions in Dy3+ doped Y4Al2O9 crystals for temperature sensing. Opt. Lett. 2012, 37, 5214–5216. [Google Scholar] [CrossRef]
- Abbas, M.T.; Khan, N.Z.; Mao, J.; Qiu, L.; Wei, X.; Chen, Y.; Khan, S.A. Lanthanide and transition metals doped materials for non-contact optical thermometry with promising approaches. Mater. Today Chem. 2022, 24, 100903. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Q.; Bu, Y.; Liu, C.S.; Liu, T.; Yan, X. Optical temperature sensing of rare-earth ion doped phosphors. RSC Adv. 2015, 5, 86219–86236. [Google Scholar] [CrossRef]
- Chen, D.; Yu, Y.; Huang, P.; Lin, H.; Shan, Z.; Wang, Y. Color-tunable luminescence of Eu3+ in LaF3 embedded nanocomposite for light emitting diode. Acta Mater. 2010, 58, 3035–3041. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, K.H.; Unithrattil, S.; Yoon, H.S.; Jang, H.G.; Im, W.B. Melilite-structure CaYAl3O7: Eu3+ phosphor: Structural and optical characteristics for near-UV LED-based white light. J. Phys. Chem. C 2012, 116, 26850–26856. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, Y.; Liu, Y.; Mi, C.; Li, G.; Han, B.; Seo, H.J. Photoluminescence properties of heavily Eu3+-doped BaCa2In6O12 phosphor for white-light-emitting diodes. J. Am. Ceram. Soc. 2015, 98, 1567–1573. [Google Scholar] [CrossRef]
- Khan, S.A.; Jalil, A.; Khan, Q.U.; Irfan, R.M.; Mehmood, I.; Khan, K.; Agathopoulos, S. New physical insight into crystal structure, luminescence and optical properties of YPO4: Dy3+∖Eu3+∖Tb3+ single-phase white-light-emitting phosphors. J. Alloys Compd. 2020, 817, 152687. [Google Scholar] [CrossRef]
- Lu, Z.; Cao, Z.; Wei, X.; Yin, M.; Chen, Y. Luminescence properties of Eu3+ doped YBO3 for temperature sensing. J. Rare Earths 2017, 35, 356–360. [Google Scholar]
- Hua, Y.; Du, P.; Yu, J.S. Synthesis and luminescent properties of Er3+-activated LaBMoO6 green-emitting phosphors for optical thermometry. Mater. Res. Bull. 2018, 107, 314–320. [Google Scholar] [CrossRef]
- Runowski, M.; Woźny, P.; Stopikowska, N.; Martín, I.R.; Lavín, V.; Lis, S. Luminescent nanothermometer operating at very high temperature—Sensing up to 1000 K with upconverting nanoparticles (Yb3+/Tm3+). ACS Appl. Mater. Interfaces 2020, 12, 43933–43941. [Google Scholar] [CrossRef]
- Cao, Z.; Wei, X.; Zhao, L.; Chen, Y.; Yin, M. Investigation of SrB4O7: Sm2+ as a multimode temperature sensor with high sensitivity. ACS Appl. Mater. Interfaces 2016, 8, 34546–34551. [Google Scholar] [CrossRef]
- Li, X.; Wei, X.; Qin, Y.; Chen, Y.; Duan, C.; Yin, M. The emission rise time of BaY2ZnO5: Eu3+ for non-contact luminescence thermometry. J. Alloys Compd. 2016, 657, 353–357. [Google Scholar] [CrossRef]
- Zhou, S.; Duan, C.; Wang, M. Origin of the temperature-induced redshift of the charge transfer band of GdVO4. Opt. Lett. 2017, 42, 4703–4706. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Jin, Y.; Zhang, R.; Yao, Q.; Hu, Y. A review and outlook of ratiometric optical thermometer based on thermally coupled levels and non-thermally coupled levels. J. Alloys Compd. 2022, 894, 162494. [Google Scholar] [CrossRef]
- Baleizão, C.; Nagl, S.; Borisov, S.M.; Schäferling, M.; Wolfbeis, O.S.; Berberan-Santos, M.N. An Optical Thermometer Based on the Delayed Fluorescence of C70. Chem.–A Eur. J. 2010, 13, 3643–3651. [Google Scholar] [CrossRef]
- Runowski, M.; Stopikowska, N.; Szeremeta, D.; Goderski, S.; Skwierczyńska, M.; Lis, S. Upconverting lanthanide fluoride core@ shell nanorods for luminescent thermometry in the first and second biological windows: β-NaYF4: Yb3+–Er3+@ SiO2 temperature sensor. ACS Appl. Mater. Interfaces 2019, 11, 13389–13396. [Google Scholar] [CrossRef]
- Soni, A.K.; Dey, R.; Rai, V.K. Stark sublevels in Tm3+–Yb3+ codoped Na2Y2B2O7 nanophosphor for multifunctional applications. RSC Adv. 2015, 5, 34999–35009. [Google Scholar] [CrossRef]
- Ximendes, E.C.; Rocha, U.; Sales, T.O.; Fernández, N.; Rodríguez, S.R.; Martín, I.R.; Jacinto, C.; Jaque, D. In vivo subcutaneous thermal video recording by supersensitive infrared nanothermometers. Adv. Funct. Mater. 2017, 27, 1702249. [Google Scholar] [CrossRef]
- Stopikowska, N.; Runowski, M.; Skwierczyńska, M.; Lis, S. Improving performance of luminescent nanothermometers based on non-thermally and thermally coupled levels of lanthanides by modulating laser power. Nanoscale 2021, 13, 14139–14146. [Google Scholar] [CrossRef]
- Bu, Y.Y.; Wang, Y.M.; Yan, X.H. Optical temperature sensing behavior of Dy3+-doped transparent alkaline earth fluoride glass ceramics. Appl. Phys. A 2017, 123, 336. [Google Scholar] [CrossRef]
- Shinde, S.L.; Nanda, K.K. Wide-range temperature sensing using highly sensitive green-luminescent ZnO and PMMA–ZnO film as a non-contact optical probe. Angew. Chem. 2013, 52, 11325–11328. [Google Scholar] [CrossRef]
- Zhu, J.; Huang, A.; Ma, H.; Chen, Y.; Zhang, S.; Ji, S.; Jin, P. Hybrid films of VO2 nanoparticles and a nickel(II)-based ligand exchange thermochromic system: Excellent optical performance with a temperature responsive colour change. New J. Chem. 2017, 41, 830–835. [Google Scholar] [CrossRef]
- Zhang, Y.; Qian, Y.; Liu, Y.; Lei, C.; Qiu, G.; Chen, G. Multivalent Metal Ion Cross-Linked Lignocellulosic Nanopaper with Excellent Water Resistance and Optical Performance. Biomacromolecules 2022, 23, 1920–1927. [Google Scholar] [CrossRef]
- Ichiba, K.; Kato, T.; Nakauchi, D.; Kawaguchi, N.; Yanagida, T. Ultraviolet–near infrared luminescence characteristics of Nd:La2Be2O5 single crystals for near-infrared emitting scintillators. J. Lumin. 2025, 277, 120938. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, J.; Shu, Y.; Fu, W.; Wang, Y.; Ye, J.; Yang, L. Design of a high-efficiency electrolytic energy conversion system based on equilibrium analysis of electric-field and flow-field through simulation and experiments. Energy Convers. Manag. 2023, 277, 116679. [Google Scholar] [CrossRef]
- Kordjamshidi, M. Application of fuzzy technique to integrate multiple occupancy scenarios into house rating schemes (HRS). Energy Build. 2013, 67, 463–470. [Google Scholar] [CrossRef]
- Azorin, J. Preparation methods of thermoluminescent materials for dosimetric applications: An overview. Appl. Radiat. Isot. 2014, 83, 187–191. [Google Scholar] [CrossRef]
- Minwal, W.P.; Luidold, S. Recycling of Rare Earth Elements by Smelting of RE-Bearing Raw Materials. Erzmetall 2023, 76, 255–260. [Google Scholar]
- Pagano, G.; Guida, M.; Tommasi, F.; Oral, R. Health effects and toxicity mechanisms of rare earth elements-Knowledge gaps and research prospects. Ecotoxicol. Environ. Saf. 2015, 115, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Medialdea, A.; Brill, D.; King, G.E.; Zander, A.; Lopez-Ramirez, M.R.; Bartz, M.; Brückner, H. Violet stimulated luminescence as an alternative for dating complex colluvial sediments in the Atacama Desert. Quat. Geochronol. 2022, 71, 101337. [Google Scholar] [CrossRef]
- Liu, H.; Liao, L.; Pan, X.; Su, K.; Shuai, P.; Yan, Z.; Mei, L. Recent research progress of luminescent materials with apatite structure: A review. Open Ceram. 2022, 10, 100251. [Google Scholar] [CrossRef]
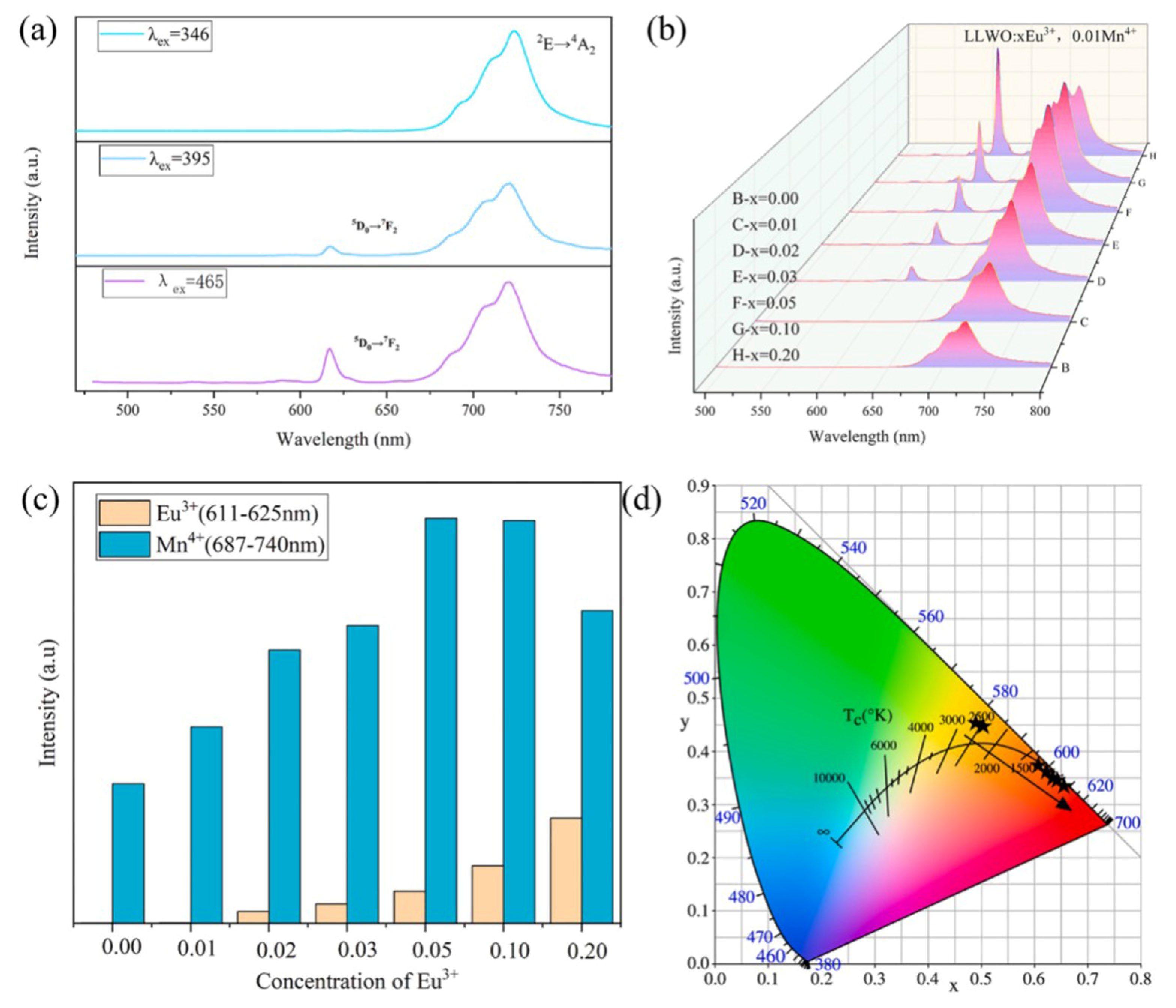
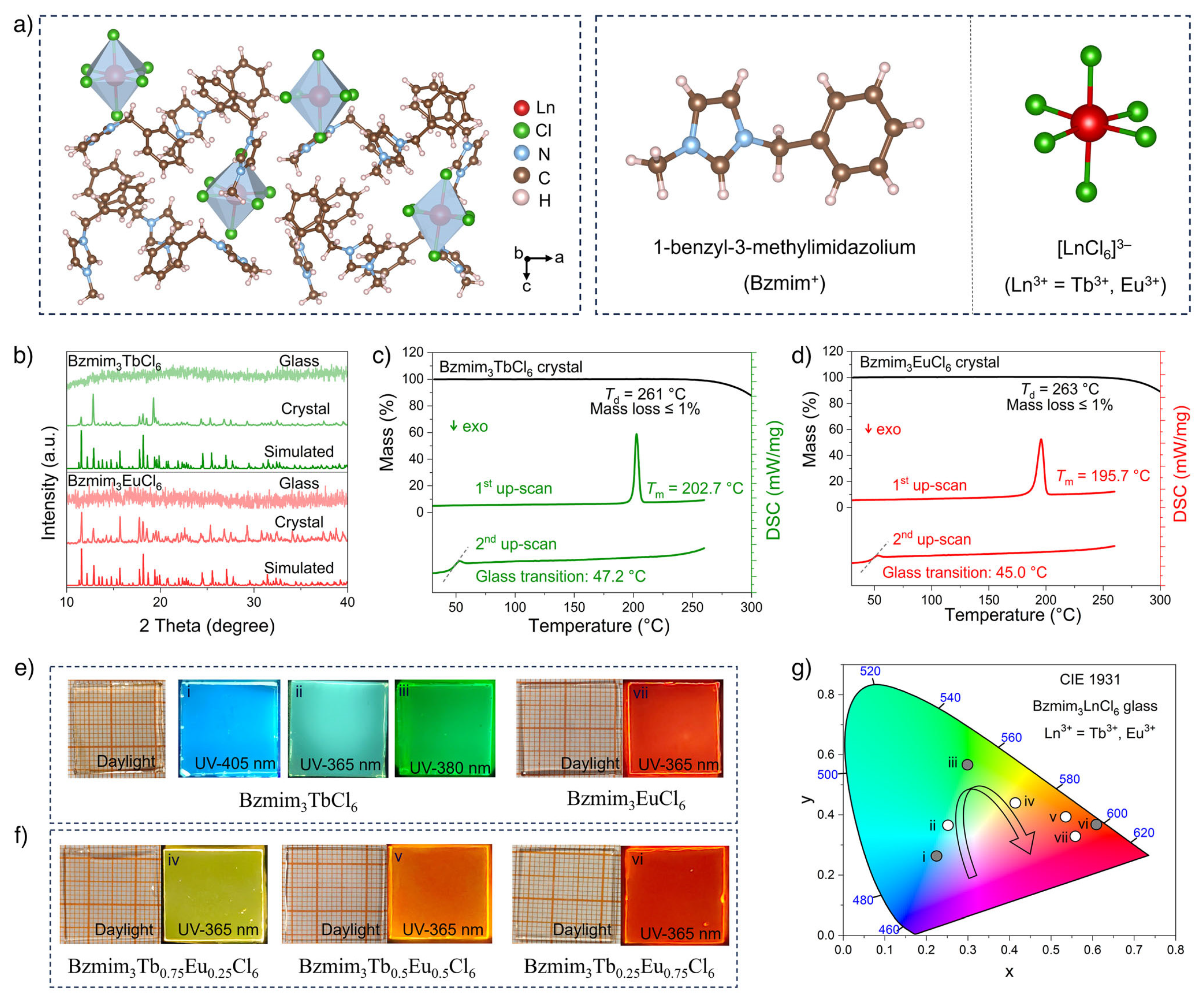

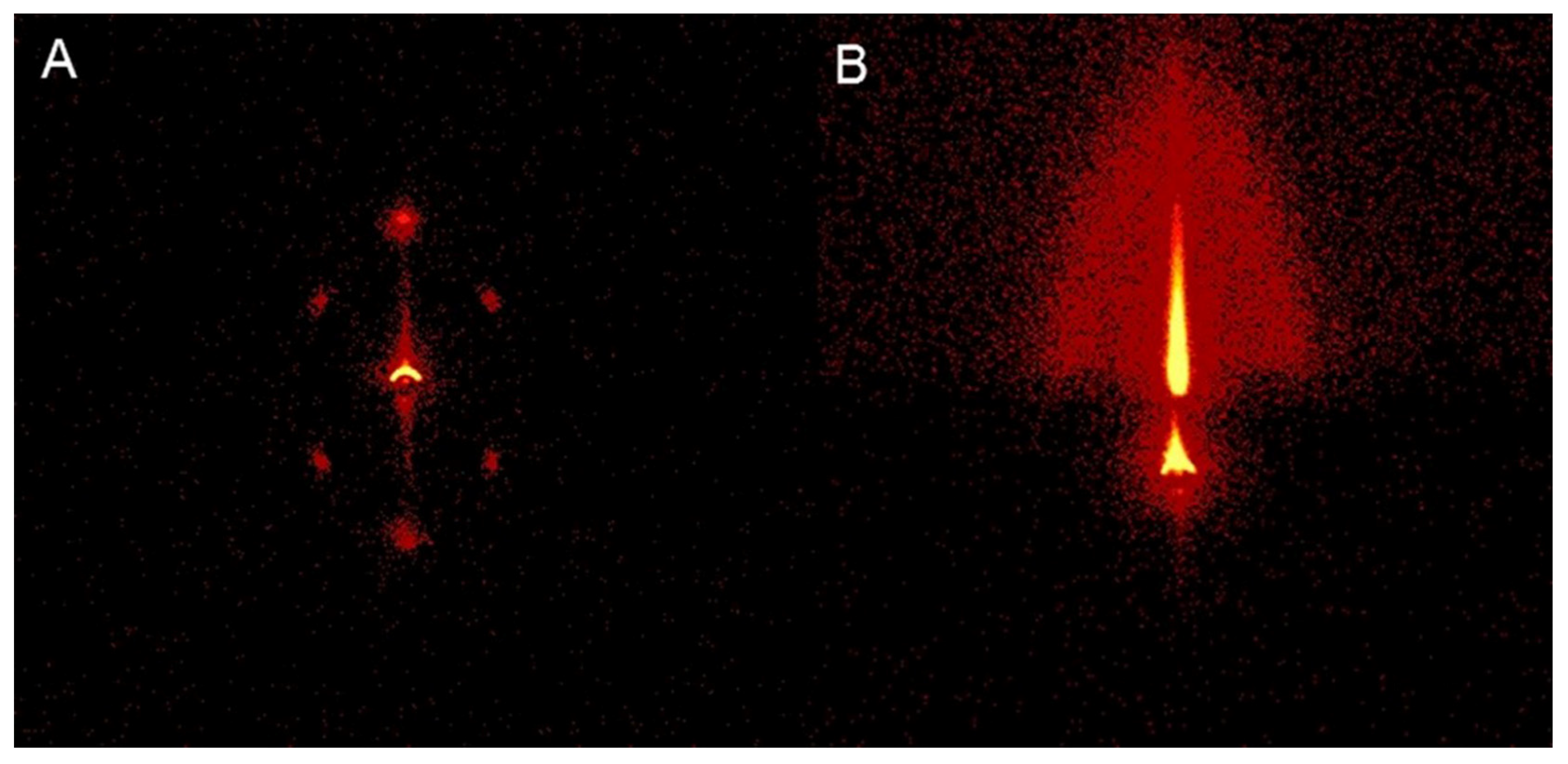
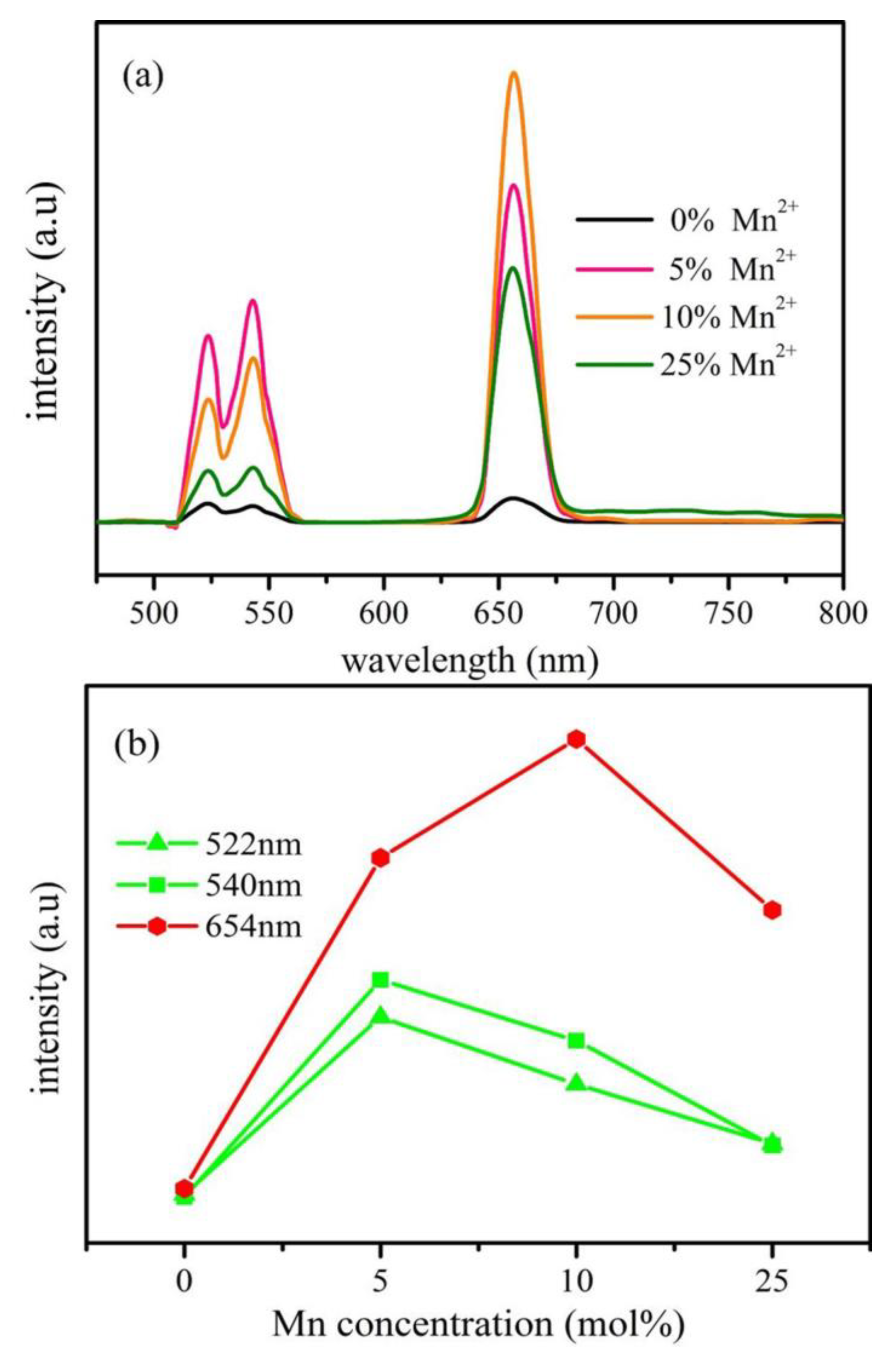
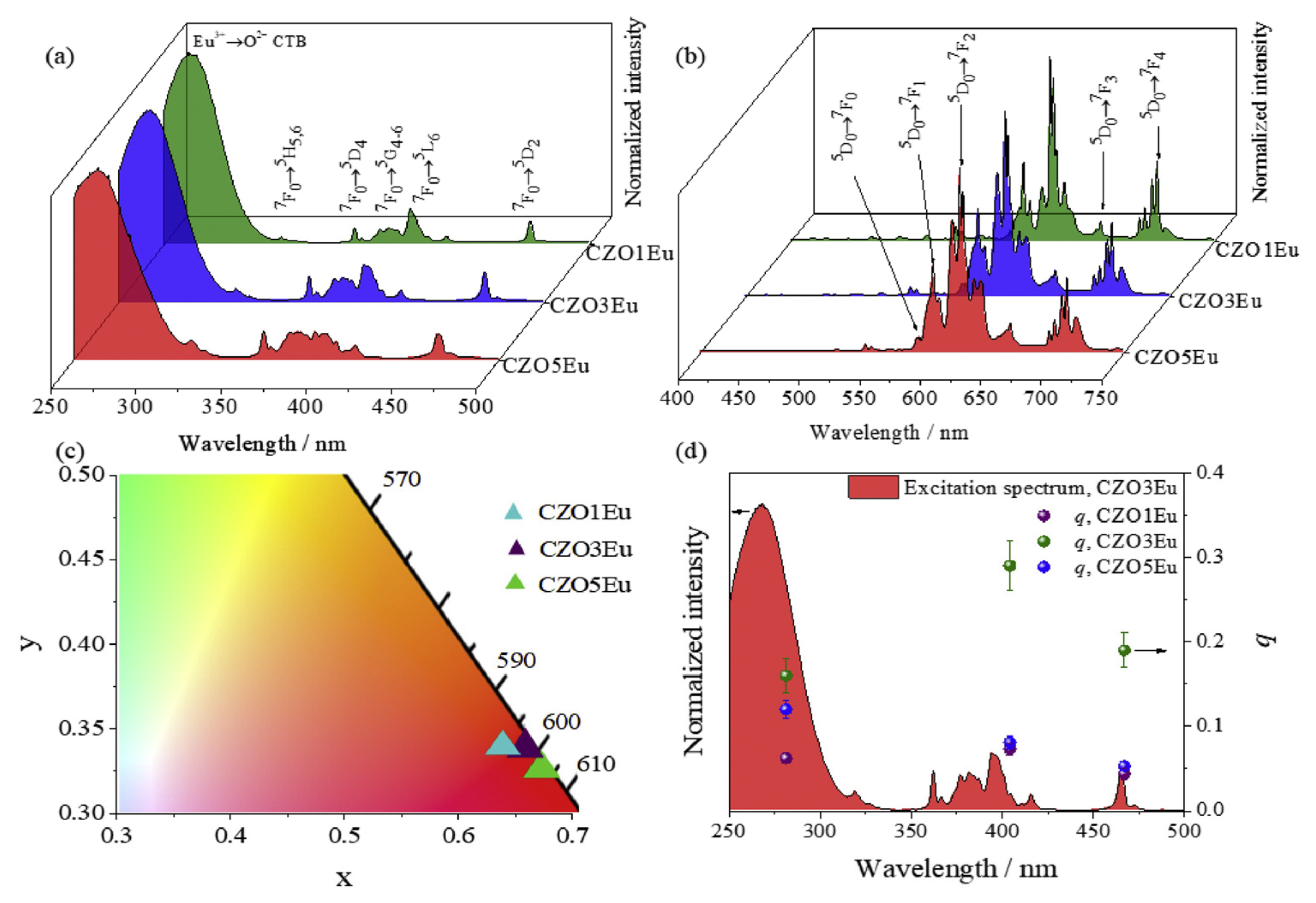
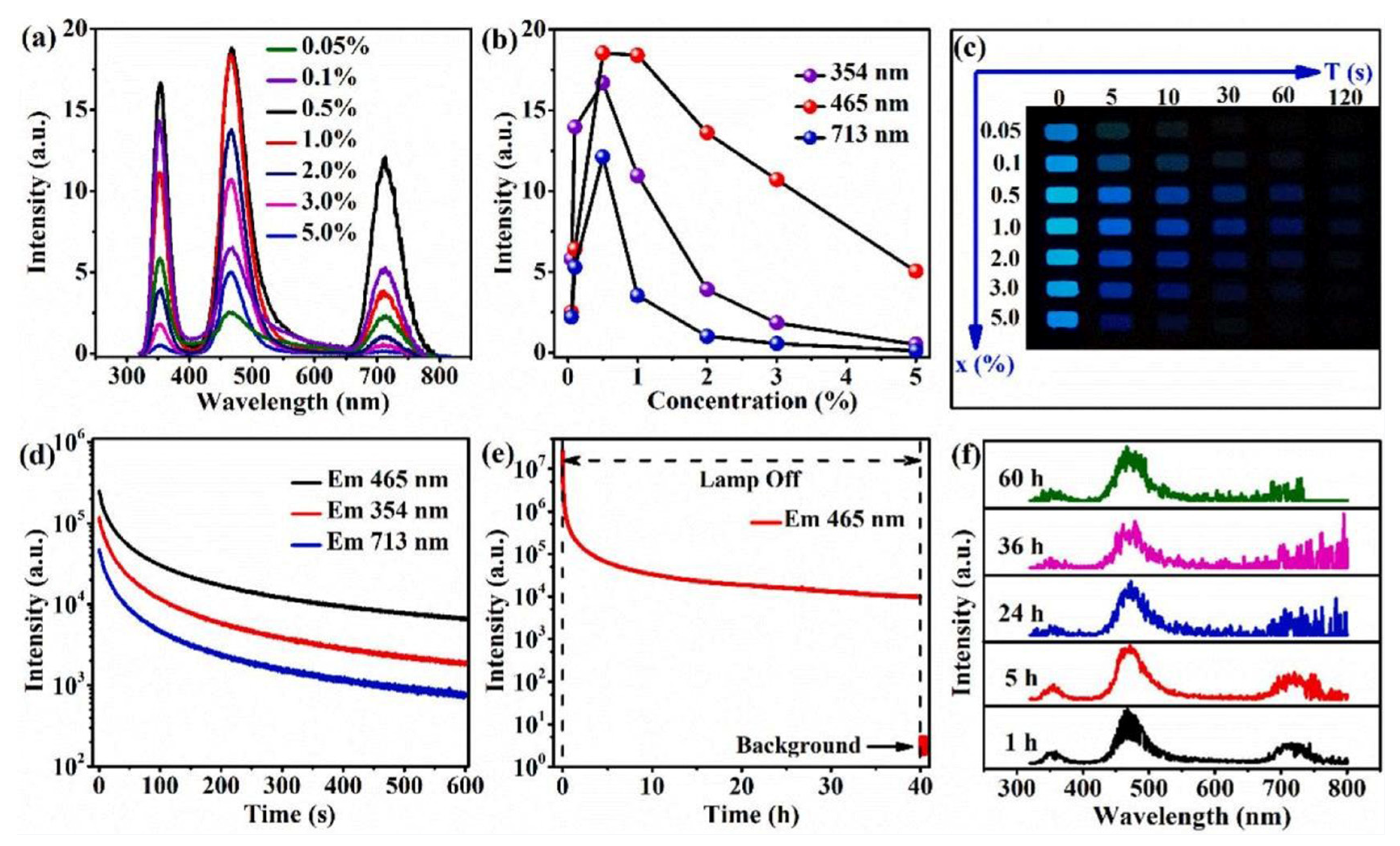
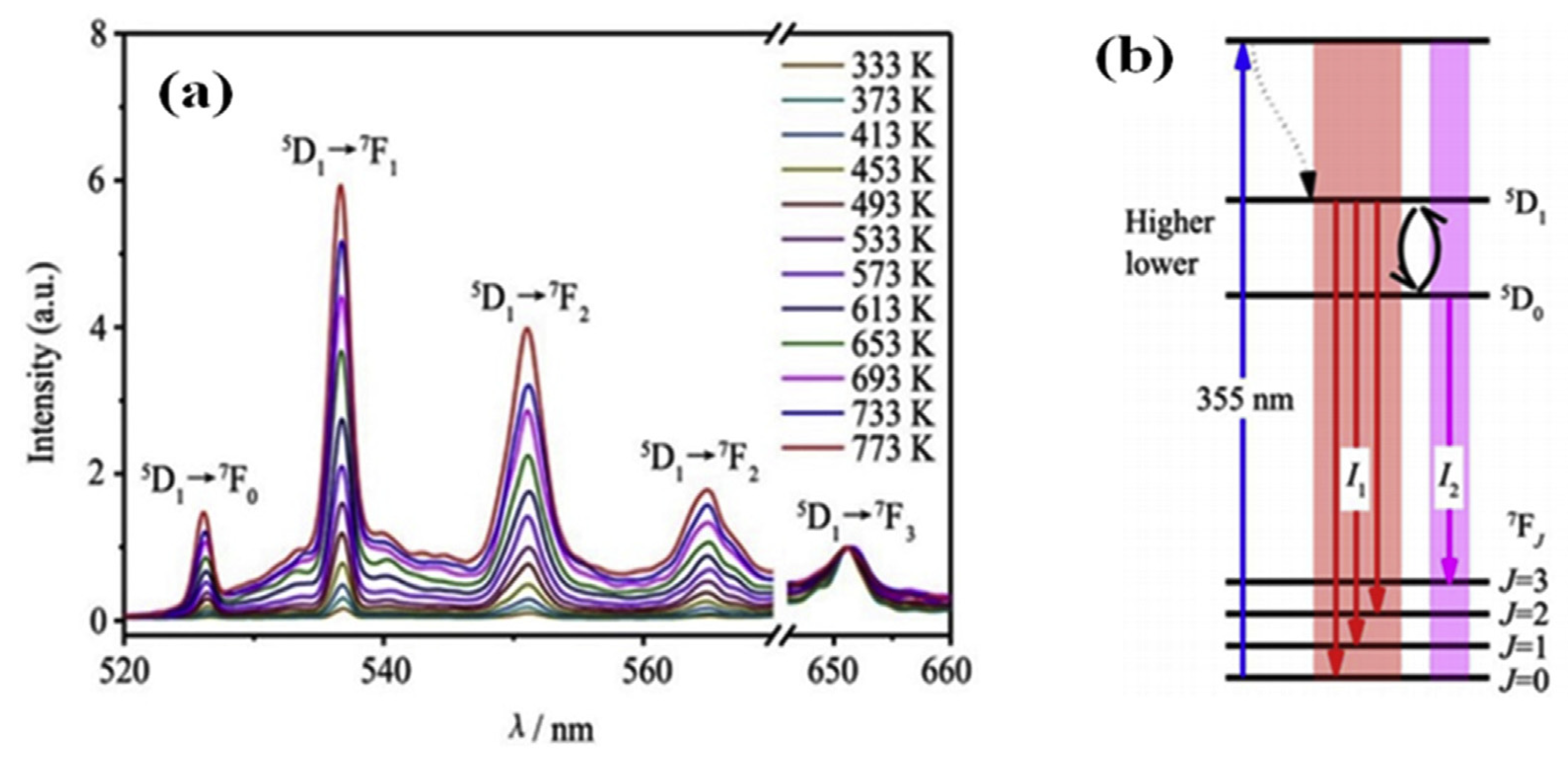
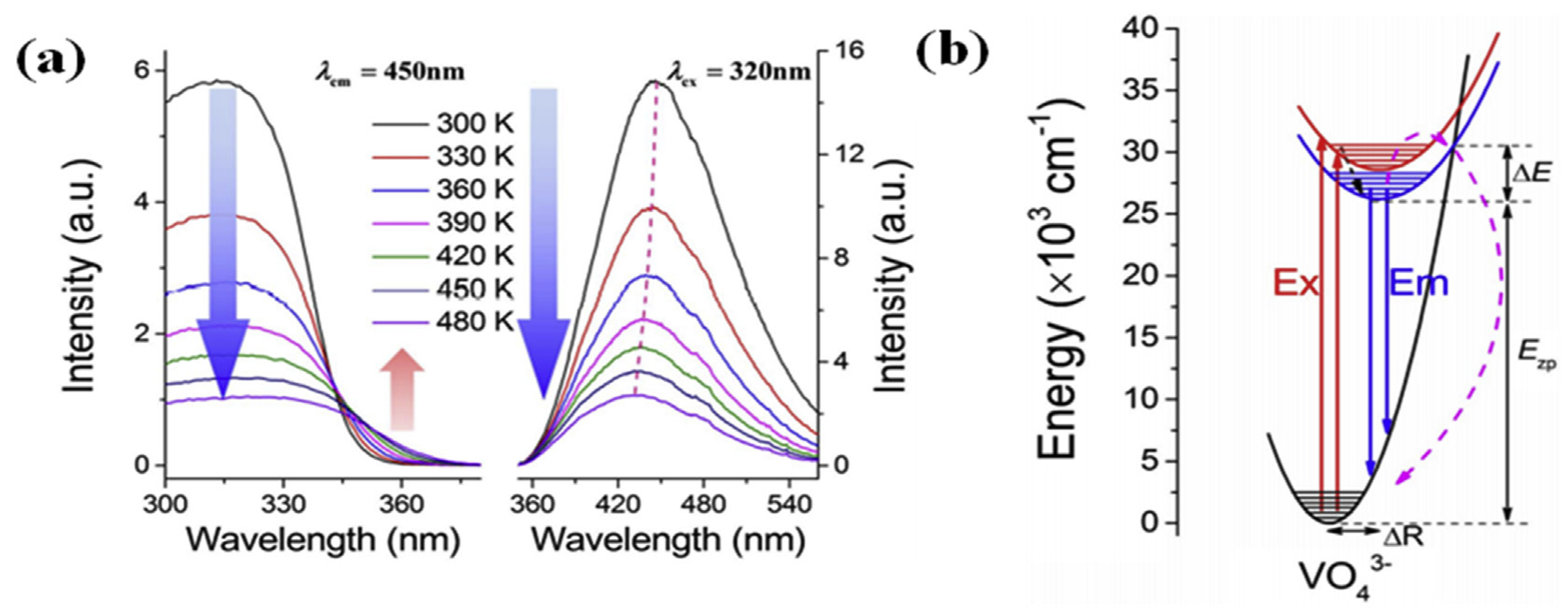
| Performance Criteria | Lanthanide Ion-Doped Oxides | Transition Metal Ion-Doped Oxides |
|---|---|---|
| Luminescence mechanism | Mainly f-f transitions within the 4f electron layer (some containing f-d transitions, such as Ce3+ and Eu2+), which are shielded by outer electrons and less affected by the matrix | Mainly the d-d transition or charge transfer transition of 3d electrons (such as between Mn4+ and ligands); 3d electrons have no outer shielding and are significantly affected by crystal fields |
| Spectral characteristic | Narrowband emission (half width usually <50 nm), fixed wavelength (such as Eu3+ red light~615 nm and Tb3+ green light~545 nm), and high color purity | Broadband emission (peak width at half maximum mostly >100 nm), with wavelength varying with the intensity of the matrix crystal field (e.g., Cr3+ can emit red to near-infrared light) |
| Quantum efficiency | When there is no spin barrier, the f-d transition quantum efficiency of Ce3+ approaches 100%. The f-f transition has a slightly lower spin barrier, but it is still higher than most TM ions | Moderate (d-d transitions are mostly spin-forbidden), such as Cr3+ with a quantum efficiency of about 70% in Al2O3, which is greatly affected by defect quenching |
| Synthesis Method | Key Technical Points | Performance Advantages |
|---|---|---|
| High-temperature solid-phase synthesis method | Raw material pretreatment, homogenization of mixed materials, and precise control of high-temperature reactions | Suitable for refractory materials (such as magnesia alumina spinel); it can maintain structural stability at high temperatures above 1600 °C and has strong chemical inertness |
| Sol-gel method | Low-temperature hydrolysis (<100 °C) forms a uniform sol, combined with heat treatment doping | Nanoparticle size deviation < 5%; controllable composition |
| Hydrothermal/solvothermal method | Crystallization at 120–200 °C regulates crystallinity and enhances photocatalytic activity | Improves material crystallinity and stability (photocatalytic efficiency increased by 3.7 times) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Liu, J.; Zhou, S.; Li, Z.; Yuan, M.; Shen, J.; Zhang, Y.; Ye, R. Methods and Applications of Lanthanide/Transition Metal Ion-Doped Luminescent Materials. Molecules 2025, 30, 3470. https://doi.org/10.3390/molecules30173470
Chen X, Liu J, Zhou S, Li Z, Yuan M, Shen J, Zhang Y, Ye R. Methods and Applications of Lanthanide/Transition Metal Ion-Doped Luminescent Materials. Molecules. 2025; 30(17):3470. https://doi.org/10.3390/molecules30173470
Chicago/Turabian StyleChen, Xiaoyi, Jiaqi Liu, Shujing Zhou, Zan Li, Min Yuan, Jinghui Shen, Yifan Zhang, and Rongrong Ye. 2025. "Methods and Applications of Lanthanide/Transition Metal Ion-Doped Luminescent Materials" Molecules 30, no. 17: 3470. https://doi.org/10.3390/molecules30173470
APA StyleChen, X., Liu, J., Zhou, S., Li, Z., Yuan, M., Shen, J., Zhang, Y., & Ye, R. (2025). Methods and Applications of Lanthanide/Transition Metal Ion-Doped Luminescent Materials. Molecules, 30(17), 3470. https://doi.org/10.3390/molecules30173470







