Between Heritage Conservation and Forensic Science: An Analytical Study of Personal Items Found in Mass Graves of the Francoism (1939–1956) (Spain)
Abstract
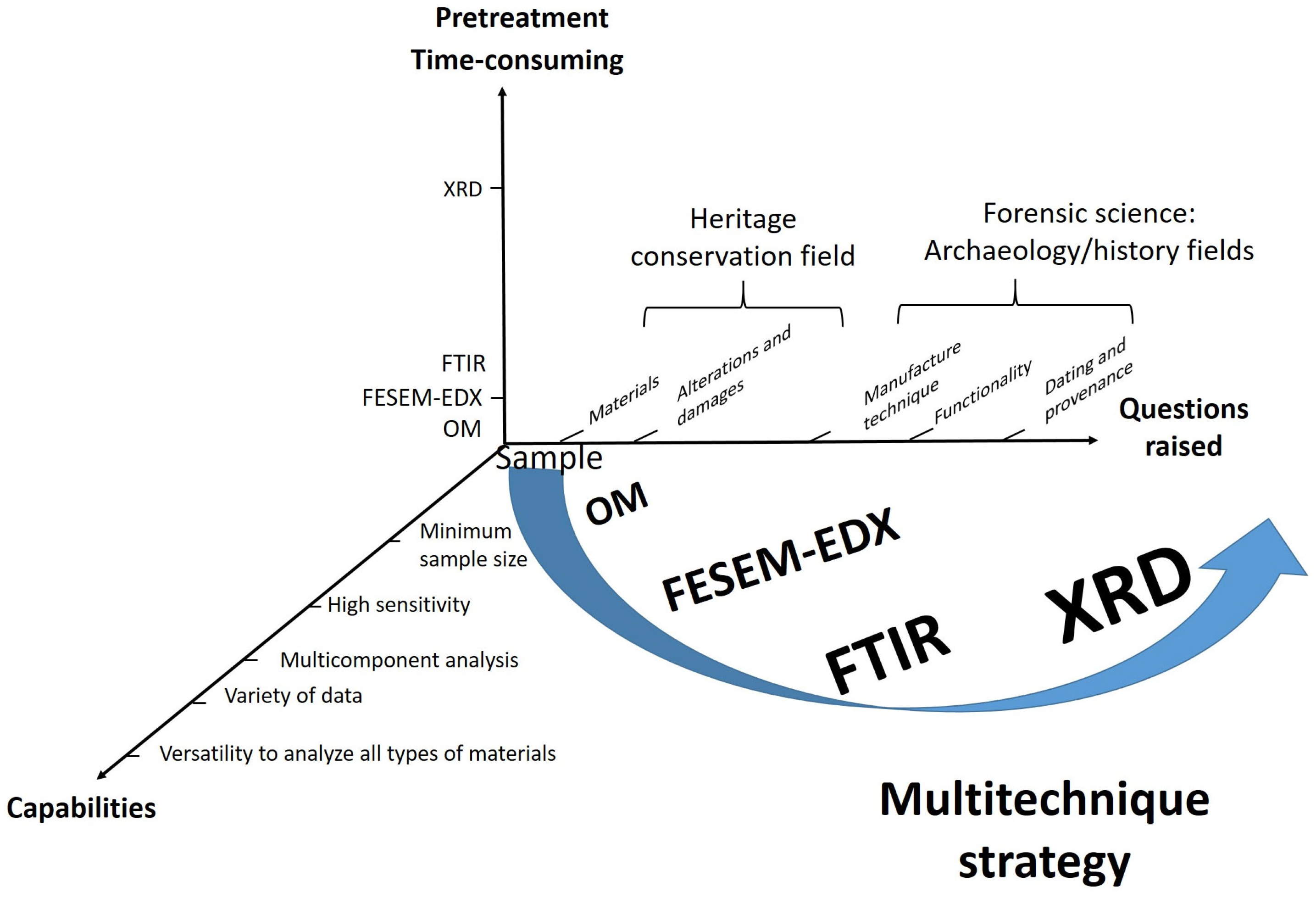
1. Introduction
2. Results
2.1. The Collection of Personal Items of the Mass Graves of Paterna
2.2. Identification of Materials
2.2.1. Elemental Composition of Metallic Objects
2.2.2. Mineralogical and Micromorphological Characterization of Inorganic Materials
2.2.3. Molecular Structure and Micromorphological Characterization of Organic Materials
2.3. Adipoceres
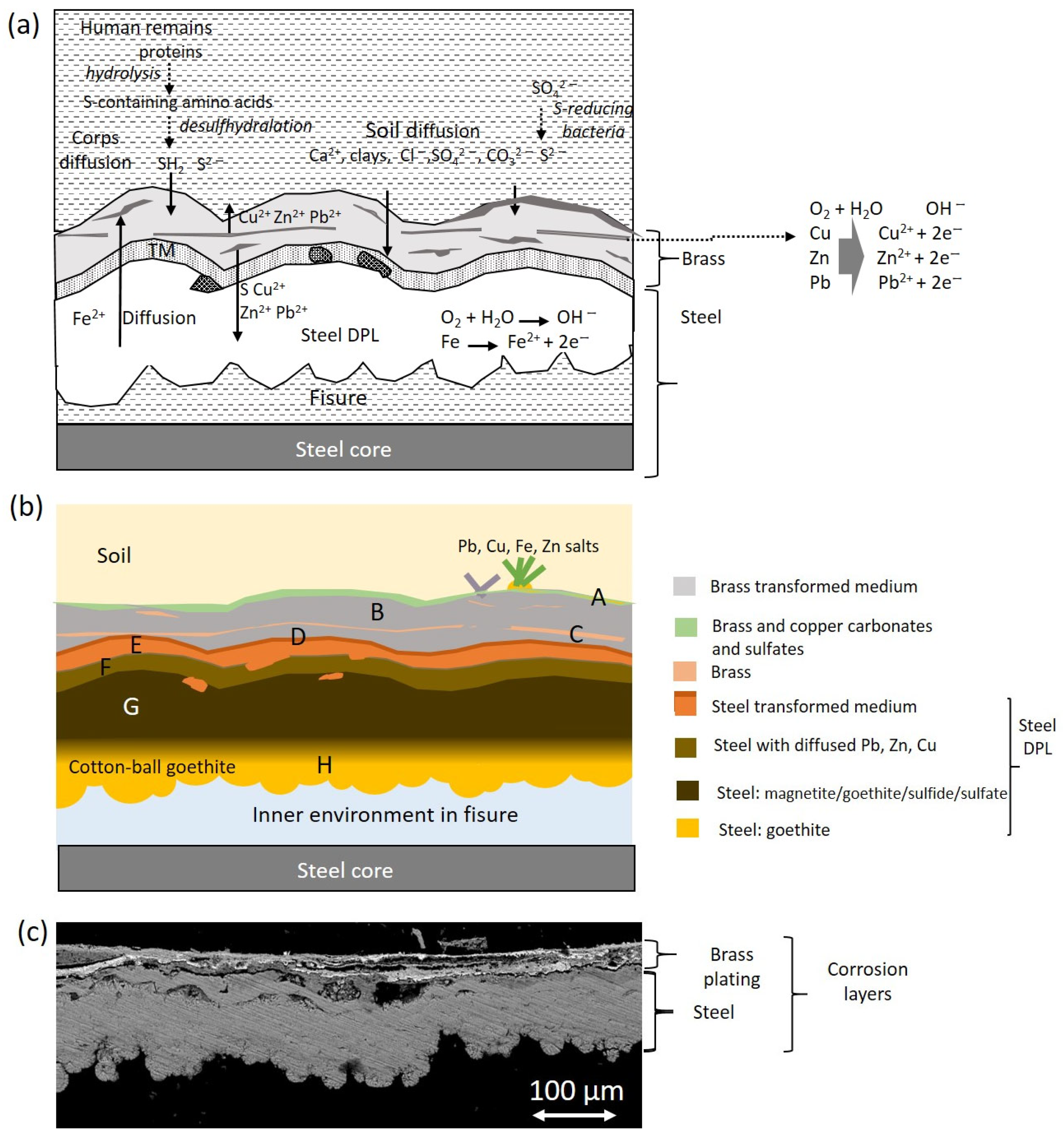
2.4. Corrosion Products
2.5. Fungal Biodeterioration
2.6. Grave Soil
3. Discussion
3.1. Connecting the Materials Identified and the Manufacturing Technique
3.2. Alteration Processes
4. Materials and Methods
4.1. Optical Microscope
4.2. FESEM–EDX
4.3. FTIR Spectroscopy
4.4. XRD
4.5. Preparation of Samples
4.6. Ethical Considerations and Procedures
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| l’ETNO | Museums of Ethnography |
| GC–MS | Gas chromatography–mass spectrometry |
| OM | Optical microscopy |
| SEM | Scanning electron microscopy |
| FESEM–EDX | Field emission X-ray microanalysis scanning electron microscopy |
| FTIR | Fourier transform infrared spectroscopy |
| XRD | X-ray diffraction |
| C8:0 | Octanodioic acid |
| C9:0 | Nonanodioic acid |
| C14:0 | Tetradecanoic acid |
| C16:0 | Hexadecanoic acid |
| C18:0 | Octadecanoic acid |
| C20:0 | Eicosanoic acid |
| ACC | Amorphous calcium carbonate |
| ZAF | Z—stopping power, backscattering factor and X-ray production power (Z), absorption (A) and fluorescence (F) |
| CF | Casein formaldehyde |
| DPL | Dense product layer |
| TM | Transition medium |
| ATR | Attenuated total reflectance |
| FR-DTGS | Fast recovery deuterated triglycine sulfate |
References
- Gabarda, V. El cementeri de Paterna com a exemple de monumentalització popular, Postguerres/Aftermaths of war. In Postguerres/Aftermaths of War; Abelló, T., Cattini, G.C., Gavin, V., Ibarz, J., Santacana, C., Solé, O., Vives, A., Eds.; Universitat de Barcelona: Barcelona, Spain, 2020; pp. 169–187. [Google Scholar]
- Ley 52/2007, de 26 de Diciembre, por la que se Reconocen y Amplían Derechos y se Establecen Medidas en Favor de Quienes Padecieron Persecución o Violencia Durante la Guerra civil y la dictadura [Law 52/2007, 26th December, Which Recognizes and Expands Rights and Establishes Measures in Favor of Those Who Suffered Persecution or Violence During the Civil War and the Dictatorship]. Available online: https://www.boe.es/eli/es/l/2007/12/26/52/con (accessed on 24 February 2025).
- Orden PRE/2568/2011, de 26 de Septiembre, por la que se Publica el Acuerdo del Consejo de Ministros de 23 de Septiembre de 2011, por el que se Ordena la Publicación en el Boletín Oficial del Estado del Protocolo de Actuación en Exhumaciones de Víctimas de la Guerra Civil y la Dictadura [Order of September 26, Which Publishes the Agreement of the Council of Ministers of September 23, 2011, Which Orders the Publication in the Official State Gazette of the Protocol for Action in Exhumations of Victims of the Civil War and the Dictatorship]. Available online: https://www.boe.es/eli/es/o/2011/09/26/pre2568 (accessed on 24 February 2025).
- Ley 14/2017, de 10 de Noviembre, de Memoria Democrática y Para la Convivencia de la Comunitat Valenciana. [Law 14/2017, of November 10, on Democratic Memory and for the Coexistence of the Valencian Community]. Available online: https://www.boe.es/eli/es-vc/l/2017/11/10/14/con (accessed on 24 February 2025).
- Decreto 1/2023, de 13 de enero, del Consell, por el cual se Desarrolla la Ley 14/2017, de 10 de Noviembre, de la Generalitat, de Memoria Democrática y para la Convivencia de la Comunitat Valenciana. [Decree 1/2023, of 13 January, of the Consell, by which Law 14/2017, of November 10, of the Generalitat, on Democratic Memory and for the Coexistence of the Valencian Community, Is Developed]. Available online: https://dogv.gva.es/es/eli/es-vc/d/2023/01/13/1/ (accessed on 24 February 2025).
- Díaz-Ramoneda, E.; Vila, A.; Sancho, S.; Calpe, A.; Iglesias, J.; Mezquida, M. Les fosses de Paterna, testimonis de la maquinària repressiva del règim franquista al País Valencià. Rev. D’arqueologia Ponent 2021, 31, 239–258. [Google Scholar] [CrossRef]
- Múgica, J.; Pasíes, T.; Canal, R. Las fosas de Paterna: Un proyecto de conservación sostenible y puesta en valor en el Museu de Prehistòria de València. PH Boletín Inst. Andal. Patrim. Histórico 2023, 31, 320–321. [Google Scholar] [CrossRef]
- Múgica, J. Las fosas de Paterna: Un proyecto de Conservación y Puesta en Valor en el Museu de Prehistòria de València. Master’s Thesis, Universitat Politècnica de València (UPV), Valencia, Spain, 25 July 2023. Available online: https://riunet.upv.es/entities/publication/4462e493-882e-45bd-8013-a8625ca3569c (accessed on 9 April 2025).
- Moreno, A.; Vizcaíno, A.; Mezquida, M.; García, M.J.; Gadea, I. (Eds.) Mass Graves of Francoism. Archaeology, Anthropology and Memory; Diputació de València-Museu de Prehistòria de València: Valencia, Spain, 2023; Available online: https://mupreva.org/web_mupreva_dedalo/publicaciones/1631/va (accessed on 9 April 2025).
- Moreno, A.; Vizcaíno, A.; Mezquida, M.; Ayán, X.M. Las Fosas del Franquismo. Arqueología, Antropología y Memoria; Diputació de València-Museu de Prehistòria de València: Valencia, Spain, 2023; Available online: http://mupreva.org/pub/1600/va (accessed on 9 April 2025).
- Moreno, A.; Mezquida, M.; Schwab, M.E. Exhumaciones de fosas comunes en el País Valenciano: 10 años de intervenciones científicas. Ebre 38. Rev. Int. Guerra Civil. 2021, 11, 125–152. [Google Scholar] [CrossRef]
- Gabarda, V. El Cost Humà de la Repressió al País Valencià (1936–1956); Universitat de València: València, Spain, 2020. [Google Scholar]
- Arqueologia de la Memòria. Arqueologia de la Memòria. Les Fosses de Paterna. Available online: https://mupreva.org/exposiciones/79/va (accessed on 9 April 2025).
- Museo de Prehistoria de Valencia. Available online: https://mupreva.org/home/?q=va (accessed on 9 April 2025).
- Moreno, A.; Ariza, E.; Mezquida, M. Tratamiento, significado y gestión de la cultura material exhumada en contextos de represión y conflicto: Las fosas del franquismo. In Arqueología y Memoria Histórica: Retos Actuales de la Investigación; Rodríguez, O., Ed.; University of Seville: Seville, Spain, 2025; pp. 57–91. [Google Scholar]
- Moses, R.J. Experimental adipocere formation: Implications for adipocere formation on buried bone. J. Forensic Sci. 2012, 57, 589–595. [Google Scholar] [CrossRef]
- Fiedler, S.; Graw, M. Decomposition of buried corpses, with special reference to the formation of adipocere. Naturwissenschaften 2003, 90, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Takatori, T.; Ishiguro, N.; Tarao, H.; Matsumiya, H. Microbial production of hydroxy and oxo fatty acids by several microorganism as a model of adipocere formation. Forensic Sci. Int. 1986, 32, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.L.; Stuart, B.H.; Dent, B.B. The effect of the burial environment on adipocere formation. Forensic Sci. Int. 2005, 154, 24–34. [Google Scholar] [CrossRef]
- Forbes, S.L.; Stuart, B.H.; Dent, B.B. The identification of adipocere in grave soils. Forensic Sci. Int. 2002, 127, 225–230. [Google Scholar] [CrossRef]
- Takatori, T. Investigations on the mechanism of adipocere formation and its relation to other biochemical reactions. Forensic Sci. Int. 1996, 80, 49–61. [Google Scholar] [CrossRef]
- Lerchi, A.; Krap, T.; Eppenberger, P.; Pedergnana, A. Implementation of adipocere fingerprinting in archaeology by applying a forensic approach. TrAC Trends Anal. Chem. 2022, 157, 116801. [Google Scholar] [CrossRef]
- Magni, P.A.; Lawn, J.; Guareschi, E.E. A practical review of adipocere: Key findings, case studies and operational considerations from crime scene to autopsy. J. Forensic Leg. Med. 2021, 78, 102109. [Google Scholar] [CrossRef] [PubMed]
- Kumar, T.M.; Monteiro, F.N.; Bhagavath, P.; Bakkannavar, S.M. Early adipocere formation: A case report and review of literature. J. Forensic Leg. Med. 2009, 16, 475–477. [Google Scholar] [CrossRef]
- Ubelaker, D.H.; Zarenko, K.M. Adipocere: What is known after over two centuries of research. Forensic Sci. Int. 2011, 208, 167–172. [Google Scholar] [CrossRef]
- Derrick, M.R.; Stulik, D.C.; Landry, J.M. Infrared Spectroscopy in Conservation Science; Getty Publications: Marina del Rey, CA, USA, 1999. [Google Scholar]
- Chalmers, H.G.M.; Edwards, M.H. (Eds.) Infrared and Raman Spectroscopy in Forensic Science; John Wiley & Sons: Hoboken, NY, USA, 2012. [Google Scholar]
- Gunasekaran, S.; Anbalagan, G.; Pandi, S. Raman and infrared spectra of carbonates of calcite structure. J. Raman Spectrosc. 2006, 37, 892–899. [Google Scholar] [CrossRef]
- Izzo, F.C.; Kratter, M.; Nevin, A.; Zendri, E. A critical review on the analysis of metal soaps in oil paintings. ChemistryOpen 2021, 10, 904–921. [Google Scholar] [CrossRef]
- Madejová, J.; Gates, W.P.; Petit, S. IR spectra of clay minerals. In Infrared and Raman Spectroscopies of Clay Minerals (Developments in Clay Science Series); Gates, W., Kloprogge, J.T., Madejova, J., Bergaya, F., Eds.; Elsevier: Oxford, UK, 2017; Volume 8, pp. 107–149. [Google Scholar]
- Pizzo, B.; Pecoraro, E.; Alves, A.; Macchioni, N.; Rodrigues, J.C. Quantitative evaluation by attenuated total reflectance infrared (ATR-FTIR) spectroscopy of the chemical composition of decayed wood preserved in waterlogged conditions. Talanta 2015, 131, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Schwanninger, M.J.C.R.; Rodrigues, J.C.; Pereira, H.; Hinterstoisser, B. Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vibrat Spectrosc. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Kostadinovska, M.; Spirovska, Z.J.; Taylor, T. A procedure for identifying cellulose fibers in paper artifacts. In Proceedings of the 4th International Virtual Conference on Advanced Scientific Results SCIECONF-2016, Ostrava, Czech Republic, 6–10 June 2016; Technical University of Ostrava: Ostrava, Czech Republic, 2016; pp. 156–162. [Google Scholar]
- Cucci, C.; Bartolozzi, G.; Marchiafava, V.; Picollo, M.; Richardson, E. Study of semi-synthetic plastic objects of historic interest using non-invasive total reflectance FT-IR. Microchem. J. 2016, 124, 889–897. [Google Scholar] [CrossRef]
- Van der Weerd, J.; Van Loon, A.; Boon, J.J. FTIR studies of the effects of pigments on the aging of oil. Stud. Conserv. 2005, 50, 3–22. [Google Scholar] [CrossRef]
- Filopoulou, A.; Vlachou, S.; Boyatzis, S.C. Fatty acids and their metal salts: A review of their infrared spectra in light of their presence in cultural heritage. Molecules 2021, 26, 6005. [Google Scholar] [CrossRef]
- Frye, G.C.; Thomas, M.M. Adsorption of organic compounds on carbonate minerals. 2. Extraction of carboxylic acids from recent and ancient carbonates. Chem. Geol. 1993, 109, 215–226. [Google Scholar] [CrossRef]
- Lu, Y.; Miller, J.D. Carboxyl stretching vibrations of spontaneously adsorbed and LB-transferred calcium carboxylates as determined by FTIR internal reflection spectroscopy. J. Colloid. Interface Sci. 2002, 256, 41–52. [Google Scholar] [CrossRef]
- Shurvell, H.F. Spectra—Structure Correlations in the Mid- and Far-Infrared. In Handbook of Vibrational Spectroscopy; Chalmers, J.M., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Watanabe, M.; Toyoda, E.; Handa, T.; Ichino, T.; Kuwaki, N.; Higashi, Y.; Tanaka, T. Evolution of patinas on copper exposed in a suburban area. Cor Sci. 2007, 49, 766–780. [Google Scholar] [CrossRef]
- Ashkenazi, D.; Inberg, A.; Langgut, D.; Hendler, N.; Cvikel, D. Brass–iron couple and brass–iron–wood ternary system of metal objects from the Akko 1 shipwreck (Israel). Cor Sci. 2016, 110, 228–241. [Google Scholar] [CrossRef]
- Aghamohammadi, H.; Jamaati, R. Effect of cold single-roll drive rolling on the microstructural evolution and mechanical properties of ferritic stainless steel. J. Mat. Res. Technol. 2024, 29, 2679–2688. [Google Scholar] [CrossRef]
- Thalib, S.; Ikhsan, M.; Fonna, S.; Huzni, S.; Ridha, S. Identification of corrosion product on medium carbon steel under the exposure of Banda Aceh’s atmosphere. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2003; Volume 352, p. 012004. [Google Scholar]
- Antunes, R.A.; Costa, I.; Faria, D.L.A. Characterization of corrosion products formed on steels in the first months of atmospheric exposure. Mat. Res. 2003, 6, 403–408. [Google Scholar] [CrossRef]
- Moreno, J.D.; Bonilla, M.; Adam, J.M.; Borrachero, M.V.; Soriano, L. Determining corrosion levels in the reinforcement rebars of buildings in coastal areas. A case study in the Mediterranean coastline. Constr. Build. Mater. 2015, 100, 11–21. [Google Scholar] [CrossRef]
- Fitzgerald, K.P.; Nairn, J.; Atrens, A. The chemistry of copper patination. Cor Sci. 1998, 40, 2029–2050. [Google Scholar] [CrossRef]
- Mindat. Available online: https://www.mindat.org/gm/1144 (accessed on 18 April 2025).
- Grevey, A.L.; Vignal, V.; Krawiec, H.; Ozga, P.; Peche-Quilichini, K.; Rivalan, A.; Mazière, F. Microstructure and long-term corrosion of archaeological iron alloy artefacts. Herit. Sci. 2020, 8, 57. [Google Scholar] [CrossRef]
- Gettens, R.J.; West Fitzhugh, E. Malachite and Green Verditer. In Artists’ Pigments: A Handbook of Their History and Characteristics; Roy, A., Ed.; Archetype: London, UK, 1993; Volume 2, pp. 183–202. [Google Scholar]
- Rruff DataBase. Available online: https://rruff.info/about/about_general.php (accessed on 5 March 2025).
- Doménech-Carbó, M.T.; Álvarez-Romero, C.; Doménech-Carbó, A.; Osete-Cortina, L.; Martínez-Bazán, M.L. Microchemical surface analysis of historic copper-based coins by the combined use of FIB-FESEM-EDX, OM, FTIR spectroscopy and solid-state electrochemical techniques. Microchem. J. 2019, 148, 573–581. [Google Scholar] [CrossRef]
- Naumann, A.; Navarro-González, M.; Peddireddi, S.; Kües, U.; Polle, A. Fourier transform infrared microscopy and imaging: Detection of fungi in wood. Fungal Genet. Biol. 2005, 42, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Fischer, G.; Braun, S.; Thissen, R.; Dott, W. FT-IR spectroscopy as a tool for rapid identification and intra-species characterization of airborne filamentous fungi. J. Microbiol. Meth. 2006, 64, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.A.; Maish, J.P. A Lydian Bed of Iron, Bronze and Copper-Technical Examination of a Metallurgical Masterpiece. Stud. Conserv. 2010, 55, 3–19. [Google Scholar] [CrossRef]
- Edwards, J. Coating and Surface Treatment Systems for Metals; Finishing Publications Ltd. and ASM International: Geauga County, OH, USA, 1997; pp. 34–38. [Google Scholar]
- Sheasby, P.G.; Pinner, R. The Surface Treatment and Finishing of Aluminum and Its Alloys, 6th ed.; ASM International & Finishing Publications: Geauga County, OH, USA, 2001; Volume 2. [Google Scholar]
- Schuman, T.P. Protective Coatings for Aluminum Alloys. In Handbook of Environmental Degradation of Materials, 3rd ed.; Kutz, M., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Liang, J.; Yue, W.; Gu, Y.; Liu, J.; Wang, C.; Ma, H. Improving Corrosion Resistance and Corrosive Wear Resistance of Aluminum Alloy Drill Pipe by Surface Nanocrystallization and Micro-arc Oxidation. J. Mater. Eng. Perform. 2018, 27, 4462–4472. [Google Scholar] [CrossRef]
- Ingo, G.M.; Riccucci, C.; Faraldi, F.; Pascucci, M.; Messina, E.; Fierro, G.; Di Carlo, G. Roman sophisticated surface modification methods to manufacture silver counterfeited coins. Appl. Surf. Sci. 2017, 421, 109–119. [Google Scholar] [CrossRef]
- Hiscox, G.D.; Hopkins, A.A. Recetario Industrial [Industrial Recipe Book]; Club de Golf Mexico: Mexico City, Mexico, 1997. [Google Scholar]
- Doménech-Carbó, M.T.; Di Turo, F.; Montoya, N.; Catalli, F.; Doménech-Carbó, A.; De Vito, C. FIB-FESEM and EMPA results on Antoninianus silver coins for manufacturing and corrosion processes. Sci. Rep. 2018, 8, 10676. [Google Scholar] [CrossRef]
- Keturakis, C.J.; Notis, B.; Blenheim, A.; Miller, A.C.; Pafchek, R.; Notis, M.R.; Wachs, I.E. Analysis of corrosion layers in ancient Roman silver coins with high resolution surface spectroscopic techniques. App Surf. Sci. 2016, 376, 241–251. [Google Scholar] [CrossRef]
- Di Fazio, M.; Di Turo, F.; Medeghini, L.; Fabrizi, L.; Catalli, F.; De Vito, C. New insights on medieval Provisini silver coins by a combination of non-destructive and micro-invasive techniques. Microchem. J. 2019, 144, 309–318. [Google Scholar] [CrossRef]
- Fabrizi, L.; Di Turo, F.; Medeghini, L.; Di Fazio, M.; Catalli, F.; De Vito, C. The application of non-destructive techniques for the study of corrosion patinas of ten Roman silver coins: The case of the medieval Grosso Romanino. Microchem. J. 2019, 145, 419–427. [Google Scholar] [CrossRef]
- Ingo, G.M.; Balbi, S.; de Caro, T.; Fragalà, I.; Angelini, E.; Bultrini, G. Combined use of SEM-EDS, OM and XRD for the characterization of corrosion products grown on silver roman coins. Appl. Phys. A 2006, 83, 493–497. [Google Scholar] [CrossRef]
- Mayer, R. Materiales y Técnicas del Arte [Materials and techniques of Art]; Ediciones Akal: Madrid, Spain, 2005; Volume 28. [Google Scholar]
- La Galvanotecnia y su Industria en España Entre los Años 1950–2005. [Electroplating and Its Industry in Spain Between 1950–2005] Institut Metalurgic de Cataluña. Available online: https://www.centrem.cat/ecomu/upfiles/aiasdestacat/fitxers/A1256.pdf (accessed on 9 March 2025).
- Julve, E. Historia de la galvanotecnia y técnicas afines. An. Química RSEQ 2009, 105, 227. [Google Scholar]
- Nudelman, F. Nacre biomineralisation: A review on the mechanisms of crystal nucleation. Semin. Cell Dev. Biol. 2015, 46, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Khouzani, M.F.; Chevrier, D.M.; Güttlein, P.; Hauser, K.; Zhang, P.; Hedin, N.; Gebauer, D. Disordered amorphous calcium carbonate from direct precipitation. Cryst. Eng. Comm. 2015, 17, 4842–4849. [Google Scholar] [CrossRef]
- Gueta, R.; Natan, A.; Addadi, L.; Weiner, S.; Refson, K.; Kronik, L. Local atomic order and infrared spectra of biogenic calcite. Angew. Chem. Int. Ed. 2007, 46, 291–294. [Google Scholar] [CrossRef]
- Chu, V.; Regev, L.; Weiner, S.; Boaretto, E. Differentiating between anthropogenic calcite in plaster, ash and natural calcite using infrared spectroscopy: Implications in archaeology. J. Archaeol. Sci. 2008, 35, 905–91186. [Google Scholar] [CrossRef]
- Beniash, E.; Aizenberg, J.; Addadi, L.; Weiner, S. Amorphous calcium carbonate transforms into calcite during sea-urchin larval spicule growth. Proc. R. Soc. Lond. Ser. B 1997, 264, 461–465. [Google Scholar] [CrossRef]
- Regev, L.; Poduska, K.M.; Addadi, L.; Weiner, S.; Boaretto, E. Distinguishing between calcites formed by different mechanisms using infrared spectrometry: Archaeological applications. J. Archaeol. Sci. 2010, 37, 3022–3029. [Google Scholar] [CrossRef]
- Doménech-Carbó, M.T.; Osete-Cortina, L. Another beauty of analytical chemistry: Chemical analysis of inorganic pigments of art and archaeological objects. ChemTexts 2016, 2, 1–50. [Google Scholar] [CrossRef][Green Version]
- Kaner, J.; Ioras, F.; Ratnasingam, J. Performance and stability of historic casein formaldehyde. e-Plastory J. Plast. Hist. 2017, 2. Available online: http://e-plastory.com (accessed on 10 March 2025).
- Erinoid Ltd. Erinoid Book of Dye Recipes; Lightpill Mills: Stroud, UK, 1951. [Google Scholar]
- Erinoid Ltd. Colour in Casein Booklet; Lightpill Mills: Stroud, UK, 1951. [Google Scholar]
- Comercio Histórico Valencia. Available online: https://comercioshistoricosdevalencia.blogspot.com/2020/06/productos-de-epoca-el-aceite-de-ricino.html (accessed on 10 March 2025).
- Algemesí en la Memoria. Available online: https://algemesienlamemoria.blogspot.com/ (accessed on 10 March 2025).
- Turgoose, S. The corrosion of archaeological iron during burial and treatment. Stud. Conserv. 1985, 30, 13–18. [Google Scholar] [CrossRef]
- Selwyn, L.S.; Sirois, P.I.; Argyropoulos, V. The corrosion of excavated archaeological iron with details on weeping and akaganeite. Stud. Conserv 1999, 44, 217–232. [Google Scholar] [CrossRef]
- Neff, D.; Dillmann, P.; Bellot-Gurlet, L.; Béranger, G. Corrosion of iron archaeological artefacts in soil: Characterisation of the corrosion system. Corros. Sci. 2005, 47, 515–535. [Google Scholar] [CrossRef]
- Chang, J.; Li, Y.; Lu, H. The Morphological Characteristics of Authigenic Pyrite Formed in Marine Sediments. J. Mar. Sci. Eng. 2022, 10, 1533. [Google Scholar] [CrossRef]
- Canfield, D.E.; Raiswell, R.; Bottrell, S. The Reactivity of Sedimentary iron minerals towards sufide. Am. J. Sci. 1992, 292, 659–683. [Google Scholar] [CrossRef]
- Burdige, D.J.; Komada, T. Iron redox cycling, sediment resuspension and role of sediments in low oxygen environments as sources of iron to water column. Mar. Chem. 2020, 223, 103793. [Google Scholar] [CrossRef]
- Doménech-Carbó, M.T.; Guasch-Ferré, N.; Álvarez-Romero, C.; Castillo-Belinchón, R.; Pérez-Mateo, S.; Buendía-Ortuño, M. Study of the Geological Context of the 7th–6th Century BC Phoenician Era Shipwreck “Mazarrón 2” (Murcia, Spain). Minerals 2024, 14, 778. [Google Scholar] [CrossRef]
- Hjelm-Hansen, N. Cleaning and stabilization of sulphide-corroded bronzes. Stud. Conserv. 1984, 29, 17–20. [Google Scholar] [CrossRef]
- Muros, V.; Scott, D.A. The occurrence of brochantite on archaeological bronzes: A case study from Lofkënd, Albania. Stud. Conserv. 2018, 63, 113–125. [Google Scholar] [CrossRef]
- Ioan, B.G.; Manea, C.; Hanganu, B.; Statescu, L.; Solovastru, L.G.; Manoilescu, I. The chemistry decomposition in human corpses. Rev. Chim. 2017, 68, 1352–1356. [Google Scholar] [CrossRef]
- Forbes, S.L. Decomposition Chemistry in a Burial Environment. In Soil Analysis in Forensic Taphonomy; Tibbett, M., Carter, D., Eds.; CRC Press, Taylor and Francis Group: New York, NY, USA, 2008; p. 203. [Google Scholar]
- Muros, V.; Parry, V.; Whyte, A. Conservation Laboratory Research Projects. Orient. Inst. News Notes 2002, 175, 1–8. [Google Scholar]
- Khoshnaw, F.; Gubner, R. (Eds.) Corrosion Atlas Case Studies. Part II: Corrosion Topics; Elsevier: Oxford, UK, 2019; pp. xliii–lxviii. [Google Scholar]
- Boyatzis, S.C.; Fragkos-Livanios, L.; Giannoulaki, M.; Filopoulou, A. Infrared spectroscopy reveals the reactivity of fatty acids on copper surfaces and its implications for cultural heritage objects. Herit. Sci. 2023, 11, 196. [Google Scholar] [CrossRef]
- Martínez, R.; Rocabayera, R. Els olis essencials. Conservació preventiva en el control microbiològic. Unicum. Rev. L’escola Super. Conserv. I Restauració Béns Cult. Catalunya 2011, 10, 135–141. Available online: https://unicum.escrbcc.cat/index.php/unicum/article/view/121/240 (accessed on 27 May 2025).
- Reed, S.J.B. Electron Microbe Analysis and Scanning Electron Microscopy in Geology; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Veritá, M.; Basso, R.; Wypysk, M.T.; Koestler, R.J. X-ray microanalysis of ancient glassy materials: A comparative study of wavelength dispersive and energy dispersive techniques. Archaeometry 1994, 36, 241–251. [Google Scholar] [CrossRef]
- Kuisma-Kursula, P. Accuracy. Precision and Detection Limits of SEM–WDS. SEM–EDS and PIXE in the Multi-Elemental Analysis of Medieval Glass. X-Ray Spectrom. 2000, 29, 111–118. [Google Scholar] [CrossRef]
- Moreno, A.; Mezquida, M.; Ariza, E. No solo cuerpos: La cultura material exhumada de las fosas del franquismo en Paterna. Sagvntvm. Papeles Lab. Arqueol. Valencia. 2021, 53, 213–235. [Google Scholar] [CrossRef]
- Moreno Martín, A.; Vizcaíno, T. Arqueología de la Memoria. Las Fosas de Paterna. Guía de la Exposición; Diputació de València, Museu de Prehistòria de València: Valencia, Spain, 2023; Available online: http://mupreva.org/pub/1632/va (accessed on 16 April 2025).








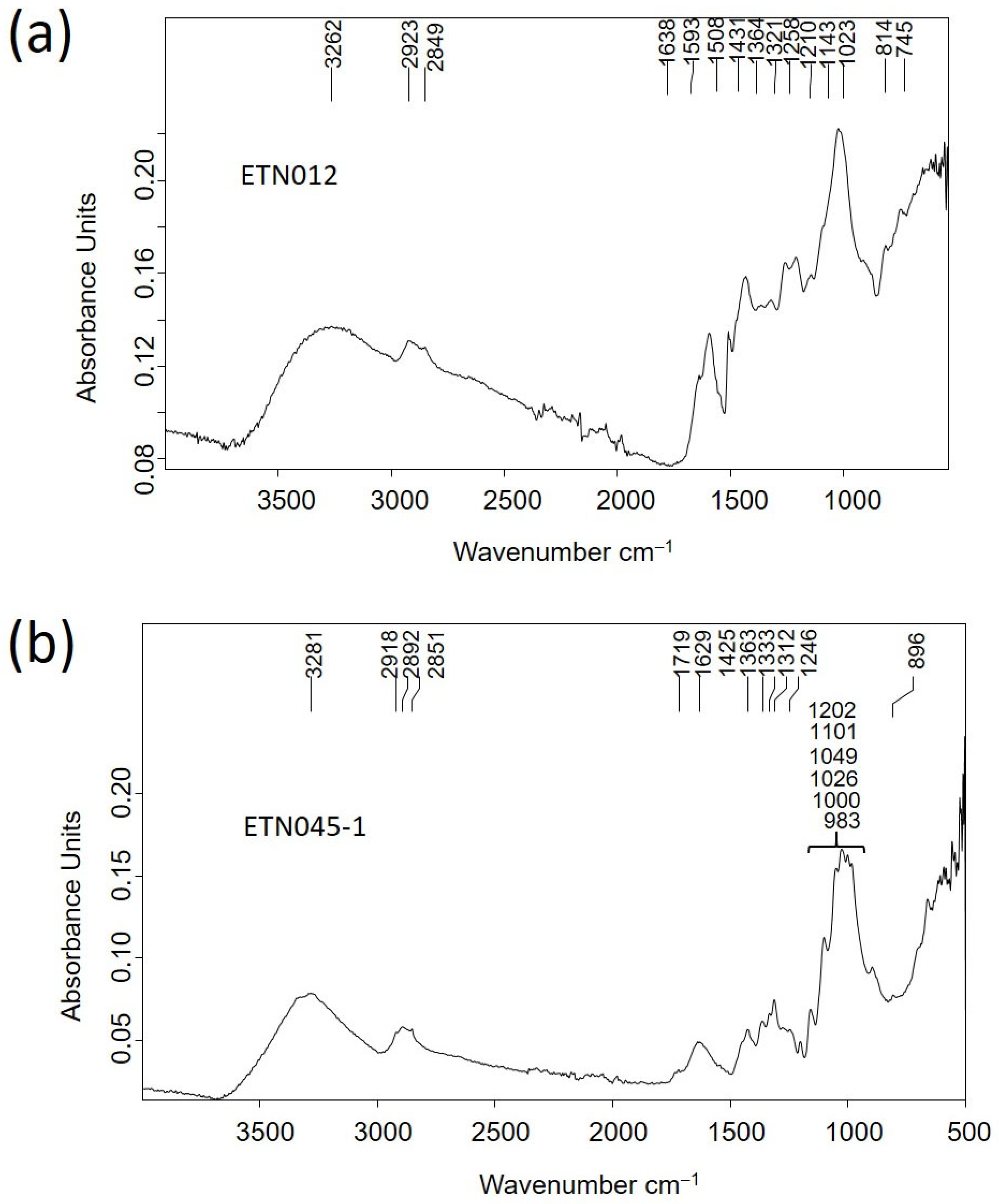


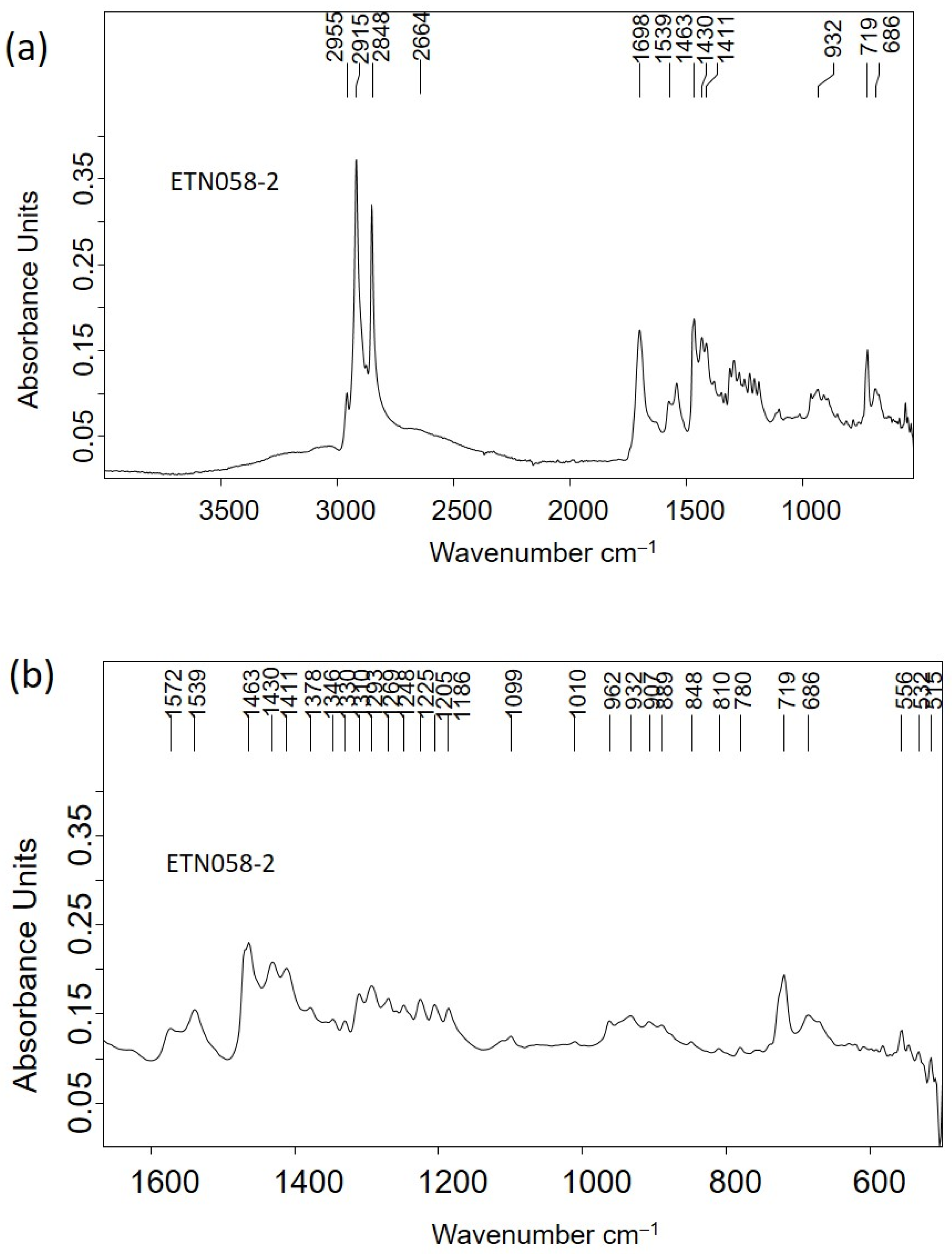
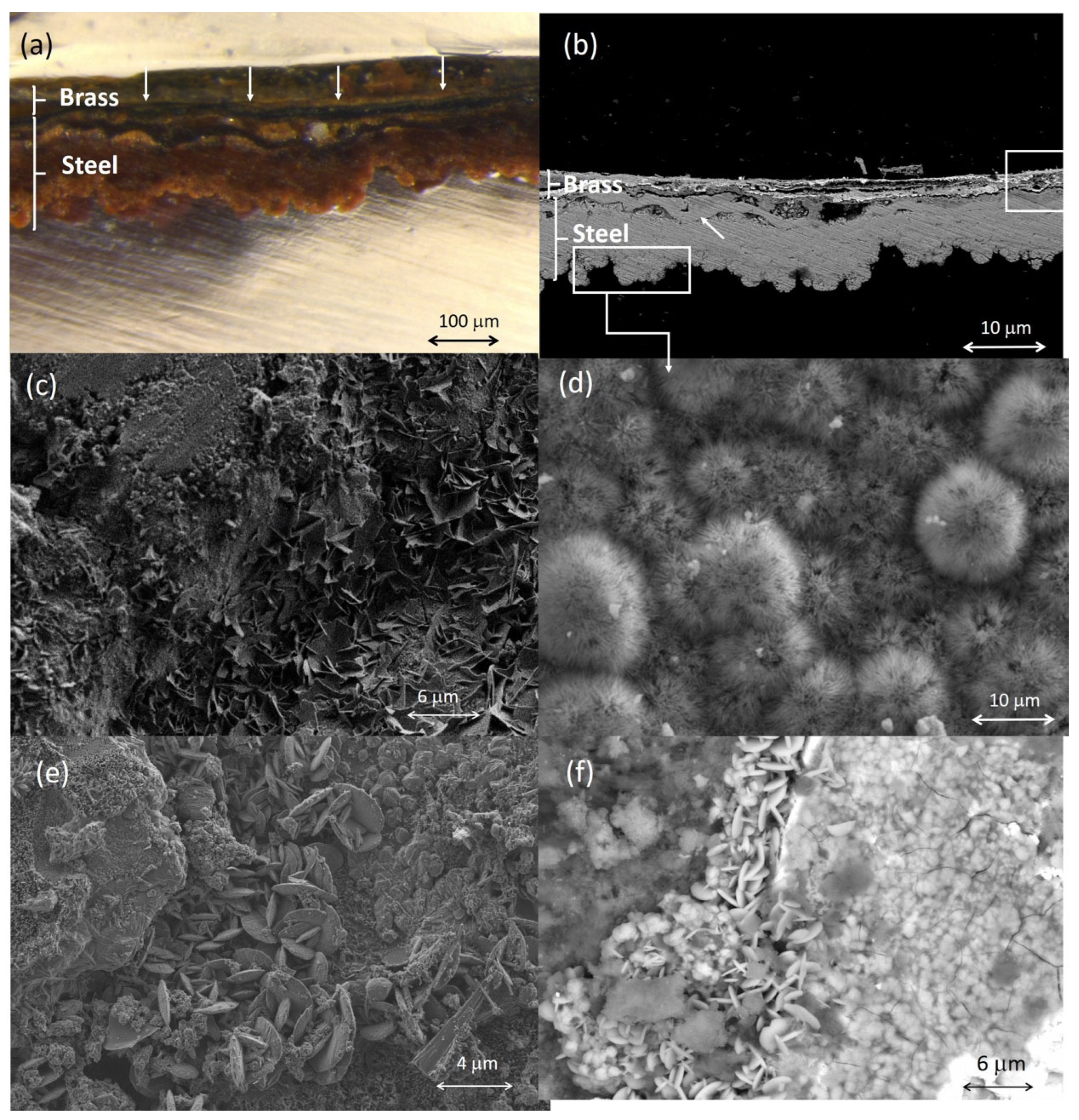
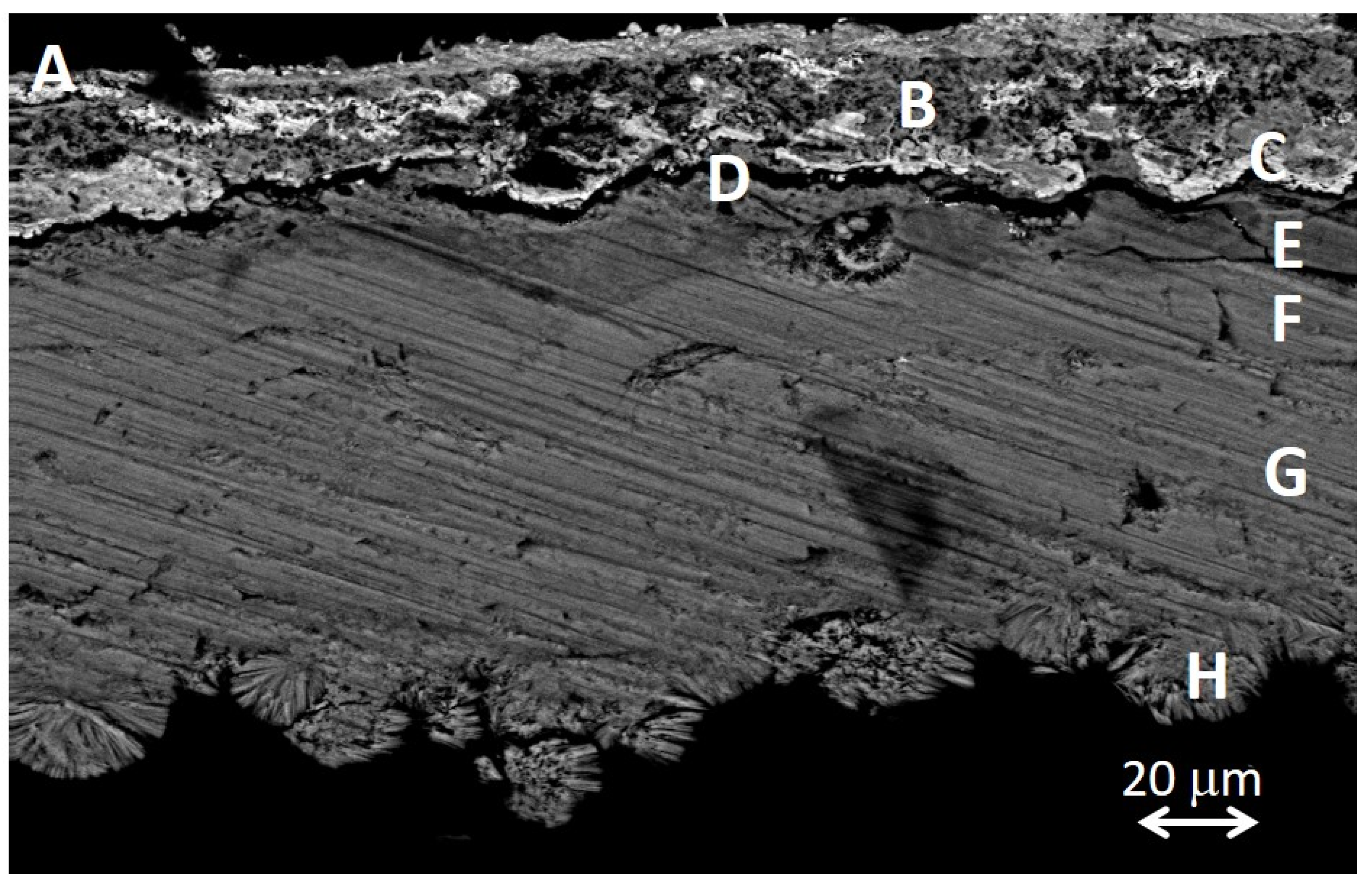
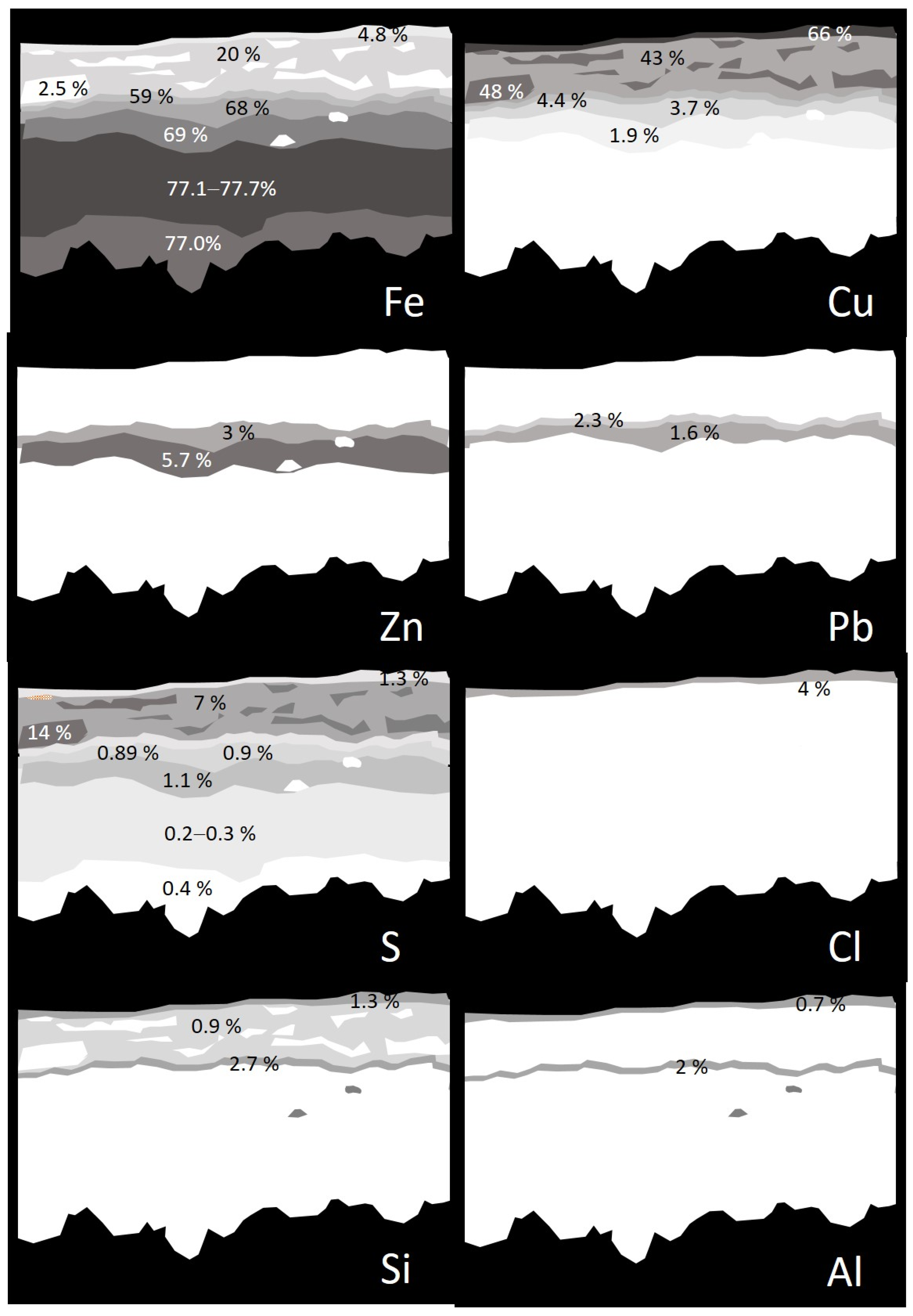


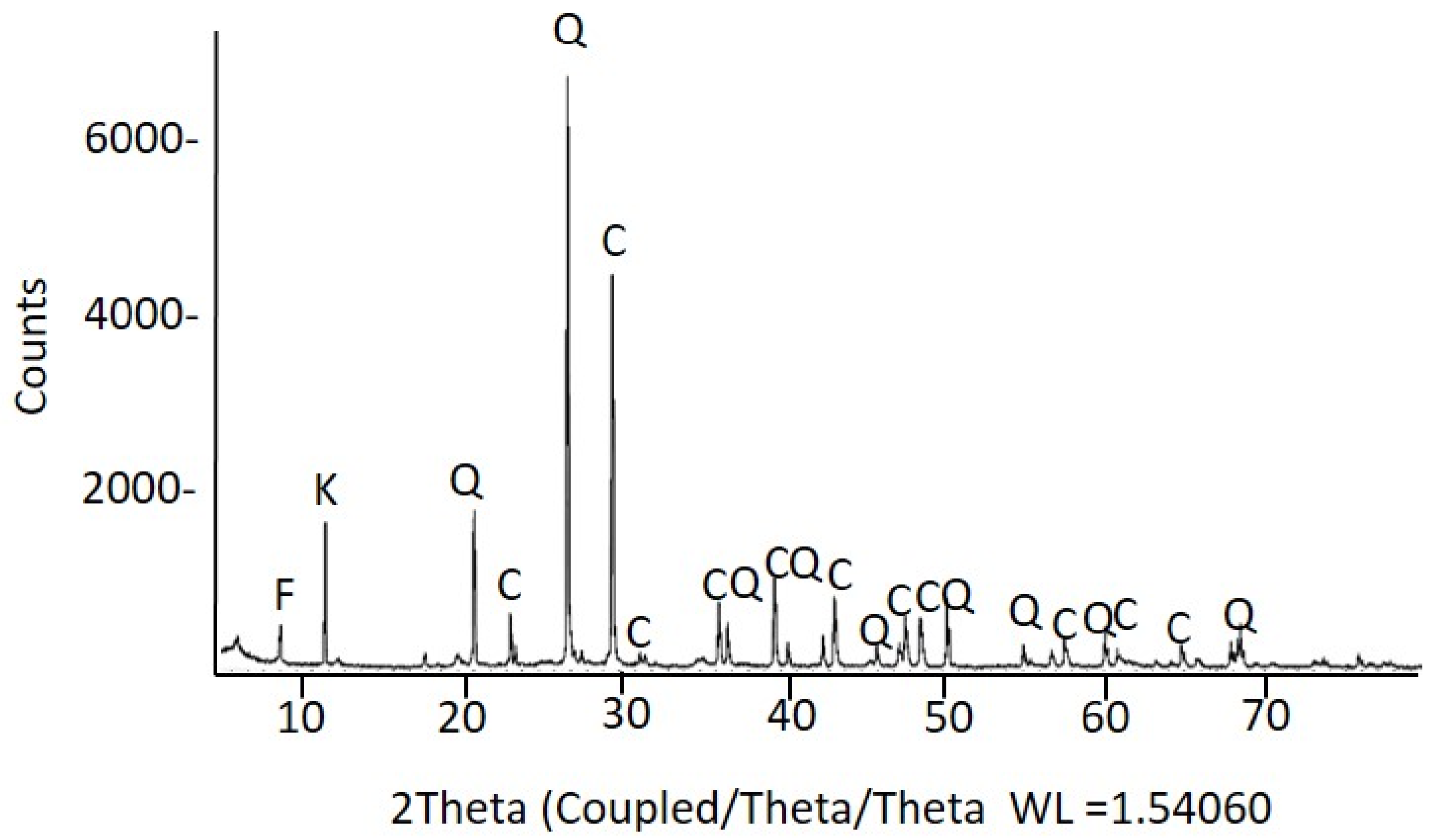

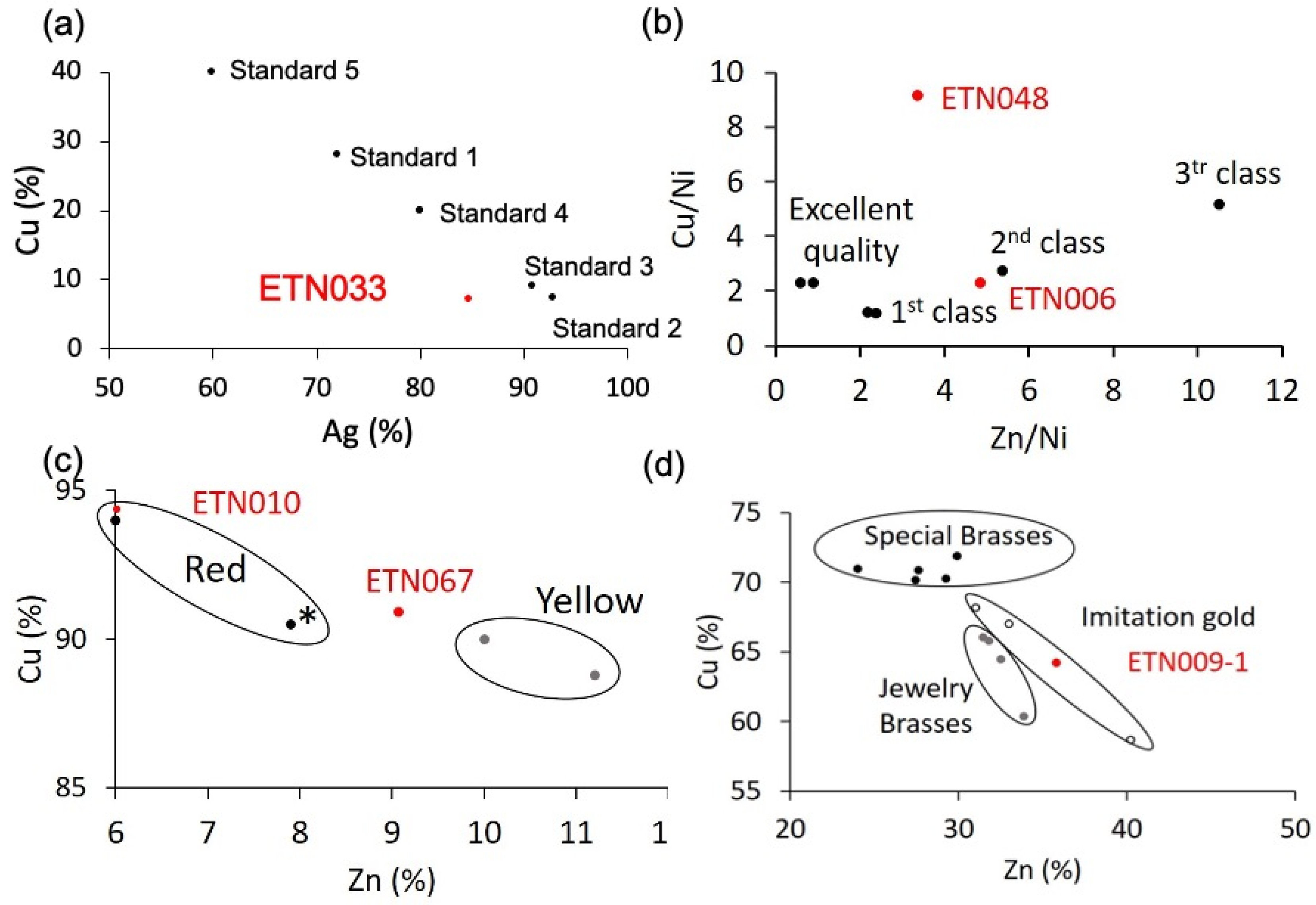
| Ref. | Elemental Composition (wt%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cu a | Zn | Ni | Pb | Sn | Fe a | Al a | Ag a | |
| ETN006 | 50.5 ± 3 | 19.1 ± 3 | 8.3 ± 0.5 | n.d. | n.d. | n.d. | n.d. | n.d. |
| ETN009-1 | 62.7 ± 0.9 | 37.3 ± 0.9 | n.d. | 5 ± 1 b | 15.9 ± 0.5 b | n.d. | n.d. | n.d. |
| ETN009-2 | n.d. | n.d. | n.d. | n.d. | n.d. | 72.1 ± 0.7 | n.d. | n.d. |
| ETN010-1 | 71.7 ± 0.5 | 4.6 ± 0.5 | n.d. | 1.51 b | n.d. | n.d. | n.d. | 4.5 b |
| ETN010-2 | n.d. | n.d. | n.d. | n.d. | n.d. | 77.7 ± 0.9 | n.d. | n.d. |
| ETN033-1 | 7.2 ± 0.4 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 84.7 ± 0.5 |
| ETN033-2 | 6.6 ± 0.5 | n.d. | n.d. | n.d. | n.d. | 2.7 ± 0.3 | n.d. | 28.3 ± 0.6 |
| ETN048 | 53.6 ± 0.9 | 11.8 ± 0.9 | 7.9 ± 0.6 | n.d. | n.d. | n.d. | n.d. | n.d. |
| ETN067 | 90.9 ± 0.7 | 9.1 ± 0.4 | n.d. | 1.6 b | n.d. | n.d. | n.d. | n.d. |
| MCP029 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 52.9 ± 0.2 a | n.d. |
| Assignment and IR Band Position (cm−1) | Material Identified |
|---|---|
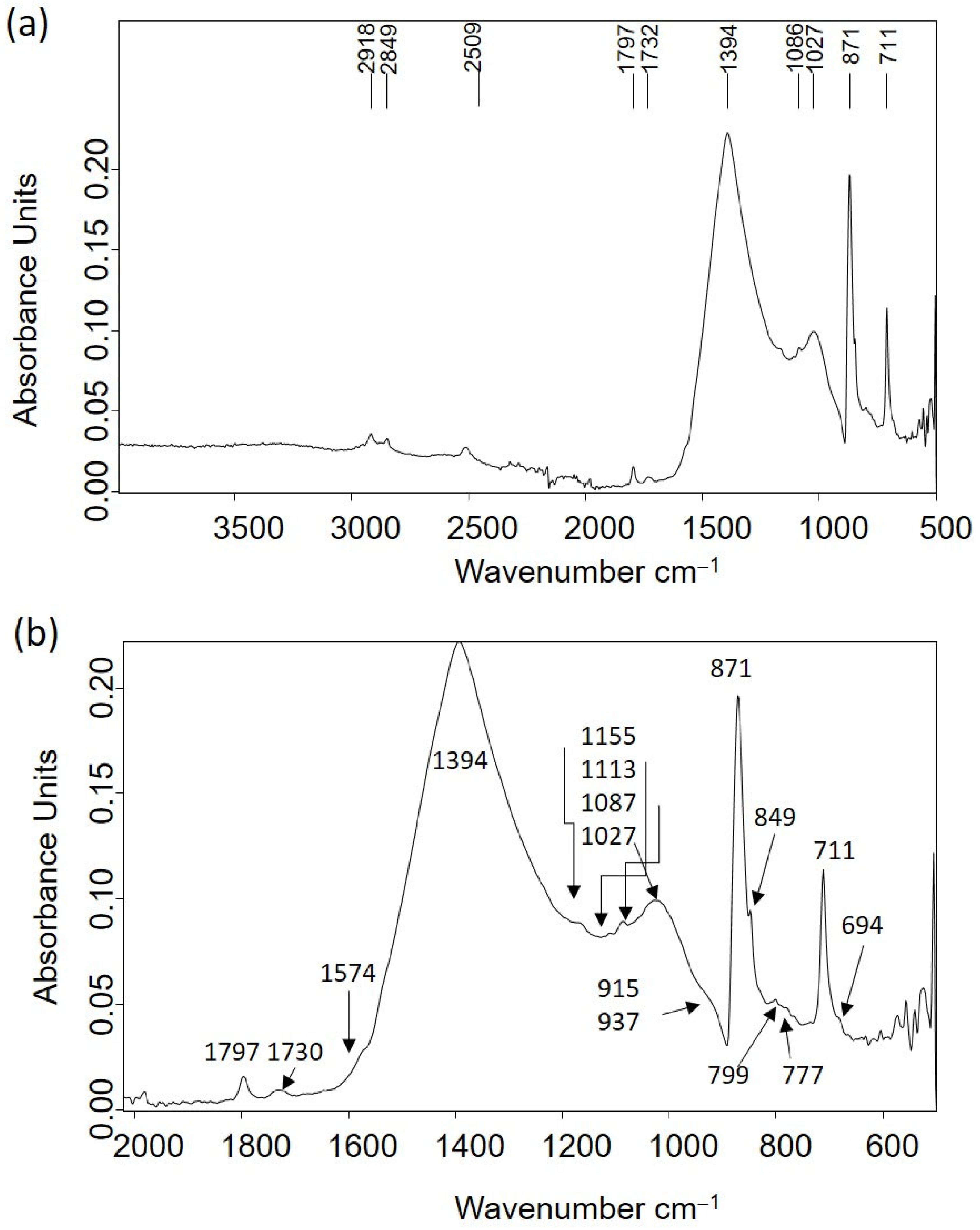
| 2ν2 + ν4 CO32− stretching (2509), ν1 + ν4 CO32− stretching (1797), ν3 CO32− asymmetric stretching (1398), ν2 CO32− asymmetric deformation (874), ν4 CO32− symmetric deformation (712) | Limestone |
| ν Si-O symmetric stretching (1113sh), ν Si-O-Si antisymmetric in-plane stretching 1027), δ (Al2OH) bending (915, 937), Si-O vibrations (748sh, 694) | Dioctahedral 1:1 clay |
| ν Si-O-Si stretching (1155sh, 1086sh), Si-O symmetric stretching (799, 777), Si-O of SiO4 symmetric bending (694) | Quartz |
| ν CH2, CH3 asymmetric and symmetric stretching (2918, 2851), ν C=O stretching (1730); calcium carboxylate stretching (1574) | Lipid matter |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doménech-Carbó, M.T.; Pasíes Oviedo, T.; Roca, R.C.; Múgica Mestanza, J. Between Heritage Conservation and Forensic Science: An Analytical Study of Personal Items Found in Mass Graves of the Francoism (1939–1956) (Spain). Molecules 2025, 30, 2783. https://doi.org/10.3390/molecules30132783
Doménech-Carbó MT, Pasíes Oviedo T, Roca RC, Múgica Mestanza J. Between Heritage Conservation and Forensic Science: An Analytical Study of Personal Items Found in Mass Graves of the Francoism (1939–1956) (Spain). Molecules. 2025; 30(13):2783. https://doi.org/10.3390/molecules30132783
Chicago/Turabian StyleDoménech-Carbó, María Teresa, Trinidad Pasíes Oviedo, Ramón Canal Roca, and Janire Múgica Mestanza. 2025. "Between Heritage Conservation and Forensic Science: An Analytical Study of Personal Items Found in Mass Graves of the Francoism (1939–1956) (Spain)" Molecules 30, no. 13: 2783. https://doi.org/10.3390/molecules30132783
APA StyleDoménech-Carbó, M. T., Pasíes Oviedo, T., Roca, R. C., & Múgica Mestanza, J. (2025). Between Heritage Conservation and Forensic Science: An Analytical Study of Personal Items Found in Mass Graves of the Francoism (1939–1956) (Spain). Molecules, 30(13), 2783. https://doi.org/10.3390/molecules30132783







