On the Diffusion of Ionic Liquids in ILs@ZIF-8 Composite Materials: A Density Functional Theory Study
Abstract
1. Introduction
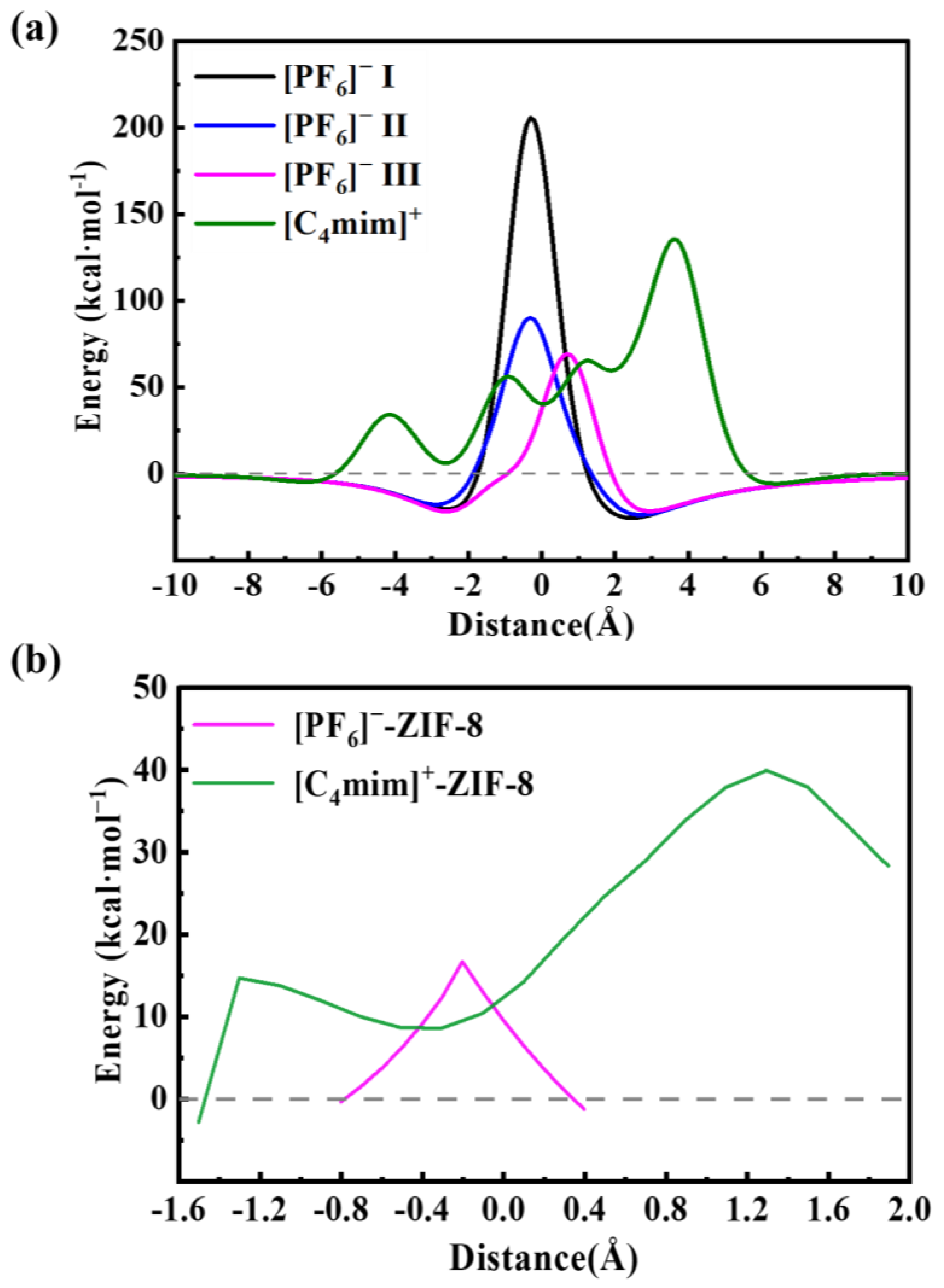
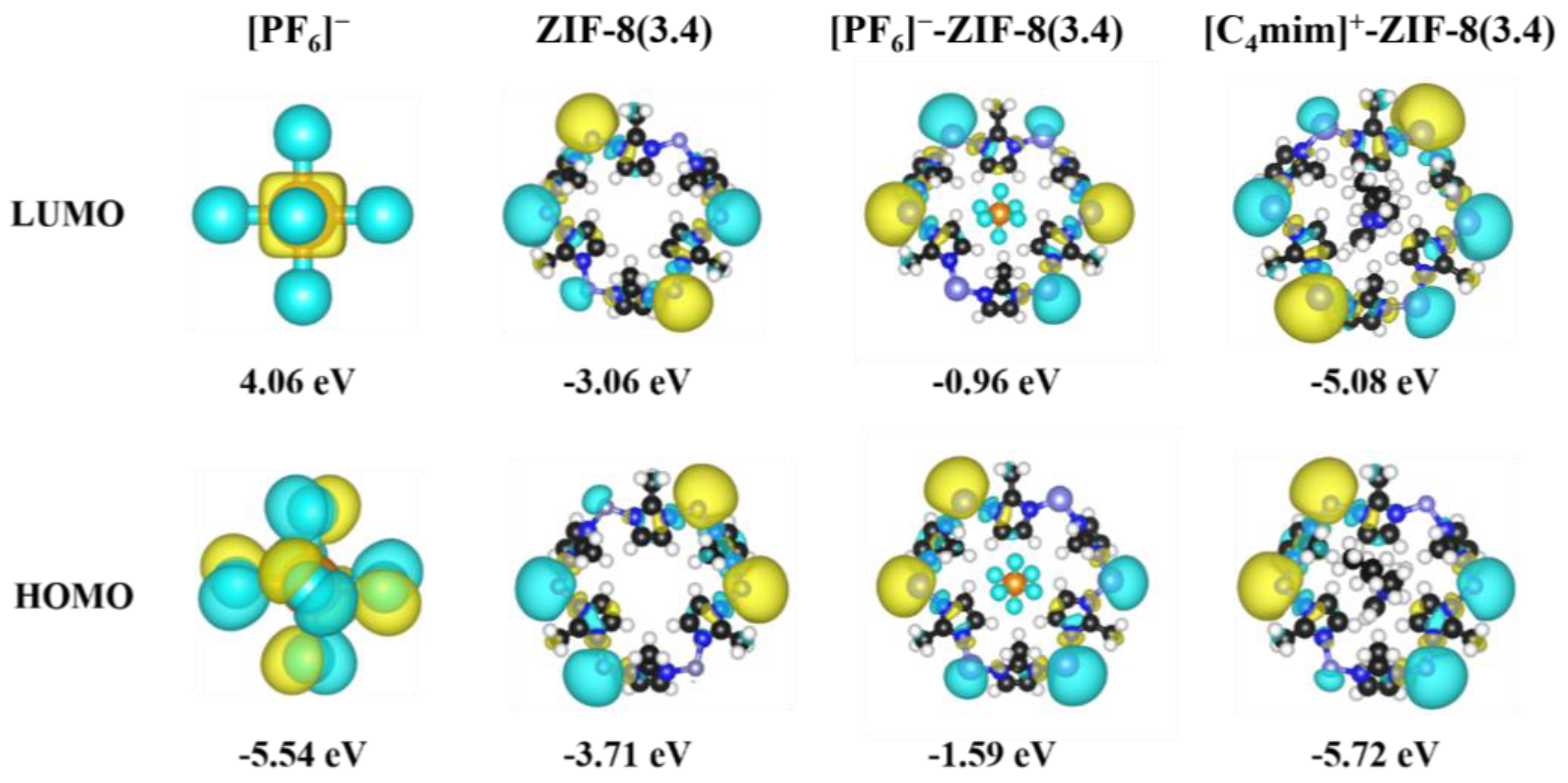
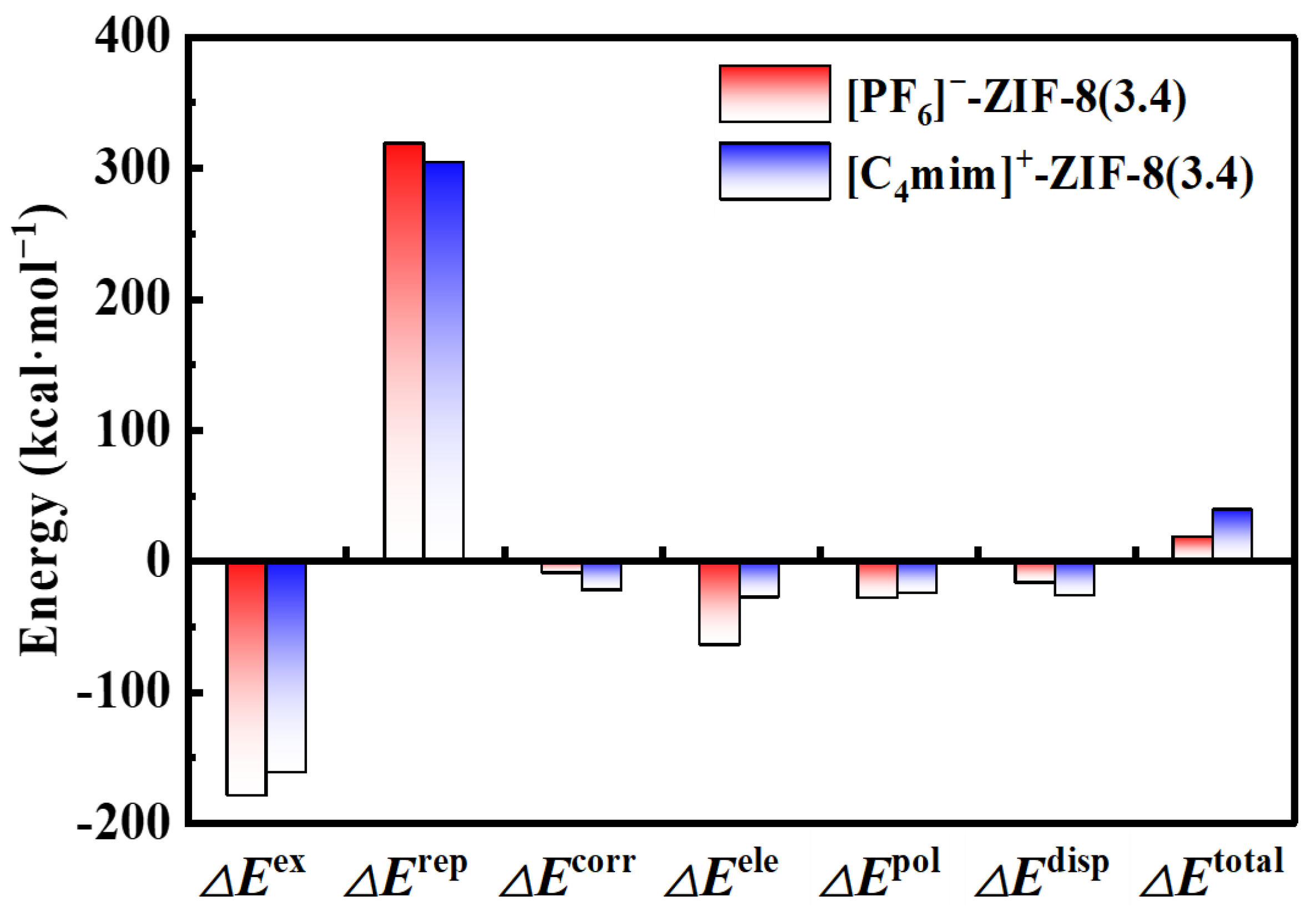
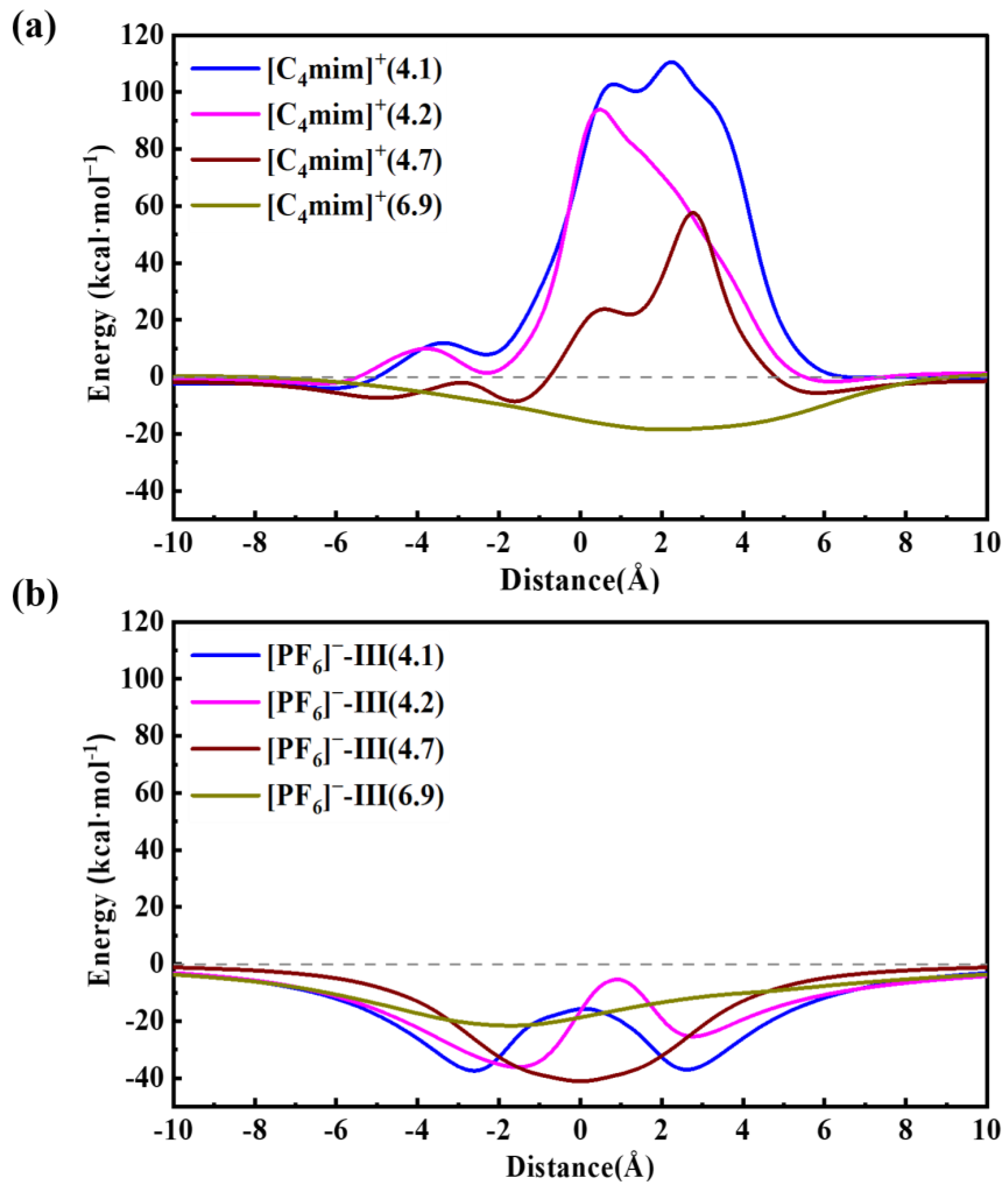
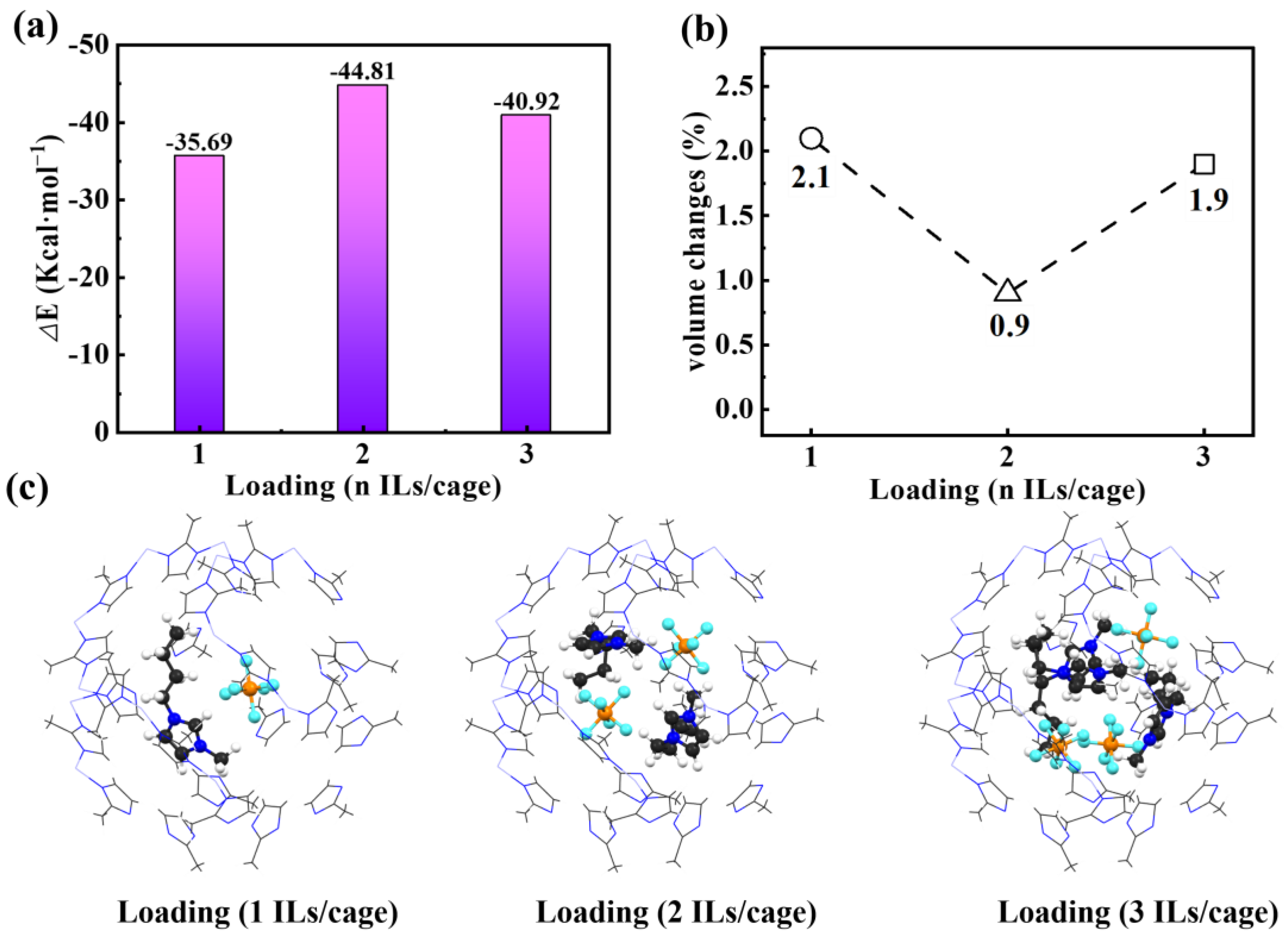
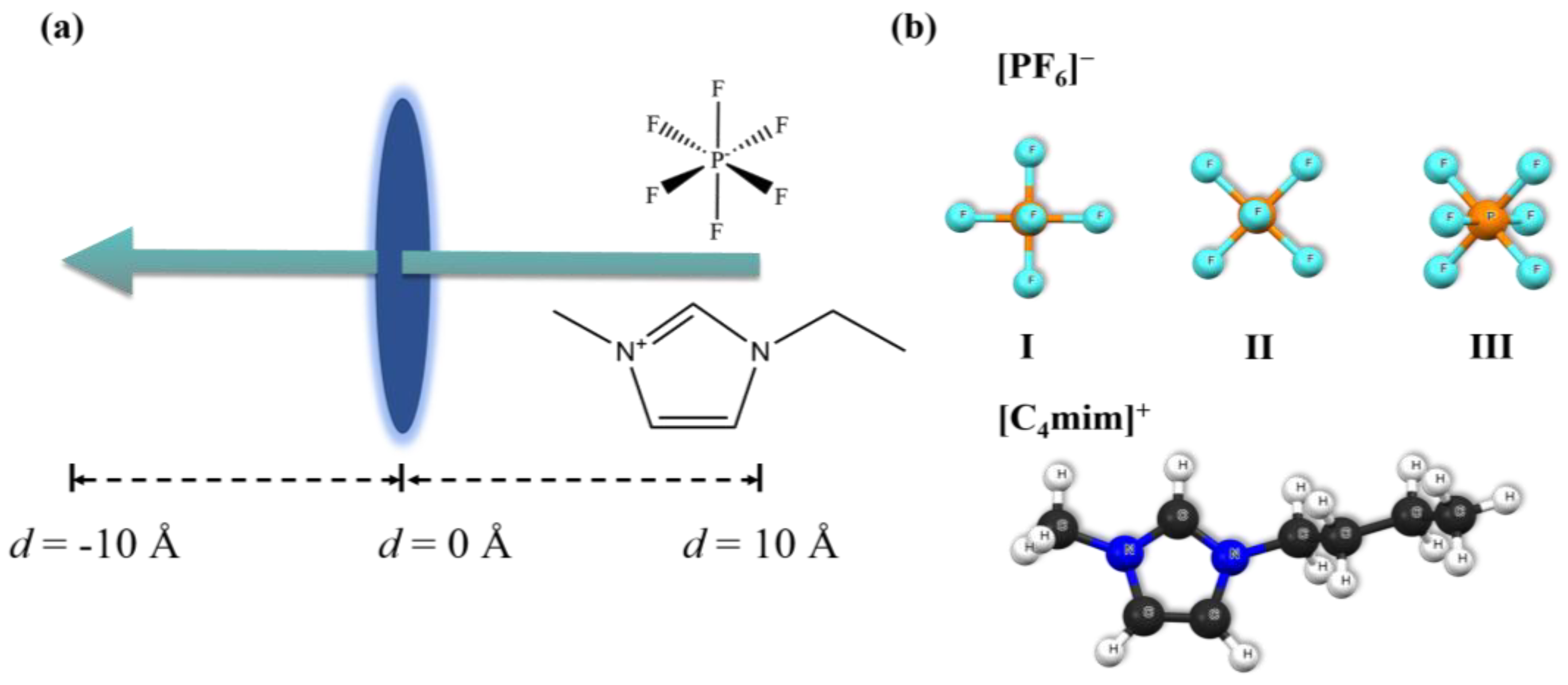
2. Results
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, X.; Chen, K.; Guo, R.; Wei, Z. Ionic Liquids Functionalized MOFs for Adsorption. Chem. Rev. 2023, 123, 10432–10467. [Google Scholar] [CrossRef] [PubMed]
- Durak, O.; Zeeshan, M.; Habib, N.; Gulbalkan, H.C.; Alsuhile, A.A.A.M.; Caglayan, H.P.; Kurtoğlu-Öztulum, S.F.; Zhao, Y.; Haslak, Z.P.; Uzun, A.; et al. Composites of porous materials with ionic liquids: Synthesis, characterization, applications, and beyond. Microporous Mesoporous Mater. 2022, 332, 111703. [Google Scholar] [CrossRef]
- Wu, K.; Miao, X.; Zhao, H.; Liu, S.; Fei, T.; Zhang, T. Selective Encapsulation of Ionic Liquids in UiO-66-NH2 Nanopores for Enhanced Humidity Sensing. ACS Appl. Nano Mater. 2023, 6, 9050–9058. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, F.; Pang, H. A Review of MOFs and Their Composites-Based Photocatalysts: Synthesis and Applications. Adv. Funct. Mater. 2021, 31, 2104231. [Google Scholar] [CrossRef]
- Friess, K.; Izák, P.; Kárászová, M.; Pasichnyk, M.; Lanč, M.; Nikolaeva, D.; Luis, P.; Jansen, J.C. A Review on Ionic Liquid Gas Separation Membranes. Membranes 2021, 11, 97. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.; Saiz, P.G.; Peřinka, N.; Wuttke, S.; Fernández de Luis, R. Printed Capacitive Sensors Based on Ionic Liquid/Metal-Organic Framework Composites for Volatile Organic Compounds Detection. Adv. Funct. Mater. 2021, 31, 2010703. [Google Scholar] [CrossRef]
- Tuffnell, J.M.; Morzy, J.K.; Kelly, N.D.; Tan, R.; Song, Q.; Ducati, C.; Bennett, T.D.; Dutton, S.E. Comparison of the ionic conductivity properties of microporous and mesoporous MOFs infiltrated with a Na-ion containing IL mixture. Dalton Trans. 2020, 49, 15914–15924. [Google Scholar] [CrossRef]
- Zeeshan, M.; Nozari, V.; Keskin, S.; Uzun, A. Structural Factors Determining Thermal Stability Limits of Ionic Liquid/MOF Composites: Imidazolium Ionic Liquids Combined with CuBTC and ZIF-8. Ind. Eng. Chem. Res. 2019, 58, 14124–14138. [Google Scholar] [CrossRef]
- Yoshida, Y.; Fujie, K.; Lim, D.W.; Ikeda, R.; Kitagawa, H. Superionic Conduction over a Wide Temperature Range in a Metal–Organic Framework Impregnated with Ionic Liquids. Angew. Chem. Int. Ed. 2019, 58, 10909–10913. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tuo, L.; Yang, K.; Jeong, H.-K.; Dai, Y.; He, G.; Zhao, W. Simultaneous enhancement of mechanical properties and CO2 selectivity of ZIF-8 mixed matrix membranes: Interfacial toughening effect of ionic liquid. J. Membr. Sci. 2016, 511, 130–142. [Google Scholar] [CrossRef]
- Khan, M.S.; Li, Y.; Yang, L.; Yan, Z.C.; Li, D.-S.; Qiu, J.; Xu, X.; Yang, H.Y. Improving capacitive deionization performance through tailored iodine-loaded ZIF-8 composites. Desalination 2024, 579, 117486. [Google Scholar] [CrossRef]
- Khan, M.S.; Leong, Z.Y.; Li, D.-S.; Qiu, J.; Xu, X.; Yang, H.Y. A mini review on metal–organic framework-based electrode materials for capacitive deionization. Nanoscale 2023, 15, 15929–15949. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Li, Y.; Li, D.-S.; Qiu, J.; Xu, X.; Yang, H.Y. A review of metal–organic framework (MOF) materials as an effective photocatalyst for degradation of organic pollutants. Nanoscale Adv. 2023, 5, 6318–6348. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, Z.; Yang, C.; Liu, J.; Shen, H.; Yang, K.; Wang, Z. PIM-based mixed-matrix membranes containing MOF-801/ionic liquid nanocomposites for enhanced CO2 separation performance. J. Membr. Sci. 2021, 636, 119581. [Google Scholar] [CrossRef]
- Zeeshan, M.; Keskin, S.; Uzun, A. Enhancing CO2/CH4 and CO2/N2 separation performances of ZIF-8 by post-synthesis modification with [BMIM][SCN]. Polyhedron 2018, 155, 485–492. [Google Scholar] [CrossRef]
- Krokidas, P.; Moncho, S.; Brothers, E.N.; Castier, M.; Economou, I.G. Tailoring the gas separation efficiency of metal organic framework ZIF-8 through metal substitution: A computational study. Phys. Chem. Chem. Phys. 2018, 20, 4879–4892. [Google Scholar] [CrossRef] [PubMed]
- Gulbalkan, H.C.; Haslak, Z.P.; Altintas, C.; Uzun, A.; Keskin, S. Assessing CH4/N2 separation potential of MOFs, COFs, IL/MOF, MOF/Polymer, and COF/Polymer composites. Chem. Eng. J. 2022, 428, 131239. [Google Scholar] [CrossRef]
- Ban, Y.; Li, Z.; Li, Y.; Peng, Y.; Jin, H.; Jiao, W.; Guo, A.; Wang, P.; Yang, Q.; Zhong, C.; et al. Confinement of Ionic Liquids in Nanocages: Tailoring the Molecular Sieving Properties of ZIF-8 for Membrane-Based CO2 Capture. Angew. Chem. Int. Ed. Engl. 2015, 54, 15483–15487. [Google Scholar] [CrossRef] [PubMed]
- Kinik, F.P.; Altintas, C.; Balci, V.; Koyuturk, B.; Uzun, A.; Keskin, S. [BMIM][PF6] Incorporation Doubles CO2 Selectivity of ZIF-8: Elucidation of Interactions and Their Consequences on Performance. ACS Appl. Mater. Interfaces 2016, 8, 30992–31005. [Google Scholar] [CrossRef] [PubMed]
- Koyuturk, B.; Altintas, C.; Kinik, F.P.; Keskin, S.; Uzun, A. Improving Gas Separation Performance of ZIF-8 by [BMIM][BF4] Incorporation: Interactions and Their Consequences on Performance. J. Phys. Chem. C 2017, 121, 10370–10381. [Google Scholar] [CrossRef]
- Ali, S.A.; Khan, A.U.; Mulk, W.U.; Khan, H.; Nasir Shah, S.; Zahid, A.; Habib, K.; Shah, M.U.H.; Othman, M.H.D.; Rahman, S. An Ongoing Futuristic Career of Metal–Organic Frameworks and Ionic Liquids, A Magical Gateway to Capture CO2; A Critical Review. Energy Fuels 2023, 37, 15394–15428. [Google Scholar] [CrossRef]
- Dong, K.; Liu, X.; Dong, H.; Zhang, X.; Zhang, S. Multiscale Studies on Ionic Liquids. Chem. Rev. 2017, 117, 6636–6695. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Dong, K.; Liu, L.; Zhang, X.; Zhang, S. Anti-electrostatic hydrogen bonding between anions of ionic liquids: A density functional theory study. Phys. Chem. Chem. Phys. 2021, 23, 7426–7433. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.T.; Jeong, H.-K.; Lee, A.S.; An, H.S.; Lee, J.S. Heteroepitaxially Grown Zeolitic Imidazolate Framework Membranes with Unprecedented Propylene/Propane Separation Performances. J. Am. Chem. Soc. 2015, 137, 12304–12311. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Prakash, M. Tuning the CO2 adsorption by the selection of suitable ionic liquids at ZIF-8 confinement: A DFT study. Appl. Surf. Sci. 2019, 491, 633–639. [Google Scholar] [CrossRef]
- Mohamed, A.M.O.; Moncho, S.; Krokidas, P.; Kakosimos, K.; Brothers, E.N.; Economou, I.G. Computational investigation of the performance of ZIF-8 with encapsulated ionic liquids towards CO2 capture. Mol. Phys. 2019, 117, 3791–3805. [Google Scholar] [CrossRef]
- Gao, W.; Zheng, W.; Sun, W.; Zhao, L. Understanding the Effective Capture of H2S/CO2 from Natural Gas Using Ionic Liquid@MOF Composites. J. Phys. Chem. C 2022, 126, 19872–19882. [Google Scholar] [CrossRef]
- Thomas, A.; Ahamed, R.; Prakash, M. Selection of a suitable ZIF-8/ionic liquid (IL) based composite for selective CO2 capture: The role of anions at the interface. RSC Adv. 2020, 10, 39160–39170. [Google Scholar] [CrossRef] [PubMed]
- Kavak, S.; Polat, H.M.; Kulak, H.; Keskin, S.; Uzun, A. MIL-53(Al) as a Versatile Platform for Ionic-Liquid/MOF Composites to Enhance CO2 Selectivity over CH4 and N2. Chem.–Asian J. 2019, 14, 3655–3667. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Jia, X.; Sun, Y.; Guo, X.; Huang, H.; Zhong, C. Pore engineering of ZIF-8 with ionic liquids for membrane-based CO2 separation: Bearing functional group effect. Green Chem. Eng. 2021, 2, 104–110. [Google Scholar] [CrossRef]
- Fairen-Jimenez, D.; Moggach, S.A.; Wharmby, M.T.; Wright, P.A.; Parsons, S.; Düren, T. Opening the Gate: Framework Flexibility in ZIF-8 Explored by Experiments and Simulations. J. Am. Chem. Soc. 2011, 133, 8900–8902. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Lively, R.P.; Zhang, K.; Johnson, J.R.; Karvan, O.; Koros, W.J. Unexpected Molecular Sieving Properties of Zeolitic Imidazolate Framework-8. J. Phys. Chem. Lett. 2012, 3, 2130–2134. [Google Scholar] [CrossRef] [PubMed]
- Zeeshan, M.; Nozari, V.; Yagci, M.B.; Isık, T.; Unal, U.; Ortalan, V.; Keskin, S.; Uzun, A. Core–Shell Type Ionic Liquid/Metal Organic Framework Composite: An Exceptionally High CO2/CH4 Selectivity. J. Am. Chem. Soc. 2018, 140, 10113–10116. [Google Scholar] [CrossRef]
- Chang, Y.; Wang, L.; Jiang, Z.; Zhang, R.; Zhu, H.; Zhang, D.; Zhu, J.; Kong, X.; Huang, H. Zwitterionic metal–organic framework with highly dispersed ionic liquid for enhancing CO2 capture. Sep. Purif. Technol. 2023, 326, 124828. [Google Scholar] [CrossRef]
- Yu, T.; Cai, Q.; Lian, G.; Bai, Y.; Zhang, X.; Zhang, X.; Liu, L.; Zhang, S. Mechanisms behind high CO2/CH4 selectivity using ZIF-8 metal organic frameworks with encapsulated ionic liquids: A computational study. Chem. Eng. J. 2021, 419, 129638. [Google Scholar] [CrossRef]
- Yu, T.; Cai, Q.; Lian, G.; Liu, L. Molecular dynamics studies on separation of CO2/CH4 by the ionic liquids encapsulated ZIF-8. J. Membr. Sci. 2022, 644, 120117. [Google Scholar] [CrossRef]
- Guo, Z.; Zheng, W.; Yan, X.; Dai, Y.; Ruan, X.; Yang, X.; Li, X.; Zhang, N.; He, G. Ionic liquid tuning nanocage size of MOFs through a two-step adsorption/infiltration strategy for enhanced gas screening of mixed-matrix membranes. J. Membr. Sci. 2020, 605, 118101. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Côté, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl. Acad. Sci. USA 2006, 103, 10186–10191. [Google Scholar] [CrossRef] [PubMed]
- Bühl, M.; Wipff, G. Insights into Uranyl Chemistry from Molecular Dynamics Simulations. ChemPhysChem 2011, 12, 3095–3105. [Google Scholar] [CrossRef] [PubMed]
- Frisch, G.A.; Trucks, M.J.; Schlegel, G.W.; Scuseria, H.B.; Robb, G.E.; Cheeseman, M.A.; Scalmani, J.R.; Barone, G.; Petersson, V. Gaussian 16, Rev. C.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Weinhold, F.; Landis, C.; Glendening, E.D. What is NBO analysis and how is it useful? Int. Rev. Phys. Chem. 2016, 35, 399–440. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Momma, K.; Izumi, F. VESTA: A three-dimensional visualization system for electronic and structural analysis. J. Appl. Crystallogr. 2008, 41, 653–658. [Google Scholar] [CrossRef]
- Su, P.; Jiang, Z.; Chen, Z.; Wu, W. Energy decomposition scheme based on the generalized Kohn-Sham scheme. J. Phys. Chem. A 2014, 118, 2531–2542. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.W.; Baldridge, K.K.; Boatz, J.A.; Elbert, S.T.; Gordon, M.S.; Jensen, J.H.; Koseki, S.; Matsunaga, N.; Nguyen, K.A.; Su, S.; et al. General atomic and molecular electronic structure system. J. Comput. Chem. 1993, 14, 1347–1363. [Google Scholar] [CrossRef]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar] [CrossRef]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [PubMed]
- Kühne, T.D.; Iannuzzi, M.; Del Ben, M.; Rybkin, V.V.; Seewald, P.; Stein, F.; Laino, T.; Khaliullin, R.Z.; Schütt, O.; Schiffmann, F.; et al. CP2K: An electronic structure and molecular dynamics software package—Quickstep: Efficient and accurate electronic structure calculations. J. Chem. Phys. 2020, 152, 194103. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Jiang, K.; Chen, Q.; Liu, L. On the Diffusion of Ionic Liquids in ILs@ZIF-8 Composite Materials: A Density Functional Theory Study. Molecules 2024, 29, 1697. https://doi.org/10.3390/molecules29081697
Liu L, Jiang K, Chen Q, Liu L. On the Diffusion of Ionic Liquids in ILs@ZIF-8 Composite Materials: A Density Functional Theory Study. Molecules. 2024; 29(8):1697. https://doi.org/10.3390/molecules29081697
Chicago/Turabian StyleLiu, Longlong, Kun Jiang, Qingjun Chen, and Lei Liu. 2024. "On the Diffusion of Ionic Liquids in ILs@ZIF-8 Composite Materials: A Density Functional Theory Study" Molecules 29, no. 8: 1697. https://doi.org/10.3390/molecules29081697
APA StyleLiu, L., Jiang, K., Chen, Q., & Liu, L. (2024). On the Diffusion of Ionic Liquids in ILs@ZIF-8 Composite Materials: A Density Functional Theory Study. Molecules, 29(8), 1697. https://doi.org/10.3390/molecules29081697






