Selective CO2 Capture from CO2/N2 Gas Mixtures Utilizing Tetrabutylammonium Fluoride Hydrates
Abstract
1. Introduction
2. Results and Discussion
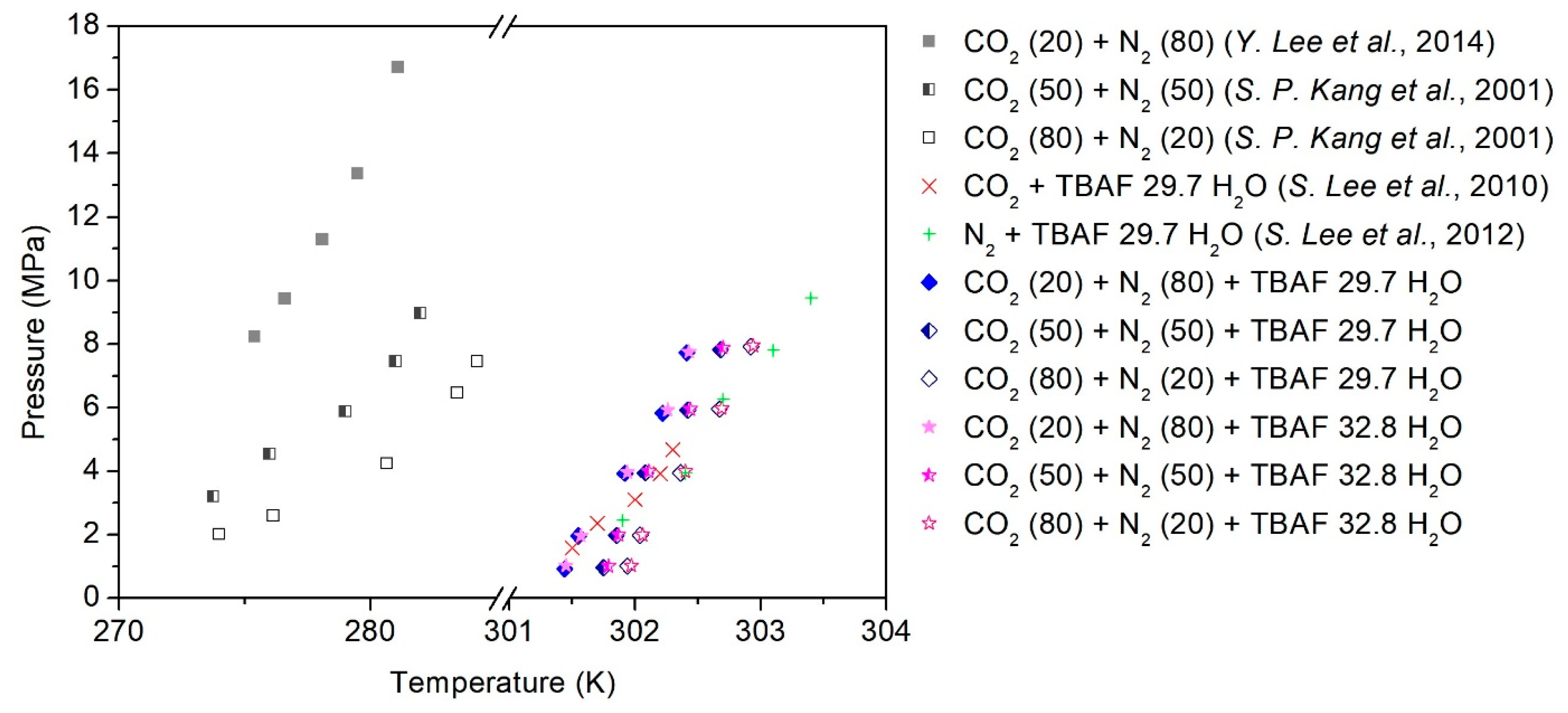
2.1. Phase Equilibria of a Semi-Clathrate Hydrate with a Secondary Gaseous Guest
2.2. Spectroscopic Analysis: Structural Identification and Guest Enclathration Behavior
2.2.1. PXRD Analysis of Semi-Clathrate Hydrates
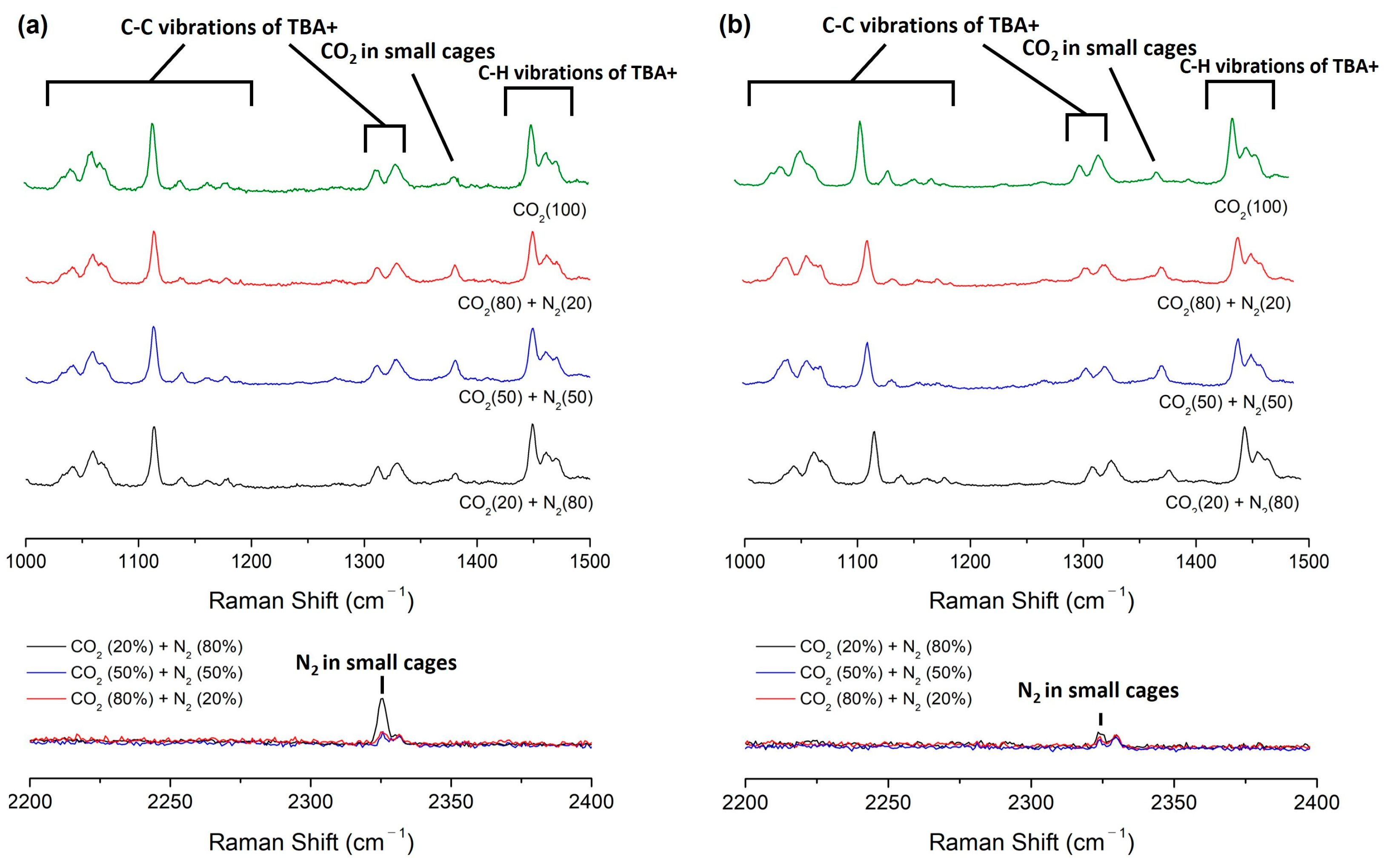
2.2.2. Raman Analysis of Semi-Clathrate Hydrates
2.3. Semi-Clathrate Hydrate-Based Gas Capture and Separation
2.3.1. Gas Uptake during Hydrate Formation
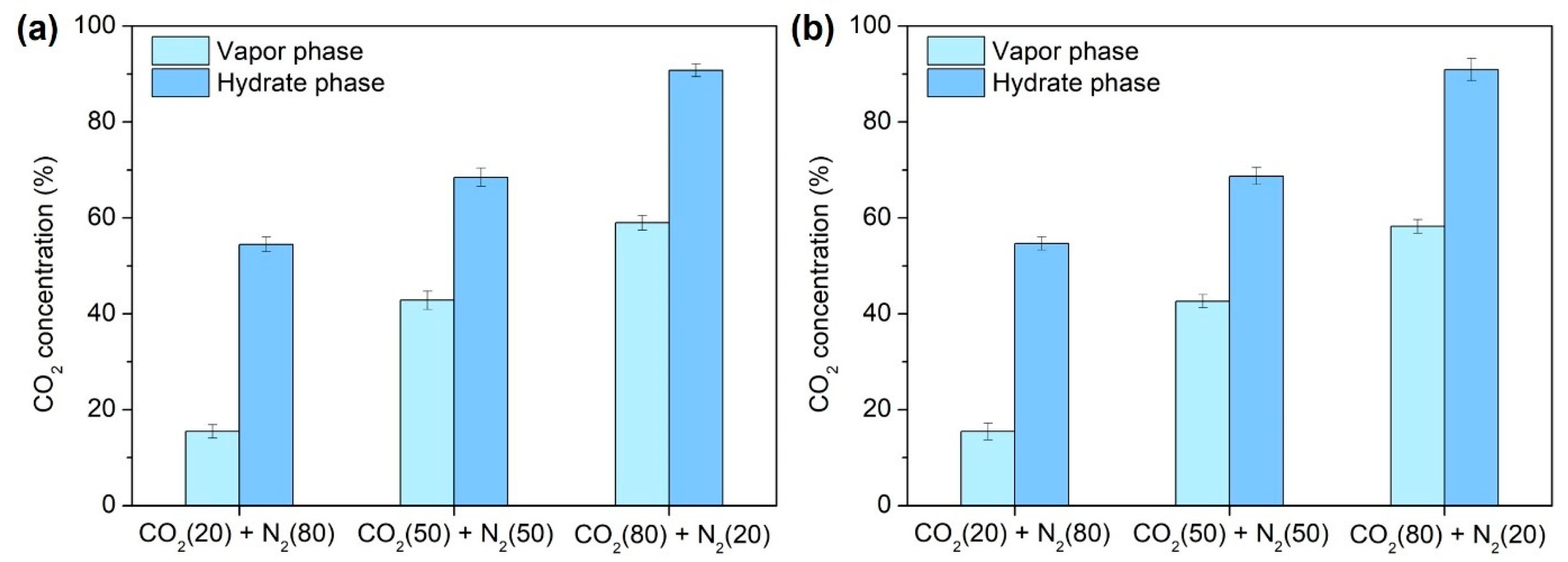
2.3.2. Separation Efficiency of Semi-Clathrate Hydrates-Based Gas Separation
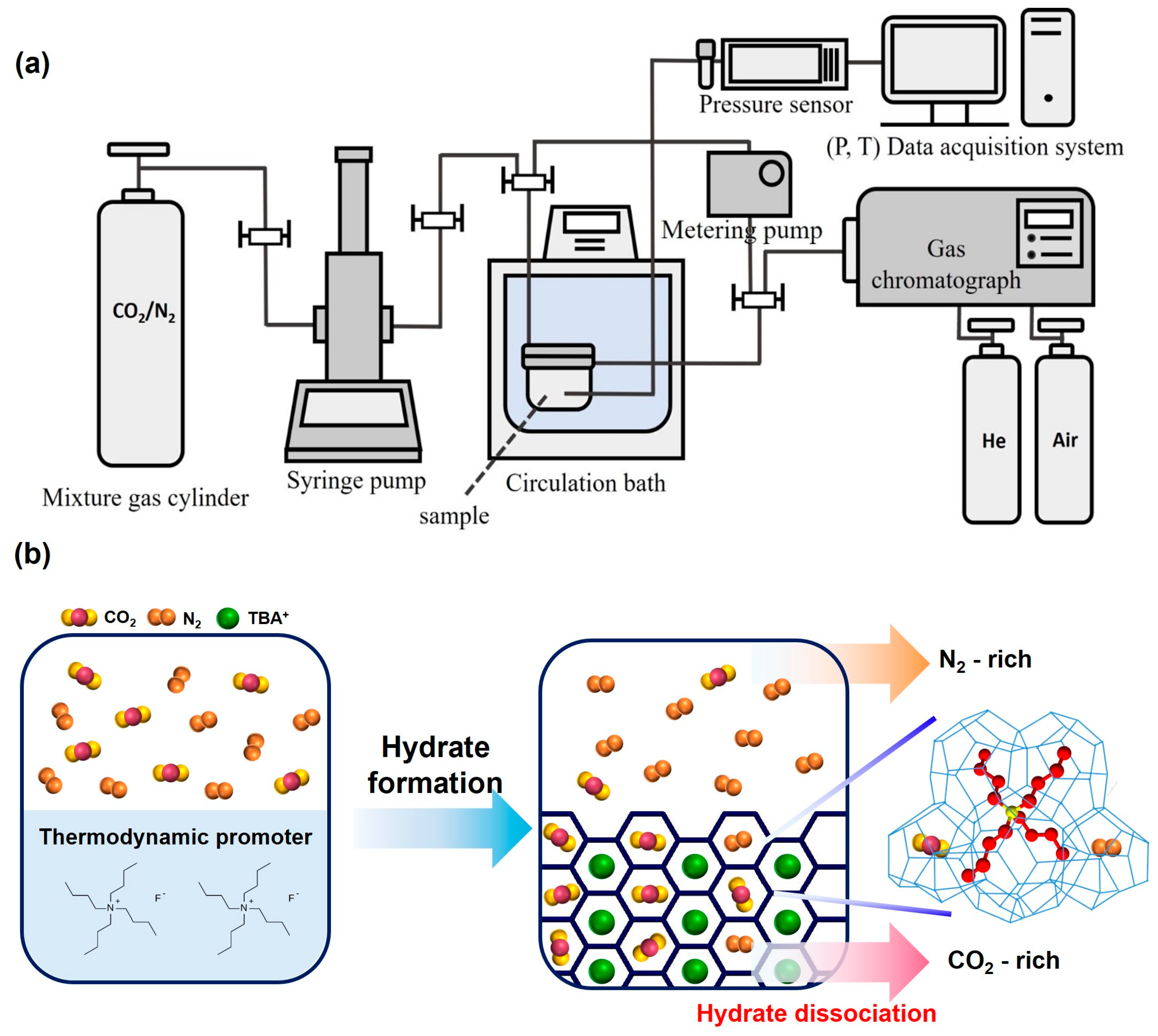
3. Materials and Methods
3.1. Materials
3.2. Phase Equilibrium Measurements of a Semi-Clathrate Hydrate
3.3. Structure and Guest Enclathration Analysis of Semi-Clathrate Hydrates
3.4. Gas Composition Analysis
3.5. Quantitative Analysis of Gas Separation Performance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sévellec, F.; Drijfhout, S.S. A novel probabilistic forecast system predicting anomalously warm 2018–2022 reinforcing the long-term global warming trend. Nat. Commun. 2018, 9, 3024. [Google Scholar] [CrossRef]
- Dlugokencky, P.T.E. Trends in Atmospheric Carbon Dioxide (Global). 2022. Available online: https://gml.noaa.gov/ccgg/trends/ (accessed on 10 March 2024).
- International Energy Agency. Energy Technology Perspectives 2020; International Energy Agency: Paris, France, 2020. [CrossRef]
- Babu, P.; Linga, P.; Kumar, R.; Englezos, P. A review of the hydrate based gas separation (HBGS) process forcarbon dioxide pre-combustion capture. Energy 2015, 85, 261–279. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, S.; Seo, D.; Moon, S.; Ahn, Y.H.; Park, Y. Highly efficient separation and equilibrium recovery of H2/CO2 in hydrate-based pre-combustion CO2 capture. Chem. Eng. J. 2024, 481, 148709. [Google Scholar] [CrossRef]
- Han, G.; Lee, W.; Kim, M.-K.; Lee, J.W.; Ahn, Y.-H. Hydrogen separation from hydrogen-compressed natural gas blends through successive hydrate formations. Chem. Eng. J. 2024, 483, 149409. [Google Scholar] [CrossRef]
- Sloan, E.D.; Koh, C.A. Clathrate Hydrates of Natural Gases, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2007; ISBN 9781420008494. [Google Scholar]
- Kang, D.W.; Lee, W.; Ahn, Y.H.; Lee, J.W. Confined tetrahydrofuran in a superabsorbent polymer for sustainable methane storage in clathrate hydrates. Chem. Eng. J. 2021, 411, 128512. [Google Scholar] [CrossRef]
- Kim, M.K.; Han, G.; Kim, H.; Yu, J.; Lee, Y.; Song, T.; Park, J.; Kim, Y.H.; Ahn, Y.H. Optimization of water-saturated superabsorbent polymers for hydrate-based gas storage. Korean J. Chem. Eng. 2023, 40, 1063–1070. [Google Scholar] [CrossRef]
- Kim, M.K.; Ahn, Y.H. Gas Hydrates for Hydrogen Storage: A Comprehensive Review and Future Prospects. Korean J. Chem. Eng. 2024, 41, 73–94. [Google Scholar] [CrossRef]
- Kang, D.W.; Lee, W.; Ahn, Y.H.; Lee, J.W. Exploring tuning phenomena of THF-H2 hydrates via molecular dynamics simulations. J. Mol. Liq. 2022, 349, 118490. [Google Scholar] [CrossRef]
- Moon, S.; Hong, S.; Lee, Y.; Lee, J.S.; Ahn, Y.H.; Park, Y. Enhancing Hydrogen Cluster Storage in Clathrate Hydrates via Defect-Mediated Lattice Engineering. J. Phys. Chem. C 2021, 125, 1767–1773. [Google Scholar] [CrossRef]
- Mahajan, D.; Tan, K.; Venkatesh, T.; Kileti, P.; Clayton, C.R. Hydrogen Blending in Gas Pipeline Networks—A Review. Energies 2022, 15, 3582. [Google Scholar] [CrossRef]
- Lee, W.; Kang, D.W.; Ahn, Y.H.; Lee, J.W. Blended hydrate seed and liquid promoter for the acceleration of hydrogen hydrate formation. Renew. Sustain. Energy Rev. 2023, 177, 113217. [Google Scholar] [CrossRef]
- Lee, W.; Kang, D.W.; Ahn, Y.H.; Lee, J.W. Rapid Formation of Hydrogen-Enriched Hydrocarbon Gas Hydrates under Static Conditions. ACS Sustain. Chem. Eng. 2021, 9, 8414–8424. [Google Scholar] [CrossRef]
- Moon, S.; Lee, Y.; Seo, D.; Lee, S.; Hong, S.; Ahn, Y.H.; Park, Y. Critical hydrogen concentration of hydrogen-natural gas blends in clathrate hydrates for blue hydrogen storage. Renew. Sustain. Energy Rev. 2021, 141, 110789. [Google Scholar] [CrossRef]
- Ahn, Y.H.; Moon, S.; Koh, D.Y.; Hong, S.; Lee, H.; Lee, J.W.; Park, Y. One-step formation of hydrogen clusters in clathrate hydrates stabilized via natural gas blending. Energy Storage Mater. 2020, 24, 655–661. [Google Scholar] [CrossRef]
- Moon, S.; Lee, S.; Ahn, Y.H.; Park, Y. Abnormal thermodynamic promotion and tuning behavior of epoxycyclopentane for its implications in CO2 storage. Chem. Eng. J. 2021, 425, 130647. [Google Scholar] [CrossRef]
- Moon, S.; Ahn, Y.H.; Kim, H.; Hong, S.; Koh, D.Y.; Park, Y. Secondary gaseous guest-dependent structures of binary neopentyl alcohol hydrates and their tuning behavior for potential application to CO2 capture. Chem. Eng. J. 2017, 330, 890–898. [Google Scholar] [CrossRef]
- Kang, D.W.; Lee, W.; Ahn, Y.H. Superabsorbent polymer for improved CO2 hydrate formation under a quiescent system. J. CO2 Util. 2022, 61, 102005. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, H.; Lee, W.; Kang, D.W.; Lee, J.W.; Ahn, Y.H. Thermodynamic and kinetic properties of CO2 hydrates and their applications in CO2 capture and separation. J. Environ. Chem. Eng. 2023, 11, 110933. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, X.; Sun, M.; Zhao, Y.; Yang, L.; Song, Y. N2O hydrate formation in porous media: A potential method to mitigate N2O emissions. Chem. Eng. J. 2019, 361, 12–20. [Google Scholar] [CrossRef]
- Ahn, Y.H.; Lim, D.; Min, J.; Kim, J.; Lee, B.; Lee, J.W.; Shin, K. Clathrate nanocage reactor for the decomposition of greenhouse gas. Chem. Eng. J. 2019, 359, 1629–1634. [Google Scholar] [CrossRef]
- Yang, Y.; Shin, D.; Choi, S.; Woo, Y.; Lee, J.W.; Kim, D.; Shin, H.Y.; Cha, M.; Yoon, J.H. Selective Encaging of N2O in N2O-N2 Binary Gas Hydrates via Hydrate-Based Gas Separation. Environ. Sci. Technol. 2017, 51, 3550–3557. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Choi, W.; Mok, J.; Seo, Y. Kinetic CO2 selectivity in clathrate-based CO2 capture for upgrading CO2-rich natural gas and biogas. Chem. Eng. J. 2019, 369, 686–693. [Google Scholar] [CrossRef]
- Trueba, A.T.; Radović, I.R.; Zevenbergen, J.F.; Peters, C.J.; Kroon, M.C. Kinetic measurements and in situ Raman spectroscopy study of the formation of TBAF semi-hydrates with hydrogen and carbon dioxide. Int. J. Hydrogen Energy 2013, 38, 7326–7334. [Google Scholar] [CrossRef]
- Mohammadi, A. The Effect of Various Concentrations of Tetra-n-butylammonium Fluoride on the Dissociation Enthalpy of Gas Hydrates. Iran. J. Energy Environ. 2022, 13, 151–157. [Google Scholar] [CrossRef]
- Pandey, G.; Poothia, T.; Kumar, A. Hydrate based carbon capture and sequestration (HBCCS): An innovative approach towards decarbonization. Appl. Energy 2022, 326, 119900. [Google Scholar] [CrossRef]
- Hashimoto, H.; Ozeki, H.; Yamamoto, Y.; Muromachi, S. CO2 Capture from Flue Gas Based on Tetra-n-butylammonium Fluoride Hydrates at near Ambient Temperature. ACS Omega 2020, 5, 7115–7123. [Google Scholar] [CrossRef]
- Shi, L.L.; Liang, D. qing Thermodynamic model of phase equilibria of tetrabutyl ammonium halide (fluoride, chloride, or bromide) plus methane or carbon dioxide semiclathrate hydrates. Fluid Phase Equilib. 2015, 386, 149–154. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, S.; Lee, J.; Seo, Y. Structure identification and dissociation enthalpy measurements of the CO2+N2 hydrates for their application to CO2 capture and storage. Chem. Eng. J. 2014, 246, 20–26. [Google Scholar] [CrossRef]
- Kang, S.P.; Lee, H.; Lee, C.S.; Sung, W.M. Hydrate phase equilibria of the guest mixtures containing CO2, N2 and tetrahydrofuran. Fluid Phase Equilib. 2001, 185, 101–109. [Google Scholar] [CrossRef]
- Lee, S.; Lee, Y.; Park, S.; Kim, Y.; Lee, J.D.; Seo, Y. Thermodynamic and spectroscopic identification of guest gas enclathration in the double tetra-n-butylammonium fluoride semiclathrates. J. Phys. Chem. B 2012, 116, 9075–9081. [Google Scholar] [CrossRef]
- Lee, S.; Lee, Y.; Park, S.; Seo, Y. Phase equilibria of semiclathrate hydrate for nitrogen in the presence of tetra-n-butylammonium bromide and fluoride. J. Chem. Eng. Data 2010, 55, 5883–5886. [Google Scholar] [CrossRef]
- Sizova, A.A.; Grintsevich, S.A.; Kochurin, M.A.; Sizov, V.V.; Brodskaya, E.N. Molecular Simulations of CO2/CH4, CO2/N2 and N2/CH4 Binary Mixed Hydrates. Colloid J. 2021, 83, 372–378. [Google Scholar] [CrossRef]
- Koh, D.Y.; Kang, H.; Kim, D.O.; Park, J.; Cha, M.; Lee, H. Recovery of methane from gas hydrates intercalated within natural sediments using CO2 and a CO2/N2 gas mixture. ChemSusChem 2012, 5, 1443–1448. [Google Scholar] [CrossRef]
- Koh, D.; Ahn, Y.; Kang, H.; Park, S.; Lee, J.Y.; Kim, S.; Lee, J.; Lee, H. One-dimensional productivity assessment for on-field methane hydrate production using CO2/N2 mixture gas. AIChE J. 2015, 61, 1004–1014. [Google Scholar] [CrossRef]
- Belosludov, V.R.; Bozhko, Y.Y.; Subbotin, O.S.; Belosludov, R.V.; Zhdanov, R.K.; Gets, K.V.; Kawazoe, Y. Influence of N2 on formation conditions and guest distribution of mixed CO2 + CH4 gas hydrates. Molecules 2018, 23, 3336. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, H.; Maruyama, K.; Yamagiwa, K.; Tajima, H. Separation processes for carbon dioxide capture with semi-clathrate hydrate slurry based on phase equilibria of CO2 + N2 + tetra-n-butylammonium bromide + water systems. Chem. Eng. Res. Des. 2019, 150, 289–298. [Google Scholar] [CrossRef]
- Ma, Z.W.; Zhang, P.; Bao, H.S.; Deng, S. Review of fundamental properties of CO2 hydrates and CO2 capture and separation using hydration method. Renew. Sustain. Energy Rev. 2016, 53, 1273–1302. [Google Scholar] [CrossRef]
- Ye, N.; Zhang, P. Phase equilibrium conditions and carbon dioxide separation efficiency of tetra-n-butylphosphonium bromide hydrate. J. Chem. Eng. Data 2014, 59, 2920–2926. [Google Scholar] [CrossRef]
- Li, S.; Fan, S.; Wang, J.; Lang, X.; Liang, D. CO2 capture from binary mixture via forming hydrate with the help of tetra-n-butyl ammonium bromide. J. Nat. Gas Chem. 2009, 18, 15–20. [Google Scholar] [CrossRef]
- Ahn, Y.H.; Kang, H.; Koh, D.Y.; Park, Y.; Lee, H. Gas hydrate inhibition by 3-hydroxytetrahydrofuran: Spectroscopic identifications and hydrate phase equilibria. Fluid Phase Equilib. 2016, 413, 65–70. [Google Scholar] [CrossRef]



| Semi-Clathrate Hydrate | Feed Gas | |||||
|---|---|---|---|---|---|---|
| CO2 (20%) + N2 (80%) | CO2 (50%) + N2 (50%) | CO2 (80%) + N2 (20%) | ||||
| T/K | P/MPa | T/K | P/MPa | T/K | P/MPa | |
| TBAF·29.7 H2O | 302.41 | 7.72 | 302.68 | 7.82 | 302.92 | 7.92 |
| 302.22 | 5.82 | 302.42 | 5.92 | 302.67 | 5.95 | |
| 301.92 | 3.93 | 302.08 | 3.95 | 302.36 | 3.94 | |
| 301.55 | 1.96 | 301.85 | 1.97 | 302.04 | 1.98 | |
| 301.44 | 0.92 | 301.75 | 0.96 | 301.94 | 1.01 | |
| TBAF·32.8 H2O | 302.43 | 7.75 | 302.70 | 7.89 | 302.94 | 7.95 |
| 302.26 | 5.92 | 302.44 | 5.95 | 302.69 | 5.97 | |
| 301.94 | 3.96 | 302.11 | 3.99 | 302.40 | 3.99 | |
| 301.57 | 1.96 | 301.87 | 1.99 | 302.06 | 1.99 | |
| 301.45 | 1.02 | 301.79 | 1.01 | 301.97 | 1.02 | |
| Feed Gas | TBAF·29.7 H2O (CO2/N2) | TBAF·32.8 H2O (CO2/N2) |
|---|---|---|
| CO2 (20%) + N2 (80%) | 124.22/63.28 | 94.79/43.91 |
| CO2 (50%) + N2 (50%) | 163.07/39.13 | 148.62/36.71 |
| CO2 (80%) + N2 (20%) | 179.69/33.63 | 158.55/27.42 |
| CO2 (100%) | 177.16/- | 159.20/- |
| Semi-Clathrate Hydrate | Feed Gas | Separation Factor | CO2 Recovery (%) |
|---|---|---|---|
| TBAF·29.7 H2O | CO2 (20%) + N2 (80%) | 11.3 | 62 |
| CO2 (50%) + N2 (50%) | 15.6 | 72 | |
| CO2 (80%) + N2 (20%) | 21.1 | 87 | |
| TBAF·32.8 H2O | CO2 (20%) + N2 (80%) | 11.4 | 63 |
| CO2 (50%) + N2 (50%) | 15.6 | 73 | |
| CO2 (80%) + N2 (20%) | 21.2 | 88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Ahn, Y.-H. Selective CO2 Capture from CO2/N2 Gas Mixtures Utilizing Tetrabutylammonium Fluoride Hydrates. Molecules 2024, 29, 1284. https://doi.org/10.3390/molecules29061284
Kim H, Ahn Y-H. Selective CO2 Capture from CO2/N2 Gas Mixtures Utilizing Tetrabutylammonium Fluoride Hydrates. Molecules. 2024; 29(6):1284. https://doi.org/10.3390/molecules29061284
Chicago/Turabian StyleKim, Hyeonjin, and Yun-Ho Ahn. 2024. "Selective CO2 Capture from CO2/N2 Gas Mixtures Utilizing Tetrabutylammonium Fluoride Hydrates" Molecules 29, no. 6: 1284. https://doi.org/10.3390/molecules29061284
APA StyleKim, H., & Ahn, Y.-H. (2024). Selective CO2 Capture from CO2/N2 Gas Mixtures Utilizing Tetrabutylammonium Fluoride Hydrates. Molecules, 29(6), 1284. https://doi.org/10.3390/molecules29061284