Abstract
Metabolic syndromes (MetS) and related cardiovascular diseases (CVDs) pose a serious threat to human health. MetS are metabolic disorders characterized by obesity, dyslipidemia, and hypertension, which increase the risk of CVDs’ initiation and development. Although there are many availabile drugs for treating MetS and related CVDs, some side effects also occur. Considering the low-level side effects, many natural products have been tried to treat MetS and CVDs. A five-cyclic triterpenoid natural product, oleanolic acid (OA), has been reported to have many pharmacologic actions such as anti-hypertension, anti-hyperlipidemia, and liver protection. OA has specific advantages in the treatment of MetS and CVDs. OA achieves therapeutic effects through a variety of pathways, attracting great interest and playing a vital role in the treatment of MetS and CVDs. Consequently, in this article, we aim to review the pharmacological actions and potential mechanisms of OA in treating MetS and related CVDs.
1. Introduction
Metabolic syndrome (MetS) is a complex cluster of metabolic disorders, including obesity, which increases the risk for the development and progression of cardiovascular diseases (CVDs) [1]. CVDs are deemed to be the main reason for death, including stroke, ischemic cardiomyopathy, and atherosclerosis [2]. Today, many effective medicines have been used in treating MetS and CVDs, such as hydrochlorothiazide and nifedipine, which are used as first-line drugs to reduce blood pressure [3]; metformin is effective in treating hyperglycemia [4]. However, the continuous use of these treatments may cause adverse reactions such as dyspnea, dizziness, vertigo, headaches, and muscle lysis [5,6].
New drugs are needed urgently to treat MetS and related CVDs without significant side effects. Recently, natural products of plant origin attracted the attention of academics because of their potent pharmacological activity and low-level side effects.
The natural product oleanolic acid (OA: 3b-hydroxyolean-12-en-28-oic acid) is a pentacyclic triterpenoid compound. It has been extracted from many species, including Olea europaea [7]. Studies on biological activity have shown that OA has a liver-protective effect, and has been listed as a liver-protective drug in China [8]. OA also has anti-inflammatory, anti-oxidant, anti-hyperglycemia, anti-hyperlipidemia, cardioprotective, anti-atherosclerotic, and some other pharmacological effects [9,10,11,12,13].
Previous research revealed the medicinal value of OA in curing MetS and related CVDs. Consequently, this review summarized the pharmacological effects and mechanisms of OA in treating Mets and related CVDs. Figure 1 summarizes OA in the treatment of MetS and related CVDs.
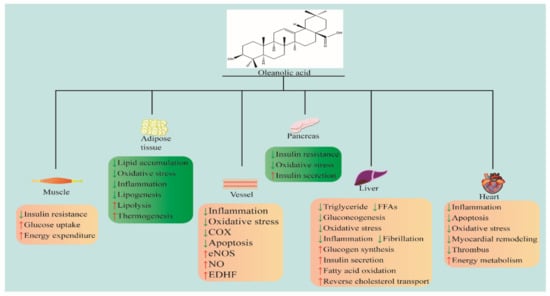
Figure 1.
OA in the treatment of MetS and CVDs. Chemical structures of OA drawn according to [14]. COX: cyclooxygenase; eNOS: endothelial nitric oxide synthase; NO: nitric oxide; FFA: free fatty acid; EDHF: endothelium-derived hyperpolarizing factor. ↓ indicates inhibition; ↑ indicates promotion.
2. Anti-Metabolic Syndromes’ Effects
2.1. Anti-Obesity
Obesity is associated with numerous diseases and a shortened life expectancy [15]. Fat production is the maturation of fat cells by which preadipocytes become adipocytes, so they play an essential part in obesity [16]. In the process, CCAAT/enhancer-binding protein (C/EBP) and peroxisome proliferator-activated receptor γ (PPARγ) are thought to be the vital early regulatory proteins for lipogenesis. Adiponectin, sterol regulatory element-binding protein 1 (SREBP1), and fatty acid synthetase (FAS) are in charge of the production of mature fat cells [17]. OA could inhibit the expression of the visceral fat-specific adipokine and downregulate PPARγ and C/EBPα to reduce the intracellular accumulation of fat in adipocytes [18]. Furthermore, OA may reduce obesity via the suppression of the adipogenic factors PPARα, SREBP1, and FAS [19]. OA has been shown to reduce the synthesis of fat and accelerate the utilization of fat through the alteration of hepatic PPARα, recombinant carnitine palmitoyltransferase 1A (CPT1A), SREBP-1, the acetyl coenzyme A carboxylase, and coupled protein 1 (UCP1) [20]. In addition, OA can reduce blood glucose and lipid levels by promoting carbohydrate and fat metabolism [21]. Another piece of research showed that OA can be effective against postmenopausal obesity by inhibiting fat synthesis acetyl-CoA carboxylase (ACC) and upregulating essential genes for estrogen production, CYP11, CYP1, and CYP17A19 [22].
Inflammation is crucial in obesity [23]; chronic inflammation in adipose tissue is primarily driven by macrophages [24] that are classified into two types: M1-type macrophages and M2-type macrophages [25]. An increase in the ratio of M1/M2-type macrophages can enhance adipocyte growth, fat storage, and adipocyte differentiation [26]. Recent research has discovered that OA was able to reduce inflammation via the inhibition of macrophage infiltration, the M1/M2 ratio in adipose tissues, reactive oxygen species (ROS), and decreasing NACHT, LRR, and PYD structural domain protein 3 (NLRP3) [27].
Resistin is an adipocyte-specific secreted factor associated with adipocyte differentiation [28]. OA could reduce resistin synthesis in vivo by stimulating the cellular signaling transcriptional repressor three signaling and interfering with the tyrosine kinase 2-transcriptional signaling sensor activator [29]. Furthermore, glucose homeostasis and adipocyte differentiation are regulated by transcription factor hepatocyte nuclear factor 1b (HNF1b) [30]. The research showed that OA could relieve glucose/lipid metabolic dysfunction via HNF1b [31].
The causes of obesity are complex, the symptoms are diverse, and multiple organs are implicated, so OA in treating obesity is far from sufficient, especially in molecular mechanisms where it is even more insufficient. Therefore, more research is needed to demonstrate the role of OA in treating obesity.
2.2. Anti-Hyperlipidemia
Hyperlipidemia is defined as elevations of the fasting total cholesterol concentration, which could directly cause some severe diseases [32]. Numerous studies have suggested that OA is beneficial in the treatment of hyperlipidemia. OA could attenuate the triglycerides (TG) in rats by reducing the fat synthesis factor sterol regulatory element and activating transcription factor 1 [33]. OA also reduces total cholesterol (TC) formation by inhibiting cholesterol acyltransferase activity [34]. A high-fat diet will increase the level of peroxisome proliferator-activated receptor gamma coactivator 1β (PGC-1β) leading to lipogenesis and very-low-density lipoprotein secretion [35]; OA could decrease serum lipids in mice via the inhibition of PGC-1β expression [36]. Clinical investigations also have shown that OA decreased serum lipids in hyperlipidemic patients [37].
Hyperlipidemia is frequently one of the risk factors for various issues. Thus, improving blood lipids is critical for human health. Recent research demonstrated that OA can decrease low-density lipoprotein-cholesterol (LDL-c), TC, and TG in mice. The process is thought to be connected to essential targets of lipid synthesis and accumulation.
2.3. Anti-Hypertension
One of the cardiovascular risk factors is hypertension [38]. Research revealed that OA was helpful in hypertension [39,40]. OA could diminish vascular resistance by promoting nitric oxide (NO) and inhibiting COX levels in isolated rat vessels [41]. OA also prevented hypertension in rats via the suppression of NO catabolism [42]. Another study indicated that OA can improve high blood pressure by increasing the expression of eNOS [43]. Meanwhile, OA increased the vasodilator endothelium-derived hyperpolarizing factor (EDHF) and NO to maintain normal blood pressure [44].
The renin–angiotensin system and atrial natriuretic peptide (ANP) are crucial to blood pressure homeostasis [45]. It was found that OA can maintain the homeostasis of blood pressure by inhibiting the renin–angiotensin system and enhancing the fluid balance [46]. OA also could increase the expression of atrial ANP, thus enhancing vascular homeostasis [47]. In addition, the diuretic and nephroprotective properties of OA could reduce hypertension [48]. Furthermore, OA could improve hypertension via upregulating the anti-oxidative stress capacity and enhancing diuretic and natriuretic functions in hypertensive rats [49].
Hypertension is one of the most prevalent systemic metabolic disorders [50]; hypertensive patients also have substantially elevated levels of lipid metabolites [51]. Numerous studies have demonstrated that reducing lipids can improve hypertension. OA was found to reduce hypertension by downregulating the expression of pro-inflammatory factor-secreting phospholipase A2 and fat synthesis factor FAS and inhibiting lipid accumulation [52].
In conclusion, the incidence of hypertension has been rising steadily over the past decade, and the effective treatment of hypertension has a positive impact on middle-age and old-age patients. OA, a natural compound, can protect vascular endothelial cells, enhance body fluid balance, and promote glucose and lipid metabolism to reduce hypertension.
2.4. Anti-Nonalcoholic Fatty Liver
Non-alcoholic fatty liver is caused by hepatic steatosis in the liver [53]. Among the pathological mechanisms, the fat overloading in the liver triggered an inflammatory cascade response and subsequently developed into steatohepatitis [54]. Recent research indicated that OA could delay the development of a nonalcoholic fatty liver by reducing inflammation, steatosis, and fibrosis in rats [55]. Furthermore, the liver could be in danger from microbial disorders and increased intestinal permeability, which may exacerbate the inflammatory responses to the nonalcoholic fatty liver [56]; research has shown that OA could treat nonalcoholic fatty liver by ameliorating intestinal barrier dysfunction and the Toll-like receptor 4 (TLR4)-associated inflammatory responses [57].
Oxidative stress induced by a hepatic lipid overload exacerbates liver injury [58]. It was discovered that OA could substantially mitigate a nonalcoholic fatty liver by ameliorating hepatic oxidative stress and decreasing lipid synthesis factor SREBP1 [59].
Liver X receptors (LXR) are highly expressed in the liver and responsible for cholesterol metabolism and homeostasis [60]; LXR primarily activates the hepatic fat synthesis pathway by activating the promoter region of SREBP-1 [61]. Research demonstrated that OA was able to improve the abnormal accumulation of fat in the liver by reducing the expression of LXR and the activity of SREBP-1, as well as increasing the expression of reverse cholesterol transport (RCT)-related genes, including ATP-binding cassette transporter protein (ABC)A1 and ABCG1 [62]. Furthermore, OA could directly inhibit the expression of the SREBP-1 protein and decrease fatty acid accumulation in the body, thus ameliorating the progress of nonalcoholic fatty liver [63].
Briefly speaking, OA inhibits fat accumulation, accelerates cholesterol transport in the liver, and suppresses hepatic inflammation and oxidative stress in the treatment of nonalcoholic fatty liver.
2.5. Anti-Diabetes Mellitus
Diabetes mellitus is a metabolic disorder characterized by elevated blood sugar, mainly caused by an absolute or relative insulin deficiency and insulin resistance, classified as type 1 and type 2, with type 2 comprising nearly 95% of cases [64]. Insulin sensitivity can be affected by oxidative stress, inflammation, and metabolic disorders.
Inflammation is significant in diabetes mellitus [65]; an inordinate increase of inflammatory factors hinders insulin receptor signaling and leads to insulin resistance [66]. Research has shown that the expression of TLR4, TLR9, interleukin 6 (IL-6), IL-18, tumor necrosis factor α (TNF-α), TNF-1, and C-reactive protein (CRP) was reduced by OA in diabetic rats [67,68,69,70]. Furthermore, OA also could improve insulin resistance by inhibiting the activity of nuclear factor-κB (NF-κB) [71].
Oxidative stress is closely associated with diabetes and causes deleterious consequences of diabetes [72]. OA could improve the antioxidant capacity in diabetic rats by attenuating the levels of NO and malonaldehyde (MDA), as well as enhancing the level of catalase (CAT) and superoxide dismutase (SOD) [73,74]. In addition, OA was able to enhance the antioxidant function of mitochondria by increasing the expression of glutathione peroxidase 4 (Gpx4) and SOD [75]. Furthermore, OA was reported to improve the mitochondrial ultrastructure and function and antioxidant capacity by inhibiting MDA and ROS levels, as well as increasing CAT, SOD, and glutathione peroxidase (GSH-px) in diabetic rats [11,76,77].
Diabetes is associated with disorders of energy metabolism [78]. Lipid accumulation and the dysregulation of glucose homeostasis are significant causes of insulin resistance [79]. It was demonstrated that OA could improve diabetes by inhibiting the level of α-glucosidase and α-amylase [80]. Meanwhile, OA was able to improve diabetes in rats by stimulating insulin secretion [81] and decreasing blood glucose and blood lipid levels [82], increasing hepatic glycogen and muscle glycogen [83]. The research indicated that OA could prevent hyperglycemia by inhibiting glucose absorption and promoting the change of glucose to glycogen [84]. Elevated blood glucose and glycated hemoglobin (HbA1c) levels (referred to as the prediabetic condition) occurred before the transition from normal to diabetic [85] and OA could improve glucose homeostasis via the reduction of blood glucose and HbA1c levels [86]. It was verified that OA affects diabetes, which was related to increasing glucose transporter-5 (GLUT-5) and GLUT-4 expressions and decreasing FAS and ACC-1 expressions [87]. In addition, OA was observed to maintain glucose homeostasis in rats by decreasing the activity of hexokinase, the expression of glycogen phosphorylase (GP), and increasing the expression of glycogen synthase (GS) [88]. Another study indicated that OA could accelerate glucose and lipid metabolism via increasing the level of PPARγ/α and its related regulators, as well as GLUT-4 and fatty acid transport protein-1 (FATP-1) proteins [89]. Furthermore, takeda G protein-coupled receptor 5 (TGR5) belongs to the g-protein-coupled receptors involved in various cellular physiological effects [90]. By activating the expression of TGR5, OA was able to decrease the blood glucose levels [91]. Based on the accumulated evidence, the imbalance of the phosphatidylinositol-3-kinase (PI3K)/protein kinase B (Akt) signaling pathway could cause the development of diabetes mellitus [92]. OA was verified to inhibit gluconeogenesis by reducing the level of Akt, forkhead box O1 (FoxO1), and glucose-6-phosphatase (G6Pase) [93]. It also exhibited that OA was able to accelerate glucose transport by increasing p-Akt levels and GS levels, as well as decreasing GP levels [94,95]. Furthermore, OA has positive effects on diabetes via increasing PI3K/Akt and AMPK phosphorylation, phosphoenolpyruvate carboxykinase (PEPCK), and G6Pase levels, as well as decreasing the level of the mammalian target of rapamycin (mTOR) [96]. It was discovered that OA could improve insulin resistance through the activation of the level of the insulin receptor substrate (IRS-1) and PI3K/Akt [97]. Moreover, OA may normalize insulin, high-density lipoprotein (HDL), IRS1, GLUT2, GLUT4, and Akt levels, and decrease TC, TG, and low-density lipoprotein (LDL) levels [98]. Furthermore, OA could decrease insulin resistance by improving β-cells [99].
High-glucose environments have been found to cause endothelial cell dysfunction [100]. Research has shown that OA attenuated human umbilical vein endothelial cells (HUVECs) function damage via activating PPARδ, increasing the phosphorylation of Akt and eNOS [101]. Furthermore, persistent hyperglycemia will change blood composition, such as erythrocyte morphology [102], and increase the production of erythropoietin (EPO) [103]. OA could improve diabetes by reducing plasma glucose, HbA1c, and EPO levels and increasing the antioxidant capacity of erythrocytes [104].
Complications caused by diabetes are also a leading cause of harm to human health, such as diabetic nephropathy [105]. Research reported that OA could protect rats against diabetic nephropathy by restoring plasma aldosterone and renal injury molecule-1 [106]. In addition, advanced glycosylation end products, such as renal N-(carboxymethyl) lysine, HbA1c, and glycosylated albumin, are also related to the development of diabetic nephropathy [107]. OA was able to inhibit diabetic nephropathy via a reduction of the level of renal N-(carboxymethyl) lysine, HbA1c, urinary albumin, and urine glycated albumin, as well as increasing the level of plasma insulin and renal creatinine clearance [108]. Furthermore, OA could also restore the damaged renal structure by increasing insulin secretion, renal units, and endothelial-selective adhesion molecules, and decreasing urinary albumin/creatinine levels [109].
There is accumulating evidence that OA cures diabetes by decreasing inflammation, reducing oxidative stress, and protecting endothelial cell function. Furthermore, OA could enhance the glucose–lipid metabolism in diabetic rats, restore blood components damaged by high glucose levels, and alleviate diabetic nephropathy problems. To summarize, OA in the treatment of diabetes mellitus has shown tremendous potential and is supported by numerous pieces of research; however, this research may require additional clinical trials to confirm. The detailed pharmacological effects of OA on metabolic syndrome are shown in Table 1.

Table 1.
Pharmacological Effects of OA in the Treatment of MetS.
3. Anti-Cardiovascular Diseases Effects
3.1. Anti-Stroke
Stroke is one of the main causes of increased mortality [110], which is affected by inflammation, oxidative stress, and nerve damage [111].
The key mechanism in the formation of ischemic stroke is oxidative stress [112], which also causes neuronal apoptosis, inflammation, and nerve injury [113]. It was reported that OA reduced cerebral ischemic stroke damage by increasing the level of mitochondrial antioxidant α-tocopherol (α-TOC) and GSH, as well as decreasing the leakage of the damage marker lactate dehydrogenase (LDH) [114]. Furthermore, OA was able to improve oxidative stress in brain-injured rats; the results showed that OA treatment significantly increased the activity of SOD, GSH-Px, mitochondrial membrane potential (MMP), and succinate dehydrogenase (SDH), and decreased MDA and LDH levels [115]. Meanwhile, OA also could restrain the blood–brain barrier indicator occludin, matrix metalloproteinase 9 (MMP9), and Evans blue leakage, and inhibit oxidative indicator dihydroethidium fluorescence and MDA expression [116]. In addition, heme oxygenase-1 (HO-1) is the most effective antioxidant response element, and glycogen synthase kinase-3β (GSK-3β) is able to regulate HO-1 in controlling oxidative stress [117]. OA attenuated cytotoxicity and ROS via the regulation of the GSK-3β/HO-1 signal in rats [118].
In general, OA from natural product sources has neuroprotective functions, such as the improvement of the blood–brain barrier, reduction of nerve injury, and cerebral edema in mice; the mechanism was primarily associated with the improvement of oxidative damage. However, it remains to be determined whether OA in stroke treatment has a more promising mechanism.
3.2. Heart Protection
Heart disease has a high mortality rate, and the number of deaths is still rising [119]. Oxidative stress is a significant reason for heart disease; the elevated expression of ROS causes cardiomyocyte dysfunction and damage [120]. Research demonstrated that OA promoted the antioxidant capacity of the heart via the reduction level of the lipid peroxidation products [121]. Furthermore, OA was able to prevent diabetic cardiomyopathy through the regulation of HO-1/Nrf2 to increase SOD and GS, as well as decrease MDA and GP [122]. Meanwhile, OA was verified to prevent CVDs by improving the inflammatory reaction, MDA, SOD, GPx, as well as heart weight in rats [123]. In addition, OA could improve myocardial apoptosis by increasing the antioxidant capacity and decreasing apoptosis signaling caspase-3 and BAX activity, increasing Bcl-2 activity [124,125].
Endothelin 1 (ET-1) aggravates the development of CVDs [126], and OA could inhibit cardiomyocyte injury through the regulation of the expression of ET-1 [12]. Furthermore, ET-1 and NF-κB modulate the fibrotic process in the heart, as well as promote the expression of fibronectin in cardiac tissues [127]. OA could improve fibrotic hearts in rats by reducing the activation of NF-κB and ET-1 [128]. Moreover, the Akt/mTOR exacerbates the pathological process of myocardial remodeling [129]; OA performed cardiac protection with the inhibition of vascular remodeling by decreasing the levels of Akt and mTOR [130]. In addition, OA possessed the ability to suppress the platelet aggregation mediated by phospholipase C, thereby aiding in the prevention of cardiovascular thrombosis [131].
Therefore, current research demonstrates that OA could treat a variety of heart diseases, as well as prevent cardiac fibrosis and the cardiac remodeling process. The mechanism includes the inhibition of inflammation, oxidative stress, and the improvement of the expression of vasoconstrictive factors.
3.3. Anti-Atherosclerosis
Atherosclerosis (AS) is the underlying pathology of CVDs [132]. OA could prevent AS by inhibiting many pathological developments, such as oxidative stress, endothelial dysfunction, and lipid deposition. Oxidative stress was deemed the critical mechanism in AS [133]. Research demonstrated that OA may safeguard HUVECs damage by inhibiting the levels of lipoprotein receptor 1 (LOX-1), ROS, as well as hypoxia-inducible factor 1 α (HIF-1α) [134]. Moreover, OA has been confirmed to alleviate HUVECs damage via the reduction in the level of ROS and LOX-1, as well as enhancing the level of Nrf2/HO-1 [135].
PPARγ is considered a ligand-activated transcription factor that regulates the glycolipid metabolism, and adiponectin promotes fatty acid biosynthesis and inhibits hepatic gluconeogenesis [136]. OA could reduce lipids and enhance high-density lipoprotein cholesterol (HDL-c) by increasing PPARγ and adiponectin Receptor 1 (AdipoR1) Levels, decreasing AdopoR2 levels [137].
Farnesoid-X-receptor (FXR) is associated with the bile metabolism [138], and angiotensin1-7 (Ang1-7) has been implicated as an AS protector [139]. OA was found to decrease the levels of lipids in rats via the regulation of the expression of FXR and Ang1-7 [13]. In addition, OA inhibited the expression of iNOS, thereby delaying the progression of aortic stenosis [140].
In conclusion, OA can reduce the area of vascular lipid plaque and treat AS by protecting HUVECs, reducing inflammatory factors and the accumulation of lipids. The detailed pharmacological effects of OA on metabolic syndrome-related cardiovascular diseases are shown in Table 2.

Table 2.
Pharmacological effects of OA in the treatment of CVDs.
4. Signaling Pathways of OA for the Treatment of MetS and CVDs
Many pharmacological effects of OA in alleviating Mets have been discovered, such as the inhibition of inflammation, reduction of adipogenesis, and improvement of insulin resistance, and they have also been thoroughly investigated; many of these mechanisms were critical pathways for the treatment of CVDs, such as PPAR, NF-κB, and Akt.
4.1. PPAR Signaling Pathway
Peroxisome proliferator-activated receptors (PPARs) belong to the nuclear receptor superfamily, and the PPAR subtypes have been identified, including PPARα, PPARγ, and PPARβ/δ [141]. There is a lot of PPARα expression in brown adipose tissue for promoting energy depletion; PPARγ has high expression levels in white adipose tissue, which could improve energy storage mainly by facilitating adipogenesis and lipotransformation. Meanwhile, PPARγ and C/EBPs are synergistically activated adipogenesis. PPARβ/δ is commonly expressed in skeletal muscle and is involved in fatty acid oxidation [142].
Studies showed that OA could reduce the expression of the visceral fat-specific adipokine adiponectin and the accumulation of fat via the downregulation of PPARγ and C/EBPα [18]. Furthermore, OA could accelerate fat utilization by reducing PPARα, inhibiting CPT1, and promoting UCP1. It was also observed that OA suppressed fat accumulation via the regulation of the level of SERBP1 and ACACA [20].
ChREBP, in combination with SREBP-1, activates adipogenic enzymes, such as ACC and FAS [143]. Research has shown that OA could inhibit SREBP-1 and ChREBP, thus reducing FAS and ACC expression; OA also promoted the expression of PPARα, promoting fat utilization and inhibiting fat production [19]. In addition, OA exhibited an endothelial cell protection effect through the regulation of PPARδ to promote the expression of Akt and eNOS [101]. Adiponectin is regarded as a marker of insulin sensitization [144]. OA can increase GLUT4 and adiponectin protein expression via the activation of PPARγ, and it also can increase the level of fatty acid transport protein 1 (FATP1) and long-chain acyl-CoA synthetase (ACSL) via the activation of PPARα, thereby enhancing insulin sensitivity and accelerating fat utilization [89]. AdipoR1 and AdipoR2 exert beneficial effects on AS; AdipoR1 and AdipoR2 have opposing roles in regulating the glucose and energy metabolism [145]. Studies demonstrated that OA could improve AS in rats by regulating the expression of PPARγ to increase AdipoR1 and decrease AdipoR2 [137].
4.2. PI3K/Akt Signaling Pathway
The metabolism of matter usually accompanies the metabolism of energy; the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PI3K/Akt) signaling pathways are vital for the regulation of growth and metabolism [146]. The PI3K belongs to lipid kinases, whose phosphorylated phosphatidylinositol is an integral part of the membrane of eukaryotic cells [147]; the downstream signaling for the PI3Ks is Akt, and research demonstrated that the PI3K/Akt signaling pathways have an important role in ameliorating diseases such as diabetes, hypertension, and cardiovascular diseases [92].
Research revealed that the Akt signaling pathway is involved in maintaining blood pressure homeostasis [148]. Studies suggested that OA could exert endothelial protective effects primarily via the mediation of PPARδ, activating the Akt and eNOS [101].
The downstream of PI3K/Akt is FoxO1, which regulates hepatic gluconeogenesis [149]. It was verified that OA could inhibit gluconeogenesis by stimulating the phosphorylation of Akt and FoxO1, as well as inhibiting the level of G6Pase [93]. Another piece of research showed that OA protected against diabetes by regulating PI3K/Akt expression to inhibit GSK3 and GS expression, as well as increase GP [94]. In addition, OA may improve insulin resistance via the reduction in the expression of mTOR via PI3K/Akt. OA can also inhibit gluconeogenesis by reducing the expression of G6Pase and PEPCK [96]. IRS signaling is a common pathological mechanism of insulin resistance [150]; OA alleviated insulin resistance by increasing the level of IRS-1, which was achieved by PI3K/Akt [97]. Furthermore, OA also ameliorated insulin resistance by inhibiting the expression of IRS-1 and Akt, as well as GLUT2 and GLUT4 [98].
Myocardial pressure overload stimulates cell surface receptors, resulting in PI3K activation and Akt phosphorylation. Although the acute activation of Akt is cardioprotective, over-activation is detrimental to heart failure [151]. In chronic heart failure, Akt phosphorylation is accompanied by an increased level of FoxO3a phosphorylation [152]. mTOR is upregulated in response to stimuli of cardiac remodeling [152]. GSK3 is another well-established PI3K/Akt signaling pathway target; the hypertrophic myocardium induces the activation of GSK3, resulting in cardiac remodeling [153]. OA revealed potent anti-myocardial hypertrophy and fibrosis activity through the inhibition of Akt activation, which decreased the expression of mTOR, FoxO3a, and GSK3 in rats [130].
4.3. NF-κB Signaling Pathway
Nuclear factor-kappa B (NF-κB) is formed by homodimers or heterodimers to coordinate the expression of hundreds of genes. Under physiological conditions, NF-κB is sequestered in the cytoplasm by interacting with any member of the inhibitor-κB (I-κB) family of protein inhibitors, for example, IκBα, IκBβ, and p100. In the presence of activating signals, IκB kinase (IKK) rapidly phosphorylates IκBβ. It promotes IκBα ubiquitination and proteasomal degradation, allowing NF-κB translocated to the nucleus upon activation, where it regulates the transcription of target genes [154].
The TLR signaling pathway is capable of activating NF-κB, which induces an inflammatory reaction [155]. OA could ameliorate inflammation by reducing serum TLR9 and NF-κB [67]. Furthermore, OA could reduce inflammatory cell infiltration by decreasing the level of TLR4/NF-κB in diabetic nephropathy rats [69]. In addition, OA was able to improve inflammation and insulin resistance by reducing TNF-α, IL-6, IRS1, and GLUT4 proteins via NF-κB [71].
The NF-κB and mitogen-activated protein kinase (MAPK) in cardiomyocytes synergistically induce inflammatory responses in cardiac tissues [156]. Research has shown that OA could decrease inflammatory factors such as IL-6, MCP-1, and TNF-α by reducing the level of NF-κB/MAPK [124]. NF-κB also promoted the process of cardiac fibrosis and mediated increased connexin synthesis and the expression of fibrous connexin in myocardial tissues in macrovascular and microvascular endothelial cell lines [157]. OA decreased the expression of IκBβ mRNA and restored the normalization of the expression of down-regulated IκBα mRNA, thereby reducing the level of NF-κB [128]. The molecular mechanisms of OA in treating MetS and CVDs are the regulation of PPAR, PI3K/Akt, and NF-κB, the pathways of which are shown in Figure 2.
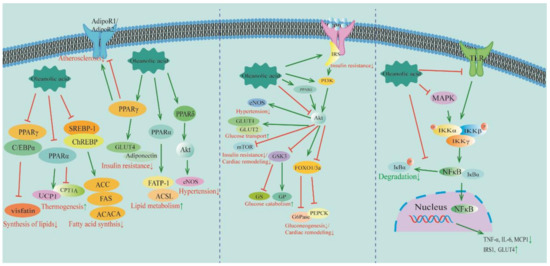
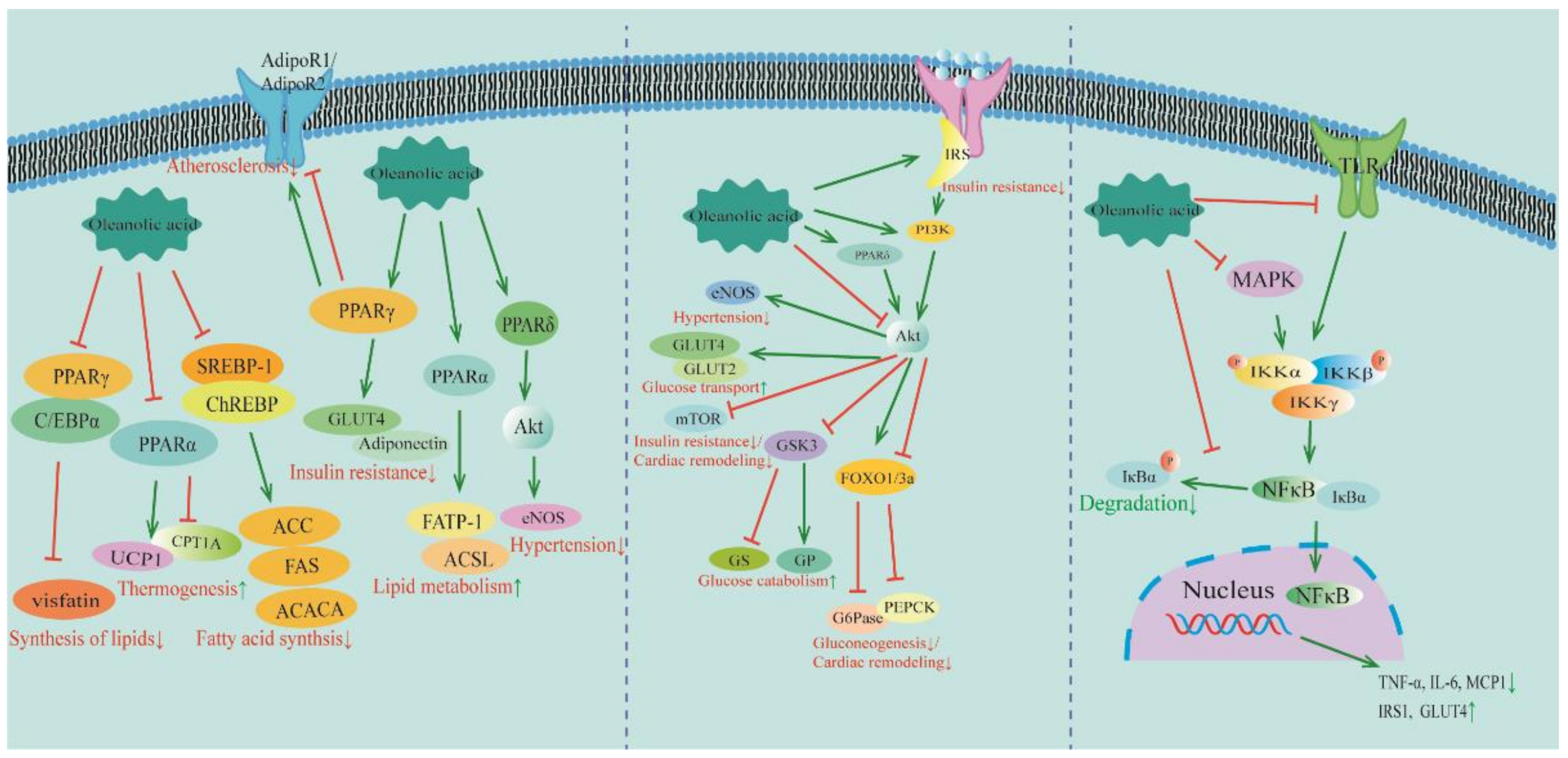
Figure 2.
The molecular mechanisms of OA in treating MetS and CVDs: regulation of PPAR, PI3K/Akt, and NF-κB pathways. Information is derived from the references in Table 1 and Table 2. PPAR: peroxisome proliferator-activated receptor; C/EBPα: CCAAT/enhancer-binding protein α; UCP1: coupled protein 1; CPT1A: carnitine palmitoyltransferase 1A; ACC: acetyl-CoA carboxylase; FAS: fat synthesis factor; ACACA: acetyl-CoA carboxylase alpha; GLUT: glucose transporter; FATP-1: fatty acid transport protein-1; ACSL: long-chain acyl-CoA synthetase; Akt: protein kinase B; eNOS: endothelial nitric oxide synthase; IRS: insulin receptor substrate; PI3K: phosphatidylinositol-3-kinase; mTOR: mammalian target of rapamycin; GSK3: glycogen synthase kinase-3; GS: glycogen synthase; GP: glycogen phosphorylase; FoxO1/3a: forkhead box O1/3a; G6Pase: glucose-6-phosphatase; PEPCK: phosphoenolpyruvate carboxykinase; MAPK: mitogen-activated protein kinase; Ikk: IκB kinase; NF-κB: nuclear factor-κB; TNF-α: tumor necrosis factor α; IL-6: interleukin 6; MCP1: monocyte chemoattractant protein-1; GLUT: glucose transporter. ↓ indicates inhibition; ↑ indicates promotion.
5. Metabolism, Bioavailability, and Clinical Potential of OA
Pharmacokinetic studies on OA are rarely reported, but are of great interest. Song et al. [158] reported a pharmacokinetic study of OA, made up of 18 healthy Chinese male volunteers receiving a single dose of 40 mg OA in a capsule formulation. The results showed that the peak plasma OA concentration was 12.1 ± 6.8 ng/mL, which appeared at 5.2 ± 2.9 h after the oral administration of OA, and the area under the concentration–time curve (AUC) was 124.3 ± 106.7 ng h/mL. In addition, Rada et al. [159]. conducted a randomized crossover control trial with nine adult male volunteers to study the pharmacokinetics of OA. The volunteers took 70 g of pomace olive oil containing 30 mg of OA. The peak concentrations of OA were reported to be 598.2 ± 176.7 ng/mL, occurring 3.0 ± 0.8 h after administration, with an AUC0-48 of 3181.9 ± 894.3 ng h/mL. Additionally, de la Torre et al. [160] conducted a pharmacokinetic investigation of OA involving the administration of 30 mL of olive oil (containing 4.7 mg of OA) to 12 volunteers. The study found that the peak plasma concentration of OA was 5.1 ± 2.1 ng/mL [160]. More recently, there has been a pharmacokinetic study of OA conducted by González et al. [161]. Twenty-two volunteers were administered a single dose of functional olive oil containing 30 mg of OA. The study revealed peak plasma OA concentrations ranging from 500–600 ng/mL, with an AUC0-∞ which was 2862.50 ± 174.50 ng h/mL.
OA has a low solubility in water, limiting its own bioavailability and therapeutic potential. Despite the low bioavailability of OA, an intact form of OA is present in tissues after oral intake (4 or 8 weeks) in mice, including the liver and brain. Consequently, these results indicate that OA can cross the blood–brain barrier and have a protective effect on nerves, and the liver was the main organ for the storage and metabolism of OA [162,163]. To address this, there have been some studies combining OA with nanomaterials or using OA as an adjuvant to improve its bioavailability. Férez et al. [164] used cyclodextrins to encapsulate OA, and the results show that OA-encapsulated compounds have advantages over unencapsulated OA in terms of chemical stability, the promotion of cell migration, and the preservation of cell viability. A solidified phospholipid complex composed of an OA–phospholipid complex and hydroxyapatite has been developed, which has increased the oral availability of OA to 240% [165]. Another piece of research developed a self-microemulsifying drug delivery system of OA and conducted pharmacokinetic studies in rats, which increased the bioavailability of OA to 507% [166].
The clinical trials on OA are summarized as follows. Luo et al. [37] designed a small-scale clinical trial to assess the hypolipidemic effect of OA. Hyperlipidemic patients were administrated with OA for four weeks (four tablets at once, three times a day). The results displayed that the TC, TG, and HDL-c levels in the serum decreased significantly. Another clinical study was conducted to assess whether the regular intake of an OA-enriched olive oil is effective in the prevention of diabetes. Prediabetic individuals (176 patients) were randomized to receive 55 mL/day of OA-enriched olive oil (equivalent-dose 30 mg OA/day) or the same oil not enriched (control group). The results showed that the intake of OA-enriched olive oil reduces the risk of developing diabetes in prediabetic patients [167]. Furthermore, a regular consumption of virgin olive oil is associated with a reduced risk of cardiovascular disease. Sanchez-Rodriguez et al. [168] aimed to assess whether the raw intake of a functional olive oil (487 parts per million of phenolic compounds and enriched with 389 parts per million of triterpenes) supplementation (30 mL per day) over three weeks would provide additional health benefits. Fifty-one healthy adults participated in a randomized, crossover, and controlled study. The results showed that the intake of functional olive oil decreased urinary 8-hidroxy-20-deoxyguanosine, plasma IL-8, and TNF-α concentrations. This study provides first-level evidence for the in vivo health benefits, reduction of DNA oxidation, and plasma inflammatory biomarkers of olive oil triterpenoids (oleanolic acid) in healthy humans.
6. Conclusions
In summary, OA is a plant-derived pentacyclic triterpenoid natural compound, which has been extensively studied to cure MetS and CVDs in recent years. The research has shown that OA has several therapeutic effects. First, OA decreases blood glucose by increasing insulin sensitivity and promoting glucose uptake and utilization. Second, OA may also inhibit the level of blood lipids and improve the lipid metabolism. In addition, OA has anti-oxidant and anti-inflammatory effects, reducing the injury to the vascular endothelium caused by inflammatory responses. Finally, it also increases NO production, promotes vasodilation, reduces blood pressure, and prevents AS. Overall, OA has various beneficial effects as a natural product for MetS and related cardiovascular diseases.
However, OA also has some objective limitations in the treatment of MetS and CVDs. First of all, although OA is considered a natural and safe drug or food supplement, there are few studies focusing on the toxic side effects of OA, which should be concerned in the future. Secondly, OA has low bioavailability and is insoluble in water. But, there have already been some studies on the combination of OA with nanomaterials, or as an adjuvant to improve the bioavailability of OA, which have achieved good results. Finally, there have been a few clinical trials on OA. There are problems concerning clinical trials such as the small number of participants, the uncertainty of molecular targets, and the short experimental cycle, and preclinical experiments should be combined to determine the efficacy, molecular targets, and safety of OA.
Author Contributions
Conceptualization: Q.L., D.Y. and Q.T.; first draft preparation: Q.L. and Y.W.; review and editing: X.L., W.C. and Q.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Key Research and Development Projects of the Hunan Provincial Science and Technology Department (No. 2022SK2011), the Science and Technology Innovation team of the Hunan Province (No. 2021RC4064), the Key Projects of the Hunan Provincial Department of Education (No. 20A379), the Graduate Innovation Project of Hunan University of Chinese Medicine (No. 2023CX163), the Excellent Youth Project of the Hunan Provincial Department of Education (No. 22B0387), and the Natural Science Foundation of the Hunan Province (No. 2022JJ40314).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Silveira Rossi, J.L.; Barbalho, S.M.; Reverete de Araujo, R.; Bechara, M.D.; Sloan, K.P.; Sloan, L.A. Metabolic syndrome and cardiovascular diseases: Going beyond traditional risk factors. Diabetes/Metab. Res. Rev. 2022, 38, e3502. [Google Scholar] [CrossRef]
- Joseph, P.; Leong, D.; McKee, M.; Anand, S.S.; Schwalm, J.D.; Teo, K.; Mente, A.; Yusuf, S. Reducing the Global Burden of Cardiovascular Disease, Part 1: The Epidemiology and Risk Factors. Circ. Res. 2017, 121, 677–694. [Google Scholar] [CrossRef] [PubMed]
- Fishel Bartal, M.; Blackwell, S.C.; Pedroza, C.; Lawal, D.; Amro, F.; Samuel, J.; Chauhan, S.P.; Sibai, B.M. Oral combined hydrochlorothiazide and lisinopril vs nifedipine for postpartum hypertension: A comparative-effectiveness pilot randomized controlled trial. Am. J. Obstet. Gynecol. 2023, 228, 571.e1–571.e10. [Google Scholar] [CrossRef] [PubMed]
- Davidsen, L.; Jensen, M.H.; Cook, M.E.; Vestergaard, P.; Knop, F.K.; Drewes, A.M.; Olesen, S.S. Metformin treatment is associated with reduced risk of hypoglycaemia, major adverse cardiovascular events, and all-cause mortality in patients with post-pancreatitis diabetes mellitus: A nationwide cohort study. Eur. J. Endocrinol. 2024, 190, 44–53. [Google Scholar] [CrossRef]
- Ward, N.C.; Watts, G.F.; Eckel, R.H. Statin Toxicity. Circ. Res. 2019, 124, 328–350. [Google Scholar] [CrossRef] [PubMed]
- Elizalde-Velázquez, G.A.; Herrera-Vázquez, S.E.; Gómez-Oliván, L.M.; García-Medina, S. Health impact assessment after Danio rerio long-term exposure to environmentally relevant concentrations of metformin and guanylurea. Chemosphere 2023, 341, 140070. [Google Scholar] [CrossRef]
- Pollier, J.; Goossens, A. Oleanolic acid. Phytochemistry 2012, 77, 10–15. [Google Scholar] [CrossRef]
- Liu, J. Pharmacology of oleanolic acid and ursolic acid. J. Ethnopharmacol. 1995, 49, 57–68. [Google Scholar] [CrossRef]
- Yoo, S.R.; Jeong, S.J.; Lee, N.R.; Shin, H.K.; Seo, C.S. Quantification Analysis and In Vitro Anti-Inflammatory Effects of 20-Hydroxyecdysone, Momordin Ic, and Oleanolic Acid from the Fructus of Kochia scoparia. Pharmacogn. Mag. 2017, 13, 339–344. [Google Scholar] [CrossRef]
- De Stefani, C.; Vasarri, M.; Salvatici, M.C.; Grifoni, L.; Quintela, J.C.; Bilia, A.R.; Degl’Innocenti, D.; Bergonzi, M.C. Microemulsions Enhance the In Vitro Antioxidant Activity of Oleanolic Acid in RAW 264.7 Cells. Pharmaceutics 2022, 14, 2232. [Google Scholar] [CrossRef]
- Wang, X.; Liu, R.; Zhang, W.; Zhang, X.; Liao, N.; Wang, Z.; Li, W.; Qin, X.; Hai, C. Oleanolic acid improves hepatic insulin resistance via antioxidant, hypolipidemic and anti-inflammatory effects. Mol. Cell Endocrinol. 2013, 376, 70–80. [Google Scholar] [CrossRef]
- Wu, D.; Zhang, Q.; Yu, Y.; Zhang, Y.; Zhang, M.; Liu, Q.; Zhang, E.; Li, S.; Song, G. Oleanolic Acid, a Novel Endothelin A Receptor Antagonist, Alleviated High Glucose-Induced Cardiomyocytes Injury. Am. J. Chin. Med. 2018, 46, 1187–1201. [Google Scholar] [CrossRef]
- Pan, Y.; Zhou, F.; Song, Z.; Huang, H.; Chen, Y.; Shen, Y.; Jia, Y.; Chen, J. Oleanolic acid protects against pathogenesis of atherosclerosis, possibly via FXR-mediated angiotensin (Ang)-(1-7) upregulation. Biomed. Pharmacother. Biomed. Pharmacother. 2018, 97, 1694–1700. [Google Scholar] [CrossRef]
- Castellano, J.M.; Ramos-Romero, S.; Perona, J.S. Oleanolic Acid: Extraction, Characterization and Biological Activity. Nutrients 2022, 14, 623. [Google Scholar] [CrossRef]
- Piché, M.E.; Tchernof, A.; Després, J.P. Obesity Phenotypes, Diabetes, and Cardiovascular Diseases. Circ. Res. 2020, 126, 1477–1500. [Google Scholar] [CrossRef]
- White, U.A.; Stephens, J.M. Transcriptional factors that promote formation of white adipose tissue. Mol. Cell Endocrinol. 2010, 318, 10–14. [Google Scholar] [CrossRef]
- Ali, A.T.; Hochfeld, W.E.; Myburgh, R.; Pepper, M.S. Adipocyte and adipogenesis. Eur. J. Cell Biol. 2013, 92, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.Y.; Kang, S.W.; Kim, J.L.; Li, J.; Lee, E.S.; Gong, J.H.; Han, S.J.; Kang, Y.H. Oleanolic acid reduces markers of differentiation in 3T3-L1 adipocytes. Nutr. Res. 2010, 30, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Djeziri, F.Z.; Belarbi, M.; Murtaza, B.; Hichami, A.; Benammar, C.; Khan, N.A. Oleanolic acid improves diet-induced obesity by modulating fat preference and inflammation in mice. Biochimie 2018, 152, 110–120. [Google Scholar] [CrossRef]
- Acin, S.; Munoz, D.L.; Guillen, A.; Soscue, D.; Castano, A.; Echeverri, F.; Balcazar, N. Triterpene-enriched fractions from Eucalyptus tereticornis ameliorate metabolic alterations in a mouse model of diet-induced obesity. J. Ethnopharmacol. 2021, 265, 113298. [Google Scholar] [CrossRef] [PubMed]
- De Melo, C.L.; Queiroz, M.G.; Fonseca, S.G.; Bizerra, A.M.; Lemos, T.L.; Melo, T.S.; Santos, F.A.; Rao, V.S. Oleanolic acid, a natural triterpenoid improves blood glucose tolerance in normal mice and ameliorates visceral obesity in mice fed a high-fat diet. Chem. Biol. Interact. 2010, 185, 59–65. [Google Scholar] [CrossRef]
- Wan, Q.; Lu, H.; Liu, X.; Yie, S.; Xiang, J.; Yao, Z. Study of oleanolic acid on the estrodiol production and the fat production of mouse preadipocyte 3T3-L1 in vitro. Hum. Cell 2015, 28, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Horng, T. Lipid Metabolism in Regulation of Macrophage Functions. Trends Cell Biol. 2020, 30, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Shen, P.; Huang, Y.; Han, L.; Ba, X.; Huang, Y.; Yan, J.; Li, T.; Xu, L.; Qin, K.; et al. Wutou decoction attenuates the synovial inflammation of collagen-induced arthritis rats via regulating macrophage M1/M2 type polarization. J. Ethnopharmacol. 2023, 301, 115802. [Google Scholar] [CrossRef] [PubMed]
- Chylikova, J.; Dvorackova, J.; Tauber, Z.; Kamarad, V. M1/M2 macrophage polarization in human obese adipose tissue. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czechoslov. 2018, 162, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zeng, H.; Xu, M.; Huang, C.; Tao, L.; Li, J.; Zhang, T.; Chen, H.; Xia, J.; Li, C.; et al. Oleanolic Acid Improves Obesity-Related Inflammation and Insulin Resistance by Regulating Macrophages Activation. Front. Pharmacol. 2021, 12, 697483. [Google Scholar] [CrossRef] [PubMed]
- Lazar, M.A. Resistin- and Obesity-associated metabolic diseases. Horm. Metab. Res. 2007, 39, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Sung, H.Y.; Kim, M.S.; Kim, J.L.; Kang, M.K.; Gong, J.H.; Park, H.S.; Kang, Y.H. Oleanolic acid suppresses resistin induction in adipocytes by modulating Tyk-STAT signaling. Nutr. Res. 2013, 33, 144–153. [Google Scholar] [CrossRef]
- Wang, X.; Wu, H.; Yu, W.; Liu, J.; Peng, J.; Liao, N.; Zhang, J.; Zhang, X.; Hai, C. Hepatocyte nuclear factor 1b is a novel negative regulator of white adipocyte differentiation. Cell Death Differ. 2017, 24, 1588–1597. [Google Scholar] [CrossRef]
- Su, S.; Wu, G.; Cheng, X.; Fan, J.; Peng, J.; Su, H.; Xu, Z.; Cao, M.; Long, Z.; Hao, Y.; et al. Oleanolic acid attenuates PCBs-induced adiposity and insulin resistance via HNF1b-mediated regulation of redox and PPARgamma signaling. Free Radic. Biol. Med. 2018, 124, 122–134. [Google Scholar] [CrossRef]
- Nelson, R.H. Hyperlipidemia as a risk factor for cardiovascular disease. Prim. Care 2013, 40, 195–211. [Google Scholar] [CrossRef]
- Yunoki, K.; Sasaki, G.; Tokuji, Y.; Kinoshita, M.; Naito, A.; Aida, K.; Ohnishi, M. Effect of dietary wine pomace extract and oleanolic acid on plasma lipids in rats fed high-fat diet and its DNA microarray analysis. J. Agric. Food Chem. 2008, 56, 12052–12058. [Google Scholar] [CrossRef]
- Lin, Y.; Vermeer, M.A.; Trautwein, E.A. Triterpenic Acids Present in Hawthorn Lower Plasma Cholesterol by Inhibiting Intestinal ACAT Activity in Hamsters. Evid. Based Complement. Altern. Med. Ecam 2011, 2011, 801272. [Google Scholar] [CrossRef]
- Lin, J.; Yang, R.; Tarr, P.T.; Wu, P.H.; Handschin, C.; Li, S.; Yang, W.; Pei, L.; Uldry, M.; Tontonoz, P.; et al. Hyperlipidemic effects of dietary saturated fats mediated through PGC-1beta coactivation of SREBP. Cell 2005, 120, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wen, X.; Zhang, W.; Wang, C.; Liu, J.; Liu, C. Hypolipidemic effect of oleanolic acid is mediated by the miR-98-5p/PGC-1beta axis in high-fat diet-induced hyperlipidemic mice. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2017, 31, 1085–1096. [Google Scholar] [CrossRef]
- Luo, H.Q.; Shen, J.; Chen, C.P.; Ma, X.; Lin, C.; Ouyang, Q.; Xuan, C.X.; Liu, J.; Sun, H.B.; Liu, J. Lipid-lowering effects of oleanolic acid in hyperlipidemic patients. Chin. J. Nat. Med. 2018, 16, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Li, D.; Hu, Y.; Zhang, L.; Li, Y.; Zhang, Z.; Li, C. Persistence of severe global inequalities in the burden of Hypertension Heart Disease from 1990 to 2019: Findings from the global burden of disease study 2019. BMC Public Health 2024, 24, 110. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Perona, J.S.; Herrera, M.D.; Ruiz-Gutierrez, V. Triterpenic compounds from “orujo” olive oil elicit vasorelaxation in aorta from spontaneously hypertensive rats. J. Agric. Food Chem. 2006, 54, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; Perona, J.S.; Ruiz-Gutierrez, V. Potential vasorelaxant effects of oleanolic acid and erythrodiol, two triterpenoids contained in ‘orujo’ olive oil, on rat aorta. Br. J. Nutr. 2004, 92, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Madlala, H.P.; Metzinger, T.; van Heerden, F.R.; Musabayane, C.T.; Mubagwa, K.; Dessy, C. Vascular Endothelium-Dependent and Independent Actions of Oleanolic Acid and Its Synthetic Oleanane Derivatives as Possible Mechanisms for Hypotensive Effects. PLoS ONE 2016, 11, e0147395. [Google Scholar] [CrossRef] [PubMed]
- Bachhav, S.S.; Patil, S.D.; Bhutada, M.S.; Surana, S.J. Oleanolic acid prevents glucocorticoid-induced hypertension in rats. Phytother. Res. PTR 2011, 25, 1435–1439. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; de Sotomayor, M.A.; Ruiz-Gutierrez, V. Pomace olive oil improves endothelial function in spontaneously hypertensive rats by increasing endothelial nitric oxide synthase expression. Am. J. Hypertens. 2007, 20, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; de Sotomayor, M.A.; Ruiz-Gutierrez, V. Effects of pomace olive oil-enriched diets on endothelial function of small mesenteric arteries from spontaneously hypertensive rats. Br. J. Nutr. 2009, 102, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Acelajado, M.C.; Hughes, Z.H.; Oparil, S.; Calhoun, D.A. Treatment of Resistant and Refractory Hypertension. Circ. Res. 2019, 124, 1061–1070. [Google Scholar] [CrossRef]
- Ahn, Y.M.; Choi, Y.H.; Yoon, J.J.; Lee, Y.J.; Cho, K.W.; Kang, D.G.; Lee, H.S. Oleanolic acid modulates the renin-angiotensin system and cardiac natriuretic hormone concomitantly with volume and pressure balance in rats. Eur. J. Pharmacol. 2017, 809, 231–241. [Google Scholar] [CrossRef]
- Kim, H.Y.; Cho, K.W.; Kang, D.G.; Lee, H.S. Oleanolic acid increases plasma ANP levels via an accentuation of cardiac ANP synthesis and secretion in rats. Eur. J. Pharmacol. 2013, 710, 73–79. [Google Scholar] [CrossRef]
- Bachhav, S.S.; Bhutada, M.S.; Patil, S.P.; Sharma, K.S.; Patil, S.D. Oleanolic Acid Prevents Increase in Blood Pressure and Nephrotoxicity in Nitric Oxide Dependent Type of Hypertension in Rats. Pharmacogn. Res. 2014, 7, 385–392. [Google Scholar] [CrossRef]
- Somova, L.O.; Nadar, A.; Rammanan, P.; Shode, F.O. Cardiovascular, antihyperlipidemic and antioxidant effects of oleanolic and ursolic acids in experimental hypertension. Phytomedicine Int. J. Phytother. Phytopharm. 2003, 10, 115–121. [Google Scholar] [CrossRef]
- Bacon, S.L.; Sherwood, A.; Hinderliter, A.; Blumenthal, J.A. Effects of exercise, diet and weight loss on high blood pressure. Sport. Med. 2004, 34, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Alwardat, N.; Di Renzo, L.; de Miranda, R.C.; Alwardat, S.; Sinibaldi Salimei, P.; De Lorenzo, A. Association between hypertension and metabolic disorders among elderly patients in North Jordan. Diabetes Metab. Syndr. 2018, 12, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, Y.; Wang, X.; Tian, Z.; Qi, D.; Li, Y.; Jiang, H. Antihypertensive activity of oleanolic acid is mediated via downregulation of secretory phospholipase A2 and fatty acid synthase in spontaneously hypertensive rats. Int. J. Mol. Med. 2020, 46, 2019–2034. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar] [PubMed]
- Ratziu, V.; Bellentani, S.; Cortez-Pinto, H.; Day, C.; Marchesini, G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J. Hepatol. 2010, 53, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Nyakudya, T.T.; Mukwevho, E.; Nkomozepi, P.; Erlwanger, K.H. Neonatal intake of oleanolic acid attenuates the subsequent development of high fructose diet-induced non-alcoholic fatty liver disease in rats. J. Dev. Orig. Health Dis. 2018, 9, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.; Rivera, L.; Furness, J.B.; Angus, P.W. The role of the gut microbiota in NAFLD. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Li, Y.; Lv, H.; Zhang, L.; Bi, C.; Dong, N.; Shan, A.; Wang, J. Oleanolic Acid Targets the Gut-Liver Axis to Alleviate Metabolic Disorders and Hepatic Steatosis. J. Agric. Food Chem. 2021, 69, 7884–7897. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Tian, R.; She, Z.; Cai, J.; Li, H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic. Biol. Med. 2020, 152, 116–141. [Google Scholar] [CrossRef]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. Plant-derived oleanolic acid ameliorates markers associated with non-alcoholic fatty liver disease in a diet-induced pre-diabetes rat model. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 1953–1962. [Google Scholar] [CrossRef]
- Repa, J.J.; Liang, G.; Ou, J.; Bashmakov, Y.; Lobaccaro, J.M.; Shimomura, I.; Shan, B.; Brown, M.S.; Goldstein, J.L.; Mangelsdorf, D.J. Regulation of mouse sterol regulatory element-binding protein-1c gene (SREBP-1c) by oxysterol receptors, LXRalpha and LXRbeta. Genes Dev. 2000, 14, 2819–2830. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, Y.; Chen, Y.; Huang, Z.; Li, M.; Jiang, W.; Chen, J.; Rao, W.; Luo, S.; Chen, Y.; et al. Dingxin Recipe IV attenuates atherosclerosis by regulating lipid metabolism through LXR-α/SREBP1 pathway and modulating the gut microbiota in ApoE (−/−) mice fed with HFD. J. Ethnopharmacol. 2021, 266, 113436. [Google Scholar] [CrossRef]
- Lin, Y.N.; Chang, H.Y.; Wang, C.C.N.; Chu, F.Y.; Shen, H.Y.; Chen, C.J.; Lim, Y.P. Oleanolic Acid Inhibits Liver X Receptor Alpha and Pregnane X Receptor to Attenuate Ligand-Induced Lipogenesis. J. Agric. Food Chem. 2018, 66, 10964–10976. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, Y.; Zuo, G.; Xu, W.; Gao, H.; Yang, Y.; Yamahara, J.; Wang, J.; Li, Y. Oleanolic Acid diminishes liquid fructose-induced Fatty liver in rats: Role of modulation of hepatic sterol regulatory element-binding protein-1c-mediated expression of genes responsible for de novo Fatty Acid synthesis. Evid. Based Complement. Altern. Med. Ecam. 2013, 2013, 534084. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.C.; Philipson, L.H. Update on diabetes classification. Med. Clin. North Am. 2015, 99, 1–16. [Google Scholar] [CrossRef]
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes mellitus and inflammation. Curr. Diabetes Rep. 2013, 13, 435–444. [Google Scholar] [CrossRef]
- Glass, C.K.; Olefsky, J.M. Inflammation and lipid signaling in the etiology of insulin resistance. Cell Metab. 2012, 15, 635–645. [Google Scholar] [CrossRef]
- Iskender, H.; Dokumacioglu, E.; Terim Kapakin, K.A.; Yenice, G.; Mohtare, B.; Bolat, I.; Hayirli, A. Effects of oleanolic acid on inflammation and metabolism in diabetic rats. Biotech. Histochem. Off. Publ. Biol. Stain Comm. 2022, 97, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Matumba, M.; Ayeleso, A.; Nyakudya, T.; Erlwanger, K.; Chegou, N.; Mukwevho, E. Long-Term Impact of Neonatal Intake of Oleanolic Acid on the Expression of AMP-Activated Protein Kinase, Adiponectin and Inflammatory Cytokines in Rats Fed with a High Fructose Diet. Nutrients 2019, 11, 226. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, Z.; Xing, H.; Kang, L.; Chen, X.; Liu, B.; Niu, K. Renoprotective effects of oleanolic acid and its possible mechanisms in rats with diabetic kidney disease. Biochem. Biophys. Res. Commun. 2022, 636, 1–9. [Google Scholar] [CrossRef]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. Plant-derived oleanolic acid ameliorates markers of subclinical inflammation and innate immunity activation in diet-induced pre-diabetic rats. Ther. Adv. Endocrinol. Metab. 2020, 11, 2042018820935771. [Google Scholar] [CrossRef]
- Li, M.; Han, Z.; Bei, W.; Rong, X.; Guo, J.; Hu, X. Oleanolic Acid Attenuates Insulin Resistance via NF-kappaB to Regulate the IRS1-GLUT4 Pathway in HepG2 Cells. Evid. Based Complement. Altern. Med. Ecam 2015, 2015, 643102. [Google Scholar] [CrossRef]
- Zhang, P.; Li, T.; Wu, X.; Nice, E.C.; Huang, C.; Zhang, Y. Oxidative stress and diabetes: Antioxidative strategies. Front. Med. 2020, 14, 583–600. [Google Scholar] [CrossRef]
- Wang, S.; Du, L.B.; Jin, L.; Wang, Z.; Peng, J.; Liao, N.; Zhao, Y.Y.; Zhang, J.L.; Pauluhn, J.; Hai, C.X.; et al. Nano-oleanolic acid alleviates metabolic dysfunctions in rats with high fat and fructose diet. Biomed. Pharmacother. 2018, 108, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Wang, S.; Gu, J.; Min, Z.; Wang, R. Effect of Nano-Oleanolic Acid Combined With Lipid-Lowering Ketones on Insulin Resistance in Rats with Gestational Diabetes. J. Biomed. Nanotechnol. 2022, 18, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.L.; Wu, H.; Liu, J.Z.; Hu, J.X.; Liao, N.; Peng, J.; Cao, P.P.; Liang, X.; Hai, C.X. Antidiabetic effect of oleanolic acid: A promising use of a traditional pharmacological agent. Phytother. Res. PTR 2011, 25, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Li, Q.; Li, Y.; Liu, Z.; Fan, Y.; Liu, Z.; Zhao, H.; Li, J.; Han, Z. Antidiabetic and antioxidant effects of oleanolic acid from Ligustrum lucidum Ait in alloxan-induced diabetic rats. Phytother. Res. PTR 2009, 23, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Dubey, V.K.; Patil, C.R.; Kamble, S.M.; Tidke, P.S.; Patil, K.R.; Maniya, P.J.; Jadhav, R.B.; Patil, S.P. Oleanolic acid prevents progression of streptozotocin induced diabetic nephropathy and protects renal microstructures in Sprague Dawley rats. J. Pharmacol. Pharmacother. 2013, 4, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Nosadini, R.; Tonolo, G. Role of oxidized low density lipoproteins and free fatty acids in the pathogenesis of glomerulopathy and tubulointerstitial lesions in type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. NMCD 2011, 21, 79–85. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Song, C.; Yuan, S.; Bian, X.; Lin, Z.; Yang, M.; Dou, K. Triglyceride-glucose index as a suitable non-insulin-based insulin resistance marker to predict cardiovascular events in patients undergoing complex coronary artery intervention: A large-scale cohort study. Cardiovasc. Diabetol. 2024, 23, 15. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Victoria Awolola, G.; Ibrahim, M.A.; Anthony Koorbanally, N.; Islam, M.S. Oleanolic acid as a potential antidiabetic component of Xylopia aethiopica (Dunal) A. Rich. (Annonaceae) fruit: Bioassay guided isolation and molecular docking studies. Nat. Prod. Res. 2021, 35, 788–791. [Google Scholar] [CrossRef]
- Teodoro, T.; Zhang, L.; Alexander, T.; Yue, J.; Vranic, M.; Volchuk, A. Oleanolic acid enhances insulin secretion in pancreatic beta-cells. FEBS Lett. 2008, 582, 1375–1380. [Google Scholar] [CrossRef]
- Gao, D.; Li, Q.; Li, Y.; Liu, Z.; Liu, Z.; Fan, Y.; Han, Z.; Li, J.; Li, K. Antidiabetic potential of oleanolic acid from Ligustrum lucidum Ait. Can. J. Physiol. Pharmacol. 2007, 85, 1076–1083. [Google Scholar] [CrossRef]
- Musabayane, C.T.; Tufts, M.A.; Mapanga, R.F. Synergistic antihyperglycemic effects between plant-derived oleanolic acid and insulin in streptozotocin-induced diabetic rats. Ren. Fail. 2010, 32, 832–839. [Google Scholar] [CrossRef]
- Khathi, A.; Masola, B.; Musabayane, C.T. Effects of Syzygium aromaticum-derived oleanolic acid on glucose transport and glycogen synthesis in the rat small intestine. J. Diabetes 2013, 5, 80–87. [Google Scholar] [CrossRef]
- Tuomilehto, J.; Lindström, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. New Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. The Effects of Plant-Derived Oleanolic Acid on Selected Parameters of Glucose Homeostasis in a Diet-Induced Pre-Diabetic Rat Model. Molecules 2018, 23, 794. [Google Scholar] [CrossRef] [PubMed]
- Molepo, M.; Ayeleso, A.; Nyakudya, T.; Erlwanger, K.; Mukwevho, E. A Study on Neonatal Intake of Oleanolic Acid and Metformin in Rats (Rattus norvegicus) with Metabolic Dysfunction: Implications on Lipid Metabolism and Glucose Transport. Molecules 2018, 23, 2528. [Google Scholar] [CrossRef] [PubMed]
- Mukundwa, A.; Langa, S.O.; Mukaratirwa, S.; Masola, B. In vivo effects of diabetes, insulin and oleanolic acid on enzymes of glycogen metabolism in the skin of streptozotocin-induced diabetic male Sprague-Dawley rats. Biochem. Biophys. Res. Commun. 2016, 471, 315–319. [Google Scholar] [CrossRef]
- Loza-Rodriguez, H.; Estrada-Soto, S.; Alarcon-Aguilar, F.J.; Huang, F.; Aquino-Jarquin, G.; Fortis-Barrera, A.; Giacoman-Martinez, A.; Almanza-Perez, J.C. Oleanolic acid induces a dual agonist action on PPARgamma/alpha and GLUT4 translocation: A pentacyclic triterpene for dyslipidemia and type 2 diabetes. Eur. J. Pharmacol. 2020, 883, 173252. [Google Scholar] [CrossRef]
- Thomas, C.; Gioiello, A.; Noriega, L.; Strehle, A.; Oury, J.; Rizzo, G.; Macchiarulo, A.; Yamamoto, H.; Mataki, C.; Pruzanski, M.; et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab. 2009, 10, 167–177. [Google Scholar] [CrossRef]
- Sato, H.; Genet, C.; Strehle, A.; Thomas, C.; Lobstein, A.; Wagner, A.; Mioskowski, C.; Auwerx, J.; Saladin, R. Anti-hyperglycemic activity of a TGR5 agonist isolated from Olea europaea. Biochem. Biophys. Res. Commun. 2007, 362, 793–798. [Google Scholar] [CrossRef]
- Huang, X.; Liu, G.; Guo, J.; Su, Z. The PI3K/AKT pathway in obesity and type 2 diabetes. Int. J. Biol. Sci. 2018, 14, 1483–1496. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.Y.; Wang, Y.P.; Cantley, J.; Iseli, T.J.; Molero, J.C.; Hegarty, B.D.; Kraegen, E.W.; Ye, Y.; Ye, J.M. Oleanolic acid reduces hyperglycemia beyond treatment period with Akt/FoxO1-induced suppression of hepatic gluconeogenesis in type-2 diabetic mice. PLoS ONE 2012, 7, e42115. [Google Scholar] [CrossRef]
- Mukundwa, A.; Mukaratirwa, S.; Masola, B. Effects of oleanolic acid on the insulin signaling pathway in skeletal muscle of streptozotocin-induced diabetic male Sprague-Dawley rats. J. Diabetes 2016, 8, 98–108. [Google Scholar] [CrossRef]
- Ngubane, P.S.; Masola, B.; Musabayane, C.T. The effects of Syzygium aromaticum-derived oleanolic acid on glycogenic enzymes in streptozotocin-induced diabetic rats. Ren. Fail. 2011, 33, 434–439. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Y.; Abdelkader, D.; Hassan, W.; Sun, H.; Liu, J. Combination therapy with oleanolic acid and metformin as a synergistic treatment for diabetes. J. Diabetes Res. 2015, 2015, 973287. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.; Gu, T.; Yamahara, J.; Li, Y. Oleanolic acid supplement attenuates liquid fructose-induced adipose tissue insulin resistance through the insulin receptor substrate-1/phosphatidylinositol 3-kinase/Akt signaling pathway in rats. Toxicol. Appl. Pharmacol. 2014, 277, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, K.; Park, S.; Mariadoss, A.V.A.; Sathiyaseelan, A.; Veeraraghavan, V.P.; Kim, S.; Wang, M.H. Chemical composition, antioxidant, and anti-diabetic activities of ethyl acetate fraction of Stachys riederi var. japonica (Miq.) in streptozotocin-induced type 2 diabetic mice. Food Chem. Toxicol. 2021, 155, 112374. [Google Scholar] [CrossRef]
- Nyakudya, T.T.; Mukwevho, E.; Erlwanger, K.H. The protective effect of neonatal oral administration of oleanolic acid against the subsequent development of fructose-induced metabolic dysfunction in male and female rats. Nutr. Metab. 2018, 15, 82. [Google Scholar] [CrossRef]
- Mohandes, S.; Doke, T.; Hu, H.; Mukhi, D.; Dhillon, P.; Susztak, K. Molecular pathways that drive diabetic kidney disease. J. Clin. Investig. 2023, 133, jci165654. [Google Scholar] [CrossRef]
- Zhang, Z.; Jiang, M.; Xie, X.; Yang, H.; Wang, X.; Xiao, L.; Wang, N. Oleanolic acid ameliorates high glucose-induced endothelial dysfunction via PPARdelta activation. Sci. Rep. 2017, 7, 40237. [Google Scholar] [CrossRef] [PubMed]
- Vilahur, G. Red Blood Cells Deserve Attention in Patients with Type 2 Diabetes. J. Am. Coll. Cardiol. 2018, 72, 781–783. [Google Scholar] [CrossRef] [PubMed]
- Bashiru Shola, O.; Olatunde Olugbenga, F. Hyperglycaemic Environment: Contribution to the Anaemia Associated with Diabetes Mellitus in Rats Experimentally Induced with Alloxan. Anemia 2015, 2015, 848921. [Google Scholar] [CrossRef] [PubMed]
- Baloyi, C.M.; Khathi, A.; Sibiya, N.H.; Ngubane, P.S. The Haematological Effects of Oleanolic Acid in Streptozotocin-Induced Diabetic Rats: Effects on Selected Markers. J. Diabetes Res. 2019, 2019, 6753541. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.; Mao, X.; Zhang, Z.; Wu, H. Classification and Differential Diagnosis of Diabetic Nephropathy. J. Diabetes Res. 2017, 2017, 8637138. [Google Scholar] [CrossRef] [PubMed]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. Preventing the onset of diabetes-induced chronic kidney disease during prediabetes: The effects of oleanolic acid on selected markers of chronic kidney disease in a diet-induced prediabetic rat model. Biomed. Pharmacother. 2021, 139, 111570. [Google Scholar] [CrossRef]
- Gugliucci, A.; Bendayan, M. Renal fate of circulating advanced glycated end products (AGE): Evidence for reabsorption and catabolism of AGE-peptides by renal proximal tubular cells. Diabetologia 1996, 39, 149–160. [Google Scholar] [CrossRef]
- Wang, Z.H.; Hsu, C.C.; Huang, C.N.; Yin, M.C. Anti-glycative effects of oleanolic acid and ursolic acid in kidney of diabetic mice. Eur. J. Pharmacol. 2010, 628, 255–260. [Google Scholar] [CrossRef]
- Lee, E.S.; Kim, H.M.; Kang, J.S.; Lee, E.Y.; Yadav, D.; Kwon, M.H.; Kim, Y.M.; Kim, H.S.; Chung, C.H. Oleanolic acid and N-acetylcysteine ameliorate diabetic nephropathy through reduction of oxidative stress and endoplasmic reticulum stress in a type 2 diabetic rat model. Nephrol. Dial. Transplant. 2016, 31, 391–400. [Google Scholar] [CrossRef]
- Hankey, G.J. Stroke. Lancet 2017, 389, 641–654. [Google Scholar] [CrossRef]
- Wu, L.; Xiong, X.; Wu, X.; Ye, Y.; Jian, Z.; Zhi, Z.; Gu, L. Targeting Oxidative Stress and Inflammation to Prevent Ischemia-Reperfusion Injury. Front. Mol. Neurosci. 2020, 13, 28. [Google Scholar] [CrossRef]
- Orellana-Urzúa, S.; Rojas, I.; Líbano, L.; Rodrigo, R. Pathophysiology of Ischemic Stroke: Role of Oxidative Stress. Curr. Pharm. Des. 2020, 26, 4246–4260. [Google Scholar] [CrossRef]
- Escobar-Peso, A.; Martínez-Alonso, E.; Masjuan, J.; Alcázar, A. Development of Pharmacological Strategies with Therapeutic Potential in Ischemic Stroke. Antioxidants 2023, 12, 2102. [Google Scholar] [CrossRef]
- Du, Y.; Ko, K.M. Oleanolic acid protects against myocardial ischemia-reperfusion injury by enhancing mitochondrial antioxidant mechanism mediated by glutathione and alpha-tocopherol in rats. Planta Med. 2006, 72, 222–227. [Google Scholar] [CrossRef]
- Rong, Z.T.; Gong, X.J.; Sun, H.B.; Li, Y.M.; Ji, H. Protective effects of oleanolic acid on cerebral ischemic damage in vivo and H2O2-induced injury in vitro. Pharm. Biol. 2011, 49, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.J.; Sun, L.L.; Ji, X.; Shi, R.; Xu, F.; Gu, J.H. Neuroprotective effects of oleanolic acid against cerebral ischemia-reperfusion injury in mice. Exp. Neurol. 2021, 343, 113785. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Kong, B.; Zheng, J.; Wang, X.; Li, L. Alprostadil Injection Attenuates Coronary Microembolization-Induced Myocardial Injury Through GSK-3β/Nrf2/HO-1 Signaling-Mediated Apoptosis Inhibition. Drug Des. Dev. Ther. 2020, 14, 4407–4422. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Zhang, Z.; Zhang, Z.; Zhu, P.; Jiang, X.; Wang, Y.; Deng, Q.; Lam Yung, K.K.; Zhang, S. Oleanolic Acid Alleviates Cerebral Ischemia/Reperfusion Injury via Regulation of the GSK-3beta/HO-1 Signaling Pathway. Pharmaceuticals 2021, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Kwan, T.W.; Wong, S.S.; Hong, Y.; Kanaya, A.M.; Khan, S.S.; Hayman, L.L.; Shah, S.H.; Welty, F.K.; Deedwania, P.C.; Khaliq, A.; et al. Epidemiology of Diabetes and Atherosclerotic Cardiovascular Disease Among Asian American Adults: Implications, Management, and Future Directions: A Scientific Statement From the American Heart Association. Circulation 2023, 148, 74–94. [Google Scholar] [CrossRef] [PubMed]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Senthil, S.; Sridevi, M.; Pugalendi, K.V. Cardioprotective effect of oleanolic acid on isoproterenol-induced myocardial ischemia in rats. Toxicol. Pathol. 2007, 35, 418–423. [Google Scholar] [CrossRef]
- Li, W.F.; Wang, P.; Li, H.; Li, T.Y.; Feng, M.; Chen, S.F. Oleanolic acid protects against diabetic cardiomyopathy via modulation of the nuclear factor erythroid 2 and insulin signaling pathways. Exp. Ther. Med. 2017, 14, 848–854. [Google Scholar] [CrossRef]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. Plant-Derived Oleanolic Acid (OA) Ameliorates Risk Factors of Cardiovascular Diseases in a Diet-Induced Pre-Diabetic Rat Model: Effects on Selected Cardiovascular Risk Factors. Molecules 2019, 24, 340. [Google Scholar] [CrossRef]
- Chan, C.Y.; Mong, M.C.; Liu, W.H.; Huang, C.Y.; Yin, M.C. Three pentacyclic triterpenes protect H9c2 cardiomyoblast cells against high-glucose-induced injury. Free Radic. Res. 2014, 48, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Mapanga, R.F.; Rajamani, U.; Dlamini, N.; Zungu-Edmondson, M.; Kelly-Laubscher, R.; Shafiullah, M.; Wahab, A.; Hasan, M.Y.; Fahim, M.A.; Rondeau, P.; et al. Oleanolic acid: A novel cardioprotective agent that blunts hyperglycemia-induced contractile dysfunction. PLoS ONE 2012, 7, e47322. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.; Yanagisawa, M. Endothelin: 30 Years From Discovery to Therapy. Hypertension 2019, 74, 1232–1265. [Google Scholar] [CrossRef] [PubMed]
- Swynghedauw, B. Molecular mechanisms of myocardial remodeling. Physiol. Rev. 1999, 79, 215–262. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Yang, Q.; Harada, M.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Pomegranate flower extract diminishes cardiac fibrosis in Zucker diabetic fatty rats: Modulation of cardiac endothelin-1 and nuclear factor-kappaB pathways. J. Cardiovasc. Pharmacol. 2005, 46, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Sciarretta, S.; Volpe, M.; Sadoshima, J. Mammalian target of rapamycin signaling in cardiac physiology and disease. Circ. Res. 2014, 114, 549–564. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.H.; Zhang, N.; Feng, H.; Zhang, N.; Ma, Z.G.; Yang, Z.; Yuan, Y.; Bian, Z.Y.; Tang, Q.Z. Oleanolic acid alleviated pressure overload-induced cardiac remodeling. Mol. Cell. Biochem. 2015, 409, 145–154. [Google Scholar] [CrossRef]
- Lee, J.J.; Jin, Y.R.; Lim, Y.; Yu, J.Y.; Kim, T.J.; Yoo, H.S.; Shin, H.S.; Yun, Y.P. Oleanolic acid, a pentacyclic triterpenoid, induces rabbit platelet aggregation through a phospholipase C-calcium dependent signaling pathway. Arch. Pharmacal Res. 2007, 30, 210–214. [Google Scholar] [CrossRef]
- Libby, P. The changing landscape of atherosclerosis. Nature 2021, 592, 524–533. [Google Scholar] [CrossRef]
- Batty, M.; Bennett, M.R.; Yu, E. The Role of Oxidative Stress in Atherosclerosis. Cells 2022, 11, 3843. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Li, G.; Wang, M.; Li, H.; Han, Z. Protective effect of oleanolic acid on oxidized-low density lipoprotein induced endothelial cell apoptosis. Biosci. Trends 2015, 9, 315–324. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiang, Q.; Wang, D.; Han, Y.; Han, Z.; Zhong, W.; Wang, C. Modulation of oxidized-LDL receptor-1 (LOX1) contributes to the antiatherosclerosis effect of oleanolic acid. Int. J. Biochem. Cell Biol. 2015, 69, 142–152. [Google Scholar] [CrossRef]
- Luan, J.; Ji, X.; Liu, L. PPARγ in Atherosclerotic Endothelial Dysfunction: Regulatory Compounds and PTMs. Int. J. Mol. Sci. 2023, 24, 4494. [Google Scholar] [CrossRef]
- Luo, H.; Liu, J.; Ouyang, Q.; Xuan, C.; Wang, L.; Li, T.; Liu, J. The effects of oleanolic acid on atherosclerosis in different animal models. Acta Biochim. Biophys. Sin. 2017, 49, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Sun, L.; Hu, X.; Wang, X.; Xu, F.; Chen, B.; Liang, X.; Xia, J.; Wang, P.; Aibara, D.; et al. Suppressing the intestinal farnesoid X receptor/sphingomyelin phosphodiesterase 3 axis decreases atherosclerosis. J. Clin. Investig. 2021, 131, 2865. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Zhang, Y.H.; Dong, X.F.; Hao, Q.Q.; Zhou, X.M.; Yu, Q.T.; Li, S.Y.; Chen, X.; Tengbeh, A.F.; Dong, B.; et al. ACE2 and Ang-(1-7) protect endothelial cell function and prevent early atherosclerosis by inhibiting inflammatory response. Inflamm. Res. 2015, 64, 253–260. [Google Scholar] [CrossRef]
- Buus, N.H.; Hansson, N.C.; Rodriguez-Rodriguez, R.; Stankevicius, E.; Andersen, M.R.; Simonsen, U. Antiatherogenic effects of oleanolic acid in apolipoprotein E knockout mice. Eur. J. Pharmacol. 2011, 670, 519–526. [Google Scholar] [CrossRef]
- Sun, C.; Mao, S.; Chen, S.; Zhang, W.; Liu, C. PPARs-Orchestrated Metabolic Homeostasis in the Adipose Tissue. Int. J. Mol. Sci. 2021, 22, 8974. [Google Scholar] [CrossRef]
- Gross, B.; Pawlak, M.; Lefebvre, P.; Staels, B. PPARs in obesity-induced T2DM, dyslipidaemia and NAFLD. Nat. Rev. Endocrinol. 2017, 13, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Dentin, R.; Girard, J.; Postic, C. Carbohydrate responsive element binding protein (ChREBP) and sterol regulatory element binding protein-1c (SREBP-1c): Two key regulators of glucose metabolism and lipid synthesis in liver. Biochimie 2005, 87, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef] [PubMed]
- Bjursell, M.; Ahnmark, A.; Bohlooly, Y.M.; William-Olsson, L.; Rhedin, M.; Peng, X.R.; Ploj, K.; Gerdin, A.K.; Arnerup, G.; Elmgren, A.; et al. Opposing effects of adiponectin receptors 1 and 2 on energy metabolism. Diabetes 2007, 56, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Katso, R.; Okkenhaug, K.; Ahmadi, K.; White, S.; Timms, J.; Waterfield, M.D. Cellular function of phosphoinositide 3-kinases: Implications for development, homeostasis, and cancer. Annu. Rev. Cell Dev. Biol. 2001, 17, 615–675. [Google Scholar] [CrossRef] [PubMed]
- Fruman, D.A.; Meyers, R.E.; Cantley, L.C. Phosphoinositide kinases. Annu. Rev. Biochem. 1998, 67, 481–507. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Su, S.; Xin, M.; Zhang, Z.; Nan, X.; Li, Z.; Lu, D. Luteolin ameliorates hypoxia-induced pulmonary hypertension via regulating HIF-2α-Arg-NO axis and PI3K-AKT-eNOS-NO signaling pathway. Phytomedicine Int. J. Phytother. Phytopharm. 2022, 104, 154329. [Google Scholar] [CrossRef] [PubMed]
- Nakae, J.; Kitamura, T.; Silver, D.L.; Accili, D. The forkhead transcription factor Foxo1 (Fkhr) confers insulin sensitivity onto glucose-6-phosphatase expression. J. Clin. Investig. 2001, 108, 1359–1367. [Google Scholar] [CrossRef]
- Honma, M.; Sawada, S.; Ueno, Y.; Murakami, K.; Yamada, T.; Gao, J.; Kodama, S.; Izumi, T.; Takahashi, K.; Tsukita, S.; et al. Selective insulin resistance with differential expressions of IRS-1 and IRS-2 in human NAFLD livers. Int. J. Obes. 2018, 42, 1544–1555. [Google Scholar] [CrossRef]
- Matsui, T.; Tao, J.; del Monte, F.; Lee, K.H.; Li, L.; Picard, M.; Force, T.L.; Franke, T.F.; Hajjar, R.J.; Rosenzweig, A. Akt activation preserves cardiac function and prevents injury after transient cardiac ischemia in vivo. Circulation 2001, 104, 330–335. [Google Scholar] [CrossRef]
- Kumarswamy, R.; Lyon, A.R.; Volkmann, I.; Mills, A.M.; Bretthauer, J.; Pahuja, A.; Geers-Knörr, C.; Kraft, T.; Hajjar, R.J.; Macleod, K.T.; et al. SERCA2a gene therapy restores microRNA-1 expression in heart failure via an Akt/FoxO3A-dependent pathway. Eur. Heart J. 2012, 33, 1067–1075. [Google Scholar] [CrossRef]
- Kerkelä, R.; Woulfe, K.; Force, T. Glycogen synthase kinase-3beta—Actively inhibiting hypertrophy. Trends Cardiovasc. Med. 2007, 17, 91–96. [Google Scholar] [CrossRef]
- Napetschnig, J.; Wu, H. Molecular basis of NF-κB signaling. Annu. Rev. Biophys. 2013, 42, 443–468. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wang, R.; Li, Y.; Cui, Y.; He, Y.; Wang, H.; Liu, Y.; Zhang, M.; Chen, Y.; Jia, M.; et al. Involvement of TLRs/NF-κB/ESE-1 signaling pathway in T-2 toxin-induced cartilage matrix degradation. Environ. Pollut. 2023, 342, 123114. [Google Scholar] [CrossRef]
- Shen, S.; Wu, G.; Luo, W.; Li, W.; Li, X.; Dai, C.; Huang, W.; Liang, G. Leonurine attenuates angiotensin II-induced cardiac injury and dysfunction via inhibiting MAPK and NF-κB pathway. Phytomedicine Int. J. Phytother. Phytopharm. 2023, 108, 154519. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Khan, Z.A.; Cukiernik, M.; Chakrabarti, S. Differential activation of NF-kappa B and AP-1 in increased fibronectin synthesis in target organs of diabetic complications. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E1089–E1097. [Google Scholar] [CrossRef]
- Song, M.; Hang, T.J.; Wang, Y.; Jiang, L.; Wu, X.L.; Zhang, Z.; Shen, J.; Zhang, Y. Determination of oleanolic acid in human plasma and study of its pharmacokinetics in Chinese healthy male volunteers by HPLC tandem mass spectrometry. J. Pharm. Biomed. Anal. 2006, 40, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Rada, M.; Castellano, J.M.; Perona, J.S.; Guinda, Á. GC-FID determination and pharmacokinetic studies of oleanolic acid in human serum. Biomed. Chromatogr. BMC 2015, 29, 1687–1692. [Google Scholar] [CrossRef]
- De la Torre, R.; Carbó, M.; Pujadas, M.; Biel, S.; Mesa, M.D.; Covas, M.I.; Expósito, M.; Espejo, J.A.; Sanchez-Rodriguez, E.; Díaz-Pellicer, P.; et al. Pharmacokinetics of maslinic and oleanolic acids from olive oil-Effects on endothelial function in healthy adults. A randomized, controlled, dose-response study. Food Chem. 2020, 322, 126676. [Google Scholar] [CrossRef]
- García-González, A.; Espinosa-Cabello, J.M.; Cerrillo, I.; Montero-Romero, E.; Rivas-Melo, J.J.; Romero-Báez, A.; Jiménez-Andreu, M.D.; Ruíz-Trillo, C.A.; Rodríguez-Rodríguez, A.; Martínez-Ortega, A.J.; et al. Bioavailability and systemic transport of oleanolic acid in humans, formulated as a functional olive oil. Food Funct. 2023, 14, 9681–9694. [Google Scholar] [CrossRef]
- Yin, M.C.; Lin, M.C.; Mong, M.C.; Lin, C.Y. Bioavailability, distribution, and antioxidative effects of selected triterpenes in mice. J. Agric. Food Chem. 2012, 60, 7697–7701. [Google Scholar] [CrossRef]
- Pozo, O.J.; Pujadas, M.; Gleeson, S.B.; Mesa-García, M.D.; Pastor, A.; Kotronoulas, A.; Fitó, M.; Covas, M.I.; Navarro, J.R.F.; Espejo, J.A.; et al. Liquid chromatography tandem mass spectrometric determination of triterpenes in human fluids: Evaluation of markers of dietary intake of olive oil and metabolic disposition of oleanolic acid and maslinic acid in humans. Anal. Chim. Acta 2017, 990, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Stelling-Férez, J.; López-Miranda, S.; Gabaldón, J.A.; Nicolás, F.J. Oleanolic Acid Complexation with Cyclodextrins Improves Its Cell Bio-Availability and Biological Activities for Cell Migration. Int. J. Mol. Sci. 2023, 24, 4860. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Yang, X.; Du, P.; Zhang, H.; Zhang, T. Dual strategies to improve oral bioavailability of oleanolic acid: Enhancing water-solubility, permeability and inhibiting cytochrome P450 isozymes. Eur. J. Pharm. Biopharm. 2016, 99, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Huang, X.; Dou, J.; Zhai, G.; Su, L. Self-microemulsifying drug delivery system for improved oral bioavailability of oleanolic acid: Design and evaluation. Int. J. Nanomed. 2013, 8, 2917–2926. [Google Scholar] [CrossRef]
- Santos-Lozano, J.M.; Rada, M.; Lapetra, J.; Guinda, Á.; Jiménez-Rodríguez, M.C.; Cayuela, J.A.; Ángel-Lugo, A.; Vilches-Arenas, Á.; Gómez-Martín, A.M.; Ortega-Calvo, M.; et al. Prevention of type 2 diabetes in prediabetic patients by using functional olive oil enriched in oleanolic acid: The PREDIABOLE study, a randomized controlled trial. Diabetes Obes. Metab. 2019, 21, 2526–2534. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Rodriguez, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Espejo-Calvo, J.A.; Gil-Extremera, B.; de la Torre, R.; Fito, M.; Covas, M.I.; Vilchez, P.; et al. Effects of Virgin Olive Oils Differing in Their Bioactive Compound Contents on Biomarkers of Oxidative Stress and Inflammation in Healthy Adults: A Randomized Double-Blind Controlled Trial. Nutrients 2019, 11, 561. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).