Abstract
Transition metal phosphides (TMPs) have been widely studied for water decomposition for their monocatalytic property for anodic or cathodic reactions. However, their bifunctional catalytic activity still remains a major challenge. Herein, hexagonal nickel-cobalt bimetallic phosphide nanoneedles with 1–3 μm length and 15–30 nm diameter supported on NF (NixCo2−xP NDs/NF) with adjusted electron structure have been successfully prepared. The overall alkaline water electrolyzer composed of the optimal anode (Ni0.67Co1.33P NDs/NF) and cathode (Ni1.01Co0.99P NDs/NF) provide 100 mA cm−2 at 1.62 V. Gibbs Free Energy for reaction paths proves that the active site in the hydrogen evolution reaction (HER) is Ni and the oxygen evolution reaction (OER) is Co in NixCo2−xP, respectively. In the HER process, Co-doping can result in an apparent accumulation of charge around Ni active sites in favor of promoting HER activity of Ni sites, and ΔGH* of 0.19 eV is achieved. In the OER process, the abundant electron transfer around Co-active sites results in the excellent ability to adsorb and desorb *O and *OOH intermediates and an effectively reduced ∆GRDS of 0.37 eV. This research explains the regulation of electronic structure change on the active sites of bimetallic materials and provides an effective way to design a stable and effective electrocatalytic decomposition of alkaline water.
1. Introduction
Hydrogen, as a clean and sustainable new energy, has become the most potential substitute for non-renewable resources due to its high energy density [1,2,3,4]. Among various hydrogen production methods, electrochemical decomposition of water is an efficient and high-purity hydrogen production method, which can effectively reduce environmental pollution in the energy production process [5,6,7]. This process is two half-reactions composed of the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER), and both of them require noble metals (Pt-based or Ru-based) to achieve excellent electrochemical performance [8,9,10,11]. However, their large-scale application is hindered by their scarcity and expensive cost. Therefore, it is imperative to develop an efficient and inexpensive electrocatalyst for the widespread commercialization of electrochemical water splitting.
Among the transition metal-based electrocatalysts, transition metal phosphides (TMPs) have been widely studied due to their abundant and inexpensive raw materials and hydrogenase-like structure, which could effectively reduce the cost of precious metal catalysts [12,13,14,15,16]. As a result, the HER and OER catalytic properties of TMPs have been thoroughly investigated, such as NiCoP nanofibers [17], Ni2P-CoP@NC [18], NiPx nanospheres [19], hollow urchin-like FeP [20], and carbon encapsulating MoP [21]. Among the common transition metal phosphides with better activity, cobalt-based phosphides exhibit better OER activity, while in HER, nickel-based phosphides exhibit better performance. However, the dual functionality of these catalysts is still a challenge for practical application. Electronic structure regulation by doping heterogeneous elements is a beneficial method to enhance the electrocatalytic performance of nickel-based or cobalt-based phosphides [22,23,24,25,26]. For instance, W, Fe-doped CoP nanosheets exhibit significant improvement in OER performance (η10 = 290 mV) owing to the adjusted electronic structure by the incorporation of duplex metal, while its HER performance still has a lot to improve (η10 = 130 mV) due to the higher H* desorption energy barrier [27]. Jiang and co-workers reported that O-incorporated NiMoP nanotubes could provide high activity for HER performance (η10 = 54 mV), which benefits from the modulated electronic structure [28]. Nevertheless, its OER performance is still unsatisfactory, requiring 520 mV to reach 10 mA∙cm−2. This may be due to the absence of an active site for OER reaction intermediates [29]. Considering that Co2+ has suitable adsorption and resolution energy for intermediate O* of OER, and NiIII could optimize H* adsorption kinetics to enhance alkaline HER performance [30,31], modulating the electronic structure of Co and Ni simultaneously will be an effective strategy to improve the bifunctional activity.
Herein, we successfully fabricated urchin-like NiCoP nanoneedles grown on nickel foam (denoted as NixCo2−xP NDs/NF). Benefiting from the optimized electronic structure via changing Ni/Co ratios, the Ni0.99Co1.01P NDs/NF electrode exhibits a low overpotential of 63 mV at 10 mA cm−2 for HER, while the overpotential of Ni0.67Co1.33P NDs/NF electrode is as low as 298 mV to attain a current density of 100 mA cm−2 in the OER process. Furthermore, the NixCo2−xP NDs/NF electrode exhibits outstanding overall water splitting performance (100 mA cm−2 at 1.62 V). In addition, DFT calculations were used to investigate electronic structure change on the active sites of bimetallic materials and provided theoretical direction to design a stable and effective electrocatalytic decomposition of alkaline water.
2. Results
2.1. Preparation and Structure Characterization of NixCo2−xP NDs/NF
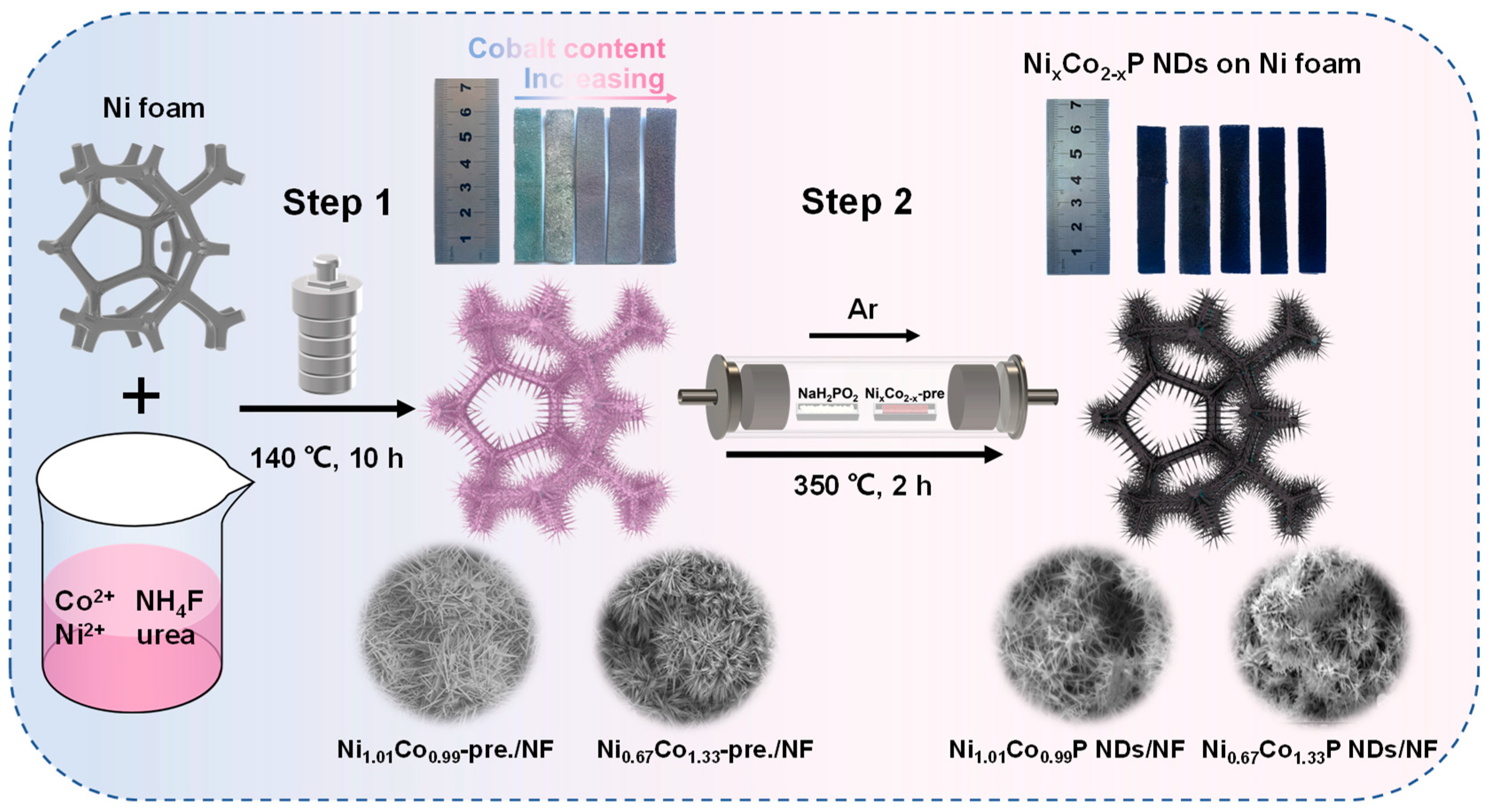
The preparation procedure of NixCo2−xP NDs/NF is shown in Figure 1. In step 1, an appropriate amount of NH4F and urea interact with cobalt/nickel salts to form needle-like precursors by rapid deposition. In order to achieve the optimum conditions, the hydrothermal reaction parameters of NixCo2−x-pre. were gradually probed by adjusting the Ni/Co ratio. As the amount of Co doping increased, the color of NixCo2−x-pre. on NF shifted from blue to pink progressively. In step 2, the subsequent phosphatization process (2) generated a sea urchin-like nanoneedle structure through the reaction of the needle-like precursor with the decomposition product PH3 of NaH2PO2 (1) [32].
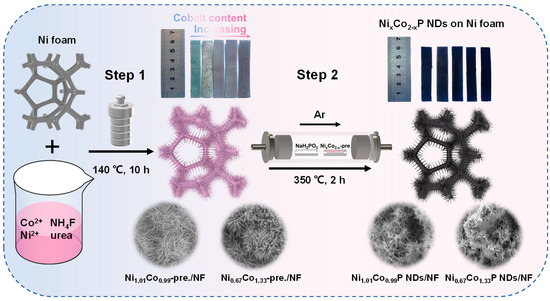
Figure 1.
Schematic illustration of the preparation of NixCo2−xP NDs/NF supported on Ni foam.
NixCo2−x-pre. converted into NixCo2−xP with heat treatment under the protection of Ar atmosphere. It can be seen from the digital picture in Figure 1 that the color of NF turns black after low-temperature phosphorization.
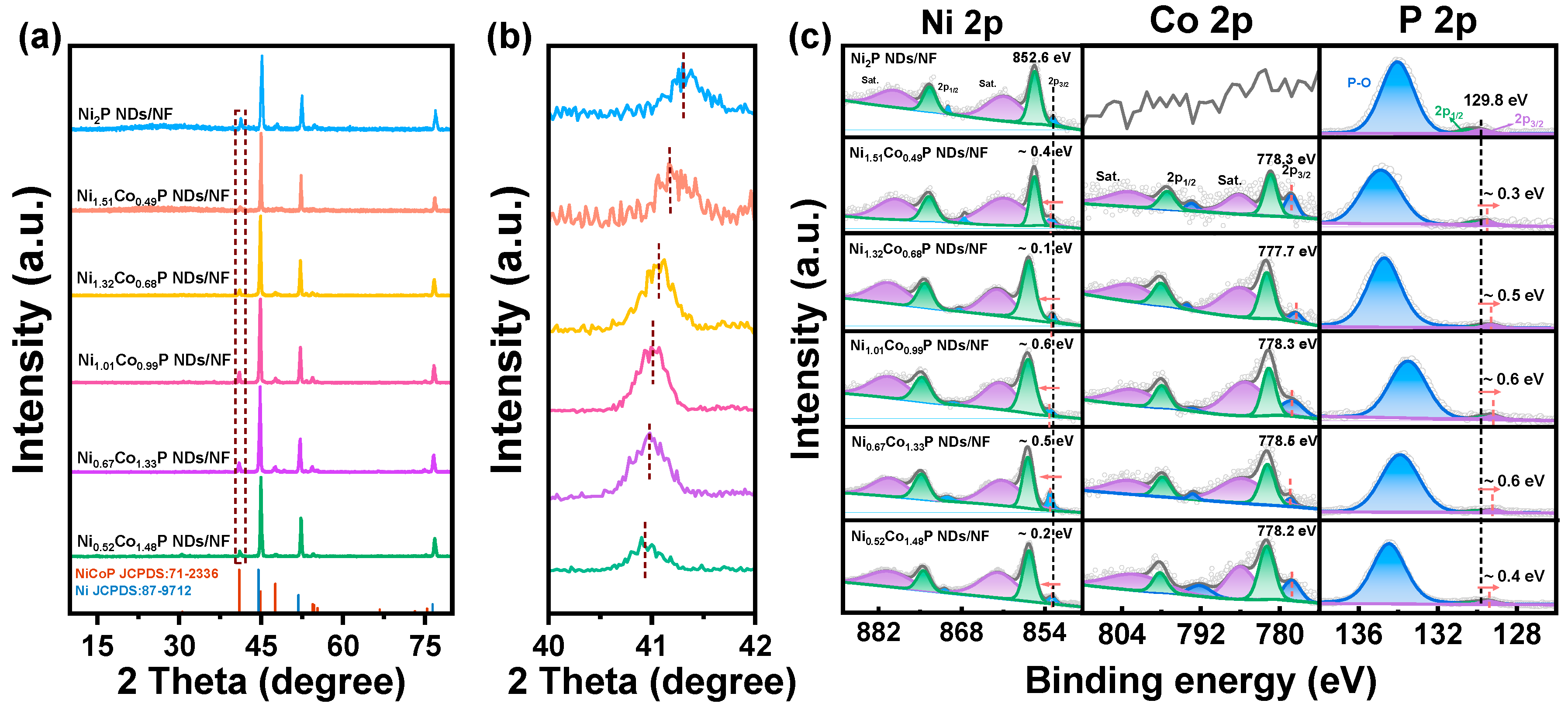
XRD was used to determine the phases of the prepared materials. To eliminate the high peak blockage of NF, the precursor powders stripped from NF via hydrothermal treatment were collected for XRD measurement (Figure S1). It can be discerned that the diffraction peaks of the precursor correspond to Ni(OH)2 (PDF #14-0017) and Co(CO3)0.5OH∙0.11H2O (PDF #48-0083), respectively [33]. As the XRD spectrum of Figure 2a depicts, the catalysts, after phosphating with different Ni/Co ratios, are all the same as the crystal structure of hexagonal Ni2P (PDF #71-2336), indicating that Co has been successfully doped into the Ni2P crystal [34]. The cell parameters are a = 5.834 Å, b = 5.834 Å, c = 3.351 Å; V = 98.8 Å3; space group P-62m (189). To determine the precise amount of Co doping in NixCo2−xP NDs, we conducted ICP–AES analysis. From Table S1, it can be concluded that the five samples present the Ni/Co atomic ratios of 1/0.326, 1/0.515, 1/0.973, 1/1.996, 1/2.845, which correspond to Ni1.51Co0.49P, Ni1.32Co0.68P, Ni1.01Co0.99P, Ni0.67Co1.33P, and Ni0.52Co1.48P, respectively. The peaks at 44.5°, 51.9°, and 76.3° are the fingerprint peaks of Ni (PDF #87-9712) [34]. It is worth noting that there is a red shift in major peaks via increasing fractions of Co (Figure 2b). That may be due to the substitution of Ni (124.6 pm) by Co (125.3 pm) with a higher atomic radius [35].
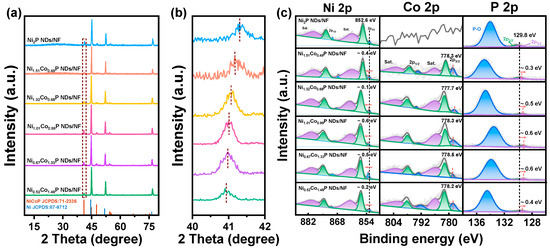
Figure 2.
(a) XRD patterns of the NixCo2−xP NDs/NF with various Ni/Co ratios; (b) XRD results at the (111) peak in the marked region of (a); (c) XPS spectra of Ni2P ND/NF and NixCo2−xP NDs/NF: Ni 2p, Co 2p, and P 2p regions.
XPS was further used to characterize the chemical states of different elements in the NixCo2−xP ND/NF. In Figure S2, the co-existence of Ni, Co, and P elements has been observed in the NixCo2−xP NDs/NF, while Co is absent in the survey of Ni2P NDs/NF. As shown in Figure 2c and Table S2, high-resolution spectra of Ni 2p and Co 2p for NixCo2−xP NDs/NF exhibit two regions and one region for P 2p. For typical samples of Ni1.01Co0.99P NDs/NF and Ni0.67Co1.33P NDs/NF, there are four fitted peaks at ~853.2 eV (2p1/2), ~869.6 eV (2p3/2), ~856.7 eV (2p1/2), and ~875.0 eV (2p3/2), respectively, which are related to Ni–P species and oxidized Ni species. The peaks at ~861.2 and ~879.6 eV can be allocated to the satellite peaks of Ni 2p [36]. In Co 2p spectra of Ni1.01Co0.99P NDs/NF and Ni0.67Co1.33P NDs/NF, the peaks at 778.3 and 778.5 eV can be fitted into Co–P, while the peaks at ~782.0/~797.9 eV and ~786.1/~802.2 eV can be recognized as the oxidized Co species and satellite peaks, respectively [37]. There are characteristic peaks of phosphate and phosphide in P 2p narrow scan spectrum. The peak at the lower binding energy of ~129.5 eV could be assigned to P 2p1/2 and P 2p3/2 of phosphide, respectively, whereas the peaks at ~132 eV could contribute to the P–O originated from the oxidation after exposure to the air [38]. The addition of Co induces a red shift in P 2p3/2 and a blue shift in Ni 2p3/2, indicating an efficient electronic modulation. The change in the Ni/Co ratio is the key reason for the binding energy shift. The electron structure of the Ni position on Ni1.01Co0.99P NDs/NF becomes the most negative, and the Co position on Ni0.67Co1.33P NDs/NF becomes the most positive, which should be highly beneficial to accelerate catalytic reactions [39].
The morphology of NixCo2−x-pre. and NixCo2−xP NDs/NF were characterized by SEM, and TEM was used to analyze the structure of NixCo2−xP NDs. Two samples with excellent electrochemical performance were selected as representative samples. The SEM images of Ni1.01Co0.99-pre. and Ni0.67Co1.33-pre. Figure 3a,d show the morphology of a sea urchin with a uniform needle shape. The Ni1.01Co0.99-pre. and Ni0.67Co1.33-pre. have an average length of approximately 2–4 μm and a diameter of 15–30 nm. In Figure 3b,e, the needle structures of Ni1.01Co0.99P and Ni0.67Co1.33P still exist; however, their surface becomes visibly rough and short in length (1–3 μm). This may be ascribed to the loss of the bonding water during the phosphating process. The one-dimensional needle shape could provide rapid transport of electrons, ions, and products of the HER and OER process and improve the contact area, which is beneficial for improving electrochemical performance [32]. HRTEM images of Ni1.01Co0.99P NDs and Ni0.67Co1.33P NDs are shown in Figure 3c,j, respectively. The same spacing of 0.220 nm was discovered to be consistent with the interplanar spacing of (111) plane for NixCo2−xP [40].

Figure 3.
SEM images of (a) Ni1.01Co0.99-pre.; (b) Ni1.01Co0.99P NDs/NF; TEM images of (c) Ni1.01Co0.99P NDs; EDX element mapping images of (d–g) Ni1.01Co0.99P NDs; SEM images of (h) Ni0.67Co1.33-pre.; (i) Ni0.67Co1.33P NDs/NF; TEM images of (j) Ni0.67Co1.33P NDs; EDX element mapping images of (k–n) Ni0.67Co1.33P NDs.
2.2. Electrocatalytic Activity for HER and OER
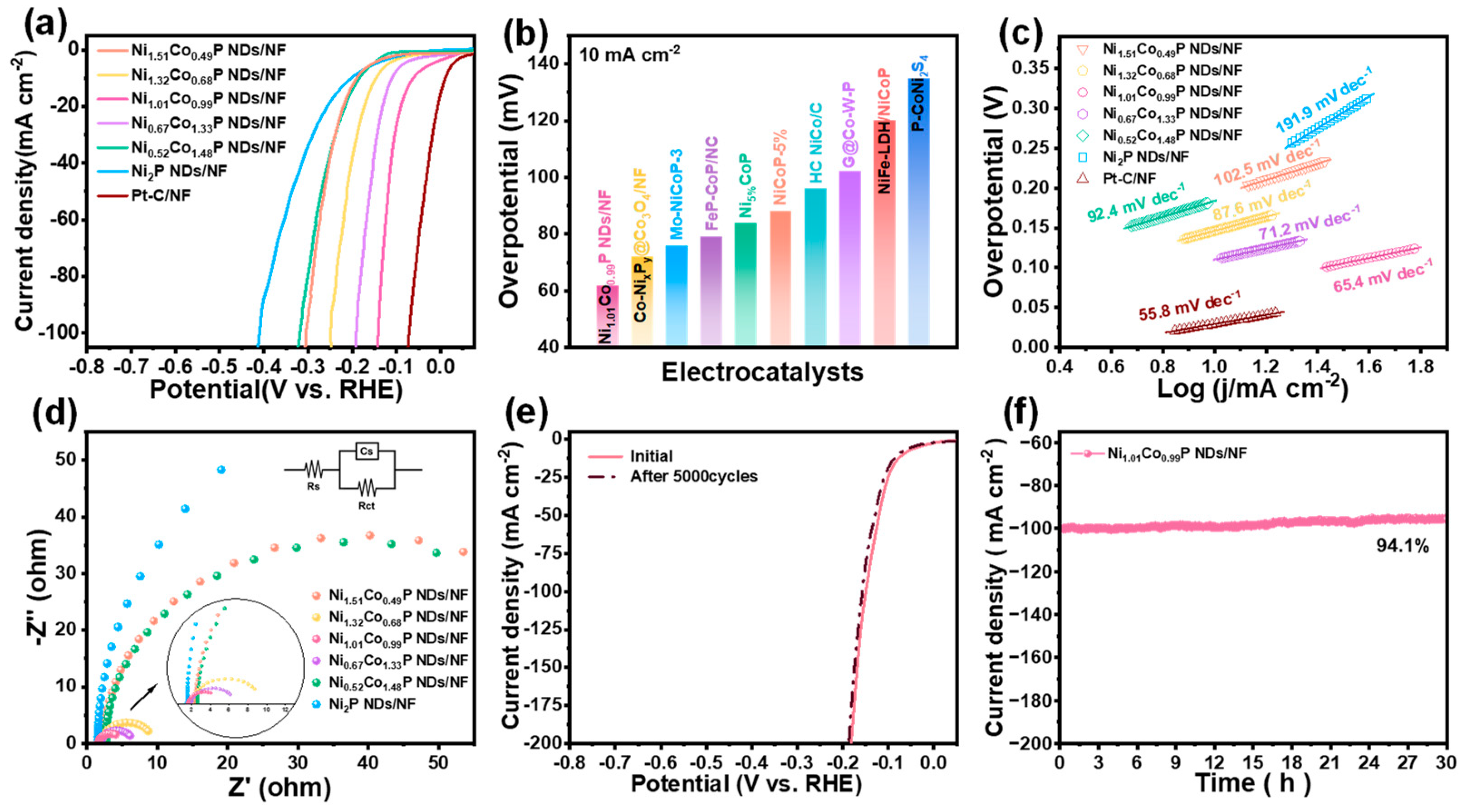
The electrocatalytic performance of all as-prepared catalysts toward HER was evaluated by linear sweep voltammetry in 1.0 M KOH solution at 5 mV∙s−1. As shown in Figure 4a, Ni1.01Co0.99P NDs/NF exhibit preferable HER performance compared with that of Ni1.51Co0.49P NDs/NF, Ni1.32Co0.68P NDs/NF, Ni0.67Co1.33P NDs/NF, Ni0.52Co1.48P NDs/NF, and Ni1.01Co0.99P NDs/NF. In addition, to achieve the current density of 10 mA∙cm−2, the Ni1.01Co0.99P NDs/NF only require an overpotential of 62 mV, which is apparently lower than that of Ni1.51Co0.49P NDs/NF (190 mV), Ni1.32Co0.68P NDs/NF (190 mV), Ni0.67Co1.33P NDs/NF (110 mV), Ni0.52Co1.48P NDs/NF (184 mV), Ni2P NDs/NF (202 mV), and previously reported TMPs catalysts (Figure 4b and Table S3) [41,42,43,44,45,46,47,48,49]. The experiment results show that Co doping could effectively improve the HER activity of Ni2P. The most optimal Ni/Co atom ratio is 1/1. Moreover, the better HER activity of Ni1.01Co0.99P NDs/NF compared with that of Ni1.01Co0.99-pre. indicates the importance of the phosphorization in the hybrid electrode. Furthermore, to probe into the consequence of Co doping on the preferred catalyst activity, we calculated the Tafel slope to analyze the reaction kinetics of the catalyst. In Figure 4c, Ni1.01Co0.99P NDs/NF exhibit the Tafel slope of 65.4 mV dec−1, which is much smaller than that of Ni1.51Co0.49P NDs/NF (102.5 mV dec−1), Ni1.32Co0.68P NDs/NF (87.6 mV dec−1), Ni0.67Co1.33P NDs/NF (71.3 mV dec−1), Ni0.52Co1.48P NDs/NF (92.4 mV dec−1), and Ni2P NDs/NF (89.5 mV dec−1). This result demonstrates Ni1.01Co0.99P NDs/NF’s excellent HER reaction kinetics and also reflects the enhanced HER activity of Ni site via the introduction of Co. Additionally, the electron transfer rate can elucidate the kinetics of electrocatalytic reactions. As a consequence, we conducted electrochemical impedance spectroscopy test for further investigation. As shown in Figure 4d, the Nyquist plot was tested at an overpotential of 0.5 V vs. RHE. The smaller semicircle represents a smaller charge transfer resistance and a faster charge transfer rate. EIS shows that the semicircle diameter of Ni1.01Co0.99P NDs/NF is the smallest, indicating that Ni1.01Co0.99P NDs/NF (Rct = 3.48 Ω) have the lowest charge transfer obstruction in the electrolyte. To further understand HER performance, the CV method was applied to derive the electrochemical active surface area (ECSA) by analyzing the measurement data of double-layer capacitance (Cdl) (Figure S4). Ni1.01Co0.99P NDs/NF also possess the highest Cdl value of 19.56 mF∙cm−2, which is 3.55, 3.05, 2.51, 2.26, and 16.16 times higher than that of Ni1.51Co0.49P NDs/NF, Ni1.32Co0.68P NDs/NF, Ni0.67Co1.33P NDs/NF, Ni0.52Co1.48P NDs/NF and Ni2P NDs/NF, respectively (Figure S5). The calculated ECSA values of all catalysts are listed in Table S4, and the ECSA of Ni1.01Co0.99P NDs/NF was also significantly higher than that of other samples. The LSV curve decays slightly after 5000 CV cycles, indicating the excellent stability of Ni1.01Co0.99P NDs/NF (Figure 4e). In addition, at the overpotential of 136 mV, the initial current density of 100 mA cm−2 decays by only 5.9%, and the current density of 500 mA cm−2 decays by 4.3% after 30 h of timing potential determination, further confirming the good stability of Ni1.01Co0.99P NDs/NF (Figure 4f and Figure S6).
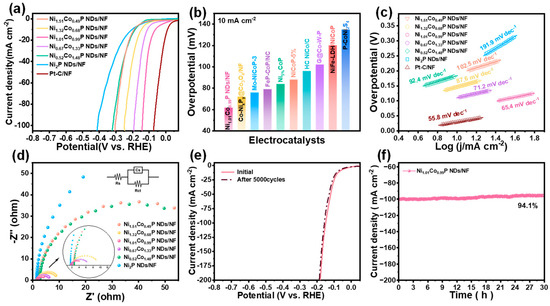
Figure 4.
Electrocatalytic performance of NixCo2−xP NDs/NF for HER with different Ni/Co ratios in 1 M KOH: (a) iR-corrected LSV profiles; (b) overpotential comparison at 10 mA cm−2 with recently reported electrocatalysts; (c) Tafel plots; (d) EIS spectra (inset shows the corresponding equivalent impedance circuit diagram); (e) comparison of LSV profiles of Ni1.01Co0.99P NDs/NF before and after 5000 CV cycles; (f) time-dependent current density curve of Ni1.01Co0.99P NDs/NF under static potential of −1.08 V for 30 h.
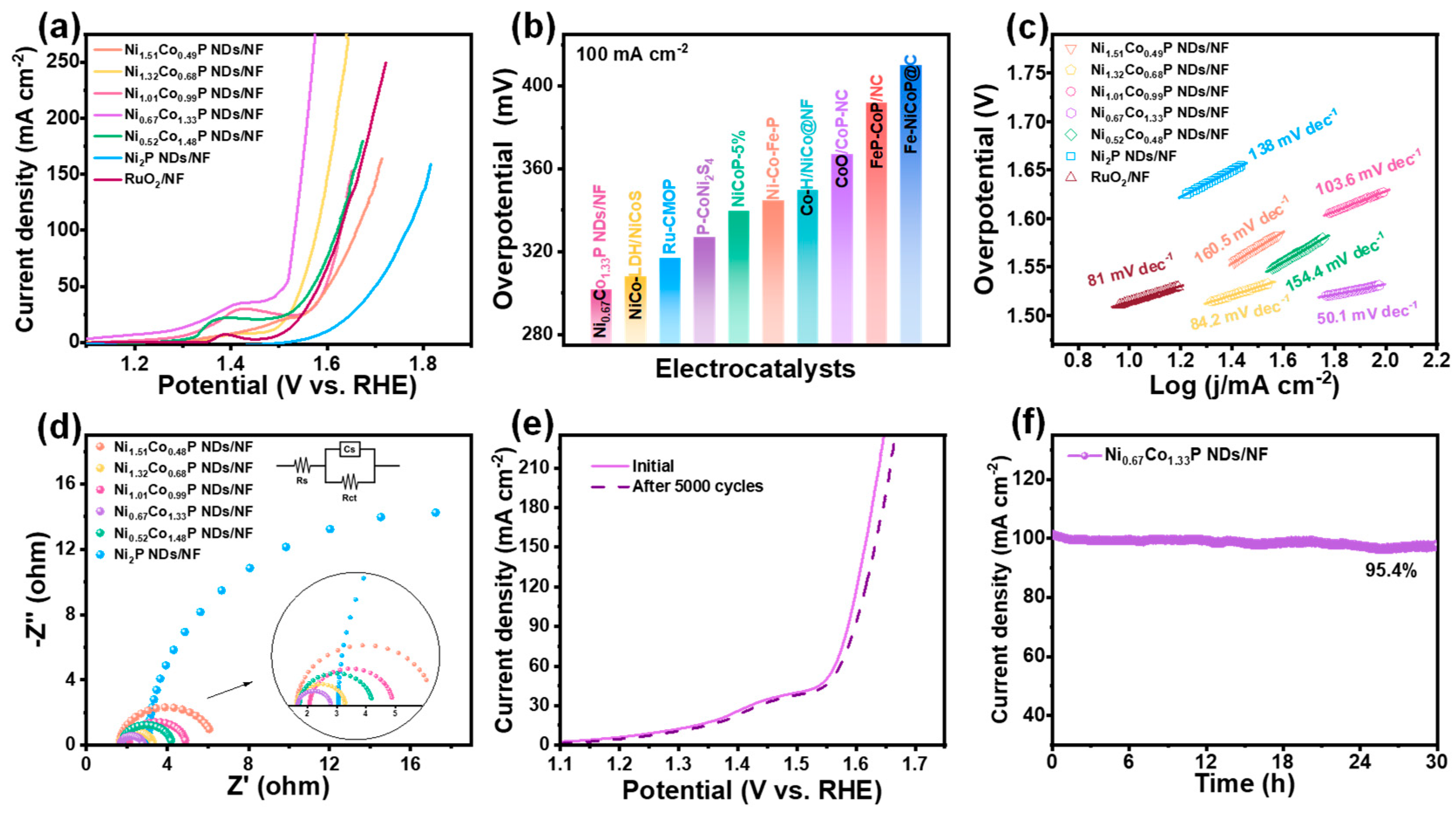
The OER performance of prepared electrocatalysts was also measured with the same electrochemical test system as the HER. It can be revealed from Figure 5a and Figure S7 that Ni0.67Co1.33P NDs/NF demonstrate exceedingly good OER performance. For Ni0.67Co1.33P NDs/NF, achieving the current density of 100 mA∙cm−2 necessitates only a 298 mV overpotential, which is much lower than that of Ni1.51Co0.49P NDs/NF (431 mV), Ni1.32Co0.68P NDs/NF (348 mV), Ni1.01Co0.99P NDs/NF (398 mV), Ni0.52Co1.48P NDs/NF (391 mV), and Ni2P NDs/NF (536 mV). That is even better than that of commercial RuO2 (397 mV). This is mainly because Ni/Co bimetallic adjusting reduces the oxygen affinity of the Co atom. Remarkably, in Figure 5b and Table S5, the overpotential at 100 mA cm−2 of Ni0.67Co1.33P NDs/NF is superior to that of most reported TMPs for OER [43,44,45,50,51,52,53,54,55]. As displayed in Figure 5c, the Ni0.67Co1.33P NDs/NF exhibit the smallest Tafel slope of 50.1 mV dec−1, obviously superior to that of RuO2 (81 mV∙dec−1), Ni1.51Co0.49P NDs/NF (160.5 mV∙dec−1), Ni1.32Co0.68P NDs/NF (84.2 mV∙dec−1), Ni1.01Co0.99P NDs/NF (103.6 mV∙dec−1), Ni0.52Co1.48P NDs/NF (154.4 mV∙dec−1), and Ni2P NDs/NF (138 mV∙dec−1). In the low-frequency range (Figure 5d), the Nyquist plots of Ni0.67Co1.33P NDs/NF indicate that the charge–transfer resistance is the lowest when compared with other semicircles. In stability testing, the performance in LSV curves remains unchanged after 5000 CV cycles (Figure 5e). Meanwhile, chronoamperometry testing at a stationary overpotential of 302 mV demonstrates the excellent stability of Ni0.67Co1.33P NDs/NF with a current density of 100 mA cm−2 maintained for 30 h. When reaching 500 mA cm−2 at a voltage of 1.32 V, the current density decays by 3.6% after 30 h (Figure 5f and Figure S10).
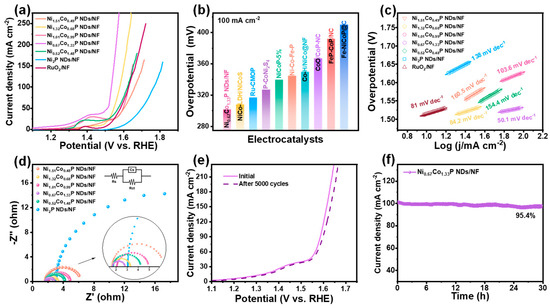
Figure 5.
Electrocatalytic performance of NixCo2−xP NDs/NF for OER with different Ni/Co ratios in 1 M KOH: (a) iR-corrected LSV profiles; (b) overpotential comparison at 100 mA cm−2 with recently reported electrocatalysts; (c)Tafel plots; (d) EIS spectra (inset shows the corresponding equivalent impedance circuit diagram); (e) comparison of LSV profiles of Ni0.67Co1.33P NDs/NF before and after 5000 CV cycle; (f) time-dependent current density curve of Ni0.67Co1.33P NDs/NF under static potential of 0.5 V for 30 h.
2.3. Overall Water Splitting Electrocatalytic Activity
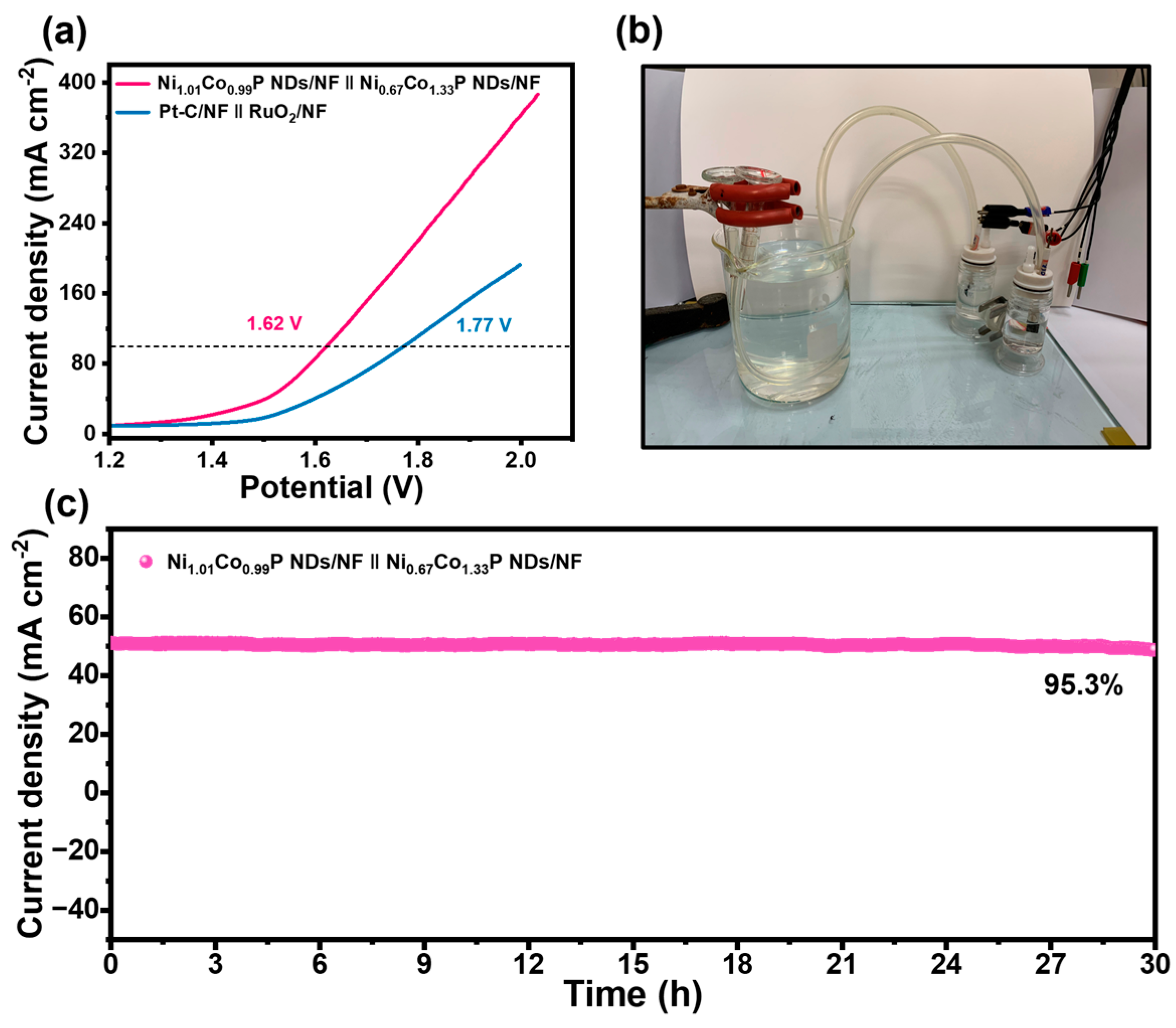
Given the results above, Ni1.01Co0.99P NDs/NF and Ni0.67Co1.33P NDs/NF were applied as competent electrocatalysts for overall water electrolysis. Ni0.67Co1.33P NDs/NF was employed as an anode, and Ni1.01Co0.99P NDs/NF as a cathode to assemble a two-electrode alkaline electrolyzer containing 1 M KOH. At the potential values of 1.62 and 1.90 V, current densities could achieve 100 and 150 mA cm−2, respectively (Figure 6a). The activity is outstanding compared with that of recent reported electrolyzers fabricated with bifunctional electrocatalysts (Table S7). In addition, the outstanding performance could fulfill the demands (1.8–2.4 V up to 200–400 mA∙cm−2) of commercial catalysts for electrolytic water. In Figure 6c and Figure S11, it is evident that there is no noticeable current variation after continuous operation at 1.57 V for 30 h compared with Pt-C/NF‖RuO2/NF electrode, which reveals that Ni1.01Co0.99P NDs/NF‖Ni0.67Co1.33P NDs/NF electrode has excellent durability. It is worthwhile mentioning that plenty of gas bubbles were generated on the surface of Ni1.01Co0.99P NDs/NF‖Ni0.67Co1.33P NDs/NF electrode.
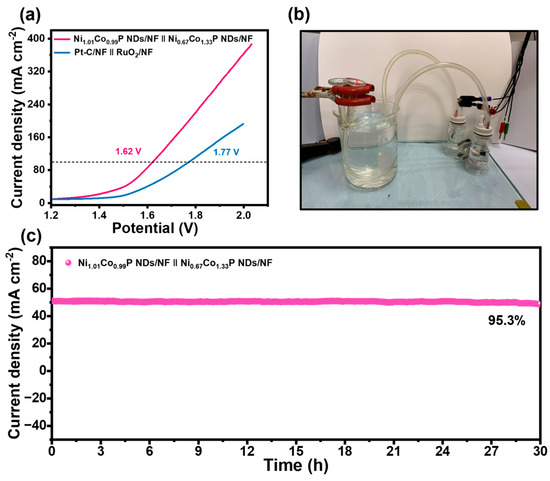
Figure 6.
(a) Overall water splitting performance of Ni1.01Co0.99P NDs/NF‖Ni0.67Co1.33P NDs/NF and Pt-C/NF‖RuO2 in 1.0 M KOH; (b) photograph of H-type alkaline water electrolytic cell using with Ni0.67Co1.33P NDs/NF and Ni1.01Co0.99P NDs/NF as the anode and cathode; (c) time-dependent current density curve for Ni1.01Co0.99P NDs/NF‖Ni0.67Co1.33P NDs/NF at a potential of 1.58 V.
2.4. DFT Calculations
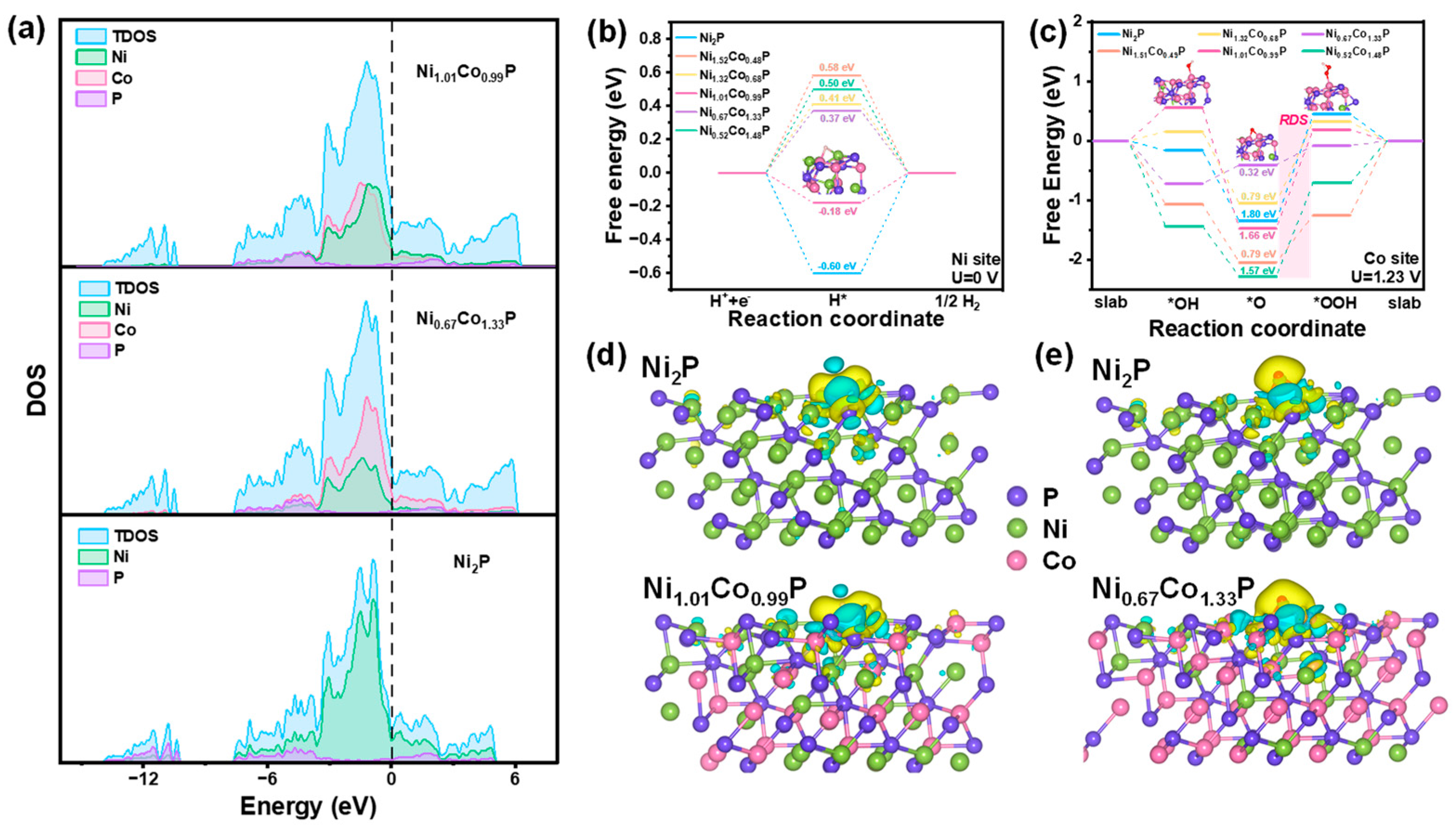
To elucidate the impact of Co-doping on the HER and OER activity, density functional theory (DFT) calculations were performed to elucidate the alterations in reaction energy and electronic structure pre- and post-incorporation of Co. The surfaces are simulated by a slab model with an exposed crystal plane of (111), and the metal atoms of Ni and Co were considered as active sites for HER and OER, respectively. The total densities of states (DOS) of Ni2P, Ni1.01Co0.99P, and Ni0.67Co1.33P are shown in Figure 7a. It is apparent that the DOS of Ni2P, Ni1.01Co0.99P, and Ni0.67Co1.33P are continuously observed near Fermi level, which implies that they possess excellent conductivity. Furthermore, the intensities of Ni1.01Co0.99P and Ni0.67Co1.33P at Fermi level are stronger than that of Ni2P, indicating the bimetal synergistic effect significantly increases the conductivity of Ni1.01Co0.99P and Ni0.67Co1.33P, which are profitable for HER and OER, respectively.
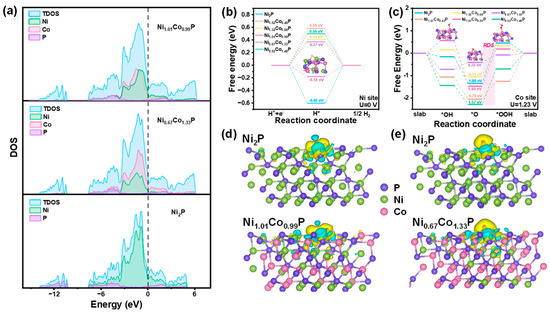
Figure 7.
(a) Total and partial electronic density of states (TDOS and PDOS) calculated for Ni1.01Co0.99P, Ni1.01Co0.99P, and Ni2P. Schematic energy profiles of different models for (b) HER pathway and (c) OER pathway. Calculated charge density differences of (d) Ni2P and Ni1.01Co0.99P for HER and (e) Ni2P and Ni0.67Co1.33P for OER. The Ni, Co, and P atoms are marked in green, rose red, and purple, respectively. The yellow and cyan regions refer to increased and decreased charge distributions, respectively.
For the HER process, the Gibbs free energy of *H is employed as the important indicator in evaluating the HER performance. The overpotential (η) at the Ni site of Ni2P, as depicted in Figure 7b, exhibits a value of 1.43 V, which decreases after the introduction of Co., and the η of Ni1.01Co0.99P (0.19 V) is smaller than that of Ni1.51Co0.49P, Ni1.32Co0.68P, Ni0.67Co1.33P, and Ni0.52Co1.48P, respectively, indicating that *H is most easily absorbed on Ni1.01Co0.99P. On the Co site, the ∆G*H value of Ni1.01Co0.99P remains the smallest. However, in comparison with the Ni site, there is little variation among all samples, suggesting a minor influence of Co on the HER activity (Figure S12). Therefore, Ni is suggested to be the main active site for all the catalysts, and the addition of Co can cooperate with Ni to improve HER performance. For OER processes, a four-step mechanism including all intermediates (*OH, *O, and *OOH) at Ni and Co sites were calculated and recorded as Ni-NixCo2−xP and Co-NixCo2−xP, respectively. As shown in Figure 7c, it is suggested that the rate-determining step (RDS) for the OER is the third step (*O conversion into *OOH), which exhibits the highest ΔG. Compared with the ΔG of RDS in Ni-NiCoP, the ΔG of RDS is only 0.37 eV on Co-Ni0.67Co1.33P. While the ΔG of RDS in Co-Ni1.51Co0.49P, Co-Ni1.01Co0.99P, Co-Ni1.32Co0.68P, and Co-Ni0.52Co1.48P are 0.97 eV, 2.03 eV, 1.37 eV, 1.57 eV, respectively (Figure S13). The calculation results show that the incorporated Co becomes the active site of OER and effectively reduces the reaction energy barrier of the RDS in the reaction, thereby enhancing the OER performance of NixCo2−xP.
Charge density difference was calculated on the same sites to evaluate the alteration of the electronic structure after Co doping in Ni2P. In Figure 7d,e, the green regions represent the charge consumption, while the yellow refers to the charge accumulation. The charge density difference of Ni2P and Ni1.01Co0.99P on the Ni site and Co site was calculated to explore the charge transfer. The charge accumulation region around the H atom on the Ni site of Ni1.01Co0.99P is more intensive than that of Ni2P and Co-Ni1.01Co0.99P, and the corresponding charge depletion region shows the trend of consumption (Figure 7d and Figure S14). This phenomenon reveals that Co doping leads to enhanced electron transfer between H and Ni atoms, thus reducing the ΔG*H of the Ni site. Then charge density difference was calculated for *O in Ni2P and Ni0.67Co1.33P (Figure 7e and Figure S11). Compared with Ni-Ni0.67Co1.33P and Ni-Ni2P, O atoms obtained more electrons in Co-Ni0.67Co1.33P. The charge loss from the Co atom in the Co-Ni0.67Co1.33P system is more significant than Ni in the Ni-Ni2P and Ni-Ni0.67Co1.33P, indicating that the appropriate doping of Co could make it an effective active site in the OER process, thus decreasing the adsorption energy of O atoms and the overpotential.
3. Experimental Section
3.1. Materials
None of the materials were further purified: Ni(NO3)2·6H2O (AR, 98%), CO(NO3)2·6H2O (AR, 99%), urea (AR, 99%), and NaH2PO2·H2O (99%) were purchased from Aladdin Reagent. The source of NH4F (AR, 99.6%), KOH (AR, 85%), HCl (AR), and ethanol (AR, 99.7%) was Sinopharm Chemical Reagent Co., Ltd, Shanghai, China. NF (99.8%, thickness 1 mm, porosity 98%, PPI 110, aperture 0.2–0.6 mm) was purchased from Sinero Energy.
3.2. Materials Preparation
Due to the oxygen in the environment, it was necessary to remove the oxide layer on NF by pretreatment first. A piece of NF () was washed using 3 M HCl, ethanol, and DI water by ultrasonicating. Then, the NF was dried in a vacuum oven at 80 °C for 6 h.
For the synthesis of NixCo2−x-pre., a hydrothermal method was employed. In order to adjust the Ni/Co molar ratio, Ni(NO3)2∙6H2O (2.25 mmol, 2 mmol, 1.5 mmol), Co(NO3)2∙6H2O (1.5 mmol, 2 mmol, 2.25 mmol), urea (15 mmol), and NH4F (7.5 mmol) were evenly dispersed in 60 mL distilled water, respectively, with continuous stirring for 30 min. The formed homogeneous solution and the NF were placed in a 100 mL autoclave Teflon-lined autoclave reactor and remained at 140 °C for 10 h in a drying oven. After washing with distilled water and ethanol, and drying at 60 °C for 10 h, the NixCo2−x precursors/NF (NixCo2−x-pre.) were obtained. Then, the prepared precursors and 2 g NaH2PO2·2H2O were placed in two porcelain boats and then heated to 350 °C in the Ar atmosphere at the rate of 2 °C∙min−1 maintained for 3 h. It is important to note that NaH2PO2·2H2O was positioned upstream of the tubular furnace. Finally, the NixCo2−xP NDs/NF were manufactured after cooling to ambient temperature, cleansing with DI water and ethanol, and drying at 80 °C for 1 h with a vacuum. According to the calculation, the loading mass of the sample on the foam nickel is about 4 mg/cm2.
3.3. Material Characterization
X-ray diffraction (XRD, DX2700, Haoyuan Instrument, Dandong, Liaoning, China) was utilized to reflect the crystalline structure between the 2θ range 10° and 80°. The precursor preparation for XRD measurement is as follows: Hydrothermal treatment was used to strip the precursor from NF. The stripped-down precursor powders were ground for 2–3 min in the agate mortar and then sprinkled into the sample slot of the glass sample holder. A cover glass was used to gently press the surface of the sample and scrape off the excess powder. The bimetallic phosphide preparation for the XRD measurement is as follows: The prepared sample was cut into squares, and a square was fixed onto the sample stage with conductive adhesive for XRD measurement. ICP-5000 was applied for analyzing inductively coupled plasma atomic emission spectroscopy (ICP–AES). Morphology and structure were determined by field emission scanning electron microscopy (FESEM; JSM-7001F, JEOL, Tokyo, Japan; Sigma500, Zeiss, Germany). Transmission electron microscopy (TEM) and high-resolution TEM (HRTEM) were used to observe the microstructure of the catalysts on a JEM-2100 F transmission electron microscope (TEM, JEOL, Tokyo, Japan). X-ray photoelectron spectroscopy (XPS) was used to determine NixCo2−xP NDs/NF surface elemental composition and valence state using a K-Alpha electron spectrometer (Thermo Scientific, Oxford, UK) with monochromatic Al Kα radiation (1486.6 eV 12 kV).
3.4. Electrochemical Measurements
Catalyst electrochemical measurements were conducted at room temperature with the application of the CHI760E electrochemical workstation in a three-electrode system. The NF (1 cm × 1 cm) supported with catalysts was applied as working electrodes, while the Ag/AgCl electrode and graphite rod (for HER) or platinum foil (for OER) were used as the reference electrode and the counter electrode, respectively. For comparison, 8.0 mg of 20 wt% Pt/C or RuO2 was dispersed in a mixture of ethanol (150 μL), deionized water (150 μL), and Nafion (5 wt%, 30 μL). After that, 110 μL of catalyst ink was painted into 1 cm × 1 cm NF (loading 4 mg/cm2). The undermentioned formula resulted in the conversion of the measured potential into the relative value of the reversible hydrogen electrode (RHE):
Before the test, cyclic voltammetry (CV) was used to test 100 cycles until the curves were stable. The measurements of linear scan voltammetry (LSV) were put into effect with the scan rate of 1 mV·s−1 in 1M KOH, and the HER and OER curves performed with 90% iR compensation. When testing the electrochemical impedance spectroscopy (EIS) with an amplitude of 5 mV, the frequency was set at the range from 10 kHz to 0.01 Hz. The double-layer capacitance (Cdl) was estimated by CV through various sweep rates (20, 40, 60, 80, 100, 120 mV∙s−1). The ECSA can be calculated based on the formula [54], where the value of the specific capacitance (CS) is 0.040 mF cm−2; the value of the Cdl is shown in Tables S4 and S6. The efficiency of water splitting was evaluated in an electrolytic cell, which is a two-electrode system with Ni1.01Co0.99P NDs/NF as the anode and Ni0.67Co1.33P NDs/NF as the cathode. The chronoamperometry method (i-t curves) was used to test the durability at a stationary overpotential. In addition, the two-electrode tests were carried out without iR compensation.
4. Conclusions
In this work, a series of overall water electrocatalysts (NixCo2−xP NDs/NF) with high catalytic performance were successfully obtained with varied Ni/Co bimetallic ratios. XPS and DFT calculation results reveal that the synergistic effect of bimetal can change the charge distribution, thereby optimizing the electronic structure of the active sites, and greatly reducing the ΔG of RDS. Ni1.01Co0.99P NDs/NF‖Ni0.67Co1.33P NDs/NF electrolyzer shows eminent catalytic performance for overall water splitting in 1 M KOH. This work will provide an effective way and theoretical direction to design a stable and effective electrocatalytic decomposition of water.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules29030657/s1. Figure S1: XRD patterns of NixCo2−x-pre. powders; Figure S2: XPS full spectra of (a) Ni1.01Co0.99P NDs/NF and (b) Ni0.67Co1.33P NDs/NF; Figure S3: The effects of phosphating on the HER activity of the Ni1.01Co0.99P NDs/NF in 1 M KOH electrolyte; Figure S4: CV curves at different scanning rates 20, 40, 60, 80, 100, 120 mV s−1 for HER in 1.0 M KOH; Figure S5: Cdl values of different catalysts for HER in 1.0 M KOH; Figure S6: Time-dependent current density curve of Ni1.01Co0.99P NDs/NF under static potential of −1.24 V for 30 h; Figure S7: The effects of phosphating on the OER activity of the Ni0.67Co1.33P NDs/NF in 1 M KOH electrolyte; Figure S8: CV curves at different scanning rates of 20, 40, 60, 80, 100, 120 mV s−1 for OER in 1.0 M KOH; Figure S9: Cdl values of different catalysts for OER in 1.0 M KOH; Figure S10: Time-dependent current density curve of Ni0.67Co1.33P NDs/NF under static potential of 1.32 V for 30 h; Figure S11: Time-dependent current density curve for Pt-C/NF‖RuO2/NF at a potential of 1.58 V; Figure S12: Reaction free-energy diagrams for HER of Co site on NixCo2−xP; Figure S13: Reaction free-energy diagrams for OER of Ni site on NixCo2−xP at 0 V; Figure S14: Calculated charge density differences of (a) Co-Ni1.01Co0.99P for HER and (b) Ni-Ni0.67Co1.33P for OER; Table S1: ICP-AES data and atomic ratio of Ni/Co in NixCo2−xP NDs/NF on Ni foam.; Table S2: The binding energy and percent of each species in XPS spectra; Table S3: Comparison of HER performance with other reported electrocatalysts; Table S4: The value of ECSA for each catalyst investigated in 1 M KOH for HER.; Table S5: Comparison of OER performance with other reported electrocatalysts; Table S6. The value of ECSA for each catalyst investigated in 1 M KOH for OER; Table S7: Comparison of overall water splitting performance with other reported electrocatalysts. References [34,37,38,41,42,43,44,45,48,49,52,55,56,57,58,59,60] are cited in the supplementary materials.
Author Contributions
H.X. and X.S. contributed equally to this work. Conceptualization, H.X. and Y.Z.; methodology, Y.Z.; software, C.W.; validation, L.D., P.L. and L.L.; formal analysis, H.X.; investigation, X.S. and H.L.; resources, X.S.; data curation, H.X. and H.L.; writing—original draft preparation, H.X.; writing—review and editing, Y.Z.; visualization, S.C.; supervision, J.S. and X.S.; project administration, L.T.; funding acquisition, Y.Z. and J.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (Nos. 51672143, 22005165, 52102362, 51808303, 22075197, and 22278290), the Natural Science Foundation of Shandong Province (ZR2021ME012 and ZR2021QB022), the Taishan Scholars Program, and the State Key Laboratory of Bio-Fibers and Eco-Textiles (Qingdao University No. ZFZ201809 and ZDKT202105).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article and supplementary materials.
Conflicts of Interest
Author Lipeng Diao was employed by the company Qingdao Hanxing New Materials Co., Ltd.
References
- Liu, D.; Xu, G.; Yang, H.; Wang, H.; Xia, B.Y. Rational Design of Transition Metal Phosphide-Based Electrocatalysts for Hydrogen Evolution. Adv. Funct. Mater. 2022, 33, 2208358. [Google Scholar] [CrossRef]
- Zang, Y.; Lu, D.-Q.; Wang, K.; Li, B.; Peng, P.; Lan, Y.-Q.; Zang, S.-Q. A pyrolysis-free Ni/Fe bimetallic electrocatalyst for overall water splitting. Nat. Commun. 2023, 14, 1792. [Google Scholar] [CrossRef]
- Guan, D.; Wang, B.; Zhang, J.; Shi, R.; Jiao, K.; Li, L.; Wang, Y.; Xie, B.; Zhang, Q.; Yu, J.; et al. Hydrogen society: From present to future. Energy Environ. Sci. 2023, 16, 4926–4943. [Google Scholar] [CrossRef]
- Chen, D.; Bai, H.; Zhu, J.; Wu, C.; Zhao, H.; Wu, D.; Jiao, J.; Ji, P.; Mu, S. Multiscale Hierarchical Structured NiCoP Enabling Ampere-Level Water Splitting for Multi-Scenarios Green Energy-to-Hydrogen Systems. Adv. Energy Mater. 2023, 13, 2200499. [Google Scholar] [CrossRef]
- Gultom, N.S.; Chen, T.-S.; Silitonga, M.Z.; Kuo, D.-H. Overall water splitting realized by overall sputtering thin-film technology for a bifunctional MoNiFe electrode: A green technology for green hydrogen. Appl. Catal. B Environ. 2023, 322, 122103. [Google Scholar] [CrossRef]
- Ma, G.; Ye, J.; Qin, M.; Sun, T.; Tan, W.; Fan, Z.; Huang, L.; Xin, X. Mn-doped NiCoP nanopin arrays as high-performance bifunctional electrocatalysts for sustainable hydrogen production via overall water splitting. Nano Energy 2023, 115, 108679. [Google Scholar] [CrossRef]
- Wu, L.; Yu, L.; Zhang, F.; McElhenny, B.; Luo, D.; Karim, A.; Chen, S.; Ren, Z. Heterogeneous Bimetallic Phosphide Ni2P-Fe2P as an Efficient Bifunctional Catalyst for Water/Seawater Splitting. Adv. Funct. Mater. 2020, 31, 2006484. [Google Scholar] [CrossRef]
- Liu, M.; Yao, Z.; Gu, J.; Li, C.; Huang, X.; Zhang, L.; Huang, Z.; Fan, M. Issues and opportunities facing hydrolytic hydrogen production materials. Chem. Eng. J. 2023, 461, 141918. [Google Scholar] [CrossRef]
- Jiang, J.; Zhou, X.L.; Lv, H.G.; Yu, H.Q.; Yu, Y. Bimetallic-Based Electrocatalysts for Oxygen Evolution Reaction. Adv. Funct. Mater. 2022, 33, 2212160. [Google Scholar] [CrossRef]
- He, X.; Han, X.; Zhou, X.; Chen, J.; Wang, J.; Chen, Y.; Yu, L.; Zhang, N.; Li, J.; Wang, S.; et al. Electronic modulation with Pt-incorporated NiFe layered double hydroxide for ultrastable overall water splitting at 1000 mA cm−2. Appl. Catal. B Environ. 2023, 331, 122683. [Google Scholar] [CrossRef]
- Zheng, X.; Yang, J.; Xu, Z.; Wang, Q.; Wu, J.; Zhang, E.; Dou, S.; Sun, W.; Wang, D.; Li, Y. Ru–Co Pair Sites Catalyst Boosts the Energetics for the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2022, 61, e202205946. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, S.; Ma, J.; Dou, M.; Wang, F. Surface engineering of MOFs as a route to cobalt phosphide electrocatalysts for efficient oxygen evolution reaction. Nano Energy 2022, 98, 107315. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, P.; Zhang, G.; Wu, S.; Chen, Z.; Ranganathan, H.; Sun, S.; Shi, Z. Mn-doped nickel-iron phosphide heterointerface nanoflowers for efficient alkaline freshwater/seawater splitting at high current densities. Chem. Eng. J. 2023, 454, 140061. [Google Scholar] [CrossRef]
- Huang, C.-J.; Xu, H.-M.; Shuai, T.-Y.; Zhan, Q.-N.; Zhang, Z.-J.; Li, G.-R. A review of modulation strategies for improving catalytic performance of transition metal phosphides for oxygen evolution reaction. Appl. Catal. B Environ. 2023, 325, 122313. [Google Scholar] [CrossRef]
- Ray, A.; Sultana, S.; Paramanik, L.; Parida, K.M. Recent advances in phase, size, and morphology-oriented nanostructured nickel phosphide for overall water splitting. J. Mater. Chem. A 2020, 8, 19196–19245. [Google Scholar] [CrossRef]
- Yin, H.; Rong, F.; Xie, Y. A review of typical transition metal phosphides electrocatalysts for hydrogen evolution reaction. Int. J. Hydrogen Energy 2024, 52, 350–375. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, X.; Huang, Z.; Fan, J.; Li, L.; Zhang, M. One-dimensional nickel-cobalt bimetallic phosphide nanostructures for the oxygen evolution reaction. Sustain. Energy Fuels. 2024, 8, 159–165. [Google Scholar] [CrossRef]
- Wen, F.; Li, C.; Zhang, T.; Pang, L.; Liu, H.; Huang, X. Nickel-cobalt bimetal phosphide integrated in nitrogen-doped carbon being a novel chainmail electrocatalyst for oxygen evolution. Int. J. Hydrogen Energy 2024, 54, 1487–1494. [Google Scholar] [CrossRef]
- Fu, Q.; Wang, X.; Han, J.; Zhong, J.; Zhang, T.; Yao, T.; Xu, C.; Cao, T.; Xi, S.; Liang, C. Phase-Junction Electrocatalysts towards Enhanced Hydrogen Evolution Reaction in Alkaline Media. Angew. Chem. Int. Ed. 2021, 133, 263–271. [Google Scholar] [CrossRef]
- Gao, P.; Gao, M.; Lei, T.; Ren, Z.; Luo, J.; Huang, Z.; Wu, A. Hollow urchin-like FeP as highly efficient hydrogen evolution catalyst. Inorg. Chem. Commun. 2023, 156, 111143. [Google Scholar] [CrossRef]
- Xiao, W.; Zhang, L.; Bukhvalow, D.; Chen, Z.; Zou, Z.; Shang, L.; Yang, X.; Yan, D.; Han, F.; Zhang, T. Hierarchical ultrathin carbon encapsulating transition metal doped MoP electrocatalysts for efficient and pH-universal hydrogen evolution reaction. Nano Energy 2020, 70, 104445. [Google Scholar] [CrossRef]
- Shuai, C.; Mo, Z.; Niu, X.; Zhao, P.; Dong, Q.; Chen, Y.; Liu, N.; Guo, R. Nickel/cobalt bimetallic phosphides derived metal-organic frameworks as bifunctional electrocatalyst for oxygen and hydrogen evolution reaction. J. Alloys Compd. 2020, 847, 156514. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Z.; Zhang, L.; Xia, Y.; Wang, H.; Liu, H.; Ge, S.; Yu, J. Manipulating the d-band centers of transition metal phosphides through dual metal doping towards robust overall water splitting. J. Mater. Chem. A 2022, 10, 22125–22134. [Google Scholar] [CrossRef]
- Wen, S.; Huang, J.; Li, T.; Chen, W.; Chen, G.; Zhang, Q.; Zhang, X.; Qian, Q.; Ostrikov, K. Multiphase nanosheet-nanowire cerium oxide and nickel-cobalt phosphide for highly-efficient electrocatalytic overall water splitting. Appl. Catal. B Environ. 2022, 316, 121678. [Google Scholar] [CrossRef]
- Yang, N.; Tian, S.; Feng, Y.; Hu, Z.; Liu, H.; Tian, X.; Xu, L.; Hu, C.; Yang, J. Introducing High-Valence Iridium Single Atoms into Bimetal Phosphides toward High-Efficiency Oxygen Evolution and Overall Water Splitting. Small 2023, 19, 2207253. [Google Scholar] [CrossRef]
- Jo, C.; Surendran, S.; Kim, M.-C.; An, T.-Y.; Lim, Y.; Choi, H.; Janani, G.; Cyril Jesudass, S.; Jun Moon, D.; Kim, J.; et al. Meticulous integration of N and C active sites in Ni2P electrocatalyst for sustainable ammonia oxidation and efficient hydrogen production. Chem. Eng. J. 2023, 463, 142314. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Y.; Zhao, Z.; Zhang, Q.-H.; Huang, L.-B.; Gu, L.; Lu, G.; Hu, J.-S.; Wan, L.-J. Steering elementary steps towards efficient alkaline hydrogen evolution via size-dependent Ni/NiO nanoscale heterosurfaces. Natl. Sci. Rev. 2020, 7, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Sun, M.; Wu, S.; Huang, B.; Lee, C.S.; Zhang, W. Oxygen-Incorporated NiMoP Nanotube Arrays as Efficient Bifunctional Electrocatalysts For Urea-Assisted Energy-Saving Hydrogen Production in Alkaline Electrolyte. Adv. Funct. Mater. 2021, 31, 2104951. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, F.; Wang, D.; Li, Z.; Wang, X.; Wang, C.; Zhang, K.; Du, Y. Amorphous/Crystalline Heterostructure Transition-Metal-based Catalysts for High-Performance Water Splitting. Coord. Chem. Rev. 2023, 475, 214916. [Google Scholar] [CrossRef]
- Zhang, W.; Han, N.; Luo, J.; Han, X.; Feng, S.; Guo, W.; Xie, S.; Zhou, Z.; Subramanian, P.; Wan, K.; et al. Critical Role of Phosphorus in Hollow Structures Cobalt-Based Phosphides as Bifunctional Catalysts for Water Splitting. Small 2021, 18, 2103561. [Google Scholar] [CrossRef]
- Wang, Z.; Guo, P.; Cao, S.; Chen, H.; Zhou, S.; Liu, H.; Wang, H.; Zhang, J.; Liu, S.; Wei, S.; et al. Contemporaneous inverse manipulation of the valence configuration to preferred Co2+ and Ni3+ for enhanced overall water electrocatalysis. Appl. Catal. B Environ. 2021, 284, 119725. [Google Scholar] [CrossRef]
- Zeng, Z.; Xu, R.; Zhao, H.; Zhang, H.; Liu, L.; Lei, Y. Exploration of nanowire- and nanotube-based electrocatalysts for oxygen reduction and oxygen evolution reaction. Mater. Today Nano 2018, 3, 54–68. [Google Scholar] [CrossRef]
- Liu, H.; Li, J.; Zhang, Y.; Ge, R.; Yang, J.; Li, Y.; Zhang, J.; Zhu, M.; Li, S.; Liu, B.; et al. Boosted water electrolysis capability of NixCoyP via charge redistribution and surface activation. Chem. Eng. J. 2023, 473, 145397. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Ge, R.; Cairney, J.M.; Zheng, R.; Khan, A.; Li, S.; Liu, B.; Dai, L.; Li, W. Tailoring the electronic structure of Ni5P4/Ni2P catalyst by Co2P for efficient overall water electrolysis. Appl. Energy 2023, 349, 121582. [Google Scholar] [CrossRef]
- Oyama, S.T.; Gott, T.; Zhao, H.; Lee, Y.-K. Transition metal phosphide hydroprocessing catalysts: A review. Catal. Today 2009, 143, 94–107. [Google Scholar] [CrossRef]
- Lan, X.; Li, G.; Jin, R.; Li, X.; Zheng, J. Carbon coated nickel cobalt phosphide with sea urchin-like structure by low temperature plasma processing for hydrogen evolution and urea oxidation. Chem. Eng. J. 2022, 450, 138225. [Google Scholar] [CrossRef]
- Du, C.; Yang, L.; Yang, F.; Cheng, G.; Luo, W. Nest-like NiCoP for Highly Efficient Overall Water Splitting. ACS Catal. 2017, 7, 4131–4137. [Google Scholar] [CrossRef]
- Hu, R.; Jiao, L.; Liang, H.; Feng, Z.; Gao, B.; Wang, X.F.; Song, X.Z.; Liu, L.Z.; Tan, Z. Engineering Interfacial Built-in Electric Field in Polymetallic Phosphide Heterostructures for Superior Supercapacitors and Electrocatalytic Hydrogen Evolution. Small 2023, 19, 2304132. [Google Scholar] [CrossRef]
- Tan, Y.; Wang, H.; Liu, P.; Shen, Y.; Cheng, C.; Hirata, A.; Fujita, T.; Tang, Z.; Chen, M. Versatile nanoporous bimetallic phosphides towards electrochemical water splitting. Energy Environ. Sci. 2016, 9, 2257–2261. [Google Scholar] [CrossRef]
- Li, J.; Wei, G.; Zhu, Y.; Xi, Y.; Pan, X.; Ji, Y.; Zatovsky, I.V.; Han, W. Hierarchical NiCoP nanocone arrays supported on Ni foam as an efficient and stable bifunctional electrocatalyst for overall water splitting. J. Mater. Chem. A 2017, 5, 14828–14837. [Google Scholar] [CrossRef]
- Lu, B.; Zang, J.; Li, W.; Li, J.; Zou, Q.; Zhou, Y.; Wang, Y. Co-doped NixPy loading on Co3O4 embedded in Ni foam as a hierarchically porous self-supported electrode for overall water splitting. Chem. Eng. J. 2021, 422, 130062. [Google Scholar] [CrossRef]
- Lin, J.; Yan, Y.; Li, C.; Si, X.; Wang, H.; Qi, J.; Cao, J.; Zhong, Z.; Fei, W.; Feng, J. Bifunctional Electrocatalysts Based on Mo-Doped NiCoP Nanosheet Arrays for Overall Water Splitting. Nano-Micro Lett. 2019, 11, 55. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Biemolt, J.; Zhao, K.; Zhao, Y.; Cao, X.; Yang, Y.; Wu, X.; Rothenberg, G.; Yan, N. A membrane-free flow electrolyzer operating at high current density using earth-abundant catalysts for water splitting. Nat. Commun. 2021, 12, 1–9. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, J.; Xie, Y.; Sun, B.; Jiang, J.; Jiang, W.-J.; Xi, S.; Yang, H.Y.; Yan, K.; Wang, S.; et al. Constructing Atomic Heterometallic Sites in Ultrathin Nickel-Incorporated Cobalt Phosphide Nanosheets via a Boron-Assisted Strategy for Highly Efficient Water Splitting. Nano Lett. 2021, 21, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.F.; Zhang, S.L.; Sim, W.L.; Gao, S.; Lou, X.W. Phosphorized CoNi2S4 Yolk-Shell Spheres for Highly Efficient Hydrogen Production via Water and Urea Electrolysis. Angew. Chem. Int. Ed. 2021, 60, 22885–22891. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wen, M.; Tian, Y.; Wu, Q.; Fu, Y. A novel structure of quasi-monolayered NiCo-bimetal-phosphide for superior electrochemical performance. J. Energy Chem. 2022, 74, 203–211. [Google Scholar] [CrossRef]
- Qi, B.; Chang, W.; Xu, Q.; Jiang, L.; An, S.; Chu, J.-F.; Song, Y.-F. Regulating Hollow Carbon Cage Supported NiCo Alloy Nanoparticles for Efficient Electrocatalytic Hydrogen Evolution Reaction. ACS Appl. Mater. Inter. 2023, 15, 12078–12087. [Google Scholar] [CrossRef]
- Wang, X.; Le, J.B.; Fei, Y.; Gao, R.; Jing, M.; Yuan, W.; Li, C.M. Self-assembled ultrasmall mixed Co–W phosphide nanoparticles on pristine graphene with remarkable synergistic effects as highly efficient electrocatalysts for hydrogen evolution. J. Mater. Chem. A 2022, 10, 7694–7704. [Google Scholar] [CrossRef]
- Zhang, H.; Li, X.; Hähnel, A.; Naumann, V.; Lin, C.; Azimi, S.; Schweizer, S.L.; Maijenburg, A.W.; Wehrspohn, R.B. Bifunctional Heterostructure Assembly of NiFe LDH Nanosheets on NiCoP Nanowires for Highly Efficient and Stable Overall Water Splitting. Adv. Funct. Mater. 2018, 28, 1706847. [Google Scholar] [CrossRef]
- Li, J.; Wang, L.; He, H.; Chen, Y.; Gao, Z.; Ma, N.; Wang, B.; Zheng, L.; Li, R.; Wei, Y.; et al. Interface construction of NiCo LDH/NiCoS based on the 2D ultrathin nanosheet towards oxygen evolution reaction. Nano Res. 2022, 15, 4986–4995. [Google Scholar] [CrossRef]
- Quan, Q.; Zhang, Y.; Wang, F.; Bu, X.; Wang, W.; Meng, Y.; Xie, P.; Chen, D.; Wang, W.; Li, D.; et al. Topochemical domain engineering to construct 2D mosaic heterostructure with internal electric field for high-performance overall water splitting. Nano Energy 2022, 101, 107566. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Gao, H.; Huang, Z.; Hao, Q.; Liu, B. Interface-induced contraction of core–shell Prussian blue analogues toward hollow Ni-Co-Fe phosphide nanoboxes for efficient oxygen evolution electrocatalysis. Chem. Eng. J 2023, 451, 138515. [Google Scholar] [CrossRef]
- Huang, Y.; Song, X.; Chen, S.; Zhang, J.; Gao, H.; Liao, J.; Ge, C.; Sun, W. Multi layer Architecture of Novel Sea Urchin like Co Hopeite to Boosting Overall Alkaline Water Splitting. Adv. Mater. Interfaces 2023, 10, 2202349. [Google Scholar] [CrossRef]
- Yu, J.; Cao, Q.; Li, Y.; Long, X.; Yang, S.; Clark, J.; Nakabayashi, M.; Shibata, N.; Delaunay, J. Defect-rich NiCeOx electrocatalyst with ultrahigh stability and low overpotential for water oxidation. ACS Catal. 2019, 9, 1605–1611. [Google Scholar] [CrossRef]
- Chen, K.; Cao, Y.; Wang, W.; Diao, J.; Park, J.; Dao, V.; Kim, G.-C.; Qu, Y.; Lee, I.-H. Effectively enhanced activity for overall water splitting through interfacially strong P-Co–O tetrahedral coupling interaction on CoO/CoP heterostructure hollow-nanoneedles. J. Mater. Chem. A 2023, 11, 3136–3147. [Google Scholar] [CrossRef]
- Kang, Y.; Wang, S.; Zhu, S.; Gao, H.; Hui, K.S.; Yuan, C.-Z.; Yin, H.; Bin, F.; Wu, X.-L.; Mai, W.; et al. Iron-modulated nickel cobalt phosphide embedded in carbon to boost power density of hybrid sodium–air battery. Appl. Catal. B Environ. 2021, 285, 119786. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, J.; Zhao, S.; Zhou, W.; Dai, R.; Zhao, X.; Chen, Z.; Sun, T.; Zhang, H.; Chen, A. Mo-doped NiCoP nanowire array grown in situ on Ni foam as a high-performance bifunctional electrocatalyst for overall water splitting. J. Alloys Compd. 2022, 918, 165802. [Google Scholar] [CrossRef]
- Liu, H.; Jin, M.; Zhan, D.; Wang, J.; Cai, X.; Qiu, Y.; Lai, L. Stacking faults triggered strain engineering of ZIF-67 derived Ni-Co bimetal phosphide for enhanced overall water splitting. Appl. Catal. B Environ. 2020, 272, 118951. [Google Scholar] [CrossRef]
- Hu, H.-S.; Li, Y.; Shao, Y.-R.; Li, K.-X.; Deng, G.; Wang, C.-B.; Feng, Y.-Y. NiCoP nanorod arrays as high-performance bifunctional electrocatalyst for overall water splitting at high current densities. J. Power Source 2021, 484, 229269. [Google Scholar] [CrossRef]
- Narasimman, R.; Jnanapriya, G.; Sujatha, S.; Ilangovan, S.A. Vertically aligned nickel-iron-phosphide nanosheets on three-dimensional porous nickel foam for overall water splitting. J. Alloys Compd. 2023, 947, 169474. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).