Applying Subcritical Water Extraction to Obtain Bioactive Compounds and Cellulose Fibers from Brewer Spent Grains
Abstract
1. Introduction
2. Results and Discussion
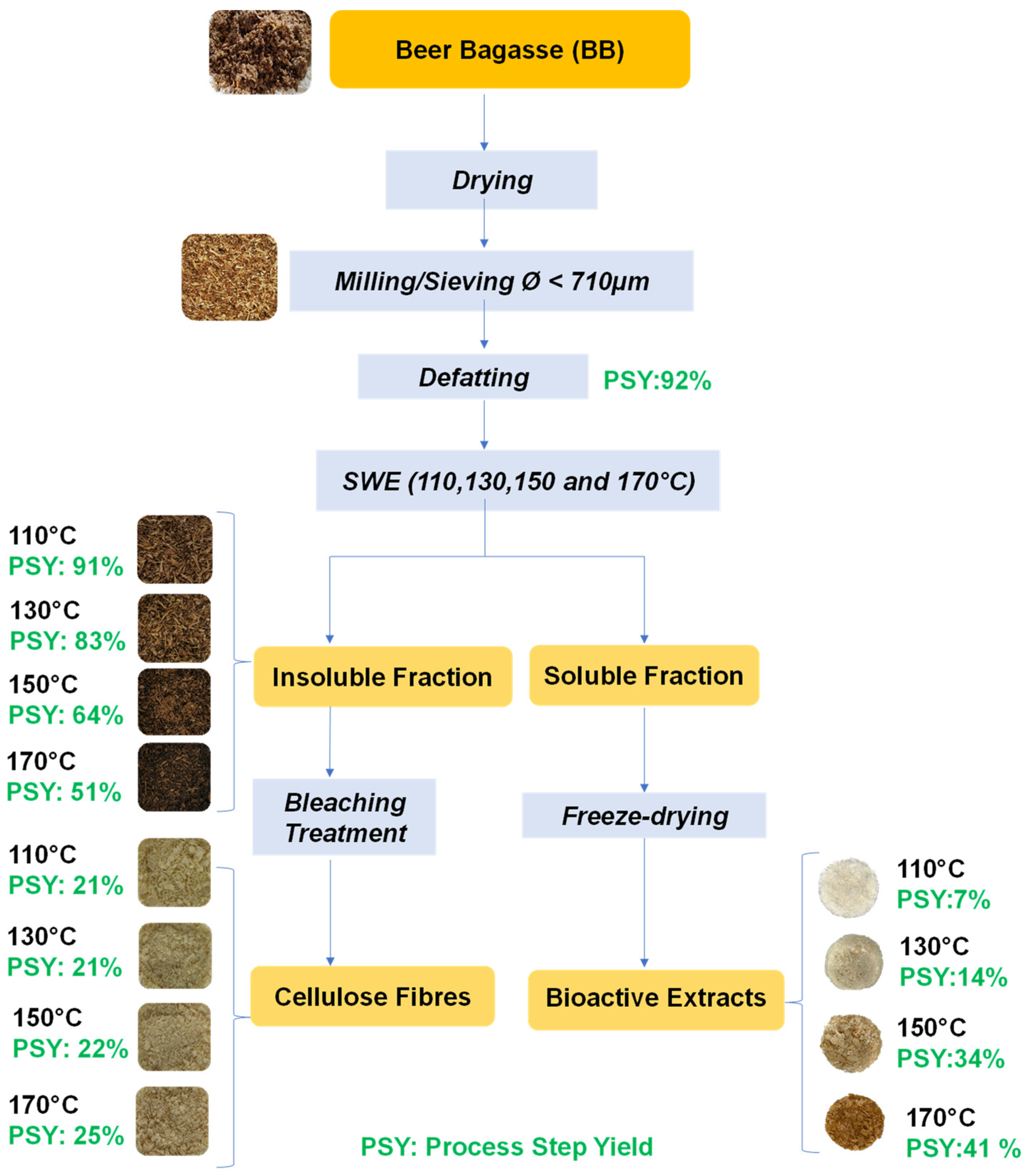
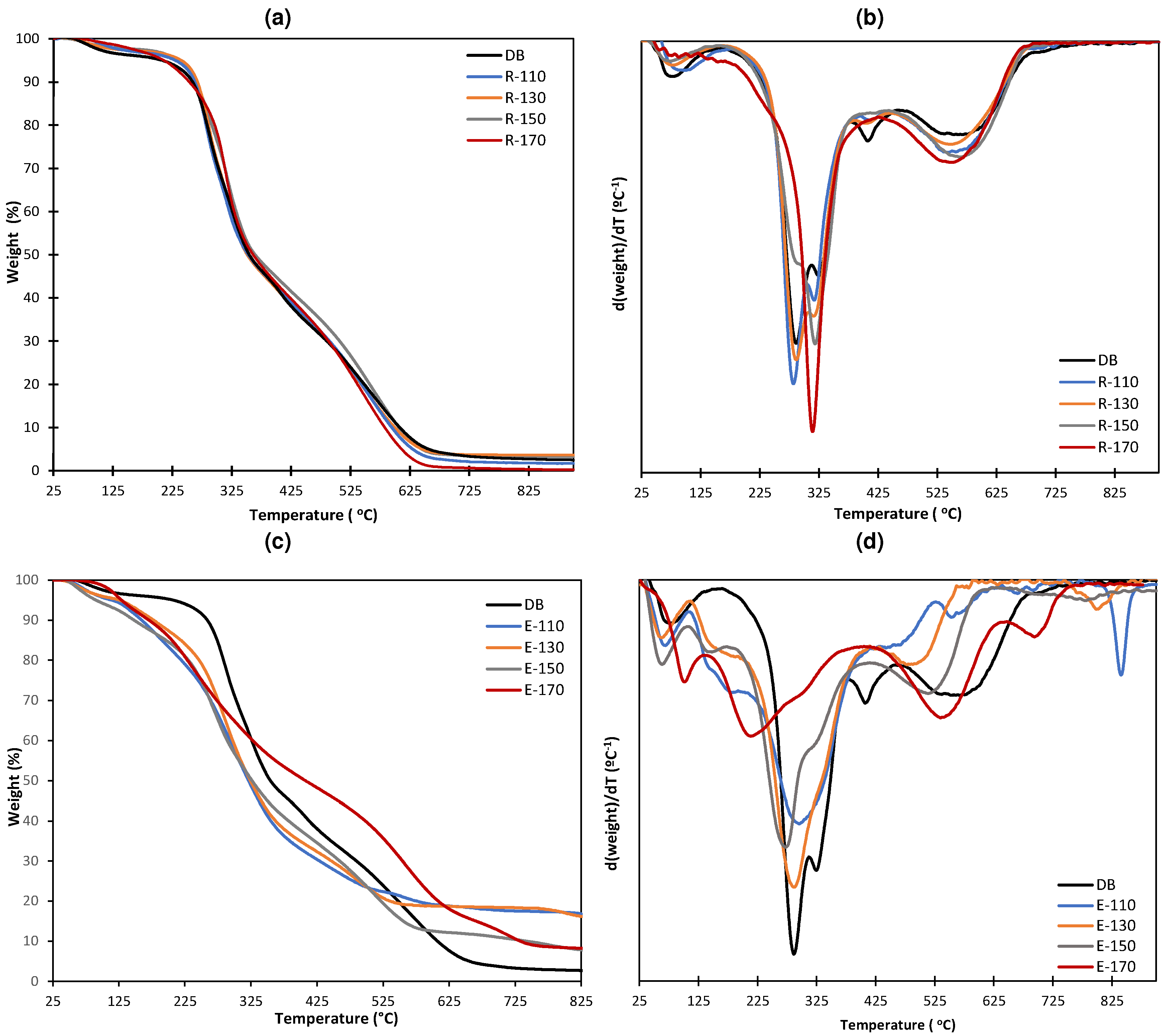
2.1. Yields and Composition of the Different SWE Fractions
2.2. Functional Properties of the SWE Extracts: Antioxidant and Antibacterial Properties
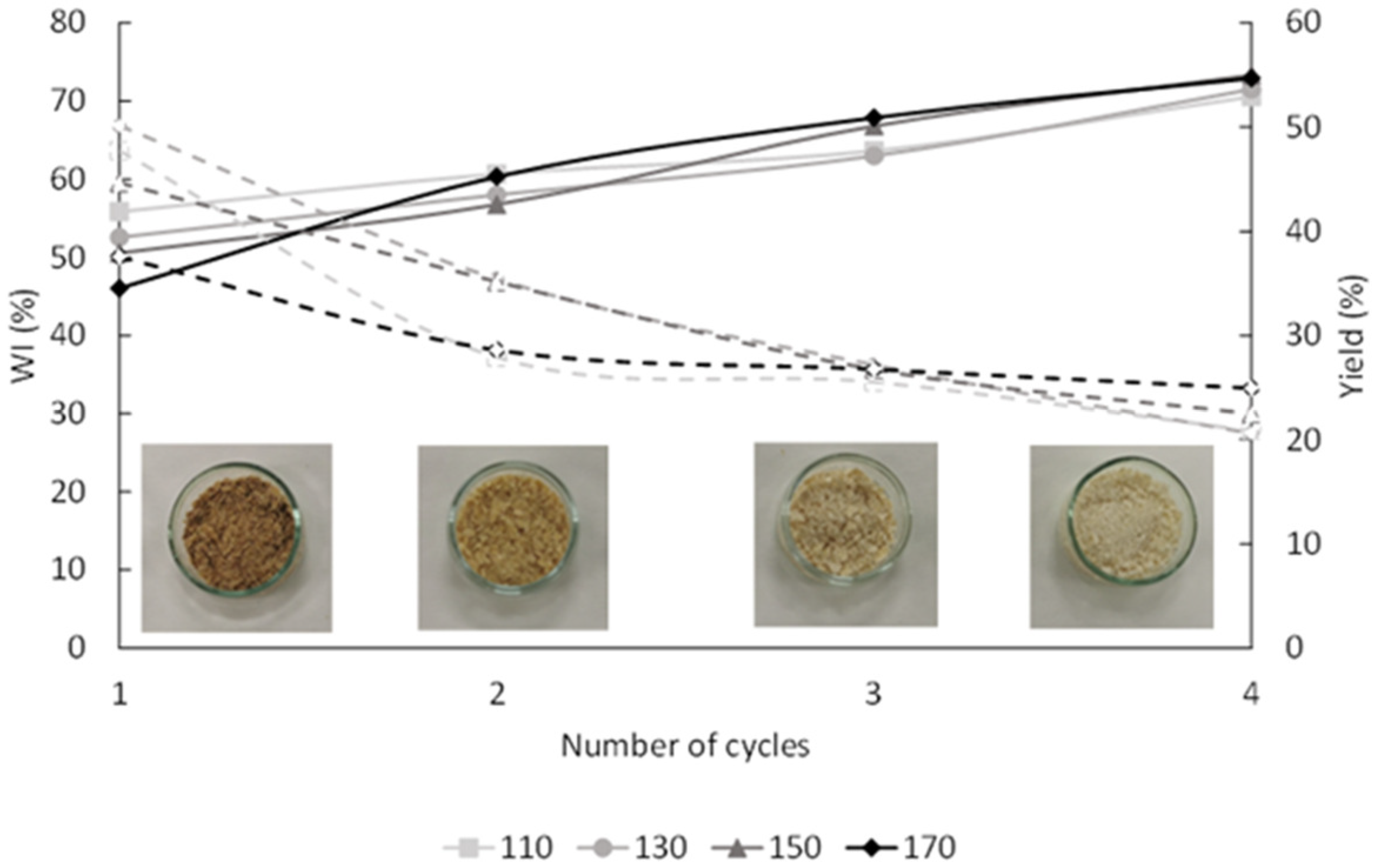
2.3. Bleaching of the Extraction Residues
3. Materials and Methods
3.1. Materials
3.2. Residue Preparation
3.3. Subcritical Water Extraction
3.4. Bleaching Process
3.5. Physico-Chemical Analysis of Beer Bagasse, Soluble, and Insoluble Fractions
3.5.1. Analysis of Structural Components in the Insoluble Fraction
3.5.2. Total Phenolic Content, Antioxidant and Antibacterial Properties of Soluble Fraction
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rodriguez, L.M.; Camina, J.L.; Borroni, V.; Pérez, E. Protein recovery from brewery solid wastes. Food Chem. 2023, 407, 134810. [Google Scholar] [CrossRef] [PubMed]
- Pokrivčak, J.; Supeková, S.C.; Lančarič, D.; Savov, R.; Tóth, M.; Vašina, R. Development of beer industry and craft beer expansion. J. Food Nutr. Res. 2019, 58, 63–74. [Google Scholar]
- Puligundla, P.; Mok, C. Recent advances in biotechnological valorization of brewers’ spent grain. Food Sci. Biotechnol. 2021, 30, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Bonifacio-Lopes, T.; Vilas-Boas, A.; Machado, M.; Costa, E.M.; Silva, S.; Pereira, R.N.; Pintado, M. Exploring the bioactive potential of brewers spent grain ohmic extracts. Innov. Food Sci. Emerg. Technol. 2022, 76, 102943. [Google Scholar] [CrossRef]
- Ikram, S.; Huang, L.; Zhang, H.; Wang, J.; Yin, M. Composition and nutrient value proposition of brewers spent grain. J. Food Sci. 2017, 82, 2232–2242. [Google Scholar] [CrossRef]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ spent grain: A review with an emphasis on food and health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- Devnani, B.; Moran, G.C.; Grossmann, L. Extraction, Composition, Functionality, and Utilization of Brewer’s Spent Grain Protein in Food Formulations. Foods 2023, 12, 1543. [Google Scholar] [CrossRef]
- Guido, L.F.; Moreira, M.M. Techniques for Extraction of Brewer’s Spent Grain Polyphenols: A Review. Food Bioprocess Technol. 2017, 10, 1192–1209. [Google Scholar] [CrossRef]
- Ngasakul, N.; Kozlu, A.; Klojdová, I.; Chockchaisawasdee, S. Applications of Deep Eutectic Solvents in the Recovery of Bioactive Compounds from Brewer Spent Grains. Food Rev. Int. 2023, epub ahead of print. [Google Scholar] [CrossRef]
- Messinese, E.; Pitirollo, O.; Grimaldi, M.; Milanese, D.; Sciancalepore, C.; Cavazza, A. By-Products as Sustainable Source of Bioactive Compounds for Potential Application in the Field of Food and New Materials for Packaging Development. Food Bioprocess Technol. 2023, 17, 606–627. [Google Scholar] [CrossRef]
- Panzella, L.; Moccia, F.; Nasti, R.; Marzorati, S.; Verotta, L.; Napolitano, A. Bioactive phenolic compounds from agri-food wastes: An update on green and sustainable extraction methodologies. Front. Nutr. 2020, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Lachos-Perez, A.B.; Brown, A.; Mudhoo, M.T.; Timko, M.A.; Rostagno, T. Forster-Carneiro. Applications of subcritical and supercritical water conditions for extraction, hydrolysis, gasification, and carbonization of biomass: A critical review. Biofuel Res. J. 2017, 4, 611–626. [Google Scholar] [CrossRef]
- Galamba, N.; Paiva, A.; Barreiros, S.; Simões, P. Solubility of polar and nonpolar aromatic molecules in subcritical water: The role of the dielectric constant. J. Chem. Theory Comput. 2019, 15, 6277–6293. [Google Scholar] [CrossRef] [PubMed]
- Wiboonsirikul, J.; Adachi, S. Extraction of functional substances from agricultural products or by-products by subcritical water treatment. Food Sci. Technol. Res. 2008, 14, 319. [Google Scholar] [CrossRef]
- Zakaria, S.M.; Kamal, S.M.M. Subcritical water extraction of bioactive compounds from plants and algae: Applications in pharmaceutical and food ingredients. Food Eng. Rev. 2016, 8, 23–34. [Google Scholar] [CrossRef]
- Abdelmoez, W.; Nakahasi, T.; Yoshida, H. Amino acid transformation and decomposition in saturated subcritical water conditions. Ind. Eng. Chem. Res. 2007, 46, 5286–5294. [Google Scholar] [CrossRef]
- Sarkar, S.; Gayen, K.; Bhowmick, T.K. Green extraction of biomolecules from algae using subcritical and supercritical fluids. Biomass Convers. Biorefinery 2022, 1–23. [Google Scholar] [CrossRef]
- Majeed, T.; Shabir, I.; Srivastava, S.; Maqbool, N.; Dar, A.H.; Jan, K.; Pandey, V.K.; Shams, R.; Bashir, I.; Dash, K.K. Valorization of food wastes by implementation of subcritical water extraction: A comprehensive review. Trends Food Sci. Technol. 2024, 143, 104316. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Sanz, M.T.; Benito-Roman, O.; Beltran, S.; Trigueros, E. Subcritical water as hydrolytic medium to recover and fractionate the protein fraction and phenolic compounds from craft brewer’s spent grain. Food Chem. 2021, 351, 129264. [Google Scholar] [CrossRef]
- Camacho-Núñez, L.; Jurado-Contreras, S.; La Rubia, M.D.; Navas-Martos, F.J.; Rodríguez-Liébana, J.A. Cellulose-Based Upcycling of Brewer´s Spent Grains: Extraction and Acetylation. J. Polymer Environ. 2023, 32, 3011–3024. [Google Scholar] [CrossRef]
- Shahabi-Ghahafarrokhi, I.; Khodaiyan, F.; Mousavi, M.; Yousefi, H. Preparation and Characterization of Nanocellulose from Beer Industrial Residues Using Acid Hydrolysis/Ultrasound. Fibers Polym. 2015, 16, 529–536. [Google Scholar] [CrossRef]
- Faulds, C.B.; Robertson, J.A.; Waldron, K.W. Effect of pH on the solubilization of brewers’ spent grain by microbial carbohydrases and proteases. J. Agric. Food Chem. 2008, 56, 7038–7043. [Google Scholar] [CrossRef] [PubMed]
- Del Río, J.C.; Prinsen, P.; Gutiérrez, A. Chemical composition of lipids in brewer’s spent grain: A promising source of valuable phytochemicals. J. Cereal Sci. 2013, 58, 248–254. [Google Scholar] [CrossRef]
- Carr, D.G.; Mammucari, R.; Foster, N.R. A review of subcritical water as a solvent and its utilisation for the processing of hydrophobic organic compounds. Chem. Eng. J. 2011, 172, 1–17. [Google Scholar] [CrossRef]
- He, L.; Zhang, X.; Xu, H.; Xu, C.; Yuan, F.; Knez, Z.; Novak, Z.; Gao, Y. Subcritical water extraction of phenolic compounds from pomegranate (Punica granatum L.) seed residues and investigation into their antioxidant activities with HPLC–ABTS+ assay. Food Bioprod. Process. 2012, 90, 215–223. [Google Scholar] [CrossRef]
- Freitas, P.A.; Martín-Pérez, L.; Gil-Guillén, I.; González-Martínez, C.; Chiralt, A. Subcritical Water Extraction for Valorisation of Almond Skin from Almond Industrial Processing. Foods 2023, 12, 3759. [Google Scholar] [CrossRef]
- Carichino, S.; Scanferla, D.; Fico, D.; Rizzo, D.; Ferrari, F.; Jordá-Reolid, M.; Martínez-García, A.; Corcione, C.E. Poly-Lactic Acid-Bagasse Based Bio-Composite for Additive Manufacturing. Polymers 2023, 15, 4323. [Google Scholar] [CrossRef]
- Ortiz, I.; Maroño, M.; Torreiro, Y. Strategy for the Design of Waste to Energy Processes Based on Physicochemical Characterisation. Waste Biomass Valorization 2020, 11, 2961–2971. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, H.; Hou, J.; Yao, Y.; Ma, Y.; Wang, X. Cellulose fibers extracted from sesame hull using subcritical water as a pretreatment. Arab. J. Chem. 2021, 14, 103178. [Google Scholar] [CrossRef]
- Qazanfarzadeh, Z.; Ganesan, A.R.; Mariniello, L.; Conterno, L.; Kumaravel, V. Valorization of brewer’s spent grain for sustainable food packaging. J. Clean. Prod. 2023, 385, 135726. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Ramos, C.; Trigueros, E.; Beltrán, S.; Sanz, M.T. Study of subcritical water scale-up from laboratory to pilot system for brewer’s spent grain valorization. Ind. Crops Prod. 2023, 191 Pt A, 115927. [Google Scholar] [CrossRef]
- Cocero, M.J.; Cabeza, A.; Abad, N.; Adamovic, T.; Vaquerizo, L.; Martínez, C.M.; Pazo-Cepeda, M.V. Understanding biomass fractionation in subcritical & supercritical water. J. Supercrit. Fluids 2018, 133, 550–565. [Google Scholar]
- Ruthes, A.C.; Martínez-Abad, A.; Tan, H.; Bulone, V.; Vilaplana, F. Sequential fractionation of feruloylated hemicelluloses and oligosaccharides from wheat bran using subcritical water and xylanolytic enzymes. Green Chem. 2017, 8, 1919–1931. [Google Scholar] [CrossRef]
- Erven, G.; Visser, R.; Merkx, D.W.H.; Strolenberg, W.; Gijsel, P.; Gruppen, H.; Kabel, M.A. Quantification of Lignin and Its Structural Features in Plant Biomass Using 13C Lignin as Internal Standard for Pyrolysis-GC-SIM-MS. Anal Chem. 2017, 89, 10907–10916. [Google Scholar] [CrossRef] [PubMed]
- Trigueros, E.; Ramos, C.; Alonso-Riaño, P.; Beltrán, S.; Sanz, M.T. Subcritical Water Treatment for Valorization of the Red Algae Residue after Agar Extraction: Scale-Up from Laboratory to Pilot Plant. Ind. Eng. Chem. Res. 2023, 62, 3503–3514. [Google Scholar] [CrossRef]
- Rogalinski, T.; Herrmann, S.; Brunner, G. Production of amino acids from bovine serum albumin by continuous sub-critical water hydrolysis. J. Supercrit. Fluids 2005, 36, 49–58. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J.A. Enzymatic Hydrolysis of Brewers’ Spent Grain Proteins and Technofunctional Properties of the Resulting Hydrolysates. J. Agric. Food. Chem. 2007, 55, 8703–8710. [Google Scholar] [CrossRef]
- Rahman, M.J.; Malunga, L.N.; Eskin, M.; Eck, P.; Thandapilly, S.J.; Thiyam-Hollander, U. Valorization of Heat-Treated Brewers’ Spent Grain Through the Identification of Bioactive Phenolics by UPLC-PDA and Evaluation of Their Antioxidant Activities. Front. Nutr. 2021, 8, 634519. [Google Scholar] [CrossRef]
- Socaci, S.A.; Farca, A.C.; Diaconeasa, Z.M.; Vodnar, D.C.; Rusu, B.; Tofan, M. Influence of the extraction solvent on phenolic content, antioxidant, antimicrobial and antimutagenic activities of brewers’ spent grain. J. Cereal Sci. 2018, 80, 180–187. [Google Scholar] [CrossRef]
- Macias-Garbett, R.; Serna-Hernández, S.O.; Sosa-Hernández, J.E.; Parra-Saldívar, R. Phenolic Compounds from Brewer’s Spent Grains: Toward Green Recovery Methods and Applications in the Cosmetic Industry. Front. Sustain. Food Syst. 2021, 5, 681684. [Google Scholar] [CrossRef]
- Birsan, R.I.; Wilde, P.; Waldron, K.W.; Rai, D.K. Recovery of Polyphenols from Brewer’s Spent Grains. Antioxidants 2019, 8, 380. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Amigo-Benavent, M.; Del Castillo, M.D.; Ibáñez, E.; Herrero, M. Neoformation of Antioxidants in Glycation Model Systems Treated under Subcritical Water Extraction Conditions. Food Res. Int. 2010, 43, 1123–1129. [Google Scholar] [CrossRef]
- Plaza, M.; Amigo-Benavent, M.; Del Castillo, M.D.; Ibáñez, E.; Herrero, M. Facts about the Formation of New Antioxidants in Natural Samples after Subcritical Water Extraction. Food Res. Int. 2010, 43, 2341–2348. [Google Scholar] [CrossRef]
- Lee, S.E.; Hwang, H.J.; Ha, J.S.; Jeong, H.S.; Kim, J.H. Screening of medicinal plant extracts for antioxidant activity. Life Sci. 2003, 73, 167–179. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Bilbao, A.; Vilches, P.; Angulo, I.; LLuis, J.; Fité, B.; Paseiro-Losada, P.; Cruz, J.M. Brewery waste as a potential source of phenolic compounds: Optimisation of the extraction process and evaluation of antioxidant and antimicrobial activities. Food Chem. 2014, 145, 191–197. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass: Laboratory Analytical Procedure (LAP); Version 07-08-2012; Technical Report; National Renewable Energy Laboratory: Golden, CO, USA, 2008. Available online: https://www.nrel.gov/docs/gen/fy13/42618.pdf (accessed on 16 October 2023).
- Sluiter, A.D.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass: Laboratory Analytical Procedure (LAP); Technical Report; National Renewable Energy Laboratory: Golden, CO, USA, 2008. Available online: https://www.nrel.gov/docs/gen/fy08/42619.pdf (accessed on 16 October 2023).
- Vismara, E.; Gastaldi, G.; Valerio, A.; Bertini, S.; Cosentino, C.; Eisle, G. Alpha cellulose from industrial and agricultural renewable sources like short flax fibres, ears of corn and wheat-straw and its transformation into cellulose acetates. J. Mater. Chem. 2009, 45, 8678–8686. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Rocha, G.J.M.; Roberto, I.C. Hydrogen peroxide bleaching of cellulose pulps obtained from brewer’s spent grain. Cellulose 2008, 15, 641–649. [Google Scholar] [CrossRef]
- Krul, E.S. Calculation of Nitrogen-to-Protein Conversion Factors: A Review with a Focus on Soy Protein. J. Am. Oil Chem. Soc. 2019, 96, 339–364. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]

| Sample | Extractive (%) | Protein (%) | Ash (%) | Lignin * (%) | Cellulose (%) | Hemicellulose (%) |
|---|---|---|---|---|---|---|
| DB | 13.1 ± 0.5 a | 22 ± 2 a | 3.71 ± 0.01 c | 9.5 ± 1.6 ab | 17 ± 2 a | 17.9 ± 0.6 d |
| R-110 | 20.9 ± 1.5 b | 28 ± 2 b | 3.12 ± 0.11 b | 9.1 ± 0.4 a | 16 ± 2 a | 15 ± 2 c |
| R-130 | 21.79 ± 0.05 b | 26.2 ± 0.2 ab | 2.79 ± 0.04 b | 11.56 ± 0.08 b | 20 ± 2 a | 14.9± 1.2 c |
| R-150 | 34.5 ± 0.9 d | 26 ± 2 b | 2.27 ± 0.11 a | 14.6 ± 0.5 c | 21 ± 2 a | 7.8 ± 1.2 b |
| R-170 | 28 ± 3 c | 35.7 ± 0.2 ab | 3.1 ± 0.2 b | 19.6 ± 0.3 d | 30 ± 3 b | 2.01 ± 0.08 a |
| E-110 | E-130 | E-150 | E-170 | |
|---|---|---|---|---|
| Protein (g/100 g extract) | 15.1 ± 0.1 a | 16.6 ± 0.2 a | 22.4 ± 1.2 b | 28.7 ± 0.6 c |
| Ashes (g/100 g extract) | 1.54 ± 0.06 c | 1.5 ± 0.1 c | 1.20 ± 0.04 b | 0.46 ± 0.01 a |
| TPC1 (mg GAE/g extract) | 16.8 ± 0.1 a | 22 ± 2 a | 17.91 ± 0.07 b | 59.1 ± 0.2 c |
| TPC2 (mg GAE/g DB) | 1.27 ± 0.08 a | 3.2 ± 0.3 ab | 6.34 ± 0.02 ab | 24.18 ± 0.08 b |
| EC50 (mg extract/mg DPPH) | 13 ± 3 a | 18 ± 2 a | 39 ± 4 b | 59 ± 0.5 c |
| MIC (mg/mL) against L. innocua | 264 | 198 | 168 | 80 |
| MIC (mg/mL) against E. coli | 234 | 204 | 162 | 140 |
| Sample | Ashes (%) | Lignin (%) | Protein (%) | Cellulose (%) | Hemicellulose (%) |
|---|---|---|---|---|---|
| BR-110-1C | 6.96 ± 0.09 a,1 | 14.69 ± 0.03 a,1 | 11.3 ± 0.5 a,3 | 52 ± 2 a,1 | 41 ± 5 a,1 |
| BR-110-2C | 5.9 ± 0.3 b,1 | 15.9 ± 0.8 a,2 | 8.2 ± 0.3 b,2 | 44 ± 2 a,1 | 33 ± 4 b,1 |
| BR-110-3C | 6.20 ± 0.08 b,1 | 13.88 ± 1.09 a,2 | 5,1 ± 0.2 c,2 | 57 ± 5 a,1 | 30 ± 3 c,1 |
| BR-110-4C | 6.2 ± 0.3 b,1 | 11.5 ± 0.9 b,1 | 1.9 ± 0.3 d,2 | 53 ± 4 a,1 | 26 ± 3 d,1 |
| BR-130-1C | 6.5 ± 0.3 b,1 | 16.2 ± 0.5 ab,2 | 21 ± 0.6 a,1 | 62 ± 3 a,1 | 28 ± 3 a,2 |
| BR-130-2C | 7.07 ± 0.07 b,2 | 17.3 ± 0.4 bc,1 | 11.3 ± 0.4 b,1 | 65 ± 4 a,2 | 31 ± 6 a,1 |
| BR-130-3C | 6.5 ± 0.3 b,1 | 17.97 ± 0.04 c,3 | 7.7 ± 0.2 c,1 | 67± 3 a,1 | 22 ± 3 ab,2 |
| BR-130-4C | 5.8 ± 0.2 a,1 | 15.6 ± 0.8 a,2 | 2.5 ± 0.1 d,1 | 62 ± 5 a,12 | 15 ± 2 b,2 |
| BR-150-1C | 5.07 ± 0.41 a,2 | 20.88 ± 0.06 a,3 | 16.2 ± 0.9 a,2 | 58 ± 6 a,1 | 15 ± 2 a,3 |
| BR-150-2C | 7.4 ± 0.5 a,2 | 15.35 ± 0.11 b,23 | 11.6 ± 0.4 b,1 | 64 ± 5 a,2 | 14 ± 2 a,2 |
| BR-150-3C | 5.5 ± 1.4 a,1 | 15 ± 2 b,2 | 7.3 ± 0.2 c,1 | 62 ± 5 a,1 | - |
| BR-150-4C | 4.9 ± 0.7 a,1 | 14.77 ± 1.02 b,2 | 2.9 ± 0.7 d,1 | 71 ± 6 a,2 | - |
| BR-170-1C | 5.5 ± 0.4 a,2 | 21.9 ± 0.3 a,4 | 6.7 ± 0.2 a,4 | 60 ± 3 a,1 | - |
| BR-170-2C | 5.7 ± 0.3 a,1 | 14.4 ± 0.2 b,3 | 4.8 ± 0.3 b,3 | 42 ± 2 c,1 | - |
| BR-170-3C | 5.7 ± 0.4 a,1 | 11.5 ± 0.2 c,1 | 2.9 ± 0.3 c,3 | 44 ± 2 c,2 | - |
| BR-170-4C | 6.2 ± 0.2 a,1 | 10.2 ± 0.2 d,1 | 1.2 ± 0.2 d,3 | 53 ± 4 b,1 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomez-Contreras, P.A.; Obando, C.; Freitas, P.A.V.d.; Martin-Perez, L.; Chiralt, A.; Gonzalez-Martinez, C. Applying Subcritical Water Extraction to Obtain Bioactive Compounds and Cellulose Fibers from Brewer Spent Grains. Molecules 2024, 29, 4897. https://doi.org/10.3390/molecules29204897
Gomez-Contreras PA, Obando C, Freitas PAVd, Martin-Perez L, Chiralt A, Gonzalez-Martinez C. Applying Subcritical Water Extraction to Obtain Bioactive Compounds and Cellulose Fibers from Brewer Spent Grains. Molecules. 2024; 29(20):4897. https://doi.org/10.3390/molecules29204897
Chicago/Turabian StyleGomez-Contreras, Paula Andrea, Catalina Obando, Pedro Augusto Vieira de Freitas, Laia Martin-Perez, Amparo Chiralt, and Chelo Gonzalez-Martinez. 2024. "Applying Subcritical Water Extraction to Obtain Bioactive Compounds and Cellulose Fibers from Brewer Spent Grains" Molecules 29, no. 20: 4897. https://doi.org/10.3390/molecules29204897
APA StyleGomez-Contreras, P. A., Obando, C., Freitas, P. A. V. d., Martin-Perez, L., Chiralt, A., & Gonzalez-Martinez, C. (2024). Applying Subcritical Water Extraction to Obtain Bioactive Compounds and Cellulose Fibers from Brewer Spent Grains. Molecules, 29(20), 4897. https://doi.org/10.3390/molecules29204897







