Visual Tracking of Hydrogen Sulfide: Application of a Novel Lysosome-Targeted Fluorescent Probe for Bioimaging and Food Safety Assessment
Abstract
1. Introduction
2. Results and Discussion
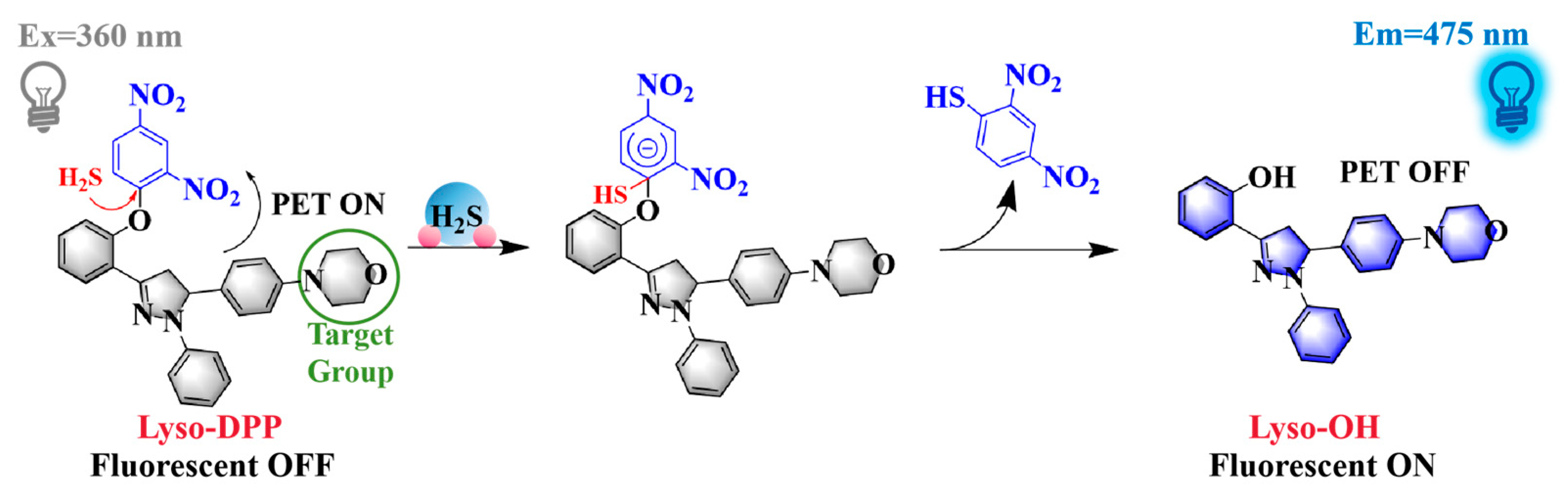
2.1. Design and Synthesis of Lyso-DPP
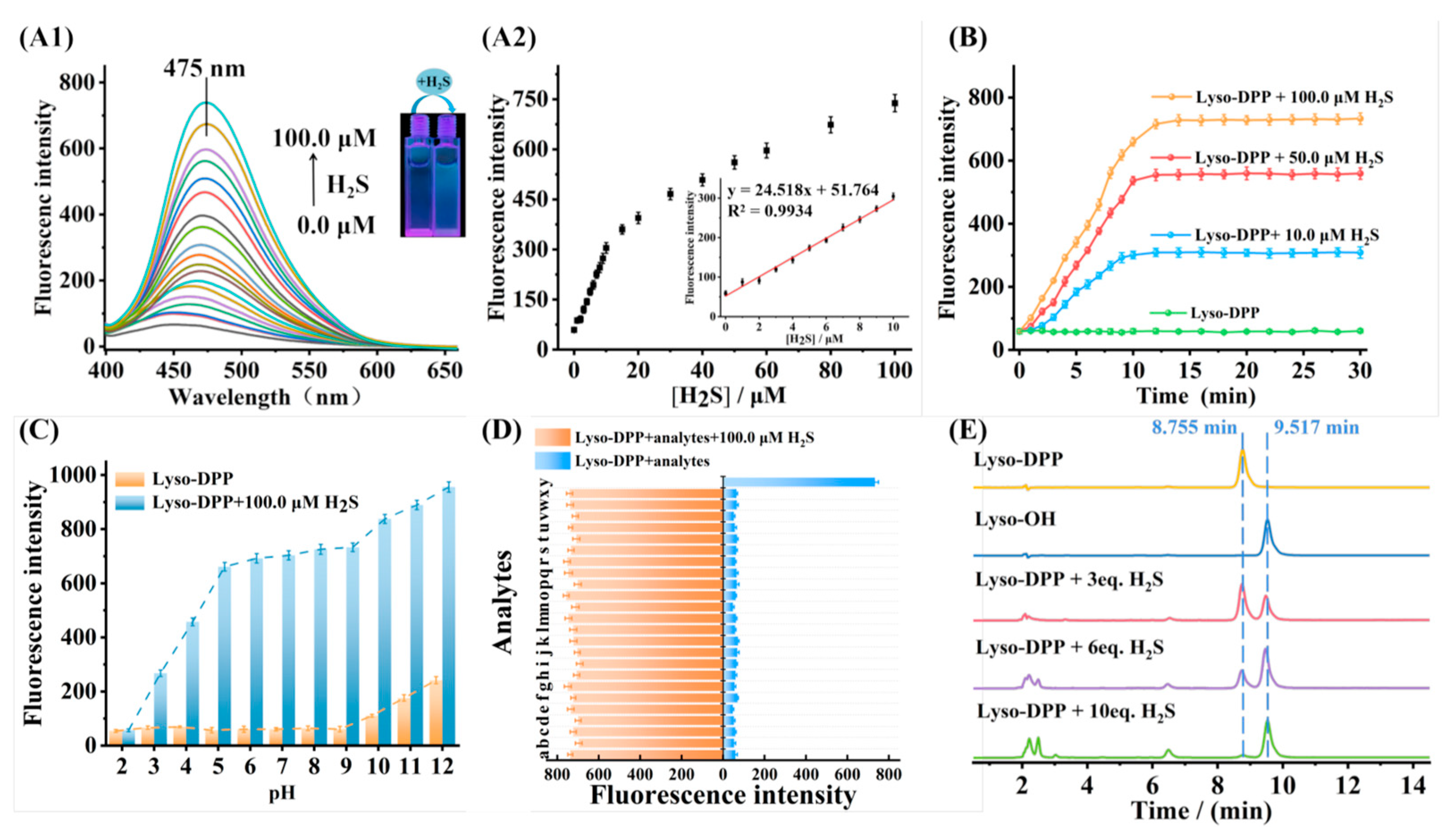
2.2. Photophysical Properties of Lyso-DPP
2.3. Kinetic Study of Lyso-DPP
2.4. pH Study of Lyso-DPP
2.5. Selectivity and Competition Study of Lyso-DPP
2.6. Sensing Mechanism
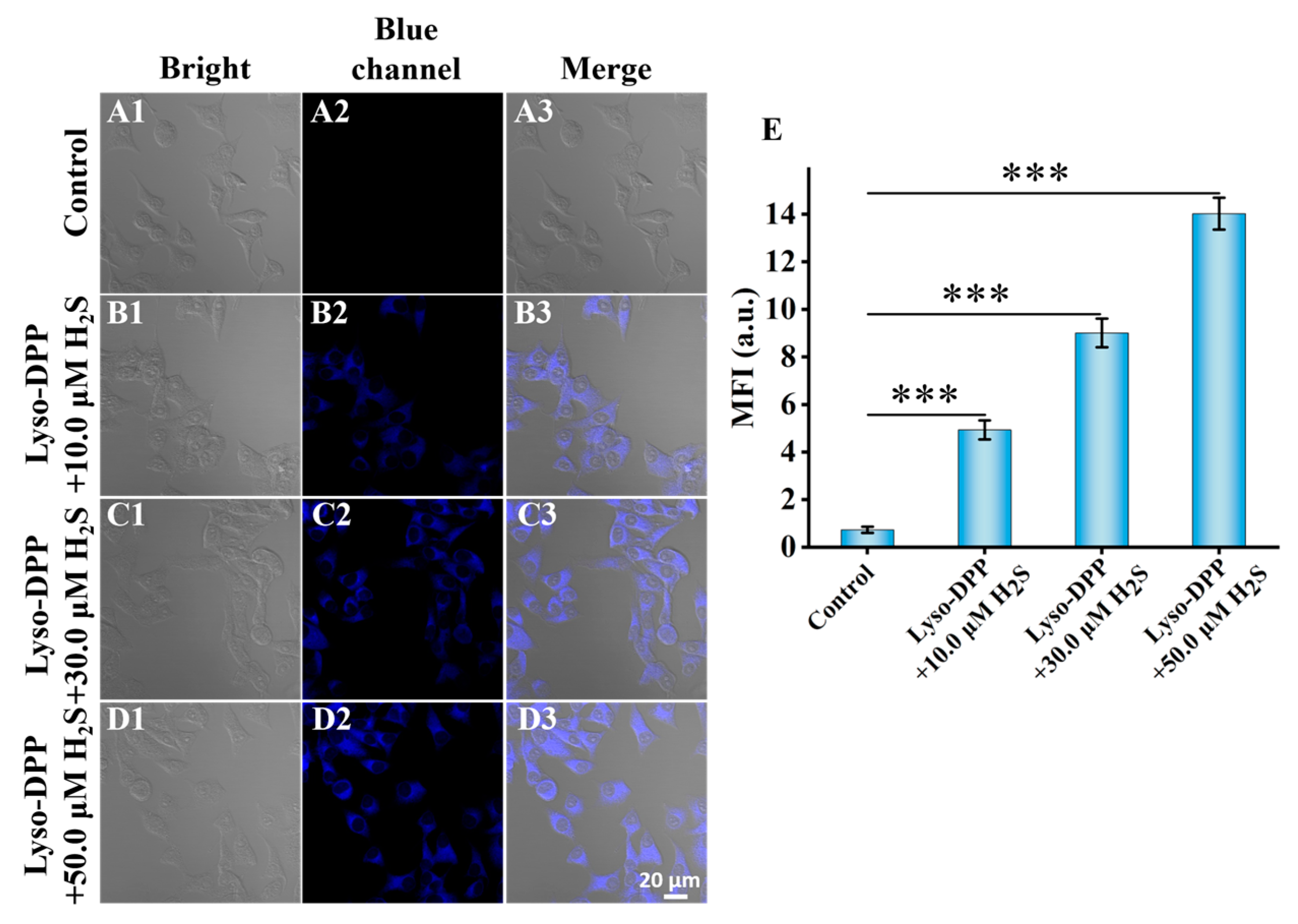
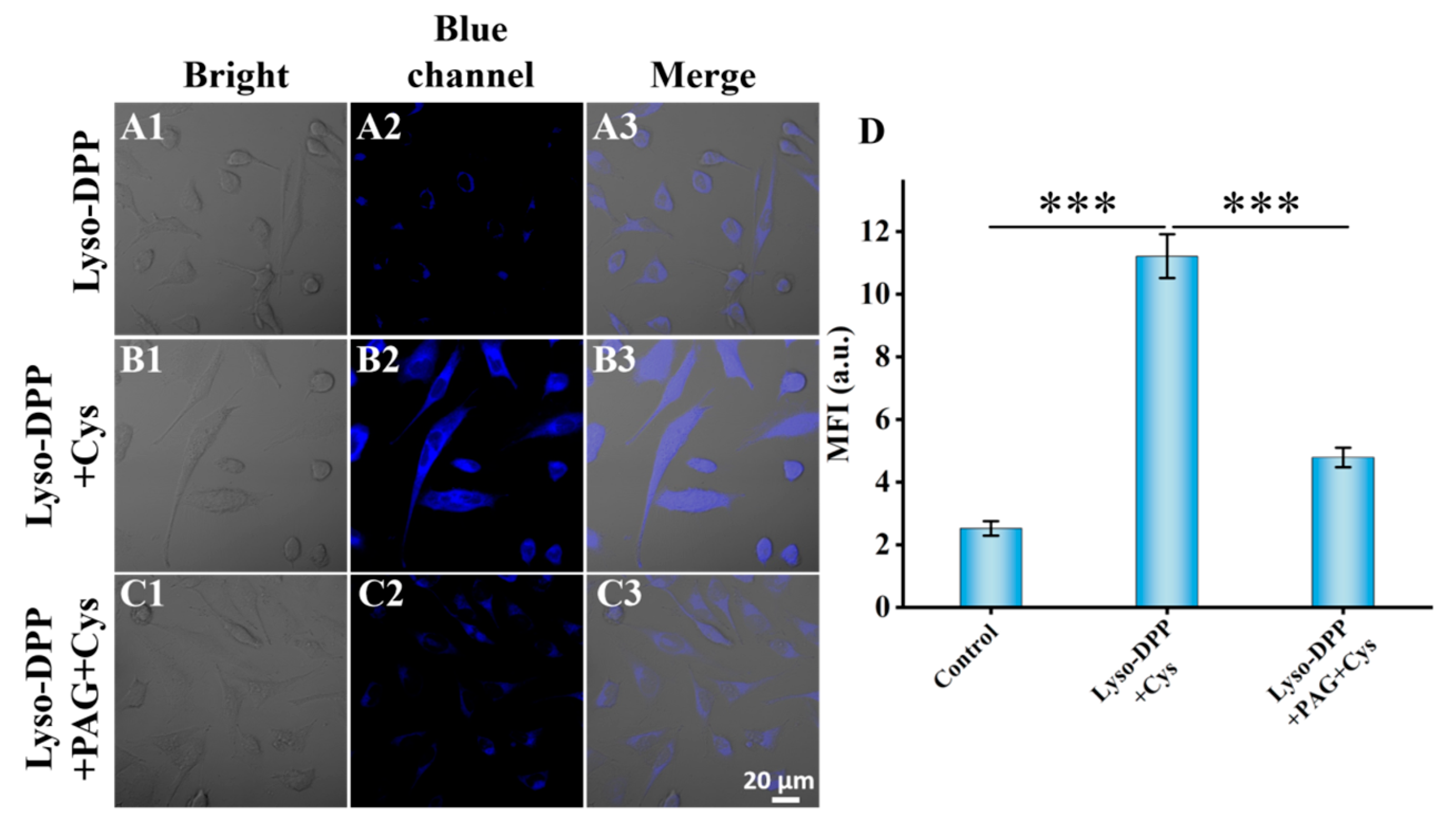
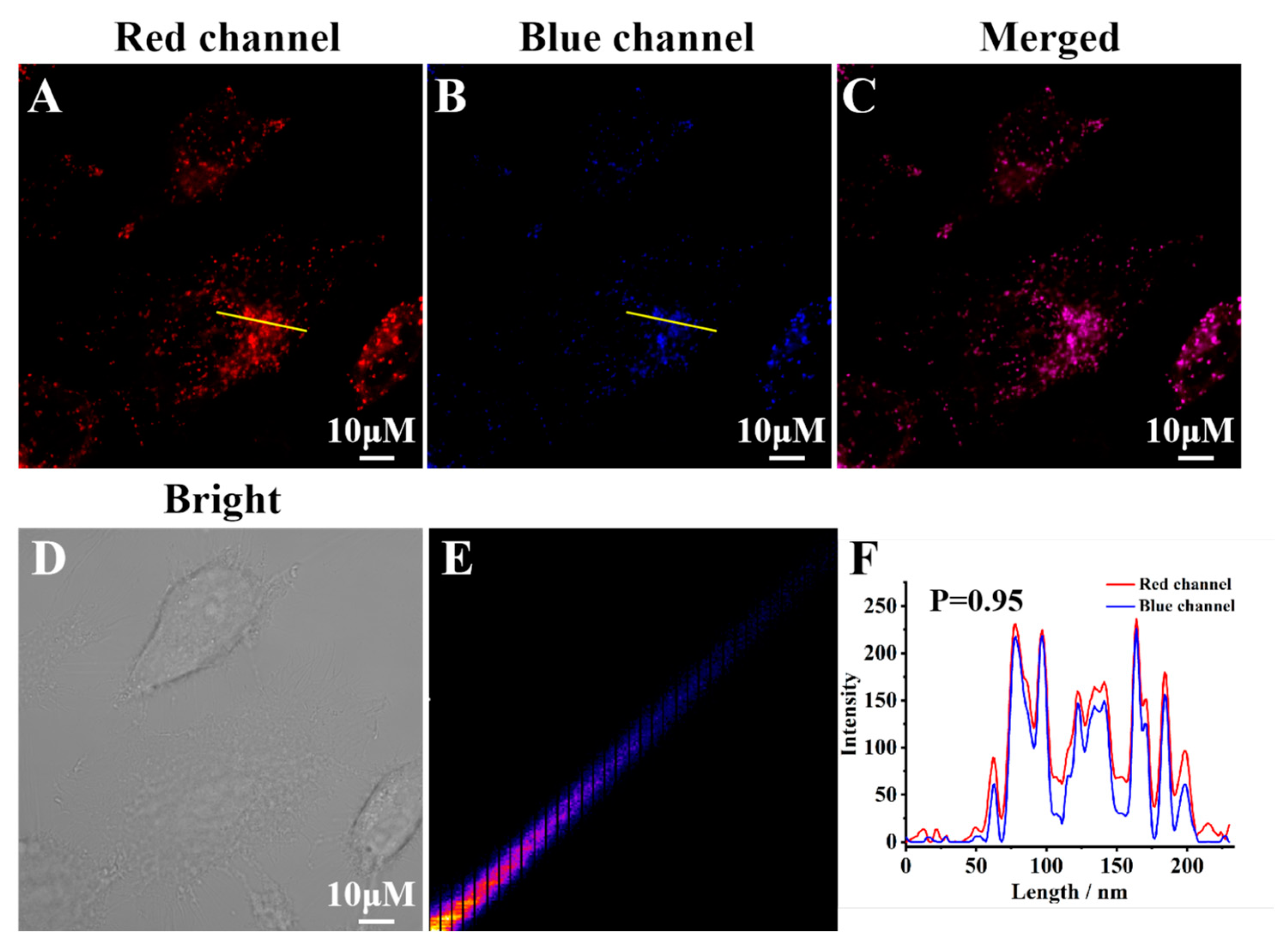
2.7. Fluorescence Imaging in Live Cells
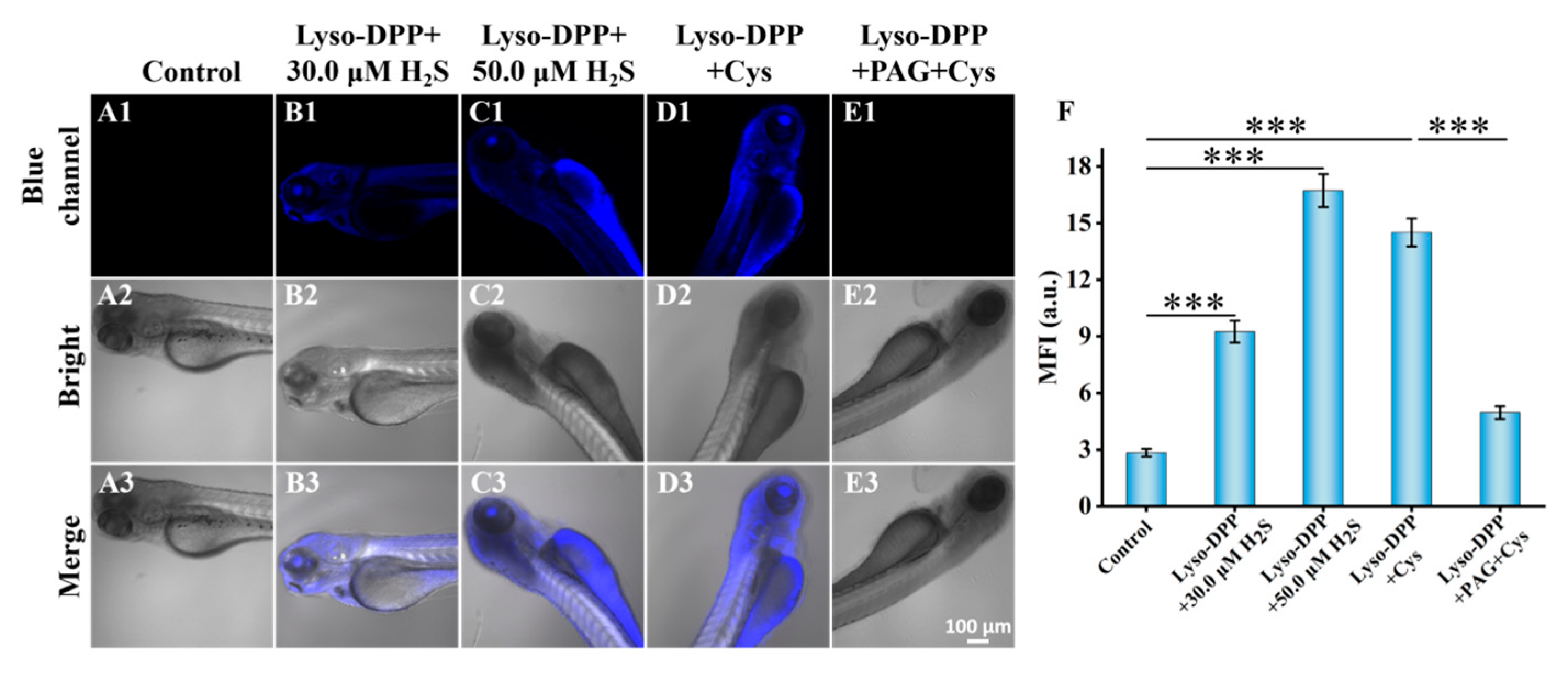
2.8. Fluorescence Imaging in Zebrafish
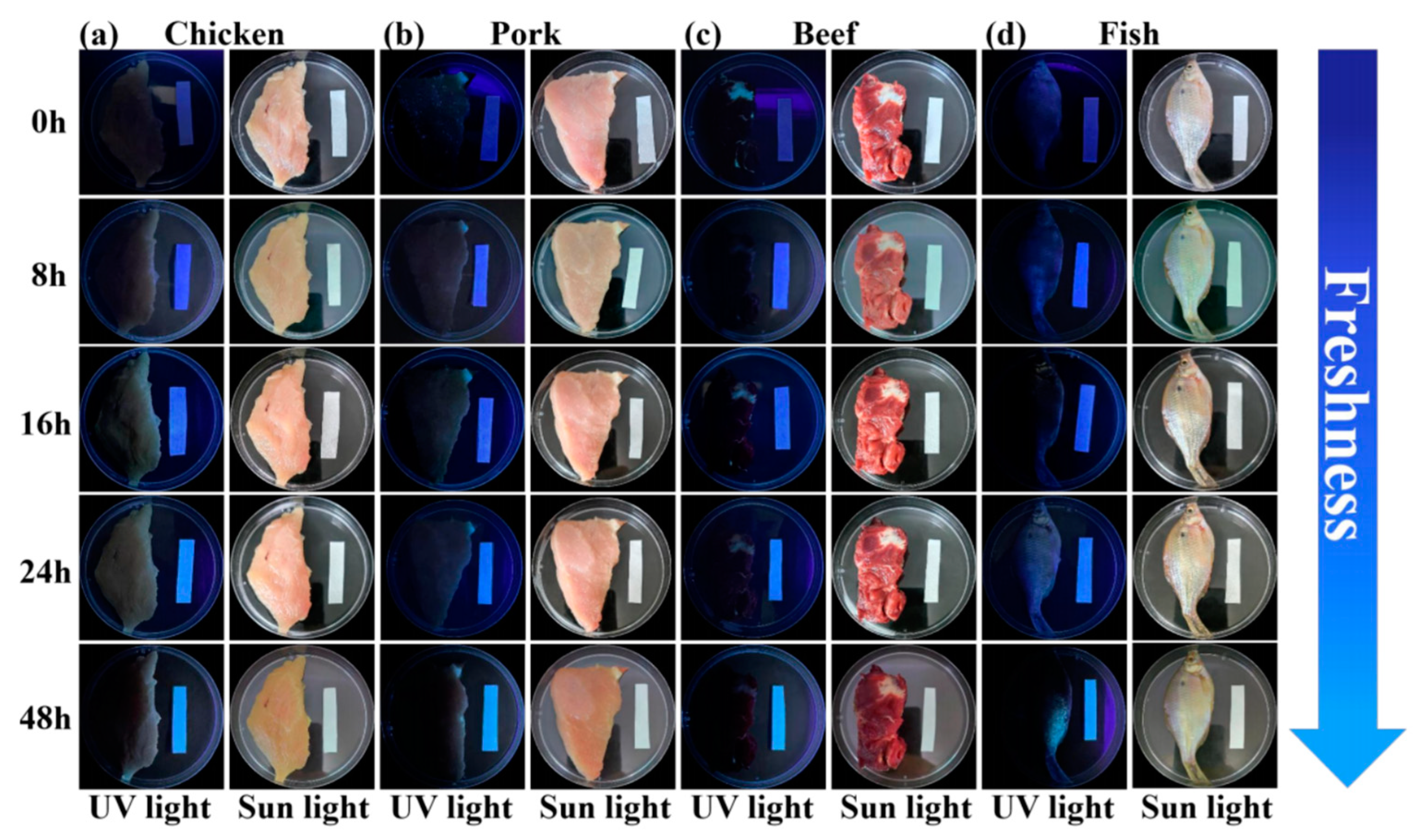
2.9. Detection of H2S and Food Samples by Means of Test Strips
3. Materials and Methods
3.1. Reagents and Apparatus
3.2. Synthesis of Lyso-OH
3.3. Synthesis of Lyso-DPP
3.4. Spectral Measurement
3.5. Cell Culture and Incubation
3.6. Zebrafish Incubation
3.7. Test Strips Test Processing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liang, B.B.; Wang, B.Y.; Ma, Q.J.; Xie, C.X.; Li, X.; Wang, S.P. A lysosome-targetable turn-on fluorescent probe for the detection of thiols in living cells based on a 1,8-naphthalimide derivative. Spectrochim Acta A 2018, 192, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Jiang, D.T.; Jiang, C.; Yao, C. A novel near-infrared ratiometric fluorescent probe targeting lysosomes for imaging HOCl in vitro and in vivo. Spectrochim Acta A 2023, 286, 121966. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.G.; Fu, A.X.; Liu, C.X.; Xi, Y.B.; Cui, S.L.; Gao, N.; Yang, L.L.; Shang, W.B.; Ma, N.N.; He, G.J.; et al. A novel lysosomal targeted near-infrared probe for ratio detection of carbon monoxide in cells and in vivo. Spectrochim Acta A 2024, 311, 123987. [Google Scholar] [CrossRef] [PubMed]
- An, K.; Fan, J.X.; Lin, B.; Han, Y.F. A lysosome-targeted fluorescent probe for fluorescence imaging of hypochlorous acid in living cells and in vivo. Spectrochim Acta A 2024, 316, 124316. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.Q.; Li, J.; Lia, S.N.; Zhou, J.X.; Cao, Z.X.; Lia, L.X.; Zheng, D.B.; Zhao, X.; Wang, W.; Deng, Y.; et al. Real-time monitoring of endogenous cysteine in LPS-induced oxidative stress process with a novel lysosome-targeted fluorescent probe. Anal. Chim. Acta. 2023, 1279, 341819. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.F.; Liang, J.P.; Fan, Z.F. A review: Small organic molecule dual/multi-organelle-targeted fluorescent probes. Talanta. 2023, 259, 124529. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, N.; Gaur, R.; Shahabuddin, S.; Chandra, P. Recent progress in lysosome-targetable fluorescent BODIPY probes for bioimaging applications. Mater. Today Proc. 2022, 62, 7082–7087. [Google Scholar] [CrossRef]
- Ou-Yang, J.; Jiang, W.-L.; Tan, K.-Y.; Liu, H.-W.; Li, S.-J.; Liu, J.; Li, Y.-F.; Li, C.-Y. Two-photon fluorescence probe for precisely detecting endogenous H2S in lysosome by employing a dual lock system. Spectrochim Acta A 2018, 260, 264–273. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, J.-L.; Wu, W.-N.; Zhao, X.-L.; Wang, Y.; Fan, Y.-C.; Xu, Z.-H. A “turn-on” chalcone-based probe for hydrogen sulfide and imaging applications in lysosomes of living cells and zebrafish. Microchem. J. 2022, 183, 107941. [Google Scholar] [CrossRef]
- Wang, W.-X.; Wang, Z.-Q.; Tan, Z.-K.; Mao, G.-J.; Chen, D.-H.; Li, C.-Y. A nitrobenzoxadiazole-based near-infrared fluorescent probe for the specific imaging of H2S in inflammatory and tumor mice. Analyst 2022, 147, 2712–2717. [Google Scholar] [CrossRef]
- Li, M.H.; Jiao, Y.; Duan, C.Y. A dual-emission fluorescence-enhanced probe for hydrogen sulfide and its application in biological imaging. New J. Chem. 2022, 46, 10184–10190. [Google Scholar] [CrossRef]
- Zou, J.H.; Song, B.; Kong, D.S.; Dong, Z.Y.; Liu, Q.; Yuan, J.L. Responsive β-Diketonate-europium(III) Complex-Based Probe for Time-Gated Luminescence Detection and Imaging of Hydrogen Sulfide In Vitro and In Vivo. Inorg. Chem. 2024, 63, 13244–13252. [Google Scholar] [CrossRef]
- Liao, L.J.; Li, Z.J.; Hu, W.; Hu, Y.; Liu, B.M.; Wang, L.S.; Wang, M.; Wang, J.Y. Design, synthesis and evaluation of a novel fluorescent probe to accurately detect H2S in lysosomes. Tetrahedron Lett. 2018, 59, 2683–2687. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Q.C.; Cai, S.T.; Ding, H.; He, S.; Zhao, L.C.; Zeng, X.S.; Gong, J. Novel near-infrared spectroscopic probe for visualizing hydrogen sulfide in lysosomes. Spectrochim Acta A 2022, 271, 120917. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.Y.; Zhang, J.Y.; Zheng, X.; Li, G.L.; Xing, C.R.; Li, P.; Yuan, J. Recent design strategies and applications of organic fluorescent probes for food freshness detection. Food Res. Int 2023, 174, 113641. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.L.; Li, Y.; Zeng, F.; Wu, S.Z. A near-infrared fluorescent probe for detection of H2S and its application in monitoring meat freshness and plant growth under aluminium-induced stress. New J. Chem. 2023, 47, 2643–2650. [Google Scholar] [CrossRef]
- Zhao, X.X.; He, F.R.; Dai, Y.P.; Ma, F.L.; Qi, Z.J. A single fluorescent probe for one- and two-photon imaging hydrogen sulfide and hydrogen polysulfides with different fluorescence signals. Dyes Pigm. 2020, 172, 107818. [Google Scholar] [CrossRef]
- Xie, L.; Fan, T.T.; Yao, R.H.; Mu, Y.Q.; Wang, R.J.; Fan, C.B.; Pu, S.Z. Highly selective near-infrared fluorescent probe with large Stokes shift and sensitivity for H2S detection in water, foodstuff and imaging in living cells. Dyes Pigm. 2022, 208, 110828. [Google Scholar] [CrossRef]
- Mu, S.A.; Han, T.H.; Zhang, X.Y.; Liu, J.; Sun, H.P.; Zhang, J.L.; Liu, X.Y.; Zhang, H.X. Exploring the Role of Mitochondrial Hydrogen Sulfide in Maintaining Polarity and mtDNA Integrity with a Multichannel Fluorescent Probe. Anal. Chem. 2023, 95, 18460–18469. [Google Scholar] [CrossRef]
- Qu, W.B.; Tian, R.W.; Yang, B.; Guo, T.Y.; Wu, Z.; Li, Y.; Geng, Z.R.; Wang, Z.L. Dual-Channel/Localization Single-Molecule Fluorescence Probe for Monitoring ATP and HOCl in Early Diagnosis and Therapy of Rheumatoid Arthritis. Anal. Chem. 2024, 96, 5428–5436. [Google Scholar] [CrossRef]
- Kong, L.X.; Lu, W.J.; Cao, X.L.; Wei, Y.C.; Sun, J.R.; Wang, Y.F. The design strategies and biological applications of probes for the gaseous signaling molecule hydrogen sulfide. J. Mater. Chem. B 2022, 10, 7924–7954. [Google Scholar] [CrossRef]
- Lin, P.X.; Jiang, S.L.; Liu, T.; Yuan, X.M.; Luo, K.; Xie, C.; Zhao, X.J.; Zhou, L.Y. Activatable fluorescent probes for early diagnosis and evaluation of liver injury. Analyst 2024, 149, 638–664. [Google Scholar] [CrossRef]
- Han, T.T.; Sun, Y.; Zhao, C.; Wang, H.-Y.; Yu, H.; Liu, Y. Mitochondrial-Targeted Ratiometric Near-Infrared Fluorescence Probe for Monitoring Nitric Oxide in Rheumatoid Arthritis. J. Med. Chem. 2024, 67, 4026–4035. [Google Scholar] [CrossRef]
- Fang, X.; Wang, S.Q.; Wang, Q.Q.; Gong, J.; Li, L.; Lua, H.L.; Xue, P. A highly selective and sensitive fluorescence probe based on BODIPY-cyclen for hydrogen sulfide detection in living cells and serum. Talanta 2024, 268, 125339. [Google Scholar] [CrossRef]
- Wang, L.; Liu, F.-Y.; Liu, H.-Y.; Dong, Y.-S.; Liu, T.-Q.; Liu, J.-F.; Yao, Y.-W.; Wan, X.-J. A novel pyrazoline-based fluorescent probe for detection of hydrazine in aqueous solution and gas state and its imaging in living cells. Sens. Actuators B-Chem. 2016, 229, 441–452. [Google Scholar] [CrossRef]
- Zhang, Y.-P.; Zhao, Y.-C.; Xue, Q.-H.; Yang, Y.-S.; Guo, H.-C.; Xue, J.-J. A novel pyrazoline-based fluorescent probe for Cu2+ in aqueous solution and imaging in live cell. Inorg. Chem. Commun. 2021, 129, 108612. [Google Scholar] [CrossRef]
- Zhang, Y.-P.; Teng, Q.; Yang, Y.-S.; Guo, H.-C.; Xue, J.-J. A novel coumarin-based pyrazoline fluorescent probe for detection of Fe3+ and its application in cells. Inorg. Chim. Acta 2021, 525, 120468. [Google Scholar] [CrossRef]
- Guo, F.-F.; Han, X.-F.; Zhao, X.-L.; Wang, Y.; Fan, Y.-C.; Wu, W.-N.; Xu, Z.-H. A ratiometric fluorescent probe for hydrogen sulfide in neat aqueous solution and its application in lysosome-targetable cell imaging. Spectrochim. Acta A 2022, 270, 120835. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, Z.Q.; Yang, W.Y.; Zhao, X.; Li, L.X.; Cao, Z.X.; Zhou, H.C.; Zheng, R.L.; Deng, Y.; Peng, C.; et al. A novel AIE-based mitochondria-targeting fluorescent probe for monitoring of the fluctuation of endogenous hypochlorous acid in ferroptosis models. Anal. Bioanal. Chem. 2024, 416, 4873–4885. [Google Scholar] [CrossRef]
- Luo, W.F.; Xue, H.Y.; Ma, J.J.; Wang, L.; Liu, W.S. Molecular engineering of a colorimetric two-photon fluorescent probe for visualizing H2S level in lysosome and tumor. Anal. Chim. Acta 2019, 1077, 273–280. [Google Scholar] [CrossRef]
- Zhang, W.S.; Xun, Q.N.; Xing, W.F.; Xu, F.; Liu, X. A Highly Selective Fluorescent Probe for Imaging Hydrogen Sulfide in Living HeLa Cells. J. Fluoresc. 2023, 33, 1603–1608. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.C.; Weng, X.S.; Yang, Z.M.; Zhao, P.; Chen, W.J.; Wu, Z.X.; Zhuang, X.W. A Chalcone-based Fluorescence Probe for H2S Detecting Utilizing ESIPT Coupled ICT Mechanism. J. Fluoresc. 2024, 34, 821–828. [Google Scholar] [CrossRef]
- Hong, L.-X.; Sun, L.; Li, C.; Zhang, R.-L.; Zhao, J.-S. Multiple Applications of a Novel Fluorescence Probe with Large Stokes Shift and Sensitivity for Rapid H2S Detection. J. Fluoresc. 2023, 34, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- Teng, Z.X.; Shangguan, H.M.; Cheng, Z.S.; Liu, L.F.; Zhang, S.H.; Li, G.L.; Qi, F.P.; Liu, X.J. Design, synthesis and application of dual-channel fluorescent probes for ratiometric detection of HClO and H2S based on phenothiazine coumarins. Spectrochim. Acta A 2024, 316, 124312. [Google Scholar] [CrossRef] [PubMed]
- Muthusamy, S.; Rajalakshmi, K.; Zhu, D.W.; Zhao, L.; Wang, S.J.; Zhu, W.H. A novel lysosome targeted fluorophore for H2S sensing: Enhancing the quantitative detection with successive reaction sites. Sens. Actuators B-Chem. 2020, 320, 128433. [Google Scholar] [CrossRef]
- Yang, W.J.; Gong, L.L.; Liu, R.X.; Wu, K.; Wang, L.H.; Fan, G.W.; Jiang, H.Q.; Li, Y.L.; Tang, Z.X. A novel HClO-activatable lysosome-targeted fluorescent probe for imaging and diagnosis of atherosclerosis. Microchem J. 2024, 196, 109694. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Liu, Y.; Ren, H.; Hou, P.; Wang, H.; Sun, J.; Liu, L.; He, C.; Chen, S. Visual Tracking of Hydrogen Sulfide: Application of a Novel Lysosome-Targeted Fluorescent Probe for Bioimaging and Food Safety Assessment. Molecules 2024, 29, 3906. https://doi.org/10.3390/molecules29163906
Liu L, Liu Y, Ren H, Hou P, Wang H, Sun J, Liu L, He C, Chen S. Visual Tracking of Hydrogen Sulfide: Application of a Novel Lysosome-Targeted Fluorescent Probe for Bioimaging and Food Safety Assessment. Molecules. 2024; 29(16):3906. https://doi.org/10.3390/molecules29163906
Chicago/Turabian StyleLiu, Likun, Yitong Liu, Haoqing Ren, Peng Hou, Haijun Wang, Jingwen Sun, Lei Liu, Chuan He, and Song Chen. 2024. "Visual Tracking of Hydrogen Sulfide: Application of a Novel Lysosome-Targeted Fluorescent Probe for Bioimaging and Food Safety Assessment" Molecules 29, no. 16: 3906. https://doi.org/10.3390/molecules29163906
APA StyleLiu, L., Liu, Y., Ren, H., Hou, P., Wang, H., Sun, J., Liu, L., He, C., & Chen, S. (2024). Visual Tracking of Hydrogen Sulfide: Application of a Novel Lysosome-Targeted Fluorescent Probe for Bioimaging and Food Safety Assessment. Molecules, 29(16), 3906. https://doi.org/10.3390/molecules29163906






