Nitrosyl and Thionitrosyl Complexes of Technetium and Rhenium and Their Reactions with Hydrotris(pyrazolyl)borates
Abstract
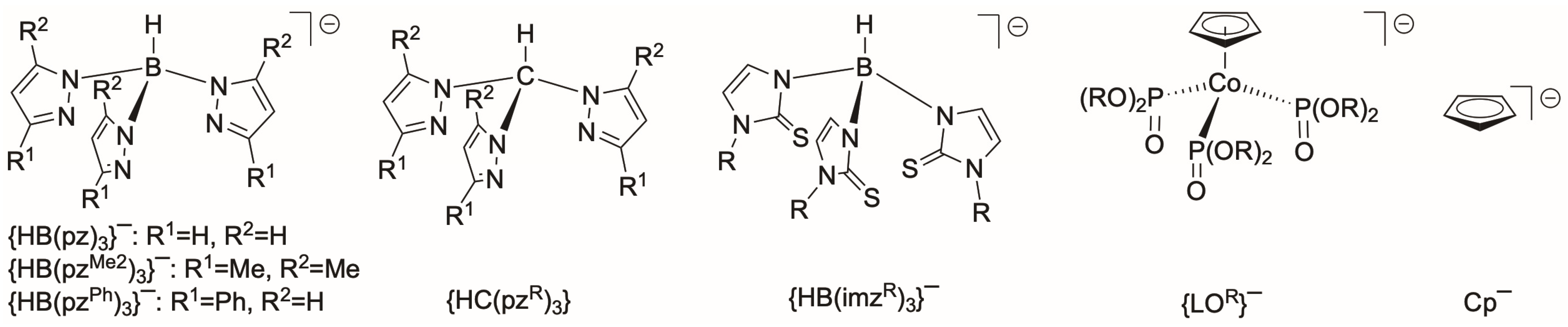
1. Introduction
2. Results and Discussion
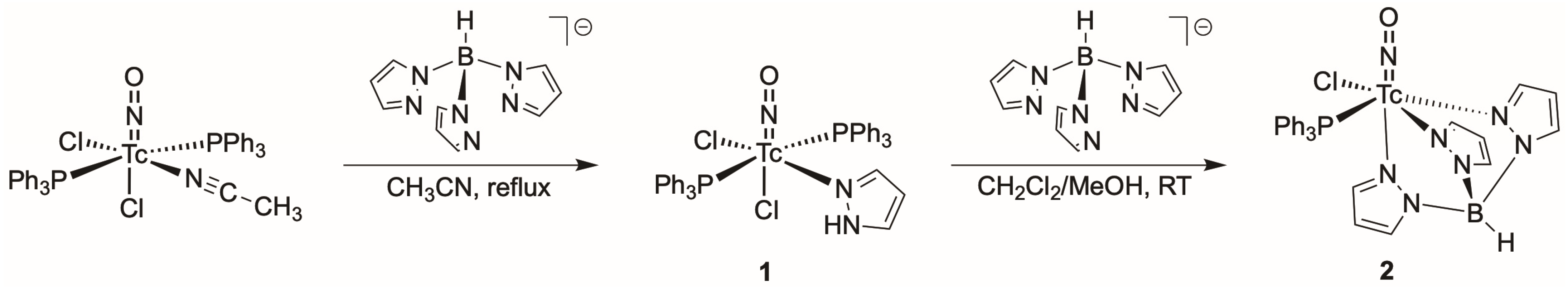
2.1. Nitrosyl Complexes of Technetium
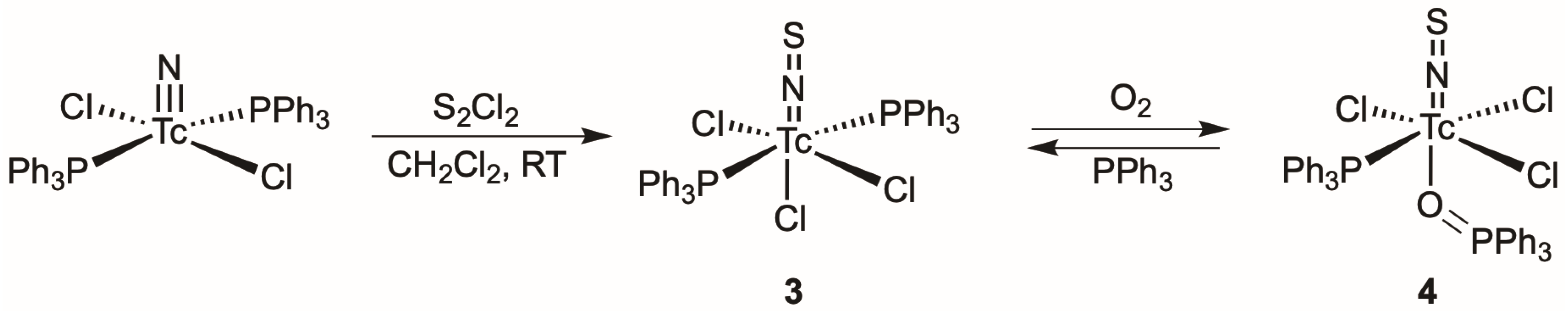
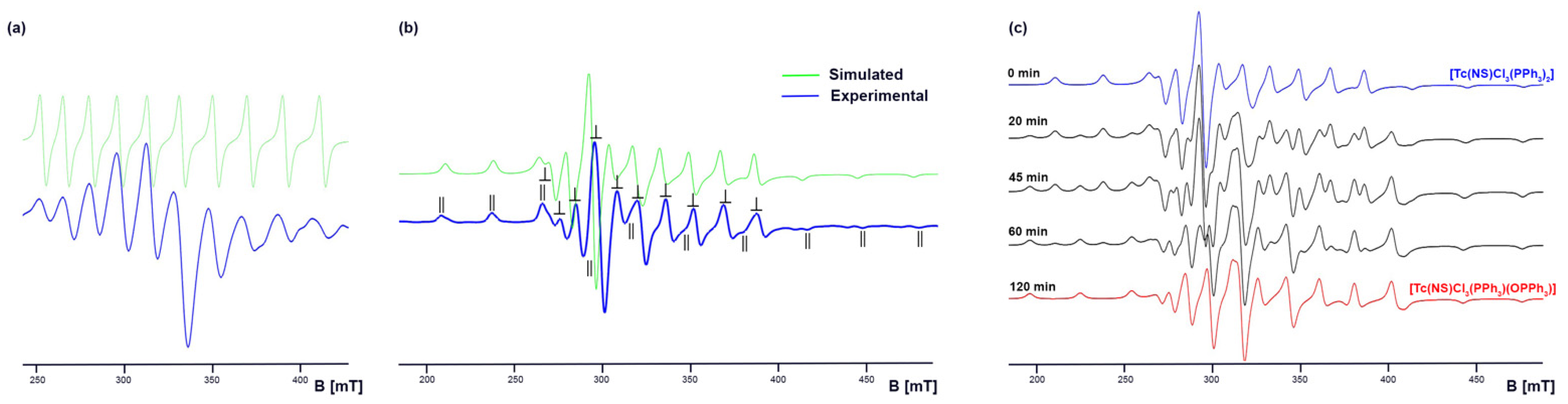
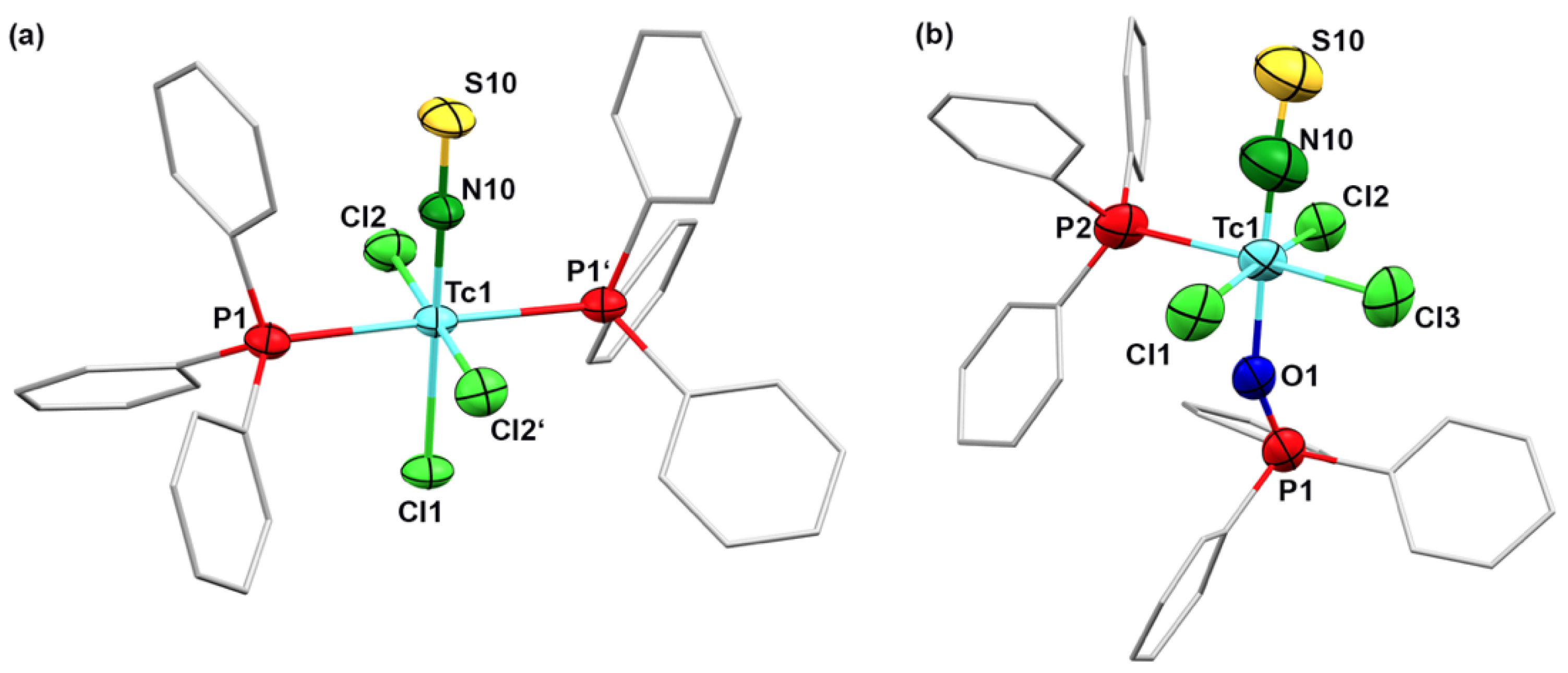
2.2. Thionitrosyl Complexes of Technetium
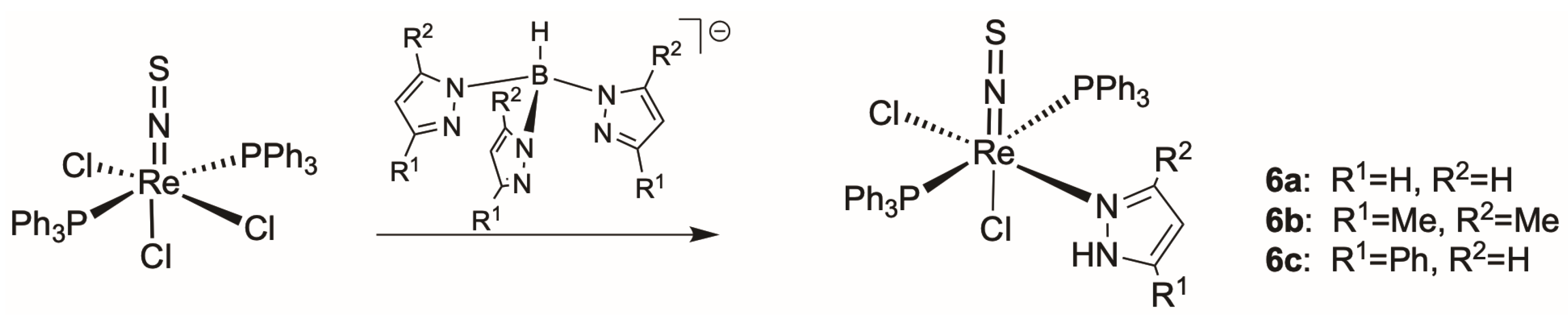
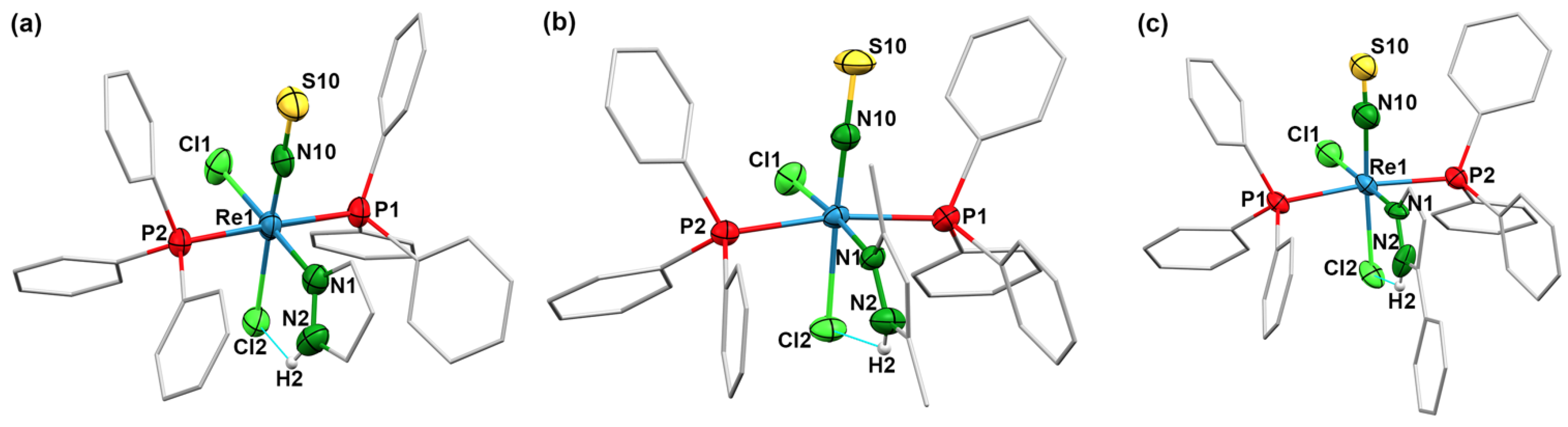
2.3. Thionitrosyl Complexes of Rhenium
3. Materials and Methods
3.1. Radiation Precaution
3.2. Syntheses
3.3. Spectroscopic and Analytical Methods
3.4. X-ray Crystallography
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trofimenko, S. Boron-Pyrazole Chemistry. J. Am. Chem. Soc. 1966, 88, 1842–1844. [Google Scholar] [CrossRef]
- Trofimenko, S. Scorpionates: The Coordination Chemistry of Polypyrazolyl-Borates Ligands; Imperial College Press: London, UK, 1999; ISBN 978-1-86094-1726. [Google Scholar] [CrossRef]
- Pettinari, C. Scorpionates II: Chelating Borate Ligands; Imperial College Press: London, UK, 2008; ISBN 978-1-86094-877-0. [Google Scholar] [CrossRef]
- Pettinari, C.; Pettinari, R.; Marchetti, F. Golden jubilee for scorpionates: Recent advances in organometallic chemistry and their role in catalysis. Adv. Organomet. Chem. 2016, 65, 175–260. [Google Scholar]
- Paulo, A.; Correia, J.D.G.; Campello, M.P.C.; Santos, I. A short ride on scorpionates: From d- to f-elements. Polyhedron 2004, 23, 331–360. [Google Scholar] [CrossRef]
- Spicer, M.D.; Reglinski, J. Soft scorpionate ligands based on imidazole-2-thione donors. Eur. J. Inorg. Chem. 2009, 40, 1553–1574. [Google Scholar] [CrossRef]
- Steel, G.; Rajasekharan-Nair, R.; Stepek, I.A.; Kennedy, A.R.; Reglinski, J.; Spicer, M.D. Observations on the steric impact of N- and S-donor scorpionate ligands. Eur. J. Inorg. Chem. 2016, 2016, 2409–2412. [Google Scholar] [CrossRef]
- Lail, M.; Pittard, K.A.; Gunnoe, T.B. Chemistry surrounding group 7 complexes that possess poly(pyrazolyl)borate ligands. Adv. Organomet. Chem. 2008, 56, 95–153. [Google Scholar]
- Thomas, J.A.; Davison, A. High oxidation state technetium and rhenium complexes of hydrotris(1-pyrazolyl)borate. Inorg. Chim. Acta 1991, 190, 231–235. [Google Scholar] [CrossRef]
- Abrams, M.J.; Davison, A. The Synthesis and Characterization of Hydrotris(1-pyrazolyl)borate Complexes of Technetium(III) and Rhenium(V) and (III). Inorg. Chim. Acta 1984, 82, 125–128. [Google Scholar] [CrossRef]
- Alberto, R.; Herrmann, W.A.; Kiprof, P.; Baumgärtner, F. Multiple bonds between main group elements and transition metals. Synthesis and reactivity of TcCl(CO)3[P(C6H5)3]2: Novel technetium complexes of 1,4,7-triazacyclononane and hydridotris(pyrazolyl)borate. Inorg. Chem. 1992, 31, 895–899. [Google Scholar] [CrossRef]
- Joachim, J.E.; Apostolidis, C.; Kanellakopoulos, B.; Maier, R.; Marques, N.; Meyer, D.; de Matos, A.P.; Nuber, B.; Rebizant, J.; Ziegler, M.L. Metallorganische Chemie des Technetiums. VIII. Technetium(I)-carbonyl-Komplexe mit Polypyrazol-1-yl-borato-Liganden im Vergleich mit seinen Mn- und Re-Homologen. J. Organomet. Chem. 1993, 448, 119–129. [Google Scholar] [CrossRef]
- Joachim, J.E.; Apostolidis, C.; Kanellakopoulos, B.; Meyer, D.; Nuber, B.; Raptis, K.; Rebizant, J.; Ziegler, M.L. Metallorganische Chemie des Technetiums XII. Photolytische CO-substitutionsreaktionen von Technetiumtricarbonylverbindungen. Synthesen und Röntgenstrukturanalysen von (C5H5)Tc(CO)3(PPh3), (C5Me5)Tc(CO)2(PPh3) und [HB(3,5-Me2Pz)3Tc(CO)2[P(OMe)3] im Vergleich mit seinen Homologen. J. Organomet. Chem. 1995, 492, 199–210. [Google Scholar]
- Domingos, A.; Marcalo, J.; Paulo, A.; de Matos, A.P.; Santos, I. Synthesis and characterization of rhenium complexes with the stabilizing ligand tetrakis(pyrazol-1-yl)borate. Inorg. Chem. 1993, 32, 5114–5118. [Google Scholar] [CrossRef]
- You, F.; Friedman, L.A.; Bassett, K.C.; Lin, Y.; Sabat, M.; Harman, D. Rhenium(I) η2-Coordinated Furan Complexes: Converting Furan into a 1,3-Carbon Dipole. Organometallics 2005, 24, 2903–2912. [Google Scholar] [CrossRef]
- Meiere, S.H.; Brooks, B.C.; Gunnoe, T.B.; Carrig, E.H.; Sabat, M.; Harman, W.D. Dihapto Coordination of Aromatic Molecules by the Asymmetric π-Bases {TpRe(CO)(L)} (Tp = hydridotris(pyrazolyl)borate; L = tBuNC, PMe3, pyridine, 1-methylimidazole, or NH3). Organometallics 2001, 20, 3661–3671. [Google Scholar] [CrossRef]
- McNeil, W.S.; DuMez, D.D.; Matano, Y.; Lovell, S.; Mayer, J.M. Synthesis and Reactivity of Aryl- and Alkyl-Rhenium(V) Imido−Triflate Compounds: An Unusual Mechanism for Triflate Substitution. Organometallics 1999, 18, 3715–3727. [Google Scholar] [CrossRef]
- Faller, J.W.; Lavoie, A.R. Diastereoselective Synthesis and Electronic Asymmetry of Chiral Nonracemic Rhenium(V) Oxo Complexes Containing the Hydrotris(1-pyrazolyl)borate Ligand. Organometallics 2000, 19, 3957–3962. [Google Scholar] [CrossRef]
- Maria, L.; Paulo, A.; Santos, I.C.; Santos, I.; Kurz, P.; Spingler, B.; Alberto, R. Very Small and Soft Scorpionates: Water Stable Technetium Tricarbonyl Complexes Combining a Bis-agostic (κ3-H, H, S) Binding Motif with Pendant and Integrated Bioactive Molecules. J. Am. Chem. Soc. 2006, 128, 14590–14598. [Google Scholar] [CrossRef] [PubMed]
- Garcia, R.; Paulo, A.; Santos, I. Rhenium and technetium complexes with anionic or neutral scorpionates: An overview of their relevance in biomedical applications. Inorg. Chim. Acta 2009, 362, 4315–4327. [Google Scholar] [CrossRef]
- Martini, P.; Pasquali, M.; Boschi, A.; Uccelli, L.; Giganti, M.; Duatti, A. Technetium Complexes and Radiopharmaceuticals with Scorpionate Ligands. Molecules 2018, 23, 2039. [Google Scholar] [CrossRef]
- Sidorenko, G.V.; Miroslavov, A.E.; Tyupina, M.Y. Technetium(I) carbonyl complexes for nuclear medicine: Coordination-chemical aspect. Coord. Chem. Rev. 2023, 476, 214911. [Google Scholar] [CrossRef]
- Armstrong, A.; Taube, H. Chemistry of trans-Aquonitrosyltetramminetechnetium(I) and Related Studies. Inorg. Chem. 1976, 15, 1904–1909. [Google Scholar] [CrossRef]
- Yang, G.A.; Heitzmann, W.; Ford, L.A.; Benson, W.R. ESR Studies of Technetium Complexes: Trans-Aquonitrosyltetramminetechnetium(II) and Nitrosylpentachlorotechnetium(II). Inorg. Chem. 1982, 21, 3242–3243. [Google Scholar] [CrossRef]
- Nicholson, T.; Chun, E.; Mahmood, A.; Mueller, P.; Davison, A.; Jones, A.G. Synthesis, spectroscopy and structural analysis of Technetium and Rhenium nitrosyl complexes. Commun. Inorg. Chem. 2015, 3, 31–39. [Google Scholar]
- Blanchard, S.S.; Nicholson, T.; Davison, A.; Davis, W.; Jones, A.G. The synthesis, characterization and substitution reactions of the mixed technetium(I) nitrosyl complex trans-trans-[(NO)(NCCH3)Cl2(PPh3)2Tc]. Inorg. Chim. Acta 1996, 244, 121–130. [Google Scholar] [CrossRef]
- Balasekaran, S.M.; Hagenbach, A.; Drees, M.; Abram, U. [TcII(NO)(trifluoroacetate)4F]2−—Synthesis and reactions. Dalton Trans. 2017, 46, 13544–13552. [Google Scholar] [CrossRef] [PubMed]
- Linder, K.E.; Davison, A.; Dewan, J.C.; Costello, C.E.; Melaknia, S. Nitrosyl complexes of technetium: Synthesis and characterization of [TcI(NO)(CNCMe3)5](PF6)2 and Tc(NO)Br2(CNCMe3)3 and the crystal structure of Tc(NO)Br2(CNCMe3)3. Inorg. Chem. 1986, 25, 2085–2089. [Google Scholar] [CrossRef]
- Ackermann, J.; Noufele, C.N.; Hagenbach, A.; Abram, U. Nitrosyltechnetium(I) Complexes with 2-(Diphenylphosphanyl)aniline. Z. Anorg. Allg. Chem. 2019, 645, 8–13. [Google Scholar] [CrossRef]
- Ackermann, J.; Hagenbach, A.; Abram, U. {Tc(NO)(Cp)(PPh3)}+—A novel technetium(I) core. Chem. Commun. 2016, 52, 10285–10288. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, J.; Abdulkader, A.; Scholtysik, C.; Jungfer, M.R.; Hagenbach, A.; Abram, U. [TcI(NO)X(Cp)(PPh3)] Complexes (X− = I−, I3−, SCN−, CF3SO3−, or CF3COO−) and Their Reactions. Organometallics 2019, 38, 4471–4478. [Google Scholar] [CrossRef]
- Abdulkader, A.; Hagenbach, A.; Abram, U. [Tc(NO)Cl(Cp)(PPh3)]—A Technetium(I) Compound with an Unexpected Synthetic Potential. Eur. J. Inorg. Chem. 2021, 2021, 3812–3818. [Google Scholar] [CrossRef]
- Schibli, R.; Marti, N.; Maurer, P.; Spingler, B.; Lehaire, M.-L.; Gramlich, V.; Barnes, C.L. Syntheses and Characterization of Dicarbonyl−Nitrosyl Complexes of Technetium(I) and Rhenium(I) in Aqueous Media: Spectroscopic, Structural, and DFT Analyses. Inorg. Chem. 2005, 44, 683–690. [Google Scholar] [CrossRef]
- Brown, D.S.; Newman, J.L.; Thornback, J.R.; Davison, A. Structure of the tetra-n-butylammonium salt of tetrachloro(methanol)nitrosyltechnetium(II) anion. Acta Cryst. 1987, C43, 1692–1694. [Google Scholar] [CrossRef]
- Brown, D.S.; Newman, J.L.; Thornback, J.R.; Pearlstein, R.M.; Davison, A.; Lawson, A. The synthesis and characterisation of the trichloronitrosyl(acetylacetonato)technetium(II) anion, a novel technetium(II) complex. Inorg. Chim. Acta 1988, 150, 193–196. [Google Scholar] [CrossRef]
- Nicholson, T.; Hirsch-Kuchma, M.; Freiberg, E.; Davison, A.; Jones, A.G. The reaction chemistry of a technetium(I) nitrosyl complex with potentially chelating organohydrazines: The X-ray crystal structure of [TcCl2(NO)(HNNC5H4N)(PPh3)]. Inorg. Chim. Acta 1998, 279, 206–209. [Google Scholar] [CrossRef]
- De Vries, N.; Cook, J.; Davison, A.; Nicholson, T.; Jones, A.G. Synthesis and characterization of a technetium(III) nitrosyl compound: Tc(NO)(Cl)(SC10H13)3. Inorg. Chem. 1990, 29, 1062–1064. [Google Scholar] [CrossRef]
- Nicholson, T.; Mahmood, A.; Limpa-Amara, N.; Salvarese, N.; Takase, M.K.; Müller, P.; Akgun, Z.; Jones, A.G. Reactions of the tridentate and tetradentate amine ligands di-(2-picolyl)(N-ethyl)amine and 2,5-bis-(2-pyridylmethyl)-2,5 diazohexane with technetium nitrosyl complexes. Inorg. Chim. Acta 2011, 373, 301–305. [Google Scholar] [CrossRef][Green Version]
- Roca Jungfer, M.; Ernst, M.J.; Hagenbach, A.; Abram, U. [{TcI(NO)(LOMe)(PPh3)Cl}2Ag](PF6) and [TcII(NO)(LOMe)(PPh3)Cl](PF6): Two Unusual Technetium Complexes with a “Kläui-type” Ligand. Z. Anorg. Allg. Chem. 2022, 648, e202100316. [Google Scholar] [CrossRef]
- Nicholson, T.L.; Mahmood, A.; Muller, P.; Davison, A.; Storm-Blanchard, S.; Jones, A.G. The synthesis and structural characterization of the technetium nitrosyl complexes [TcCl(NO)(SC5H4N)(PPh3)2] and [Tc(NO)(SC5H4N)2(PPh3)]. Inorg. Chim. Acta 2011, 365, 484–486. [Google Scholar] [CrossRef][Green Version]
- Balasekaran, S.M.; Spandl, J.; Hagenbach, A.; Köhler, K.; Drees, M.; Abram, U. Fluoridonitrosyl Complexes of Technetium(I) and Technetium(II). Synthesis, Characterization, Reactions, and DFT Calculations. Inorg. Chem. 2014, 53, 5117–5128. [Google Scholar] [CrossRef]
- Nicholson, T.; Hirsch-Kuchma, M.; Shellenbarger-Jones, A.; Davison, A.; Jones, A.G. The synthesis and characterization of a technetium nitrosyl complex with cis-{2-pyridyl,diphenylphosphine} coligands. The X-ray crystal structure of [TcCl2(NO)(pyPPh2-P,N) (pyPPh2-P)]. Inorg. Chim. Acta 1998, 267, 319–322. [Google Scholar] [CrossRef]
- Grunwald, A.C.; Scholtysik, C.; Hagenbach, A.; Abram, U. One Ligand, One Metal, Seven Oxidation States: Stable Technetium Complexes with the “Kläui Ligand”. Inorg. Chem. 2020, 59, 9396–9405. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, T.L.; Mahmood, A.; Refosco, F.; Tisato, F.; Müller, P.; Jones, A.G. The synthesis and X-ray structural characterization of mer and fac isomers of the technetium(I) nitrosyl complex [TcCl2(NO)(PNPpr)]. Inorg. Chim. Acta 2009, 362, 3637–3640. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nicholson, T.; Müller, P.; Davison, A.; Jones, A.G. The synthesis and characterization of a cationic technetium nitrosyl complex: The X-ray crystal structure of [TcCl(NO)(DPPE)2](PF6) · CH2Cl2. Inorg. Chim. Acta 2006, 359, 1296–1298. [Google Scholar] [CrossRef]
- Machura, B. Structural and spectroscopic properties of rhenium nitrosyl complexes. Coord. Chem. Rev. 2005, 249, 2277–2307. [Google Scholar] [CrossRef]
- Dilworth, J.R. Rhenium chemistry—Then and Now. Coord. Chem. Rev. 2021, 436, 213822. [Google Scholar] [CrossRef]
- Mahmood, A.; Akgun, Z.; Peng, Y.; Mueller, P.; Jiang, Y.; Berke, H.; Jones, A.G.; Nicholson, T. The synthesis and characterization of rhenium nitrosyl complexes. The X-ray crystal structures of [ReBr2(NO)(NCMe)3], [Re(NO)(N5)](BPh4)2] and [ReBr2(NO)(NCMe){py-CH2-NHCH2CH2-N(CH2-py)2}]. Inorg. Chim. Acta 2013, 405, 455–460. [Google Scholar] [CrossRef]
- Abram, U.; Ortner, K.; Hübener, R.; Voigt, A.; Caballho, R.; Vazquez-Lopez, E. Darstellung, Strukturen und EPR-Spektren der Rhenium(II)-Nitrosylkomplexe [Re(NO)Cl2(PPh3)(OPPh3)(OReO3)], [Re(NO)Cl2(OPPh3)2(OReO3)] und [Re(NO)Cl2(PPh3)3] [ReO4]. Z. Anorg. Allg. Chem. 1998, 624, 1662–1668. [Google Scholar] [CrossRef]
- Agbossou, F.; O’Connor, E.J.; Garner, C.M.; Quiros Mendez, N.; Fernandez, J.M.; Patton, A.T.; Ramsden, J.A.; Gladysz, J.A. Cyclopentadienyl Rhenium Complexes. Inorg. Synth. 1992, 29, 211–225. [Google Scholar]
- Seidel, S.N.; Prommesberger, M.; Eichenseher, S.; Meyer, O.; Hampel, F.; Gladysz, J.A. Syntheses and structural analyses of chiral rhenium containing amines of the formula (η5-C5H5)Re(NO)(PPh3)((CH2)nNRR′) (n = 0, 1). Inorg. Chim. Acta 2010, 363, 533–548. [Google Scholar] [CrossRef]
- Bernasconi, C.F.; Bhattacharya, S.; Wenzel, P.J.; Olmstead, M.M. Kinetic and Thermodynamic Acidity of [Cp(NO)(PPh3)Re(2,5-dimethyl-3-thienyl)carbene]+. Transition State Imbalance and Intrinsic Barriers. Organometallics 2006, 25, 4322–4330. [Google Scholar] [CrossRef]
- Dilsky, S.; Schenk, W.A. Diastereomeric Halfsandwich Rhenium Complexes Containing Hemilabile Phosphane Ligands. Eur. J. Inorg. Chem. 2004, 2004, 4859–4870. [Google Scholar] [CrossRef]
- Pandey, K.K. Coordination Chemistry of Thionitrosyl (NS), Thiazate (NSO−), Disulfidothionitrate (S,N−), Sulfur Monoxide (SO), and Disulfur Monoxide (S,O) Ligands. Progr. Inorg. Chem. 1992, 40, 445–502. [Google Scholar]
- Døsing, A. The electronic structure and photochemistry of transition metal thionitrosyl complexes. Coord. Chem. Rev. 2016, 306, 544–557. [Google Scholar] [CrossRef]
- Anhaus, J.; Siddiqi, Z.A.; Roesky, H.W. Reaction of Tetrasulfurtetranitride with Rhenium(VII)-chloronitride. The Crystal Structure of [Ph4As+]2[Cl4Re(NS)(NSCl)2−] · CH2Cl2. Z. Naturforsch. 1985, 40b, 740–744. [Google Scholar] [CrossRef]
- Dirican, D.; Pfister, N.; Wozniak, M.; Braun, T. Reactivity of Binary and Ternary Sulfur Halides towards Transition-Metal Compounds. Chem. Eur. J. 2020, 31, 6945–6963. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Neumüller, B.; Dehnicke, K. (PPh4)2[(SN)ReCl3(μ-N)(μ-NSN)ReCl3(THF)]—Ein Nitrido-Thionitrosyl-Dinitridosulfato-Komplex des Rheniums. Z. Anorg. Allg. Chem. 2000, 626, 1268–1270. [Google Scholar] [CrossRef]
- Reinel, M.; Höcher, T.; Abram, U.; Kirmse, R. Ein Beitrag zu Rhenium(II)-, Osmium(II)- und Technetium(II)-Thionitrosylkomplexe vom Typ [M(NS)Cl4py] : Darstellung, Strukturen und EPR-Spektren. Z. Anorg. Allg. Chem. 2003, 629, 853–861. [Google Scholar] [CrossRef]
- Voigt, A.; Abram, U.; Kirmse, R. Darstellung, Strukturen und EPR-Spektren der Rhenium(II)-Thionitrosylkomplexe trans-[Re(NS)Cl3(MePh2P)2] und trans-[Re(NS)Br3(Me2PhP)2]. Z. Anorg. Allg. Chem. 1999, 625, 1658–1663. [Google Scholar] [CrossRef]
- Hauck, H.-G.; Willing, W.; Müller, U.; Dehnicke, K. [ReCl2(NS)(NSCl)(Pyridin)2], ein Thionitrosyl-chlorthionitrenkomplex des Rheniums. Z. Anorg. Allg. Chem. 1986, 534, 77–84. [Google Scholar] [CrossRef]
- Hübener, R.; Abram, U.; Strähle, J. Isothiocyanato complexes of rhenium II. Synthesis, characterization and structures of ReN(NCS)2(Me2PhP)3 and Re(NS)(NCS)2(Me2PhP)3. Inorg. Chim. Acta 1994, 216, 223–228. [Google Scholar] [CrossRef]
- Ritter, S.; Abram, U. Gemischtligand-Komplexe des Rheniums. VI. Darstellung und Strukturen der Rhenium Thionitrosyl-Komplexe mer-[Re(NS)Cl2(Me2PhP)3] · CH2Cl2 und trans-[Re(NS)Cl3(Me2PhP)2]. Z. Anorg. Allg. Chem. 1994, 620, 1223–1228. [Google Scholar]
- Ritter, S.; Abram, U. Gemischtligandkomplexe des Rheniums. IX. Reaktionen am Nitridoliganden von [ReN(Me2PhP)(Et2dtc)2]. Synthese, Charakterisierung und Kristallstrukturen von [Re(NBCl3)(Me2PhP)(Et2dtc)2], [Re(NGaCl3)(Me2PhP)(Et2dtc)2] und [Re(NS)Cl(Me2PhP)2(Et2dtc)]. Z. Anorg. Allg. Chem. 1995, 622, 965–973. [Google Scholar] [CrossRef]
- Ruf, C.; Behrens, U.; Lork, E.; Mews, R. Reactions of halides with trans-[Re(CO)4(MeCN)(NS)][AsF6]2: Syntheses and structure of trans-[Re(CO)4(Cl)(NS)][AsF6] and [(OC)5ReNS–NS–N[Re(CO)5]S{N–SNRe(CO)5}–CH2–CH2][AsF6]2, an unusual trinuclear bis(thiazyl)rhenium complex. Chem. Commun. 1996, 939–940. [Google Scholar] [CrossRef]
- Baldas, J.; Bonnyman, J.; Mackay, M.F.; Williams, G.A. Structural studies of technetium complexes. V. The preparation and crystal structure of Dichlorobis(diethyldithiocarbamato)thionitrosyltechnetium(III). Austr. J. Chem. 1984, 37, 751–759. [Google Scholar] [CrossRef]
- Kaden, L.; Lorenz, B.; Kirmse, R.; Stach, J.; Behm, H.; Beurskens, P.T.; Abram, U. Synthesis, characterization and x-ray molecular and crystal structure of Tc(NS)Cl3(Me2PhP)(Me2PhPO)-a first example of mixed phosphine/phosphine oxide coordination. Inorg. Chim. Acta 1990, 169, 43–48. [Google Scholar] [CrossRef]
- Baldas, J.; Colmanet, S.F.; Williams, G.A. Preparation and Structure of Dibromobis(N,N-diethyldithiocarbamato)-thionitrosyltechnetium(III). Austr. J. Chem. 1991, 44, 1125–1132. [Google Scholar] [CrossRef]
- Lu, J.; Clarke, M.J. Modulation of Tc–NX (X = O or S) bonds by π-acceptor ligands. J. Chem. Soc. Dalton Trans. 1992, 1243–1248. [Google Scholar] [CrossRef]
- Abram, U.; Schulz Lang, E.; Abram, S.; Wegmann, J.; Dilworth, J.R.; Kirmse, R.; Woolins, J.D. Technetium(V) and rhenium(V) nitrido complexes with bis(diphenyl-thiophosphoryl)amide, N(SPPh2)2−. J. Chem. Soc. Dalton Trans. 1997, 623–630. [Google Scholar] [CrossRef]
- Hiller, W.; Hübener, R.; Lorenz, B.; Kaden, L.; Findeisen, M.; Stach, J.; Abram, U. Structural and spectroscopic studies on mer-dichlorotris(dimethylphenylphosphine)(thionitrosyl)technetium(I), mer-[Tc(NS)Cl2(Me2PhP)3]. Inorg. Chim. Acta 1991, 181, 161–165. [Google Scholar] [CrossRef]
- Lu, J.; Clarke, M.J. Sulfur atom transfer with reduction of a [TcVI≡N]3+ core to a [TcI-N≡S]2+ core. Crystal structure of mer-dichlorotris(4-picoline)(thionitrosyl)technetium. Inorg. Chem. 1990, 29, 4123–4125. [Google Scholar] [CrossRef]
- Abram, U.; Hübener, R.; Wollert, R.; Kirmse, R.; Hiller, W. Synthesis, characterization and reactions of [Tc(NS)X4]− complexes (X = Cl, Br, NCS). Inorg. Chim. Acta 1993, 206, 9–14. [Google Scholar] [CrossRef]
- Dressler, K. Ultraviolett- und Schumannspektren der neutralen und ionisierten Moleküle PO, PS, NS, P2. Helv. Phys. Acta 1955, 28, 563–590. [Google Scholar]
- O’Hare, P.A.G. Dissociation Energies, Enthalpies of Formation, Ionization Potentials, and Dipole Moments of NS and NS+. J. Chem. Phys. 1970, 52, 2992–2996. [Google Scholar] [CrossRef]
- Mews, R. The Thionitrosyl Cation NS+ as a Synthetic Reagent. Angew. Chem. Int. Ed. Engl. 1976, 15, 691–692. [Google Scholar] [CrossRef]
- Clegg, W.; Glemser, O.; Harms, K.; Hartmann, G.; Mews, R.; Noltemeyer, M.; Sheldrick, G.M. Crystal structures of thionitrosyl hexafluoroantimonate(V) and thionitrosyl undecafluorodiantimonate(V) at 293 K and of thionitrosyl undecafluorodiantimonate(V) at 121.5 K: The effect of thermal motion on the apparent NS bond length. Acta Cryst. 1981, 37b, 548–552. [Google Scholar] [CrossRef]
- Nowak, D.; Sawallisch, T.; Hagenbach, A.; Abram, U. Thionitrosyl Complexes of Rhenium and Technetium with PPh3 and Chelating Ligands—Synthesis and Reactivity. Inorganics 2024, 12, 210. [Google Scholar] [CrossRef]
- Ernst, M.J.; Abdulkader, A.; Hagenbach, A.; Claude, G.; Roca Jungfer, M.; Abram, U. [Tc(NO)(Cp)(PPh3)Cl] and [Tc(NO)(Cp)(PPh3)(NCCH3)](PF6), and Their Reactions with Pyridine and Chalcogen Donors. Molecules 2024, 29, 1114. [Google Scholar] [CrossRef] [PubMed]
- Wendlandt, D.; Bauche, J.; Luc, P. Hyperfine structure in Tc I: Experiment and theory. J. Phys. B At. Mol. Phys. 1977, 10, 1989–2002. [Google Scholar] [CrossRef]
- Mikhalev, V.A. 99Tc NMR Spectroscopy. Radiochemistry 2005, 47, 319–333. [Google Scholar] [CrossRef]
- Abram, U.; Lorenz, L.; Kaden, L.; Scheller, D. Nitrido Complexes of Technetium with Tertiary Phosphines and Arsines. Polyhedron 1988, 7, 285–289. [Google Scholar] [CrossRef]
- O’Connell, L.A.; Pearlstein, R.M.; Davison, A.; Thornback, J.R.; Kronauge, J.F.; Jones, A.G. Technetium-99 NMR spectroscopy: Chemical shift trends and long range coupling effects. Inorg. Chim. Acta 1989, 161, 39–43. [Google Scholar] [CrossRef]
- Roca Jungfer, M.; Elsholz, L.; Abram, U. Technetium Hydrides Revisited: Syntheses, Structures, and Reactions of [TcH3(PPh3)4] and [TcH(CO)3(PPh3)2]. Organometallics 2021, 40, 3095–3112. [Google Scholar] [CrossRef]
- Trofimenko, S. Recent Advances in Poly(pyrazolyl)borate (Scorpionate) Chemistry. Chem. Rev. 1993, 93, 943–980. [Google Scholar] [CrossRef]
- Khan, M.M.T.; Roy, P.S.; Venkatasubramanian, K.; Khan, N.H. An unexpected cleavage of the boron-nitrogen bond in the coordinated dihydrobis(1-pyrazolyl)borate: Synthesis and crystal structure of [RuII(pz)2(pzH)3(DMSO)] and synthesis of [RuIICl2(pzH)2(DMSO)2]. Inorg. Chim. Acta 1990, 176, 49–55. [Google Scholar] [CrossRef]
- Kharbani, D.; Deb, D.K.; Mawnai, I.L.; Kurbah, S.D.; Sarkar, B.; Rymmai, E.K. Pyrazole cleavage of tris(3,5-dimethylpyrazolyl)borate with Ruthenium(II) complexes: Synthesis, structural characterization and DFT studies. J. Mol. Struct. 2017, 1133, 264–270. [Google Scholar] [CrossRef]
- Zhao, Q.; Dou, T.; Guan, X.-T.; Xu, T.; Chen, M.-J.; Zhang, J.; Chen, X. B–N Cleavage in (9-BBN)bis(pyrazolyl)borate NiII Complexes. Eur. J. Inorg. Chem. 2019, 2019, 3724–3730. [Google Scholar] [CrossRef]
- Kückmann, T.I.; Abram, U. Oxorhenium(V) and Tricarbonylrhenium(I) Complexes with Substituted Pyrazoles as Products of the Degradation of Hydrotrispyrazolylborates. Z. Anorg. Allg. Chem. 2004, 630, 783–785. [Google Scholar] [CrossRef]
- Baldas, J. The chemistry of technetium nitrido complexes. In Technetium and Rhenium Their Chemistry and Its Applications; Yoshihara, K., Omori, T., Eds.; Topics in Current Chemistry; Springer: Berlin/Heidelberg, Germany, 1996; pp. 37–76. [Google Scholar]
- Bolzati, C.; Dolmella, A. Nitrido Technetium-99m Core in Radiopharmaceutical Applications: Four Decades of Research. Inorganics 2020, 8, 3. [Google Scholar] [CrossRef]
- Weil, J.A.; Bolton, J.R.; Wertz, J.E. Electron Paramagnetic Resonance—Elementary Theory and Practical Applications; John Wiley & Sons: New York, NY, USA, 1994; Appendix G; p. 533. [Google Scholar]
- Abram, U.; Kirmse, R.; Köhler, K.; Lorenz, B.; Kaden, L. Tc(NX)Y3(Me2PhP)2 Complexes (X = O or S.; Y = Cl or Br). Preparation, Characterization and EPR Studies. Inorg. Chim. Acta 1987, 129, 15–20. [Google Scholar] [CrossRef]
- Claude, G.; Weh, D.; Hagenbach, A.; Figueroa, J.S.; Abram, U. Rhenium Complexes with p-Fluorophenylisocyanide. Z. Anorg. Allg. Chem. 2023, 649, e202200320. [Google Scholar] [CrossRef]
- Claude, G.; Genz, J.; Weh, D.; Roca Jungfer, M.; Hagenbach, A.; Gembicky, M.; Figueroa, J.S.; Abram, U. Mixed-Isocyanide Complexes of Technetium under Steric and Electronic Control. Inorg. Chem. 2022, 61, 16163–16176. [Google Scholar] [CrossRef] [PubMed]
- Coppens, P. The Evaluation of Absorption and Extinction in Single-Crystal Structure Analysis. In Crystallographic Computing; Muksgaard: Copenhagen, Denmark, 1979. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Cryst. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Cryst. 2020, 53, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Trofimenko, S. Boron-Pyrazole Chemistry. II. Poly(1-pyrazolyl borates). J. Am. Chem. Soc. 1967, 89, 3170–3177. [Google Scholar] [CrossRef]
- Thomas, S.; Young, C.G.; Broce, B.; Templeton, J.L. Potassium Tris(3,5-dimethylpyrazolyl)hydroborate, KTp*. Inorg. Synth. 2002, 33, 219–221. [Google Scholar]

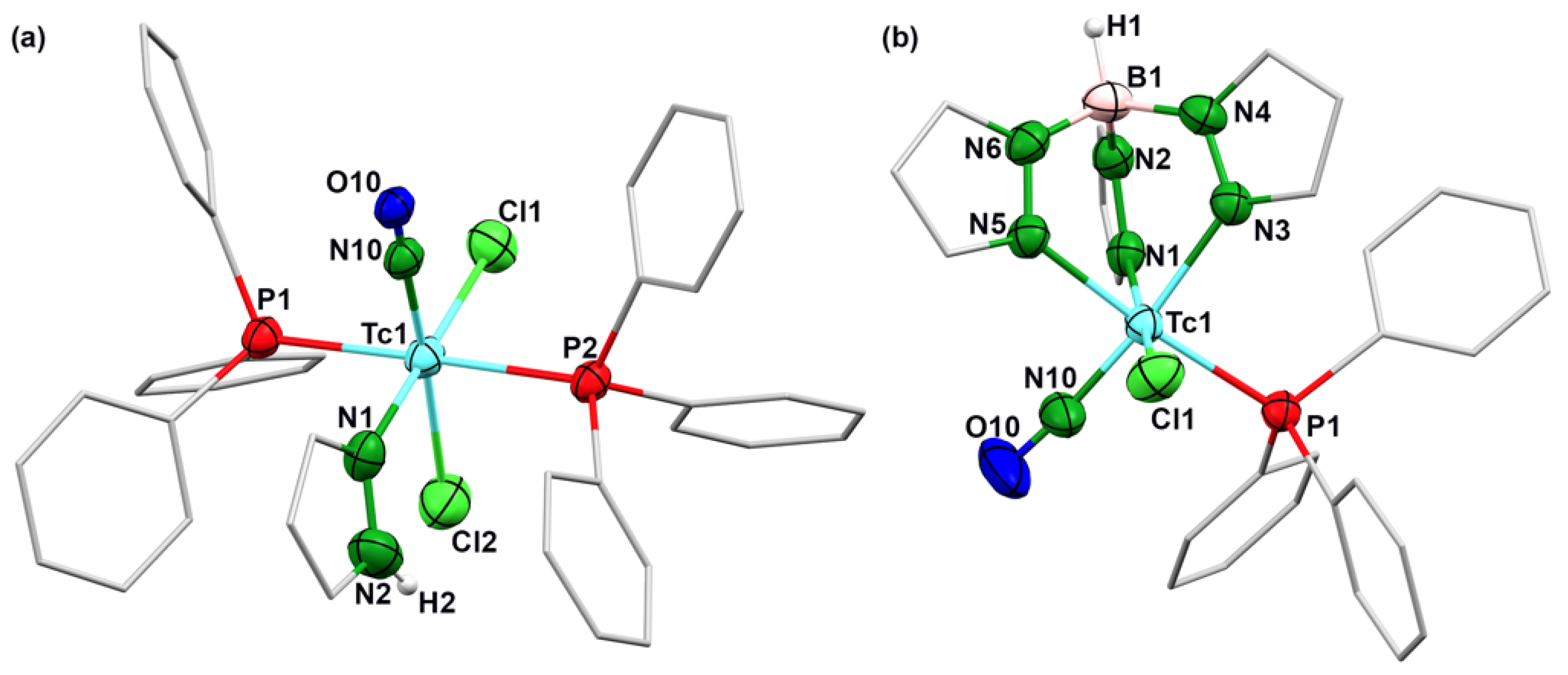


| Tc1–N10 | N10–O10 | Tc1–Cl1 | Tc1–Cl2 | Tc1–P1 | Tc1–P2 | Tc1–N1 | Tc1–N3 | Tc1–N5 | Tc1–N10–O10 | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.816(4) | 0.946(4) | 2.447(1) | 2.443(2) | 2.474(2) | 2.473(1) | 2.4149(4) | - | - | 178.4(5) |
| 2 | 1.764(7) | 1.13(1) | 2.425(2) | - | 2.4417(5) | - | 2.152(2) | 2.172(1) | 2.135(2) | 174.3(5) |
| g0 | a0Tc | g‖ | g⊥ | A‖Tc | A⊥Tc | A‖P | Reference | |
|---|---|---|---|---|---|---|---|---|
| [Tc(NS)Cl3(PPh3)2] (3) | 2.011 | 164 | 1.955 | 2.0455 | 270 | 128 | - | This work |
| [Tc(NS)Cl3(PPh3)(OPPh3)] (4) | 2.009 | 166 | 1.978 | 1.999 | 290 | 134 | - | This work |
| [Tc(NS)Cl3(PMe2Ph)2] | 2.045 | 133 | 2.027 | 2.038 | 219 | 101 | 19 (a) | [70] |
| [Tc(NS)Cl3(PMe2Ph)(OPMe2Ph)] | 2.032 | 149 | 2.027 | 2.039 | 237 | 106 | 24 (b) | [67] |
| [Tc(NO)Cl3(PMe2Ph)2] | 2.045 | 125 | 2.034 | 2.053 | 215 | 88 | 19 (a) | [93] |
| Tc1–N10 | N10–S10 | Tc1–Cl1 | Tc1–Cl2 | Tc1–Cl3 | Tc1–P1/P2 | Tc1–O1 | P1–O1 | Tc1–O1–P1 | Tc1–N10–S10 | |
|---|---|---|---|---|---|---|---|---|---|---|
| 3 | 1.78(1) | 1.51(1) | 2.443(3) | 2.357(2) | - | 2.570(2) | - | - | - | 180 |
| 4 | 1.68(2) | 1.55(2) | 2.346(6) | 2.368(6) | 2.356(6) | 2.552(7) | 2.09(1) | 1.50(1) | 154.4(9) | 174(2) |
| Tc1–N10 | N10–S10 | Tc1–Cl1 | Tc1–N1 | Tc1–N3 | Tc1–N5 | Tc1–N7 | Tc1–N10–S10 | N10–Tc1–Cl1 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1.733(2) | 1.572(2) | 2.4252(6) | 2.134(2) | 2.135(2) | 2.147(2) | 2.137(2) | 179.4(2) | 179.66(8) | |||||||
| Hydrogen Bonds | |||||||||||||||
| H10–Cl1 | H12–Cl1 | H14–Cl1 | H16–Cl1 | H2–Cl1 | H4–Cl1 | H6–Cl1 | H8–Cl1 | ||||||||
| 2.851 | 2.687 | 2.851 | 2.904 | 2.867 | 2.864 | 2.868 | 2.825 | ||||||||
| H10–Cl2 | H12–Cl2 | H14–Cl2 | H16–Cl2 | ||||||||||||
| 2.507 | 2.678 | 2.478 | 2.439 | ||||||||||||
| Re–N10 | N10–S10 | Re1–Cl1 | Re1–Cl2 | Re1–N1 | Cl2…H2 | Re1–N10–S10 | N10–Re1–Cl2 | N2–H2…Cl2 | |
|---|---|---|---|---|---|---|---|---|---|
| 6a | 1.738(8) | 1.558(7) | 2.438(2) | 2.488(4) | 2.142(5) | 2.536 | 178.3(3) | 174.8(2) | 118.9 |
| 6b | 1.737(5) | 1.578(5) | 2.445(2) | 2.468(1) | 2.158(4) | 2.396 | 173.0(3) | 176.6(2) | 125.5 |
| 6c | 1.98(2) | 1.36(2) | 2.441(8) | 2.449(4) | 2.18(2) | 2.501 | 175(2) | 171(1) | 124.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sawallisch, T.E.; Abdulkader, A.; Nowak, D.; Hagenbach, A.; Abram, U. Nitrosyl and Thionitrosyl Complexes of Technetium and Rhenium and Their Reactions with Hydrotris(pyrazolyl)borates. Molecules 2024, 29, 3865. https://doi.org/10.3390/molecules29163865
Sawallisch TE, Abdulkader A, Nowak D, Hagenbach A, Abram U. Nitrosyl and Thionitrosyl Complexes of Technetium and Rhenium and Their Reactions with Hydrotris(pyrazolyl)borates. Molecules. 2024; 29(16):3865. https://doi.org/10.3390/molecules29163865
Chicago/Turabian StyleSawallisch, Till Erik, Abdullah Abdulkader, Domenik Nowak, Adelheid Hagenbach, and Ulrich Abram. 2024. "Nitrosyl and Thionitrosyl Complexes of Technetium and Rhenium and Their Reactions with Hydrotris(pyrazolyl)borates" Molecules 29, no. 16: 3865. https://doi.org/10.3390/molecules29163865
APA StyleSawallisch, T. E., Abdulkader, A., Nowak, D., Hagenbach, A., & Abram, U. (2024). Nitrosyl and Thionitrosyl Complexes of Technetium and Rhenium and Their Reactions with Hydrotris(pyrazolyl)borates. Molecules, 29(16), 3865. https://doi.org/10.3390/molecules29163865







