1. Introduction
Surfactants, a class of amphiphilic compounds, have significant implications in various industries [
1,
2,
3], pharmaceuticals [
4], and everyday consumer products [
5,
6] due to their unique properties and versatile applications [
7,
8,
9]. These molecules possess a hydrophilic head and a hydrophobic tail, allowing them to reduce surface tension and facilitate the formation of stable interfaces between immiscible phases.
Gemini surfactants, a relatively new class of surfactants, have received significant attention in years [
10,
11]. They are characterized by a molecular structure consisting of two hydrocarbon chains linked by a spacer [
12]. The presence of two hydrophobic tails imparts distinctive properties to Gemini surfactants, making them particularly intriguing for research [
13,
14] and applications [
12,
15]. The length and nature of the spacer between the hydrocarbon chains can significantly influence their behavior and functionality [
16,
17,
18].
The investigation of Gemini surfactants and their properties holds great importance. Understanding the intermolecular interactions, self-assembly processes, and structural characteristics of these surfactants is crucial for their effective utilization and design of novel surfactant-based materials. Although traditional experimental methods provide valuable insights, they often have limitations in accessing detailed atomic-level information. Computational chemistry investigations have emerged as powerful tools to overcome these limitations. By employing first-principles calculations [
19], we can delve into the intricate details of Gemini surfactants. However, due to the relatively large number of atoms and conformations involved in Gemini surfactants, accurate first-principle calculation studies require high computational demands, resulting in a lack of systematic computational research on Gemini surfactants. Density Functional Theory (DFT) computations have been applied to study the effects of the carbon chain length of Gemini surfactants on the inhibition of the acid–rock reaction rate, showing that the longer carbon chain is more easily overturned on the rock surface. Molecule Dynamics (MD) simulations also indicate that the surfactants with longer carbon chains can aggregate easily on the surface [
20]. Though computational studies have explored the effect of chain lengths, the influence of the length of the linker and the properties and behaviors of different conformers remain insufficiently characterized.
In this study, we aim to unravel the effects of varying the number of carbon atoms in the central spacer on the properties of surfactants. By examining the structures and properties of these conformers at the atomic level, we can gain fundamental insights that will enhance our understanding of micellization processes, providing valuable information for future explorations in this area. Specifically, we focus on (CH3(CH2)11)(CH3)2-N+-(CH2)n-N+(CH3(CH2)11)(CH3)2 structures, where n = 2, 3, or 4 and, as such, are designated as 12-2-12, 12-3-12, and 12-4-12. By conducting comprehensive first-principles computational investigations, we gained insights into the structural characteristics, electronic properties, and coupling integrations of these surfactants. Our results indicate that the lengths of the spacer in Gemini surfactants influence the favorable conformations and sequential coupling behaviors. This research contributes to optimal spacer length design to achieve desired properties such as critical micelle concentration (CMC), micelle size, and stability.
2. Results and Discussion
2.1. Structural Characteristics of Three Conformers of 12-2-12, 12-3-12, 12-4-12 Surfactants
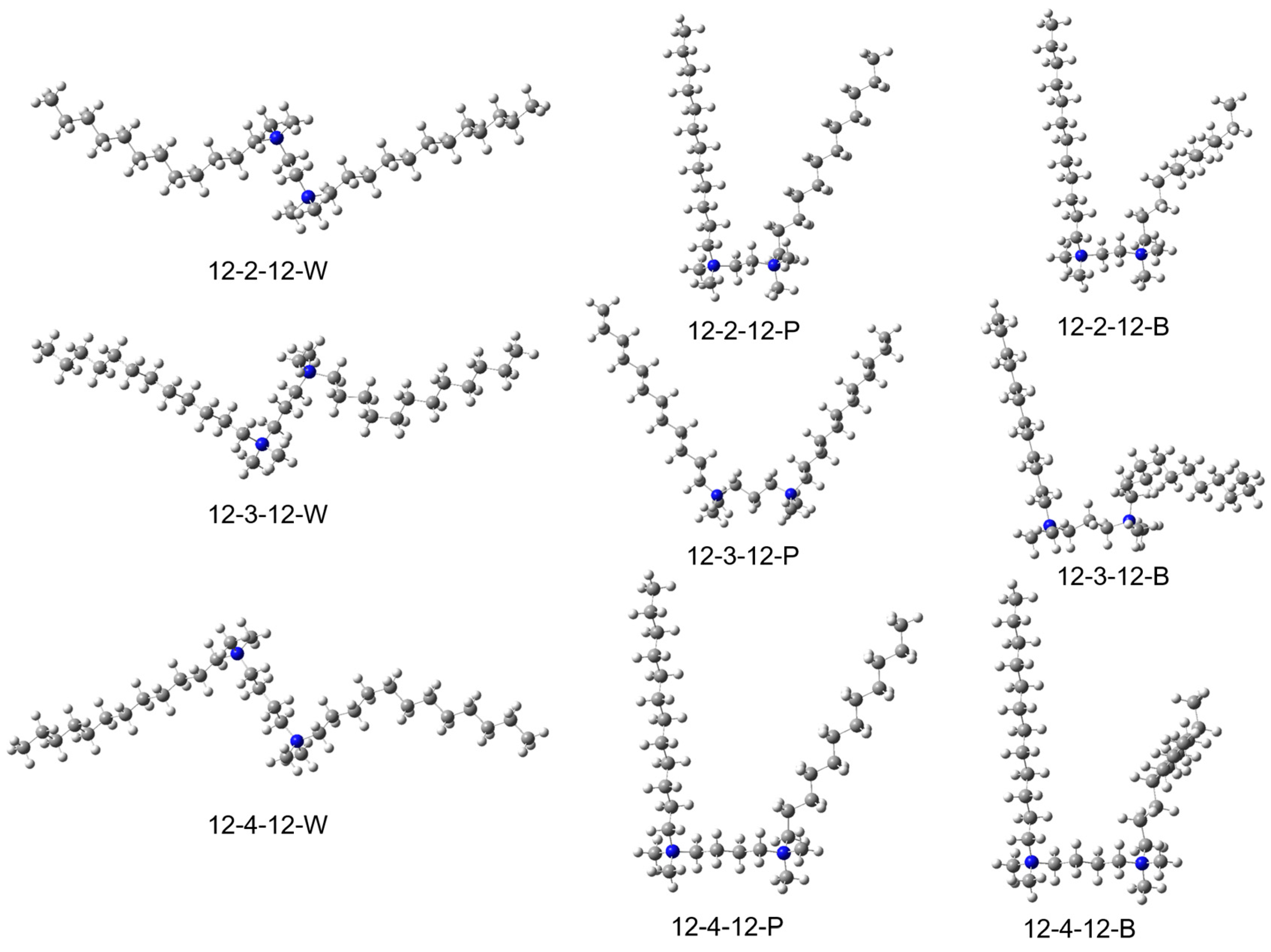
The Gemini surfactants studied here (CH3(CH2)11)(CH3)2-N+-(CH2)n-N+(CH3(CH2)11)(CH3)2) have two quaternized ammines (-N+-) separated by a linker of two to four carbons (+-(CH2)n-, n = 2, 3 or 4). Each nitrogen also has two methyl groups ((CH3)2-) and one dodecyl chain (CH3(CH2)11-). Due to the existence of long tails of 12-n-12 surfactants, one surfactant can have many conformers.
We optimized three different conformers for three different linker lengths (
n = 2, 3, 4), including wing-shaped (W), parallel-shaped (P), and one straight–bent shape (B) (
Figure 1). The two tails extend in opposite directions and away from each other in wing-shaped surfactants (W). Therefore, W presents the largest expansion lengths among three conformers for each pair of 12-n-12 surfactants: 29.88 Å for 12-2-12, 30.92 Å for 12-3-12, 31.93 Å for 12-4-12. The parallel-shaped surfactants (P) have two tails extending in the same direction (
Table 1). The tails run parallel to each other, resulting in relatively smaller molecular sizes compared with the other conformers. The longest side of P-surfactants are one tail of 18.36 Å for 12-2-12, the distance of the end of two tails (the longest sides) of 25.14 Å for 12-3-12, one tail of 18.35 Å for 12-4-12 (
Table 1). The straight–bent-shaped surfactants (B) features one tail extending in one direction, while the other tail is twisted in the middle, folding in another direction. These results in B surfactants having a size in between P and W surfactants, but very close to that of P because the longest side of B conformers are often the straight tails, so the molecule sizes are closer to that of P (
Table 1).
As shown in
Table 2, from analyzing the relative energies of optimized 12-2-12, 12-3-12, and 12-4-12 structures in wing (W), parallel (P), and bent (B) conformations, we observed distinct stability patterns among different conformers. For 12-2-12, the W surfactant emerged as the most stable conformer, followed by the P surfactant, with the B conformer being the least stable. However, the energy difference is a modest 2.84 kcal/mol, suggesting a relatively low energy barrier for conformation transitions. In the case of 12-3-12, the P surfactant exhibited the highest stability, followed by the W surfactant, while the B surfactant was the least stable, with a relative large energy difference of 5.93 kcal/mol, nearly double that of 12-2-12 (2.84 kcal/mol). Similar to 12-2-12, for 12-4-12, the W surfactant ranked as the most stable, followed by the P surfactant, and the B surfactant as the least stable, with an energy difference of 3.52 kcal/mol. In summary, the consistently preferred conformations across these surfactants are the W and P conformations. The reason for this could be, first, that the two tails of W surfactants achieve maximum expansion, thereby minimizing steric repulsion in the structure; on the other hand, P surfactants, while maximizing the overlap of the two tails, achieve stability through parallel stacking, maximizing conjugations of C-H bonds. Therefore, structural stability is a delicate balance between minimizing steric repulsion and maximizing conjugations. Bent surfactants lack a clear advantage in either aspect, resulting in relatively lower stability. Additionally, we observed a similar stability pattern in surfactants with an even number (2 and 4) of carbon atoms in the linker, which is different from surfactants with an odd number (3) of carbon atoms in the linker.
Furthermore, when comparing the most stable conformations of surfactants for each linker length, we find that the size of the most stable conformations for 12-2-12 (W conformer) is 29.88 Å, for 12-3-12 (P conformer) is 25.14 Å, and for 12-4-12 (W conformer) is 31.93 Å. The trend in sizes is 12-4-12 > 12-2-12 > 12-3-12, with the size trend of 12-4-12 and 12-2-12 being more pronounced than that of 12-3-12. This correlates to some extent with our experimental results, where the size order of micelles of 12-3-12 is the smallest compared with 12-4-12 and 12-2-12 [
21].
2.2. Electronic Properties and Raman of Three Conformers of 12-2-12, 12-3-12, 12-4-12 Surfactants
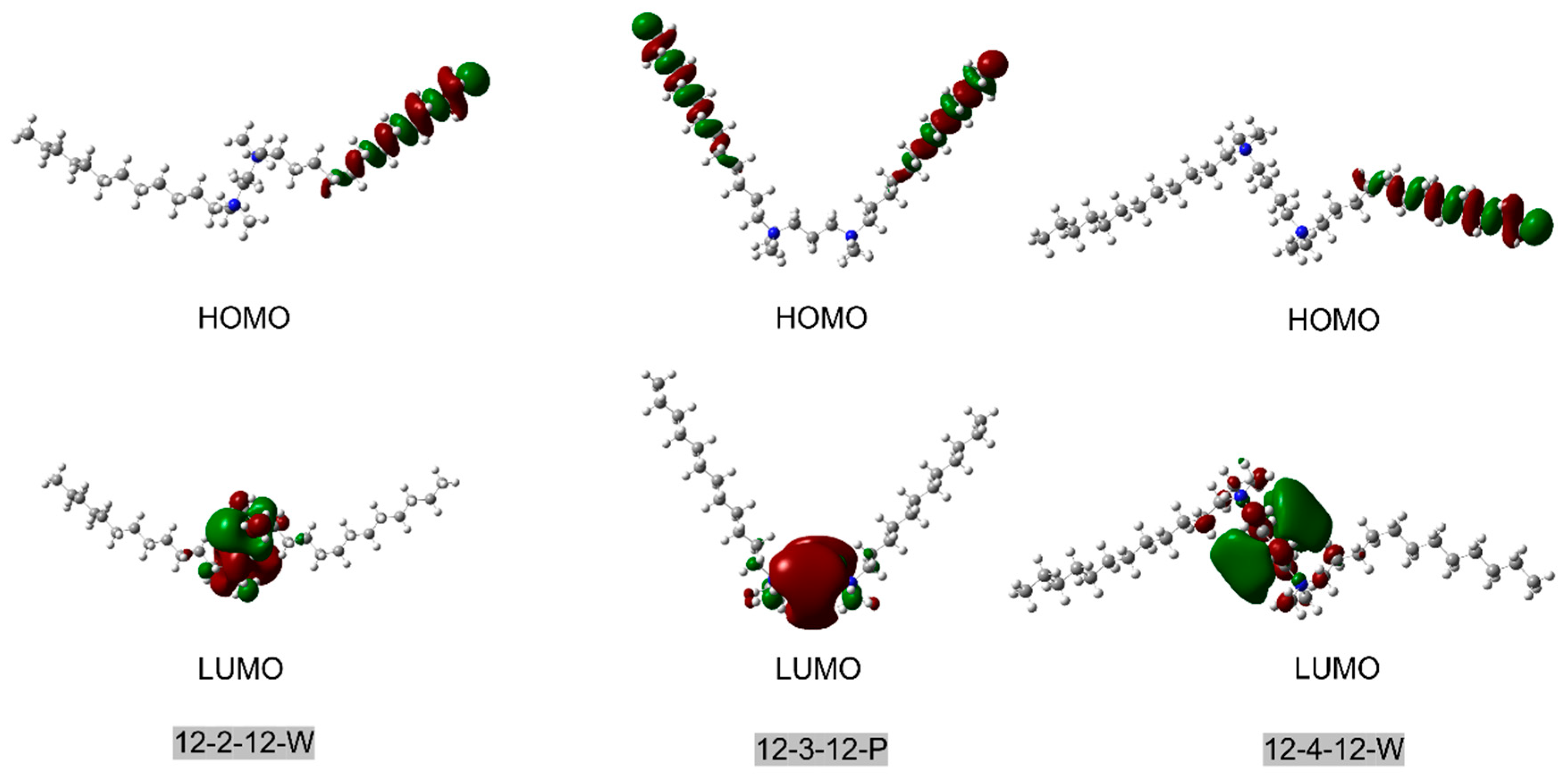
We also computed the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) of nine surfactant conformers (
Figure 2 and
Figure S1 (Supplementary Materials)). We found that only in the parallel structure of the surfactant (such as 12-3-12 P in
Figure 2) are the HOMO orbitals uniformly distributed between the end of two tails. In the wing-shaped surfactant, where the structure is stretched out, and in the bent-shaped surfactant, where one tail is bent, the HOMO is primarily localized on one tail or the straight tail. This may be because only the tails of the parallel surfactant have high conjugation, allowing the HOMO to be distributed across both tails. This also aligns with our earlier discussion that high conjugation is an important factor contributing to the stability of the parallel structure.
Meanwhile, the LUMO of different conformers is entirely concentrated around the two nitrogen (-N+-) atoms of the linker for all structures, representing the active sites of surfactants toward chemical reactions. This is mainly attributed to the positive charge distributed on the nitrogen of the linker of surfactants.
The HOMO-LUMO gap values of different conformers do not differ significantly (
Table 3). Among them, the 12-3-12 conformers exhibit the largest difference in HOMO-LUMO gap values, with the maximum being 6.865 kcal/mol, occurring between the 12-3-12-P and 12-3-12-B conformers. All calculated surfactants and conformers display relatively large HOMO-LUMO gaps (>98.574 kcal/mol), indicating that they show insulator properties.
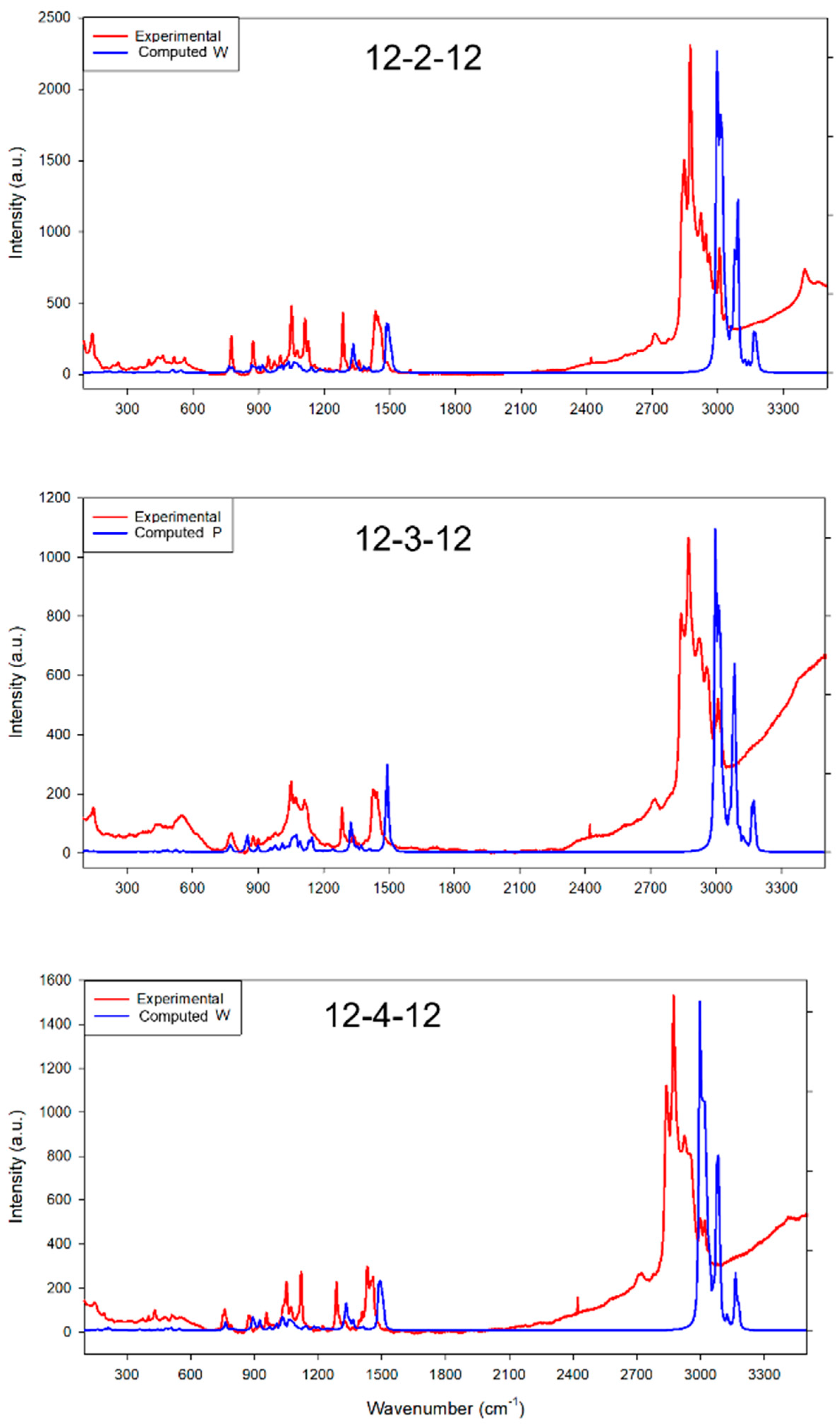
Furthermore, we computed the Raman spectra for all three conformations of 12-2-12, 12-3-12, and 12-4-12 to provide a comprehensive comparison with their respective experimental spectra (
Figure 3 and
Figure S2). The computational Raman values exhibit a slight redshift compared to the experimental values. In the wavenumber range of 1200 to 1600 cm
−1, the calculated values for the two characteristic peaks of all three surfactants closely match the experimental results. Simultaneously, the calculated values for the three peaks in the 2700–3300 range also align well with the experimental findings. In the 900–1200 cm
−1 range, the experimental results reveal a distinct presence of small peaks, which is close to what can be found in the computed Raman Spectra for P conformations. However, other conformers also exhibit some small peaks in this range. Due to the very close conformation energies of W, P, and B conformations, the experimental system likely involves a mixture of all three conformations, and, potentially, some dimerizations of three conformers. This complexity is reflected in the fact that it is challenging to directly confirm the presence of individual conformers through Raman spectra.
2.3. Coupling Interactions of Different Conformers of 12-4-12 Surfactants
One of the primary focuses of our investigation was the coupling integrations of surfactant structures. For most of the experimental results reporting Gemini surfactants with linkers of four or more atoms, we examined the interactions between the 12-4-12 surfactant molecules to investigate how different conformers form aggregates in various environments.
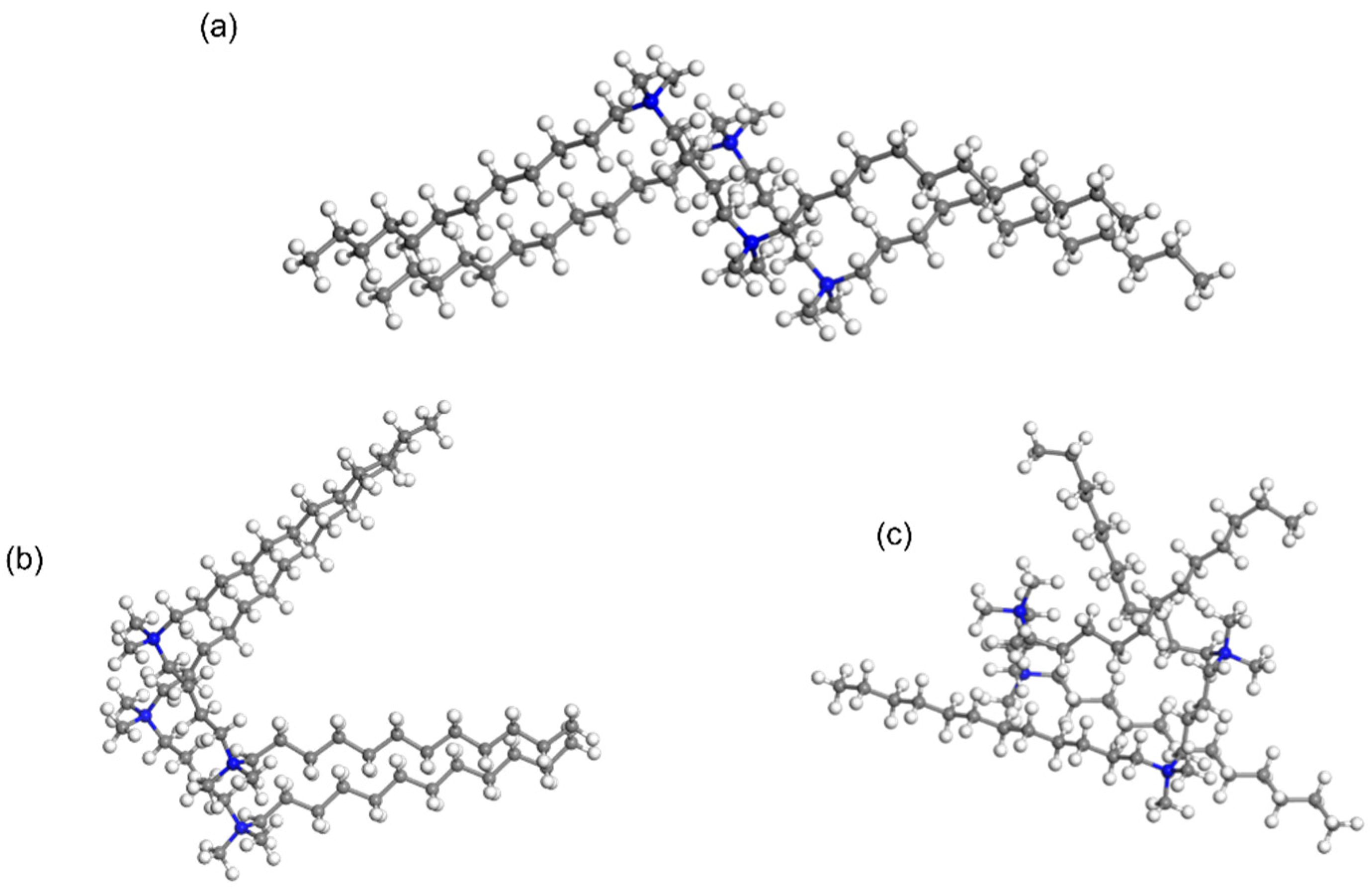
We employed a forcefield-based Grand Canonical Monte Carlo (GCMC) method to simulate the dynamic behavior of surfactants. Using the fix-loading approach, we immobilized one surfactant molecule while manipulating another structurally identical surfactant to explore the most favorable adsorption geometries of the paired surfactants. This approach allowed us to discern the pairing modes of the two surfactants. Notably, for the wing-shaped surfactant, the most stable pairing involved two molecules arranged side by side in a parallel fashion, which maximizes the conjugation between C-H bonds (
Figure 4). Confirming a similar coupling behavior in the parallel-shaped surfactant, we observed a preference for parallel stacking of the two P-surfactant molecules, with the linker and tails aligning in the same direction.
In contrast, the bent-shape surfactants exhibited an entirely distinct coupling behavior (
Figure 4). In the case of B-shape surfactants, two surfactants exhibited an opposing orientation, embedding into the space between each other’s tails. This unique conformation resulted in a mutual insertion of the two surfactants, effectively reducing the overall occupied volume. The distinct adsorption patterns of W-shape, P-shape, and B-shape surfactants provide valuable insights into the coupling behavior of these molecules.
Moreover, we calculated that the average interaction energy for the pairing of wing (W) surfactants is 13.535 kcal/mol, for P surfactants is 14.860 kcal/mol, and for B surfactants is 17.345 kcal/mol, following the order of B > P > W. Considering that the pairing interactions of 12-4-12 (B > P > W) and the order of conformer relative energy (W > P > B) is the opposite, when surfactants aggregate, the energy differences between different conformers become smaller, almost being eliminated. The results not only shed light on the diverse pairing tendencies of these structurally different surfactants, but also hint at their potential to form aggregates with unique structural and functional properties.
3. Methods
3.1. Computational Methods
The computations were carried out by Becke, 3-parameter, Lee–Yang–Parr (B3LYP) functional [
22], and the triple basis set 6-311+G (d,p) [
23] in the Gaussian 16 program [
4]. All geometric parameters were optimized with no atoms frozen. The frequencies of the molecules were computed after optimization, and no imaginary modes were found for any structures, confirming that all structures were local minima and optimized.
Grand Canonical Monte Carlo (GCMC) simulations [
23,
24,
25,
26] in the Sorption module of Materials Studio 2019 [
27] were conducted to evaluate the absorption behavior of Gemini surfactants. GCMC is a statistical–mechanical approach, in which the adsorption process is investigated relying on random sampling and probabilistic interpretation in the sorbent framework. We included one Gemini surfactant in the unit cell to calculate the average loading (/mol) and average interaction energy (kcal/mol) among surfactants with the same geometries, where more substantial loading and higher interaction energy indicate stronger interactions among them. The computations were equilibrated for 100,000 GCMC steps, and data were collected for another 1,000,000 production steps to obtain the average amount adsorbed. Fixed loading computations were carried out to determine the most stable interaction geometries of a pair of surfactants with the same structure. All GCMC simulations were carried out at a temperature of 298 K and a fixed pressure of 101.33 kPa with the Metropolis Monte Carlo method [
23] and COMPASS (Condensed-phase Optimized Molecular Potentials for Atomistic Simulation Studies) forcefield [
28,
29].
3.2. Experimental Methods
The Gemini surfactants were kindly provided by Dr. Steven Bachofer.
Two spectroscopic techniques, Raman and FTIR spectroscopy, were employed to analyze the surfactant samples.
All Raman spectra were obtained using a Thermo Scientific DXR Raman Microscope (Thermo Fisher Scientific, Waltham, MA, USA). The laser wavelength employed was 532 nm with a power of 2.0 mW. The aperture was a 50 µm slit. Surfactant samples were placed on a glass microscope slide and analyzed without any modification.
The FTIR spectra were obtained using a Thermo Scientific Nicolet iS50 FT-IR Spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). The optical velocity was set to 0.4747. The resolution was 4.000. The sample was placed on the ATR stage and analyzed directly.
4. Conclusions
Our first-principles computational investigation provides a detailed understanding of the structural characteristics, electronic properties, and coupling behavior of 12-4-12, 12-3-12, and 12-2-12 surfactants through a combination of computations and experimental verifications. The study explored three conformations of each surfactant, and we found that 12-2-12-W, 12-3-12-P, and 12-4-12-W are the most stable conformations for each surfactant based on optimized energy. However, the conformation energies difference of W, P, and B conformations are rather small; therefore, the experimental system likely involves a mixture of all three conformations, and even some dimerization of three conformers. The comparison of Raman spectra between experimental and computational results further corroborates this conclusion.
We also applied GCMC simulations to reveal subtle differences in stability and pairing tendencies among different conformers, shedding light on the factors influencing their assembly behavior. Overall, this study investigates the structural, electronic, and pairing behavior of Gemini surfactants with different spacer lengths and different conformers from the atomic level, laying the groundwork for both theoretical and experimental research in this area.
Supplementary Materials
The following supporting information can be downloaded at:
https://www.mdpi.com/article/10.3390/molecules29122880/s1, Figure S1: Highest Occupied Molecular (HOMO, left hand side) and Lowest Unoccupied Molecular (LUMO, right hand side) orbitals of 12-2-12 P, 12-2-12 B, 12-3-12 W, 12-3-12 B, and 12-4-12 P, 12-4-12 B conformers; Figure S2: Comparison of Experimental Raman Spectra for 12-2-12, 12-3-12, and 12-4-12 Surfactants and Computational Raman Spectra for 12-2-12 P, 12-2-12 B, 12-3-12 W, 12-3-12 B, 12-4-12 P and 12-4-12 B conformers.
Author Contributions
Conceptualization, R.D.S. and N.M.-K.; methodology, S.L., R.D.S. and N.M.-K.; investigation, S.L., D.W., N.M.-K., R.D.S.; resources, R.D.S. and N.M.-K.; data curation, S.L. and D.W.; writing—original draft preparation, S.L.; writing—review and editing, S.L., N.M.-K. and R.D.S.; visualization, S.L. and D.W.; supervision, R.D.S., N.M.-K. and S.L.; project administration, N.M.-K. and R.D.S.; funding acquisition, N.M.-K., R.D.S. and S.L.; All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Welch Foundation (m-0200), the National Science Foundation (# 0820845), and the start-up fund of S. L. provided by Texas Woman’s University (TWU), National Science Foundation under Grant No. 1953448. S.L. thanks the Texas Advanced Computing Center (TACC) for their support in computational resources for this work. N.M.K. also thanks the TWU Jane Nelson Institute for Women’s Leadership institute (JNIWL), grant #JNIWL 3606.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Dave, N.; Joshi, T. A concise review on surfactants and its significance. Int. J. Appl. Chem. 2017, 13, 663–672. [Google Scholar] [CrossRef]
- Hayes, D.G.; Smith, G.A. Biobased surfactants: Overview and industrial state of the art. In Biobased Surfactants; Elsevier: Amsterdam, The Netherlands, 2019; pp. 3–38. [Google Scholar]
- Tripathy, D.B.; Mishra, A.; Clark, J.; Farmer, T. Synthesis, chemistry, physicochemical properties and industrial applications of amino acid surfactants: A review. Comptes Rendus Chim. 2018, 21, 112–130. [Google Scholar] [CrossRef]
- Sekhon, B.S. Surfactants: Pharmaceutical and medicinal aspects. J. Pharm. Technol. Res. Manag. 2013, 1, 43–68. [Google Scholar] [CrossRef]
- Falbe, J. Surfactants in Consumer Products: Theory, Technology and Application; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Kralova, I.; Sjöblom, J. Surfactants used in food industry: A review. J. Dispers. Sci. Technol. 2009, 30, 1363–1383. [Google Scholar] [CrossRef]
- De, S.; Malik, S.; Ghosh, A.; Saha, R.; Saha, B. A review on natural surfactants. RSC Adv. 2015, 5, 65757–65767. [Google Scholar] [CrossRef]
- Halliday, H. Surfactants: Past, present and future. J. Perinatol. 2008, 28, S47–S56. [Google Scholar] [CrossRef]
- Ivanković, T.; Hrenović, J. Surfactants in the environment. Arh. Za Hig. Rada I Toksikol. 2010, 61, 95–109. [Google Scholar] [CrossRef]
- Menger, F.M.; Keiper, J.S. Gemini surfactants. Angew. Chem. Int. Ed. 2000, 39, 1906–1920. [Google Scholar] [CrossRef]
- Menger, F.M.; Littau, C. Gemini-surfactants: Synthesis and properties. J. Am. Chem. Soc. 1991, 113, 1451–1452. [Google Scholar] [CrossRef]
- Sharma, R.; Kamal, A.; Abdinejad, M.; Mahajan, R.K.; Kraatz, H.-B. Advances in the synthesis, molecular architectures and potential applications of gemini surfactants. Adv. Colloid Interface Sci. 2017, 248, 35–68. [Google Scholar] [CrossRef]
- Xie, Y.; He, X. Asymmetric Gemini surfactants as corrosion inhibitors for carbon steel in acidic medium: Experimental and theoretical studies. Colloids Surf. Physicochem. Eng. Asp. 2023, 660, 130850. [Google Scholar] [CrossRef]
- Layn, K.M.; Debenedetti, P.G.; Prud’homme, R.K. A theoretical study of Gemini surfactant phase behavior. J. Chem. Phys. 1998, 109, 5651–5658. [Google Scholar] [CrossRef][Green Version]
- Numin, M.S.; Hassan, A.; Jumbri, K.; Eng, K.K.; Borhan, N.; Daud, N.M.R.N.M.; Suhor, F.; Wahab, R.A. A recent review on theoretical studies of Gemini surfactant corrosion inhibitors. J. Mol. Liq. 2022, 368, 120649. [Google Scholar] [CrossRef]
- Pal, N.; Saxena, N.; Mandal, A. Synthesis, characterization, and physicochemical properties of a series of quaternary gemini surfactants with different spacer lengths. Colloid. Polym. Sci. 2017, 295, 2261–2277. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J.; Wang, Y.; Yan, H.; Li, P.; Thomas, R.K. Effect of the nature of the spacer on the aggregation properties of gemini surfactants in an aqueous solution. Langmuir 2004, 20, 53–56. [Google Scholar] [CrossRef]
- Sikirić, M.; Primožič, I.; Talmon, Y.; Filipović-Vinceković, N. Effect of the spacer length on the association and adsorption behavior of dissymmetric gemini surfactants. J. Colloid Interface Sci. 2005, 281, 473–481. [Google Scholar] [CrossRef]
- Engel, E. Density Functional Theory; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Zhao, F.; Wang, S.; Guo, J. Experimental and molecular dynamics simulation study on the effects of the carbon chain length of Gemini surfactants on the inhibition of the acid-rock reaction rate. Langmuir 2021, 37, 5232–5241. [Google Scholar] [CrossRef]
- Sutorius, T.M.M.; Aquilar, D.; Nembaware, H.; Bachofer, S.; Sheardy, R.D. Gemini Surfactants with Short Linkers: Thermodynamics of Micelle Formation. J. Therm. Anal. Calorim. 2024. [Google Scholar] [CrossRef]
- Tirado-Rives, J.; Jorgensen, W.L. Performance of B3LYP density functional methods for a large set of organic molecules. J. Chem. Theory Comput. 2008, 4, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.P.; Uvdal, P. New scale factors for harmonic vibrational frequencies using the B3LYP density functional method with the triple-ζ basis set 6-311+ G (d, p). J. Phys. Chem. A 2005, 109, 2937–2941. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, P.; Tanaka, H.; Kaneko, K.; Terzyk, A.P.; Do, D.D. Grand canonical Monte Carlo simulation study of methane adsorption at an open graphite surface and in slitlike carbon pores at 273 K. Langmuir 2005, 21, 5639–5646. [Google Scholar] [CrossRef]
- Woo, H.-J.; Dinner, A.R.; Roux, B. Grand canonical Monte Carlo simulations of water in protein environments. J. Chem. Phys. 2004, 121, 6392–6400. [Google Scholar] [CrossRef]
- Lin, S.; Jacoby, K.A.; Gu, J.; Vega-Santander, D.R.; Hernández-Maldonado, A.J.; Chen, Z. Zeolite-templated carbons as effective sorbents to remove methylsiloxanes and derivatives: A computational screening. Green Energy Environ. 2021, 6, 884–892. [Google Scholar] [CrossRef]
- Akkermans, R.L.; Spenley, N.A.; Robertson, S.H. Monte Carlo methods in materials studio. Mol. Simul. 2013, 39, 1153–1164. [Google Scholar] [CrossRef]
- Sun, H. COMPASS: An ab initio force-field optimized for condensed-phase applications overview with details on alkane and benzene compounds. J. Phys. Chem. B 1998, 102, 7338–7364. [Google Scholar] [CrossRef]
- Sun, H.; Ren, P.; Fried, J. The COMPASS force field: Parameterization and validation for phosphazenes. Comput. Theor. Polym. Sci. 1998, 8, 229–246. [Google Scholar] [CrossRef]
Figure 1.
Computational optimized structures of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations.
Figure 1.
Computational optimized structures of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations.
Figure 2.
HOMO and LUMO orbitals 12-2-12 W, 12-3-12 P, and 12-4-12 W conformations, where the isovalue was set as 0.02.
Figure 2.
HOMO and LUMO orbitals 12-2-12 W, 12-3-12 P, and 12-4-12 W conformations, where the isovalue was set as 0.02.
Figure 3.
Comparison of experimental Raman spectra for 12-2-12, 12-3-12, and 12-4-12 surfactants and computational Raman spectra for 12-2-12 W, 12-3-12 P, and 12-4-12 W conformations.
Figure 3.
Comparison of experimental Raman spectra for 12-2-12, 12-3-12, and 12-4-12 surfactants and computational Raman spectra for 12-2-12 W, 12-3-12 P, and 12-4-12 W conformations.
Figure 4.
Geometries of paired 12-4-12 with wing (a), parallel (b), and bent (c) conformations computed by the Sorption module.
Figure 4.
Geometries of paired 12-4-12 with wing (a), parallel (b), and bent (c) conformations computed by the Sorption module.
Table 1.
Lengths of the longest sides of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations; unit is Å.
Table 1.
Lengths of the longest sides of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations; unit is Å.
| Surfactant | Wing Conformation | Parallel Conformation | Bent Conformation |
|---|
| 12-2-12 | 29.88 | 18.36 | 18.56 |
| 12-3-12 | 30.92 | 25.14 | 26.17 |
| 12-4-12 | 31.93 | 18.35 | 18.35 |
Table 2.
The relative energy of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations, where the lowest energy of each conformer was set as 0; unit is kcal/mol.
Table 2.
The relative energy of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations, where the lowest energy of each conformer was set as 0; unit is kcal/mol.
| Surfactant | Wing Conformation | Parallel Conformation | Bent Conformation |
|---|
| 12-2-12 | 0 | 1.38 | 2.84 |
| 12-3-12 | 3.47 | 0 | 5.93 |
| 12-4-12 | 0 | 1.04 | 3.52 |
Table 3.
The HOMO-LUMO gaps of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations; unit is kcal/mol.
Table 3.
The HOMO-LUMO gaps of 12-2-12, 12-3-12, and 12-4-12, with wing (W), parallel (P), and bent (B) conformations; unit is kcal/mol.
| Surfactant | Wing Conformation | Parallel Conformation | Bent Conformation |
|---|
| 12-2-12 | 99.340 | 98.574 | 99.014 |
| 12-3-12 | 105.640 | 99.754 | 106.619 |
| 12-4-12 | 110.685 | 109.016 | 110.660 |
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).