Simple Method for the Determination of THC and THC-COOH in Human Postmortem Blood Samples by Gas Chromatography—Mass Spectrometry
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals Reagents and Standards
3.2. Specimens
3.3. Analytical Procedure
- Sample pretreatment and liquid-liquid extractionBlood plasma was separated by centrifugation of whole blood when it was possible, and 1 mL of plasma was placed in a screw-capped round bottom glass tube. First, 20 µL methanolic solution of 10 µg/mL of each internal standard (Δ9-THC-D3 and Δ9-THC-COOH-D3) were added. The tube was closed, and its contents mixed. Afterwards, 1 mL of Milli-Q water and 500 µL of 2 M NaOH is added, and the mixture is again stirred. Subsequently, a liquid-liquid extraction was performed with 5 mL of a mixture (9:1, v/v) of hexane-ethyl acetate.The tube is agitated for 10 min to cause the migration of components in the two phases, then it is centrifuged for 10 min, and the organic phase containing the THC is transferred and evaporated to dryness. The dried residue was reconstituted with 40 µL of methanol and injected in the GC-MS system.In order to determine Δ9-THC-COOH, a second extraction is needed starting from the aqueous phase.1 mL of a 0.1 N HCl solution and 200 mL of an acetic acid solution were added, in order to acidify the mixture. Then, it was mixed by agitation for 30 s, and 5 mL of the hexane-ethyl acetate mixture (9:1, v/v) was added again. The tube is agitated for 10 min, centrifuged, and the organic phase transferred to a conical bottom tube with cap and evaporated for later derivatization.The derivatization is carried out by adding a mixture of 40 µL of BSTFA-TCMS (99:1) to the Δ9-THC-COOH dry residue and incubating at 100 °C for 20 min.
- GC-MSGC-MS analyses were performed in an Agilent 6890 Gas Chromatograph equipped with a 7683B automatic liquid sampler, coupled with an Agilent 5973 mass selective quadrupole detector (Agilent Technologies, Las Rozas, Madrid, Spain). The GC injection port was set at 250 °C in splitless mode (purge time 0.75 min). The GC was equipped with an Agilent 19091S-133U 5% phenylmethylsyloxane capillary column, 30 m × 0.25 mm. i.d., 0.50 µm film thickness (purchased by Agilent Technologies, Las Rozas, Madrid, Spain). The oven temperature was held at 90 °C for 1 min, then at 35 °C/min to 200 °C, then at 10 °C/min to 260 °C and held 15 min. Helium was used as carrier gas at a flow of 1 mL/min. The mass detector operated in electron ionization at 70 eV. Initially, a mixture of standards of all the compounds was analyzed in full scan mode (mass range 50–550 amu). Quantifier and qualifier ions used for each analyte were selected based on their abundance and mass-to-charge ratio (m/z). Owing to their reproducibility and lack of interferences, high-mass ions were selected whenever possible. Upon the selection of ions, the mass analyzer was operated in selected ion monitoring (SIM) acquisition mode. All diagnostic ions and retention times are listed in Table 5.
3.4. Method Validation
- Limits of detection, lower limit of quantitation, and specificityThe sensitivity of the method was determined by the calculation of the limit of detection (LOD) and the lower limit of quantitation (LLOQ). LOD was determined by an empirical method that consists of analyzing a series of plasma samples containing decreasing amounts of the analytes. LOD was the lowest concentration that presented a S/N > 3 for at least three diagnostic ions for each substance. The LLOQ was the lowest concentration of analytes in a sample that can be determined with appropriate precision and accuracy. Specificity was studied analyzing 10 negative plasma samples.
- LinearityThe linearity of the method for each compound was studied in the range 0.05–1.5 µg/mL for Δ9-THC and 0.08–1.5 µg/mL for Δ9-THC-COOH, performing 5 extractions and analyses for each level. Calibration curves were built by linear regression of the area ratio of each substance with the internal standard (IS) vs. the concentration of each analyte. Curves with a quadratic regression coefficient (R2) higher than 0.99 were satisfactory.
- Precision and accuracyPrecision, expressed as the coefficient of variation (CV) of the measured values, was expected to be less than 15% at all concentrations, except for the LLOQ for which 20% was acceptable [27]. It was studied on 5 replicate analyses at three levels: 0.1, 0.5, and 1 µg/mL. In the same way, accuracy was evaluated using the mean relative error (MRE), which had to be less than 15% of the theoretical values at each concentration level except for the LLOQ, for which 20% was acceptable [37].
- RecoveryThe recovery of an analyte is the detector response obtained from an amount of the analyte added to and extracted from the biological matrix compared to the detector response obtained for the true concentration of the pure authentic standard [38]. Recovery of the analyte must be optimized to ensure that the extraction is efficient and reproducible. Recovery does not need to be 100%, but the degree of recovery of an analyte and internal standard must be consistent and reproducible. The recovery of the method was examined by comparing the analytical results for extracted samples at 2 levels of concentration (0.1 and 1 µg/mL) 5 times within 3 days versus samples spiked with the standards after the extraction step, where unextracted standards represent 100% recovery.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- European Drug Report 2019: Trends and Developments; Publications Office of the European Union: Luxembourg, 2019; Available online: https://www.emcdda.europa.eu/publications/edr/trends-developments/2019_en (accessed on 22 March 2023).
- European Monitoring Centre for Drugs and Drug Addiction. European Drug Report 2022: Trends and Developments; Publications Office of the European Union: Luxembourg, 2022; Available online: https://www.emcdda.europa.eu/publications/edr/trends-developments/2022_en (accessed on 22 March 2023).
- Horwood, L.J.; Fergusson, D.M.; Hayatbakhsh, M.R.; Najman, J.M.; Coffey, C.; Patton, G.C.; Silins, E.; Hutchinson, D.M. Cannabis use and educational achievemet: Findings from three Australasian cohort studies. Drug Alcohol Depend. 2010, 110, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Bryan, A.D.; Schmiege, S.J.; Magnan, R.E. Marijuana use and risky sexual behavior among high-risk adolescents: Trajectories, risk factors, and event-level relationships. Dev. Psychol. 2012, 48, 1429–1442. [Google Scholar] [CrossRef]
- Bell, R.; Wechsler, H.; Johnston, L. Correlates of college student marijuana use: Results of a US National Survey. Addiction 1997, 92, 571–581. [Google Scholar] [CrossRef]
- Mehling, R. Drugs. In The Straight Facts. Marijuana; Chelsea House Publishers: Philadelphia, PA, USA, 2003. [Google Scholar]
- Hanisch, S.; Paulke, A.; Toennes, S.W. Investigation of a recently detected THCCOOH isomer: Post mortem findings and comparison with D8-THCCOOH. Forensic Sci. Int. 2015, 257, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Musshoff, F.; Madea, B. Review of Biologic Matrices (Urine, Blood, Hair) as Indicators of Recent or Ongoing Cannabis Use. Ther. Drug Monit. 2006, 28, 155–163. [Google Scholar] [CrossRef]
- Bach, A.; Fleischer, H.; Wijayawardena, B.; Thurow, K. Automation System for the Flexible Sample Preparation for Quantification of D9-THC-D3, THC-OH and THC-COOH from Serum, Saliva and Urine. Appl. Sci. 2022, 12, 2838. [Google Scholar] [CrossRef]
- Frei, P.; Frauchiger, S.; Scheurer, E.; Mercer-Chalmers-Bender, K. Quantitative determination of five cannabinoids in blood and urine by gas chromatography tandem mass spectrometry applying automated on-line solid phase extraction. Drug Test Anal. 2022, 14, 1223–1233. [Google Scholar] [CrossRef]
- Teixeira, H.; Verstraete, A.; Proença, P.; Corte-Real, F.; Monsanto, P.; Vieira, D.N. Validated method for the simultaneous determination of Δ9-THC and Δ9-THC-COOH in oral fluid, urine and whole blood using solid-phase extraction and liquid chromatography–mass spectrometry with electrospray ionization. Forensic Sci. Int. 2007, 170, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Sobolesky, P.M.; Smith, B.E.; Hubbard, J.A.; Stone, J.; Marcotte, T.D.; Grelotti, D.J.; Grant, I.; Fitzgerald, R.L. Validation of a liquid chromatography-tandem mass spectrometry method for analyzing cannabinoids in oral fluid. Clin. Chim. Acta 2019, 491, 30–38. [Google Scholar] [CrossRef]
- Gustafson, R.; Moolchan, E.; Barnes, A.; Levine, B.; Huestis, M. Validated method for the simultaneous determination of Δ9-tetrahydrocannabinol (THC), 11-hydroxy-THC and 11-nor-9-carboxy-THC in human plasma using solid phase extraction and gas chromatography–mass spectrometry with positive chemical ionization. J. Chromatogr. B 2003, 798, 145–154. [Google Scholar] [CrossRef]
- Lowe, R.H.; Karschner, E.L.; Schwilke, E.W.; Barnes, A.J.; Huestis, M.A. Simultaneous quantification of Δ9-tetrahydrocannabinol, 11-hydroxy-Δ9-tetrahydrocannabinol, and 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid in human plasma using two-dimensional gas chromatography, cryofocusing, and electron impact-mass spectrometry. J. Chromatogr. A 2007, 1163, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Rosado, T.; Fernandes, L.; Barroso, M.; Gallardo, E. Sensitive determination of THC and main metabolites in human plasma by means of microextraction in packed sorbent and gas chromatography–tandem mass spectrometry. J. Chromatogr. B 2017, 1043, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Scheidweiler, K.B.; Newmeyer, M.N.; Barnes, A.J.; Huestis, M.A. Quantification of cannabinoids and their free and glucuronide metabolites in whole blood by disposable pipette extraction and liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2016, 1453, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Hädener, M.; Weinmann, W.; Schürch, S.; König, S. Development of a rapid column-switching LC-MS/MS method for the quantification of THCCOOH and THCCOOH-glucuronide in whole blood for assessing cannabis consumption frequency. Anal. Bioanal. Chem. 2016, 408, 1953–1962. [Google Scholar] [CrossRef]
- Castro, A.L.; Tarelho, S.; Melo, P.; Franco, J.M. A fast and reliable method for quantitation of THC and its 2 main metabolites in whole blood by GC–MS/MS. Forensic Sci. Int. 2018, 289, 344–351. [Google Scholar] [CrossRef]
- Palazzoli, F.; Citti, C.; Licata, M.; Vilella, A.; Manca, L.; Zoli, M.; Vandelli, M.A.; Forni, F.; Cannazza, G. Development of a simple and sensitive liquid chromatography triple quadrupole mass spectrometry (LC–MS/MS) method for the determination of cannabidiol (CBD), Δ9-tetrahydrocannabinol (THC) and its metabolites in rat whole blood after oral administration of a single high dose of CBD. J. Pharm. Biomed. Anal. 2018, 150, 25–32. [Google Scholar] [CrossRef]
- Andrews, R.; Paterson, S. A validated method for the analysis of cannabinoids in post-mortem blood using liquid–liquid extraction and two-dimensional gas chromatography–mass spectrometry. Forensic Sci. Int. 2012, 222, 111–117. [Google Scholar] [CrossRef]
- Hubbard, J.A.; Smith, B.E.; Sobolesky, P.M.; Kim, S.; Hoffman, M.A.; Stone, J.; Huestis, M.A.; Grelotti, D.J.; Grant, I.; Marcotte, T.D.; et al. Validation of a liquid chromatography tandem mass spectrometry (LC-MS/MS) method to detect cannabinoids in whole blood and breath. Clin. Chem. Lab. Med. 2019, 58, 673–681. [Google Scholar] [CrossRef]
- Giroud, C.; Ménétrey, A.; Augsburger, M.; Buclin, T.; Sanchez-Mazas, P.; Mangin, P. Δ9-THC, 11-OH-Δ9-THC and Δ9-THCCOOH plasma or serum to whole blood concentrations distribution ratios in blood samples taken from living and dead people. Forensic Sci. Int. 2001, 123, 159–164. [Google Scholar] [CrossRef]
- Hoffman, M.A.; Trochta, A.; Gary, R.D.; Fitzgerald, R.L.; McIntyre, I.M. An evaluation of postmortem concentrations of Δ9-tetrahydrocannabinol (THC) and 11-nor-9-carboxy-Δ9-tetrahydrocannabinol (THCCOOH). Forensic Sci. Int. 2020, 315, 110414. [Google Scholar] [CrossRef]
- Wu, F.; Jensen, T.L.; McMillin, G.A. Detection of In Utero Cannabis Exposure in Umbilical Cord Tissue by a Sensitive Liquid Chromatography-Tandem Mass Spectrometry Method. Methods Mol. Biol. 2018, 1872, 211–222. [Google Scholar] [CrossRef]
- Han, E.; Choi, H.; Lee, S.; Chung, H.; Song, J.M. A study on the concentrations of 11-nor-Δ (9)-tetrahydrocannabinol-9-carboxylic acid (THCCOOH) in hair root and whole hair. Forensic Sci. Int. 2011, 210, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Minoli, M.; Angeli, I.; Ravelli, A.; Gigli, F.; Lodi, F. Detection and quantification of 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid in hair by GC/MS/MS in Negative Chemical Ionization mode (NCI) with a simple and rapid liquid/liquid extraction. Forensic Sci. Int. 2012, 218, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Prego-Meleiro, P.; Lendoiro, E.; Concheiro, M.; Cruz, A.; López-Rivadulla, M.; de Castro, A. Development and validation of a liquid chromatography tandem mass spectrometry method for the determination of cannabinoids and phase I and II metabolites in meconium. J. Chromatogr. A 2017, 1497, 118–126. [Google Scholar] [CrossRef]
- Mantovani, C.D.C.; e Silva, J.P.; Forster, G.; de Almeida, R.M.; Diniz, E.M.D.A.; Yonamine, M. Simultaneous accelerated solvent extraction and hydrolysis of 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid glucuronide in meconium samples for gas chromatography-mass spectrometry analysis. J. Chromatogr. B 2018, 1074–1075, 1–7. [Google Scholar] [CrossRef]
- Han, E.; Park, Y.; Kim, E.; In, S.; Yang, W.; Lee, S.; Choi, H.; Lee, S.; Chung, H.; Song, J.M. Simultaneous analysis of Δ(9)-tetrahydrocannabinol and 11-nor-9-carboxy-tetrahydrocannabinol in hair without different sample preparation and derivatization by gas chromatography-tandem mass spectrometry. J. Pharm. Biomed. Anal. 2011, 55, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Kala, M.; Kochanowski, M. Determination of D9-Tetrahydrocannabinol (9THC) and THCCOOH in Blood and Urine Using Gas Chromatography Negative Ion Chemical Ionization Mass Spectrometry (GC-MS-NCI). Chem. Anal. 2006, 51, 65–78. [Google Scholar]
- Balcells Olivero, M. Toxicologia del cannabis. Adicciones 2000, 12, 169–174. [Google Scholar] [CrossRef]
- Lee, D.; Huestis, M.A. Current knowledge on cannabinoids in oral fluid. Drug Test. Anal. 2013, 6, 88–111. [Google Scholar] [CrossRef]
- De Cannabis, G.C. Guias Clínicas Socidrogalcohol Basadas en la Evidencia Científica. Socidrogalcohol. Available online: https://pnsd.sanidad.gob.es/profesionales/publicaciones/catalogo/bibliotecaDigital/publicaciones/pdf/2022/20220119_SOCIDROGALCOHOL_guia_cannabis.pdf (accessed on 22 February 2023).
- De Oliveira, K.; Scanferla, D.; Silva, J.; Madia, M.; Bando, E.; Junior, M.M.; Marchioni, C.; Mossini, S. Quantitative analysis of Δ9-THC-COOH in Human Urine by the Liquid-Liquid Extraction technique and Gas Chromatography-Mass Spectrometry: Adaptation, Optimization and Validation. Braz. J. Anal. Chem. 2021, 9, 229–239. [Google Scholar] [CrossRef]
- Mahmoud, A.; ElSohly, E. Marijuana and the Cannabinoids; Humana Press: Humana Totowa, NJ, USA, 2007. [Google Scholar] [CrossRef]
- Huestis, M.A.; Smith, M.L. Cannabinoid Markers in Biological Fluids and Tissues: Revealing Intake. Trends Mol. Med. 2018, 24, 156–172. [Google Scholar] [CrossRef] [PubMed]
- Bioanalytical Method Validation Guidance for Industry. U.S. Department of Health and Human Services. Food and Drug Administration. May 2018. Available online: https://www.fda.gov/files/drugs/published/Bioanalytical-Method-Validation-Guidance-for-Industry.pdf (accessed on 22 February 2023).
- Álvarez-Freire, I.; Rodríguez, T.M.; Barrera, A.B.; Cabarcos-Fernández, P.; Tabernero-Duque, M. Determination of levetiracetam in plasma: Comparison of gas chromatography-mass spectrometry technique and Abbot® Architect system. Microchem. J. 2020, 160, 105715. [Google Scholar] [CrossRef]
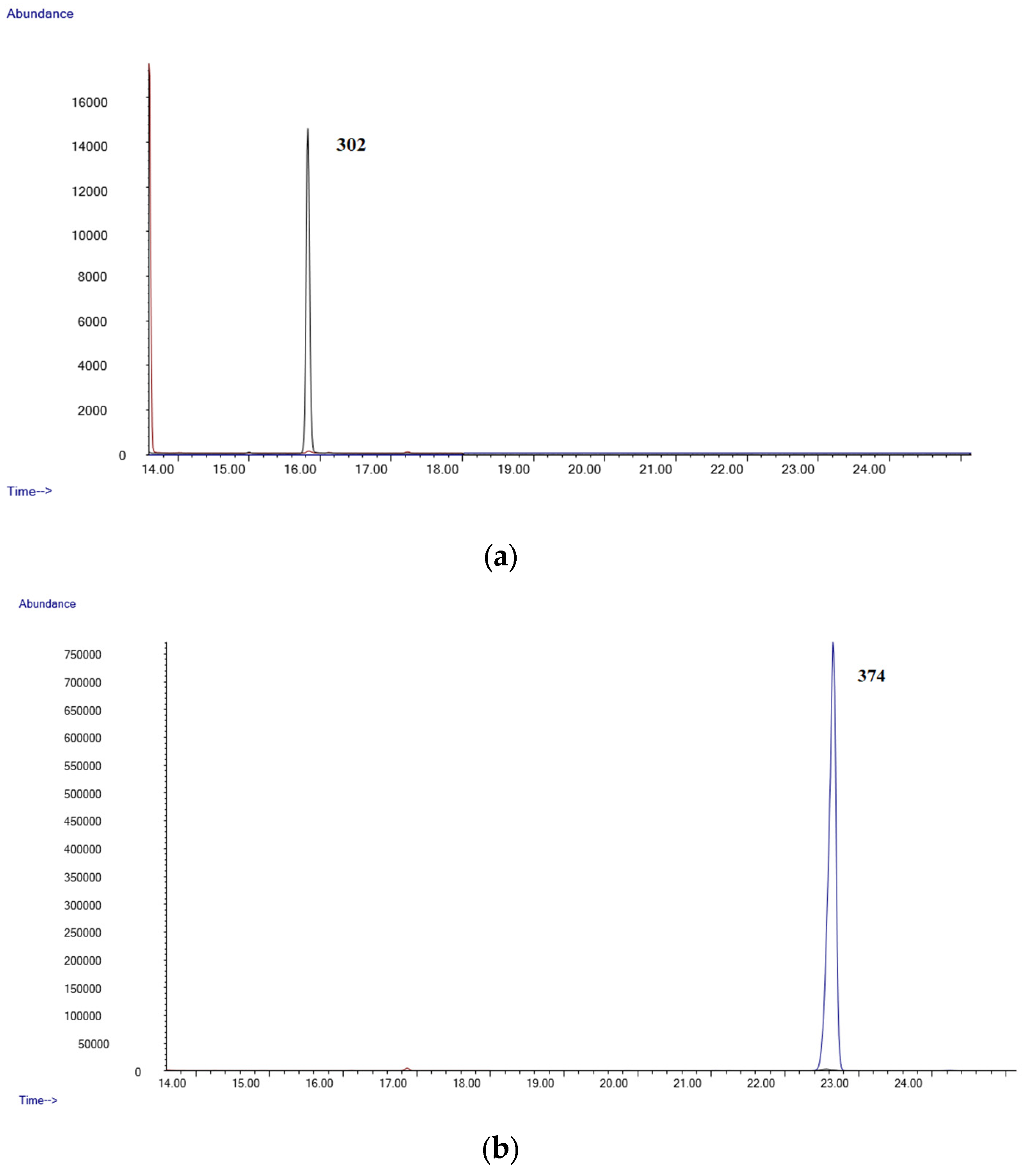
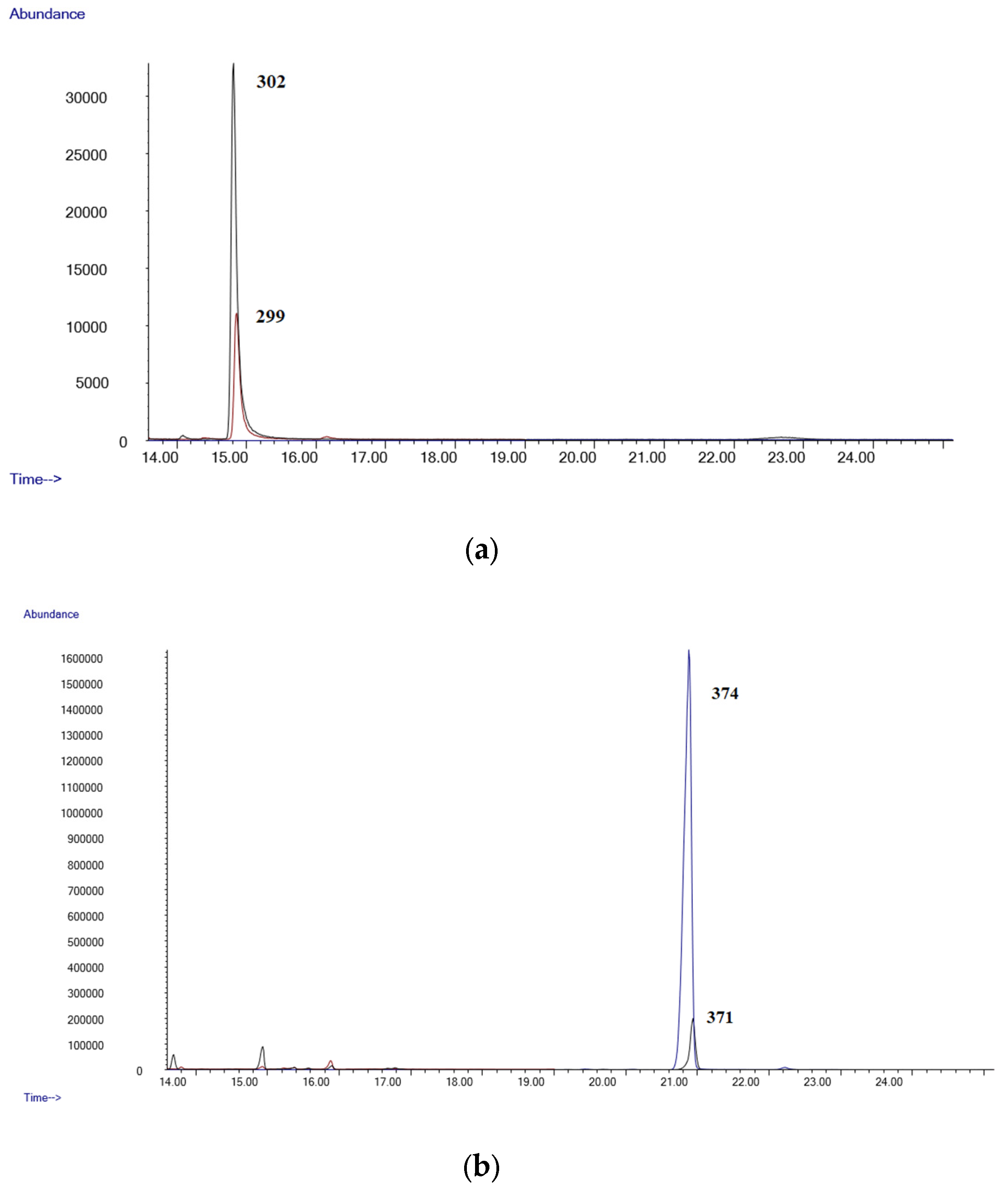
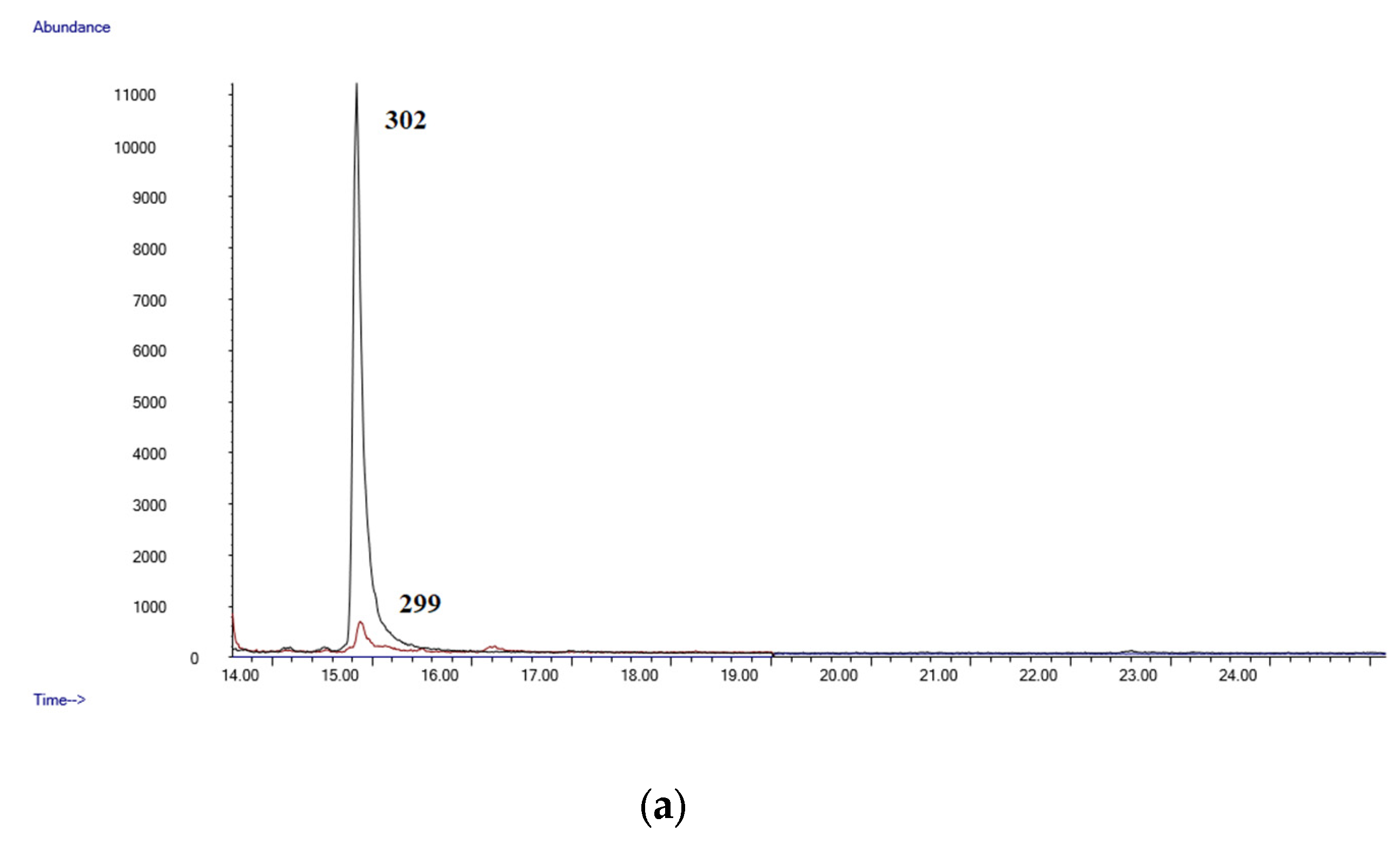
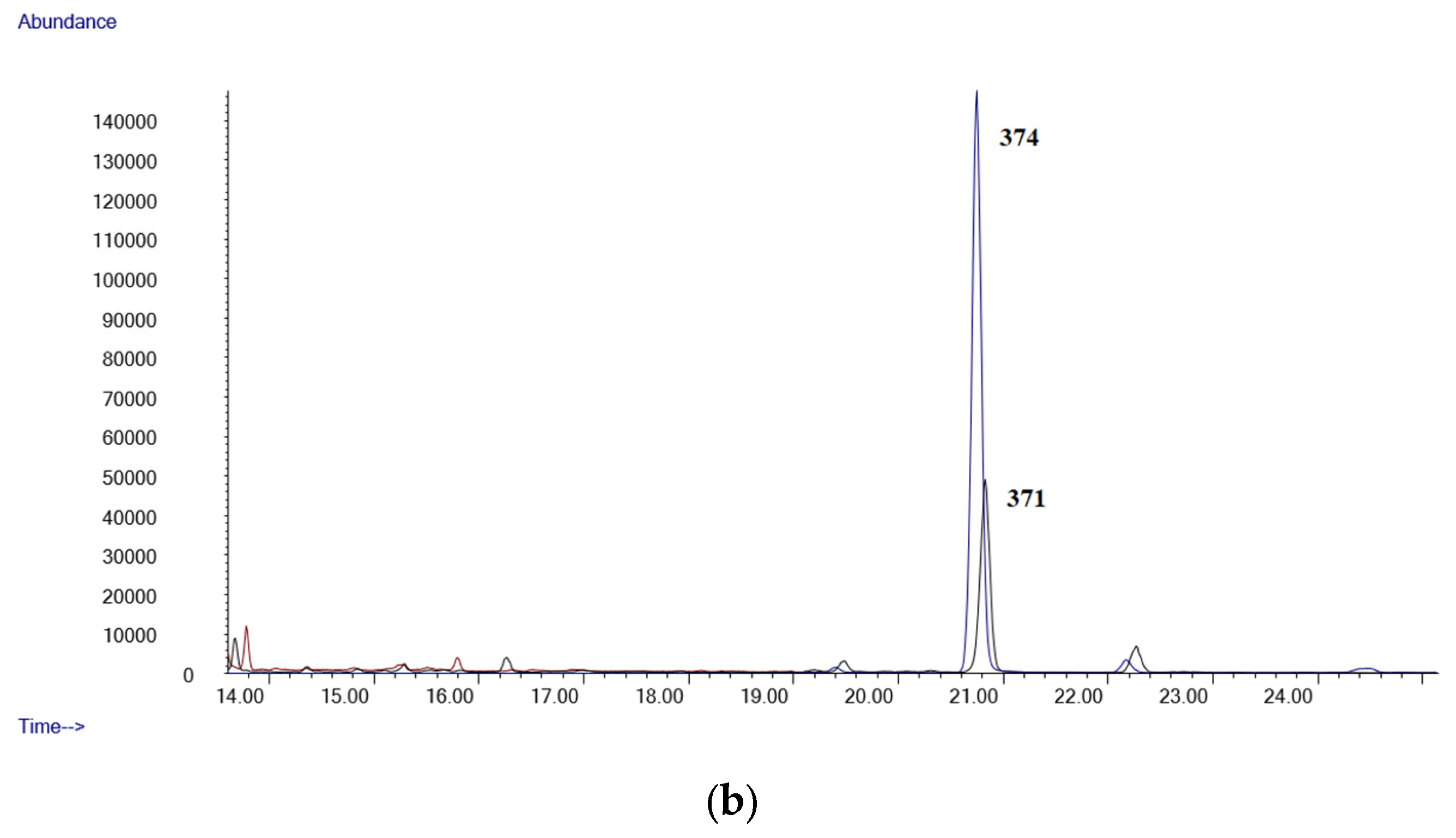
| LOD (µg/mL) | LLOQ (µg/mL) | Slope | Intercept | R2 Coef | |
|---|---|---|---|---|---|
| Δ9-THC | 0.02 | 0.05 | 4.455 | −0.474 | 0.991 |
| Δ9-THC-COOH | 0.04 | 0.08 | 1.754 | −0.234 | 0.992 |
| Intraday (n = 5) | Interday (n = 5) | ||||
|---|---|---|---|---|---|
| Concentration (µg/mL) | CV (%) | Relative Mean Error (%) | CV (%) | Relative Mean Error (%) | |
| Δ9-THC | 0.1 | 0.43 | 10.14 | 5.71 | 13.45 |
| 0.5 | 1.08 | 0.65 | 10.30 | 10.10 | |
| 1 | 2.12 | 7.74 | 1.13 | −3.13 | |
| Δ9-THC-COOH | 0.1 | 12.92 | −8.78 | 13.40 | 10.41 |
| 0.5 | 1.75 | 12.48 | 12.36 | −4.51 | |
| 1 | 6.58 | 13.80 | 8.87 | −0.12 | |
| Intraday (n = 5) | Interday (n = 5) | ||||
|---|---|---|---|---|---|
| Concentration (µg/mL) | Mean Recovery (%) | CV (%) | Mean Recovery (%) | CV (%) | |
| Δ9-THC | 0.1 | 82.95 | 10.22 | 83.54 | 5.79 |
| 1 | 81.51 | 7.28 | 80.69 | 8.28 | |
| Δ9-THC-COOH | 0.1 | 95.41 | 3.83 | 94.21 | 4.18 |
| 1 | 81.01 | 9.81 | 86.12 | 6.82 | |
| Case Number | Gender | Age | Cause of Death | [Δ9-THC] (µg/mL) | [Δ9-THC-COOH] (µg/mL) | Other Detected Substances |
|---|---|---|---|---|---|---|
| 1 | M | 37 | Traffic Accident | <LLOQ | 0.26 | - |
| 2 | M | 48 | Drug Overdose | - | 0.28 | Methadone, Benzodiacepines |
| 3 | M | 50 | Natural Death | - | 0.30 | |
| 4 | M | 42 | Traffic Accident | - | 0.12 | Ethanol |
| 5 | M | 42 | Natural Death | - | 0.14 | Ethanol, Paracetamol |
| 6 | M | 23 | Suicide. Hanging | - | 0.22 | - |
| 7 | F | 45 | Natural Death | - | 0.83 | - |
| 8 | M | 23 | Traffic Accident | 0.11 | 0.37 | Ethanol |
| 9 | M | 40 | Drug Overdose | - | 0.26 | Cocaine, Benzoilecgonine |
| 10 | M | 53 | Natural Death | 0.12 | 0.53 | Methadone |
| 11 | M | 46 | Suicide. Hanging | - | 0.17 | - |
| 12 | M | 52 | Drug Overdose | - | 0.19 | Ethanol, Methadone, Benzodiacepines |
| 13 | M | 43 | Natural Death | 0.11 | 0.83 | - |
| 14 | M | 57 | Drowning | - | 0.26 | - |
| 15 | M | 61 | Drowning | - | 0.32 | - |
| 16 | M | 40 | Drug Overdose | - | 0.20 | Benzoilecgonine, Citalopram |
| 17 | M | 48 | Drug Overdose | - | 0.38 | Ethanol, Cocaine, Benzoilecgonine, Methadone |
| 18 | F | 37 | Drowning | 0.11 | 0.28 | - |
| 19 | M | 40 | Drug Overdose | 0.12 | 0.22 | Cocaine, Benzoilecgonine |
| 20 | M | 35 | Suicide. Intoxication | - | 0.16 | Venlafaxine, Cocaine, Benzoilecgonine, Bupropion |
| 21 | M | 60 | Gas Intoxication | 0.14 | 3.26 | Carboxyhemoglobin |
| 22 | M | 53 | Natural Death | - | 0.40 | - |
| 23 | M | 19 | Traffic Accident | - | 3.11 | - |
| 24 | M | 37 | Suicide. Precipitation | - | 3.38 | Ethanol, Cocaine Benzoilecgonine |
| 25 | M | 47 | Drug Overdose | 0.11 | 1.77 | Cocaine, Benzoilecgonine |
| 26 | M | 48 | Drug Overdose | - | 0.65 | Ethanol, Cocaine, Benzoilecgonine |
| 27 | M | 35 | Drug Overdose | 0.13 | 1.14 | Ethanol, Methadone, Gabapentine |
| 28 | M | 23 | Traffic Accident | - | 0.43 | Ethanol |
| 29 | F | 22 | Suicide. Hanging | 0.12 | 0.25 | |
| 30 | M | 39 | Natural Death | 0.11 | 0.32 | |
| 31 | F | 18 | Traffic Accident | 0.13 | 1.15 | - |
| 32 | M | 59 | Drug Overdose | - | 0.15 | Benzodiacepines, methadone, chlometiazole, trazodone |
| 33 | M | 38 | Suicide. Hanging | - | 0.36 | Cocaine and metabolites |
| 34 | F | 43 | Drug Overdose | 0.12 | 0.16 | Cocaine and metabolites, methadone |
| 35 | M | 47 | Traffic Accident | - | 0.37 | - |
| 36 | F | 18 | Suicide. Precipitation | 0.12 | 0.25 | Ethanol |
| 37 | F | 38 | Traffic Accident. Pedestrian | - | 0.20 | Ethanol, Cocaine and metabolites |
| 38 | M | 39 | Drug Overdose | - | 0.50 | Cocaine and metabolites |
| 39 | F | 42 | Stabbed | - | 0.32 | Ethanol |
| 40 | M | 60 | Natural Death | 0.17 | 0.21 | - |
| 41 | M | 44 | Natural Death | - | 0.22 | - |
| Retention Time (min) | Quantifier Ion (m/z) | Qualifiers Ions (m/z) | |
|---|---|---|---|
| THC | 14.80 | 299 | 271, 314 |
| THC-D3 | 14.80 | 302 | 274, 317 |
| THC-COOH | 20.4 | 371 | 473, 488 |
| THC-COOH-D3 | 20.4 | 374 | 476, 491 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Álvarez-Freire, I.; Valeiras-Fernández, A.; Cabarcos-Fernández, P.; Bermejo-Barrera, A.M.; Tabernero-Duque, M.J. Simple Method for the Determination of THC and THC-COOH in Human Postmortem Blood Samples by Gas Chromatography—Mass Spectrometry. Molecules 2023, 28, 3586. https://doi.org/10.3390/molecules28083586
Álvarez-Freire I, Valeiras-Fernández A, Cabarcos-Fernández P, Bermejo-Barrera AM, Tabernero-Duque MJ. Simple Method for the Determination of THC and THC-COOH in Human Postmortem Blood Samples by Gas Chromatography—Mass Spectrometry. Molecules. 2023; 28(8):3586. https://doi.org/10.3390/molecules28083586
Chicago/Turabian StyleÁlvarez-Freire, Ivan, Anxa Valeiras-Fernández, Pamela Cabarcos-Fernández, Ana María Bermejo-Barrera, and María Jesús Tabernero-Duque. 2023. "Simple Method for the Determination of THC and THC-COOH in Human Postmortem Blood Samples by Gas Chromatography—Mass Spectrometry" Molecules 28, no. 8: 3586. https://doi.org/10.3390/molecules28083586
APA StyleÁlvarez-Freire, I., Valeiras-Fernández, A., Cabarcos-Fernández, P., Bermejo-Barrera, A. M., & Tabernero-Duque, M. J. (2023). Simple Method for the Determination of THC and THC-COOH in Human Postmortem Blood Samples by Gas Chromatography—Mass Spectrometry. Molecules, 28(8), 3586. https://doi.org/10.3390/molecules28083586








