Thymosin α1 and Its Role in Viral Infectious Diseases: The Mechanism and Clinical Application
Abstract
1. Introduction
2. Overview of Tα1
2.1. Properties of Tα1
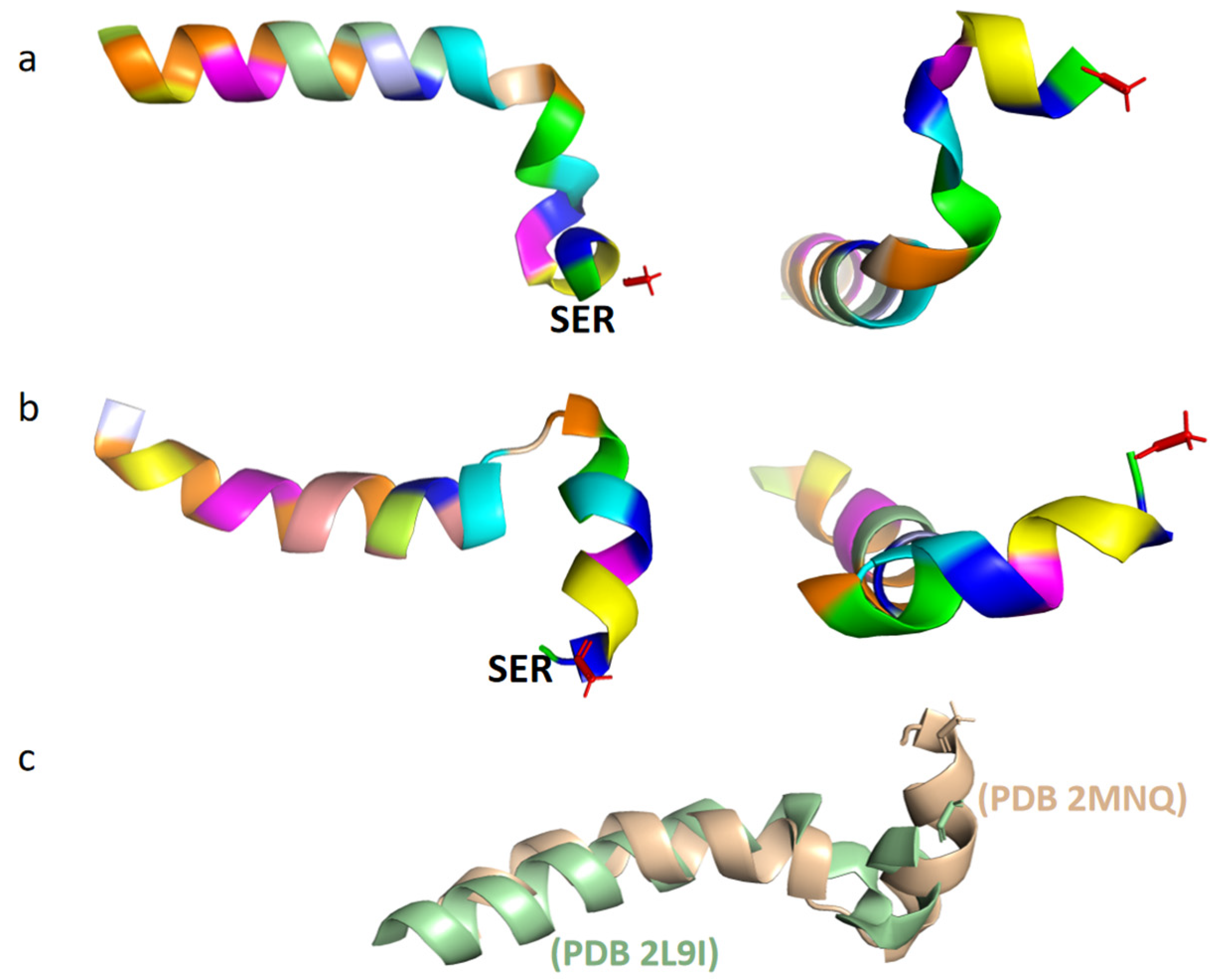
2.2. The Structure of Tα1
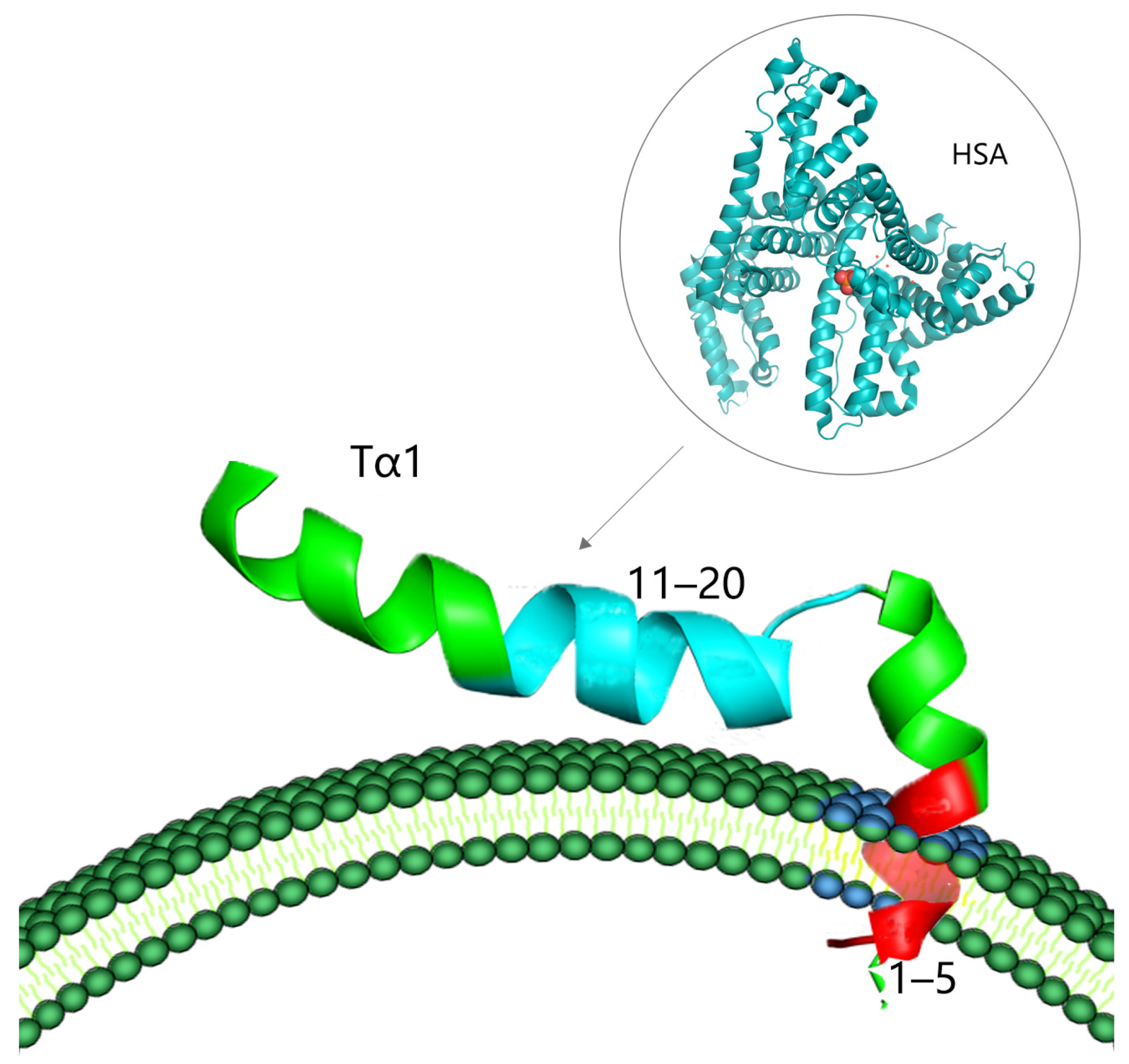
2.3. The Protein Binding Properties and Biosafety of Tα1
2.4. The Immunomodulatory Mechanism of Tα1
2.4.1. The Effects of Tα1 on Immune Cell
2.4.2. The Effects of Tα1 on Inflammation Related Signaling Pathways
3. The Application of Tα1 in Viral Diseases and Its Complications
3.1. COVID-19
3.2. Other Viral Diseases and Its Complications
3.2.1. Hepatitis B
3.2.2. Hepatitis C
3.2.3. AIDS
3.2.4. Sepsis
| Disease | Dosage Regimen | Subjects with Tα1/Subjects | Main Results | Published Year |
|---|---|---|---|---|
| HBV | Tα1 + entecavir | 345/690 |
| 2018 [33] |
| Tα1 | 228/468 |
| 2021 [92] | |
| Tα1 | 146/558 |
| 2016 [93] | |
| Tα1 + PEG-IFN-α-2a | 26/51 |
| 2012 [94] | |
| Tα1 | 25/40 |
| 2010 [95] | |
| Tα1 + lavmidine | 34/67 |
| 2008 [96] | |
| HCV | Tα1 | 12/24 |
| 2010 [38] |
| Tα1 + IFN | 22/41 |
| 2003 [97] | |
| AIDS | Tα1 | 6/12 |
| 1996 [98] |
| Tα1 | 13/20 |
| 2003 [99] |
4. Conclusions & Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yue, S.; Zheng, X.; Zheng, Y. Cell-type-specific role of lamin-B1 in thymus development and its inflammation-driven reduction in thymus aging. Aging Cell 2019, 18, e12952. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; He, K.; Hoffman, R.D.; Ying, Y.; Tao, N.; Guo, W.; Shen, J.; Liu, X.; Li, M.; Yan, M.; et al. Thymosin Beta 15 Alters the Spatial Development of Thymic Epithelial Cells. Cells 2022, 11, 3679. [Google Scholar] [CrossRef]
- Xu, M.; Gan, T.; Ning, H.; Wang, L. MicroRNA Functions in Thymic Biology: Thymic Development and Involution. Front. Immunol. 2018, 9, 2063. [Google Scholar] [CrossRef]
- Gramenzi, A.; Cursaro, C.; Andreone, P.; Bernardi, M. Thymalfasin: Clinical pharmacology and antiviral applications. Biodrugs 1998, 9, 477–486. [Google Scholar] [CrossRef]
- Shi, D.; Shui, Y.; Xu, X.; He, K.; Yang, F.; Gao, J. Thymic function affects breast cancer development and metastasis by regulating expression of thymus secretions PTMα and Tβ15b1. Transl. Oncol. 2021, 14, 100980. [Google Scholar] [CrossRef]
- Goldstein, A.L.; Slater, F.D.; White, A. Preparation, assay, and partial purification of a thymic lymphocytopoietic factor (thymosin). Proc. Natl. Acad. Sci. USA 1966, 56, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Ying, Y.; Lin, C.; Tao, N.; Hoffman, R.D.; Shi, D.; Chen, Z. Thymosin β4 and actin: Binding modes, biological functions and clinical applications. Curr. Protein Pept. Sci. 2023, 24, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, A.L.; Guha, A.; Zatz, M.M.; Hardy, M.A.; White, A. Purification and Biological Activity of Thymosin, a Hormone of the Thymus Gland. Proc. Natl. Acad. Sci. USA 1972, 69, 1800–1803. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, A.L.; Low, T.L.; McAdoo, M.; McClure, J.; Thurman, G.B.; Rossio, J.; Lai, C.Y.; Chang, D.; Wang, S.S.; Harvey, C.; et al. Thymosin alpha1: Isolation and sequence analysis of an immunologically active thymic polypeptide. Proc. Natl. Acad. Sci. USA 1977, 74, 725–729. [Google Scholar] [CrossRef]
- Haritos, A.A.; Tsolas, O.; Horecker, B.L. Distribution of prothymosin alpha in rat tissues. Proc. Natl. Acad. Sci. USA 1984, 81, 1391–1393. [Google Scholar] [CrossRef]
- Lao, X.; Liu, M.; Chen, J.; Zheng, H. A Tumor-Penetrating Peptide Modification Enhances the Antitumor Activity of Thymosin Alpha 1. PLoS ONE 2013, 8, e72242. [Google Scholar] [CrossRef] [PubMed]
- Barcia, M.G.; Castro, J.M.; Jullien, C.D.; González, C.G.; Freire, M. Prothymosin α is phosphorylated by casein kinase-2. FEBS Lett. 1992, 312, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Low, T.L.; Goldstein, A.L. The chemistry and biology of thymosin. II. Amino acid sequence analysis of thymosin alpha1 and polypeptide beta1. J. Biol. Chem. 1979, 254, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Haritos, A.; Yialouris, P.; Heimer, E.; Felix, A.; Rosemeyer, M. On the molecular size of thymosins. FEBS Lett. 1987, 218, 107–112. [Google Scholar] [CrossRef]
- Elizondo-Riojas, M.-A.; Chamow, S.M.; Tuthill, C.W.; Gorenstein, D.G.; Volk, D.E. NMR structure of human thymosin alpha-1. Biochem. Biophys. Res. Commun. 2011, 416, 356–361. [Google Scholar] [CrossRef]
- Hoch, K.; Volk, D. Structures of Thymosin Proteins. Vitam. Horm. 2016, 102, 1–24. [Google Scholar] [CrossRef]
- Mandaliti, W.; Nepravishta, R.; Vallebona, P.S.; Pica, F.; Garaci, E.; Paci, M. New studies about the insertion mechanism of Thymosin α1 in negative regions of model membranes as starting point of the bioactivity. Amino Acids 2016, 48, 1231–1239. [Google Scholar] [CrossRef]
- Armirotti, A.; Tomati, V.; Matthes, E.; Veit, G.; Cholon, D.M.; Phuan, P.-W.; Braccia, C.; Guidone, D.; Gentzsch, M.; Lukacs, G.L.; et al. Bioactive Thymosin Alpha-1 Does Not Influence F508del-CFTR Maturation and Activity. Sci. Rep. 2019, 9, 10310. [Google Scholar] [CrossRef]
- Giuliani, C.; Napolitano, G.; Mastino, A.; Di Vincenzo, S.; D’Agostini, C.; Grelli, S.; Bucci, I.; Singer, D.S.; Kohn, L.D.; Monaco, F.; et al. Thymosin-alpha1 regulates MHC class I expression in FRTL-5 cells at tran-scriptional level. Eur. J. Immunol. 2000, 30, 778–786. [Google Scholar] [CrossRef]
- Grottesi, A.; Sette, M.; Palamara, A.T.; Rotilio, G.; Garaci, E.; Paci, M. The conformation of peptide thymosin α1 in solution and in a membrane-like environment by circular dichroism and NMR spectroscopy. a possible model for its interaction with the lymphocyte membrane. Peptides 1998, 19, 1731–1738. [Google Scholar] [CrossRef]
- Nepravishta, R.; Mandaliti, W.; Eliseo, T.; Vallebona, P.S.; Pica, F.; Garaci, E.; Paci, M. Thymosin α1 inserts N terminus into model membranes assuming a helical conformation. Expert Opin. Biol. Ther. 2015, 15 (Suppl. S1), S71–S81. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Chan, J.; Dragoi, A.-M.; Gong, X.; Ivanov, S.; Li, Z.-W.; Chuang, T.; Tuthill, C.; Wan, Y.; Karin, M.; et al. Activation of IKK by thymosin α1 requires the TRAF6 signalling pathway. EMBO Rep. 2005, 6, 531–537. [Google Scholar] [CrossRef]
- Mandaliti, W.; Nepravishta, R.; Vallebona, P.S.; Pica, F.; Garaci, E.; Paci, M. Thymosin α1 Interacts with Exposed Phosphatidylserine in Membrane Models and in Cells and Uses Serum Albumin as a Carrier. Biochemistry 2016, 55, 1462–1472. [Google Scholar] [CrossRef]
- Mandaliti, W.; Nepravishta, R.; Pica, F.; Vallebona, P.S.; Garaci, E.; Paci, M. Thymosin α1 Interacts with Hyaluronic Acid Electrostatically by Its Terminal Sequence LKEKK. Molecules 2017, 22, 1843. [Google Scholar] [CrossRef]
- Li, W.; Song, L.; Wu, S.; Xue, X.; Zhang, L.; He, L.; Han, W.; Wang, Q.; Ling, R.; Zhang, W.; et al. Expression, Purification and Characterization of A Novel Soluble Human Thymosin Alpha1 Concatemer Exhibited A Stronger Stimulation on Mice Lymphocytes Proliferation and Higher Anti-tumor Activity. Int. J. Biol. Sci. 2011, 7, 618–628. [Google Scholar] [CrossRef]
- Rost, K.L.; Wierich, W.; Masayuki, F.; Tuthill, C.W.; Horwitz, D.L.; Herrmann, W.M. Pharmacokinetics of thymosin alpha1 after subcutaneous injection of three dif-ferent formulations in healthy volunteers. Int. J. Clin. Pharmacol. Ther. 1999, 37, 51–57. [Google Scholar] [PubMed]
- Ancell, C.D.; Phipps, J.; Young, L. Thymosin alpha-1. Am. J. Health Pharm. 2001, 58, 886–888. [Google Scholar] [CrossRef]
- Këlliçi, S.; Burazeri, G. Thymosin alpha1: A promising molecule for important clinical applications. Med. Arh. 2009, 63, 48–50. [Google Scholar] [PubMed]
- Garaci, E.; Pica, F.; Serafino, A.; Balestrieri, E.; Matteucci, C.; Moroni, G.; Sorrentino, R.; Zonfrillo, M.; Pierimarchi, P.; Vallebona, P.S. Thymosin α1 and cancer: Action on immune effector and tumor target cells. Ann. N. Y. Acad. Sci. 2012, 1269, 26–33. [Google Scholar] [CrossRef] [PubMed]
- King, R.; Tuthill, C. Immune Modulation with Thymosin Alpha 1 Treatment. Vitam. Horm. 2016, 102, 151–178. [Google Scholar] [CrossRef]
- Garaci, E. Thymosin 1: A Historical Overview. Ann. N. Y. Acad. Sci. 2007, 1112, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Garaci, E.; Favalli, C.; Pica, F.; Vallebona, P.S.; Palamara, A.T.; Matteucci, C.; Pierimarchi, P.; Serafino, A.; Mastino, A.; Bistoni, F.; et al. Thymosin Alpha 1: From Bench to Bedside. Ann. N. Y. Acad. Sci. 2007, 1112, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Shi, Y.; Zhou, J.; Sun, Y.; Piao, H.; Jiang, W.; Ma, A.; Chen, Y.; Xu, M.; Xie, W.; et al. Combination of entecavir with thymosin alpha-1 in HBV-related compensated cirrhosis: A prospective multicenter randomized open-label study. Expert Opin. Biol. Ther. 2018, 18 (Suppl. S1), 61–69. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Chen, F.-D.; Zhou, L.; Gong, X.-G.; Han, Q.-F. Proliferative and anti-proliferative effects of thymosin α1 on cells are associated with manipulation of cellular ROS levels. Chem. Interact. 2009, 180, 383–388. [Google Scholar] [CrossRef]
- Hong, K.S.; Pagan, K.; Whalen, W.; Harris, R.; Yang, J.; Stout-Delgado, H.; Cho, S.J. The Role of Glutathione Reductase in Influenza Infection. Am. J. Respir. Cell Mol. Biol. 2022, 67, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Chien, R.-N.; Liaw, Y.-F. Thymalfasin for the treatment of chronic hepatitis B. Expert Rev. Anti Infect. Ther. 2004, 2, 9–16. [Google Scholar] [CrossRef]
- Chen, M.; Jiang, Y.; Cai, X.; Lu, X.; Chao, H. Combination of Gemcitabine and Thymosin alpha 1 exhibit a better anti-tumor effect on nasal natural killer/T-cell lymphoma. Int. Immunopharmacol. 2021, 98, 107829. [Google Scholar] [CrossRef]
- Grandini, E.; Cannoletta, F.; Scuteri, A.; Fortini, C.; Loggi, E.; Cursaro, C.; Riili, A.; Di Donato, R.; Gramenzi, A.; Bernardi, M.; et al. Immunological modifications during treatment with thymosin α1 plus antiviral therapy in chronic hepatitis C. Ann. N. Y. Acad. Sci. 2010, 1194, 147–152. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, Z.; Zhou, C.; Yao, H.; Li, M.; Xu, C. The modulation of thymosin alpha 1 in the maturation, differentiation and function of murine bone marrow-derived dendritic cells in the absence or presence of tumor necrosis factor-alpha. Int. Immunopharmacol. 2004, 4, 539–546. [Google Scholar] [CrossRef]
- Romani, L.; Bistoni, F.; Gaziano, R.; Bozza, S.; Montagnoli, C.; Perruccio, K.; Pitzurra, L.; Bellocchio, S.; Velardi, A.; Rasi, G.; et al. Thymosin α 1 activates dendritic cells for antifungal Th1 resistance through Toll-like receptor signaling. Blood 2004, 103, 4232–4239. [Google Scholar] [CrossRef]
- Moretti, S.; Oikonomou, V.; Garaci, E.; Romani, L. Thymosin α1: Burying secrets in the thymus. Expert Opin. Biol. Ther. 2015, 15 (Suppl. S1), S51–S58. [Google Scholar] [CrossRef] [PubMed]
- Romani, L.; Bistoni, F.; Perruccio, K.; Montagnoli, C.; Gaziano, R.; Bozza, S.; Bonifazi, P.; Bistoni, G.; Rasi, G.; Velardi, A.; et al. Thymosin α1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood 2006, 108, 2265–2274. [Google Scholar] [CrossRef] [PubMed]
- Romani, L.; Moretti, S.; Fallarino, F.; Bozza, S.; Ruggeri, L.; Casagrande, A.; Aversa, F.; Bistoni, F.; Velardi, A.; Garaci, E. Jack of all trades: Thymosin α1 and its pleiotropy. Ann. N. Y. Acad. Sci. 2012, 1269, 1–6. [Google Scholar] [CrossRef]
- Pierluigi, B.; D’angelo, C.; Fallarino, F.; Moretti, S.; Zelante, T.; Bozza, S.; De Luca, A.; Bistoni, F.; Garaci, E.; Romani, L. Thymosin α1: The regulator of regulators? Ann. N. Y. Acad. Sci. 2010, 1194, 1–5. [Google Scholar] [CrossRef]
- Wu, L.; Luo, P.-P.; Tian, Y.-H.; Chen, L.-Y.; Zhang, Y.-L. Clinical efficacy of thymosin alpha 1 combined with multi-modality chemotherapy and its effects on immune function of patients with pulmonary tuberculosis complicated with diabetes. Pak. J. Med. Sci. 2021, 38, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Hadden, J.W.; Verastegui, E.; Hadden, E. IRX-2 and Thymosin 1 (Zadaxin) Increase T Lymphocytes in T Lymphocytopenic Mice and Humans. Ann. N. Y. Acad. Sci. 2007, 1112, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Keam, S.; Megawati, D.; Patel, S.K.; Tiwari, R.; Dhama, K.; Harapan, H. Immunopathology and immunotherapeutic strategies in severe acute respiratory syndrome coronavirus 2 infection. Rev. Med. Virol. 2020, 30, e2123. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef]
- Xiong, W.; Liu, Z.-G.; Xiong, L.; Xiong, M.-C.; Lei, G.-H.; Wu, Y.; Zhao, Q. CD3(+), CD4(+), and CD8(+) Expression in Cells in Peripheral Blood of Silicosis Pa-tients and Intervention Effect of Thymalfasin. Ann. Clin. Lab. Sci. 2019, 49, 368–371. [Google Scholar]
- Zanetti, G.; Peserico, A.; Grosso, G.; Biffanti, S.; Sartori, E. Enhanced prevalence of red blood cell macrocytosis in psoriatic patients. A sign of ethanol abuse? Acta Derm. Venereol. Suppl. 1989, 146, 196–198. [Google Scholar]
- Peng, X.; Zhang, P.; Wang, X.; Chan, J.; Zhu, M.; Jiang, M.; Tuthill, C.; Wan, Y.; Dragoi, A.M.; Chu, W.-M. Signaling Pathways Leading to the Activation of IKK and MAPK by Thymosin 1. Ann. N. Y. Acad. Sci. 2007, 1112, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Serafino, A.; Andreola, F.; Pittaluga, E.; Krasnowska, E.K.; Nicotera, G.; Sferrazza, G.; Vallebona, P.S.; Pierimarchi, P.; Garaci, E. Thymosin α1 modifies podosome architecture and promptly stimulates the expression of podosomal markers in mature macrophages. Expert Opin. Biol. Ther. 2015, 15 (Suppl. S1), S101–S116. [Google Scholar] [CrossRef] [PubMed]
- Eisenbarth, S.C. Dendritic cell subsets in T cell programming: Location dictates function. Nat. Rev. Immunol. 2019, 19, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 2004, 4, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, A.; Paul, S. Involvement of mitogen-activated protein kinases in the signal transduction pathway of bone marrow-derived macrophage activation in response to in vitro treatment with thymosin alpha 1. Int. Immunopharmacol. 2002, 2, 47–58. [Google Scholar] [CrossRef]
- Li, M.; Feurino, L.W.; Li, F.; Wang, H.; Zhai, Q.; Fisher, W.E.; Chen, C.; Yao, Q. Thymosinα1 stimulates cell proliferation by activating ERK1/2, JNK, and increasing cytokine secretion in human pancreatic cancer cells. Cancer Lett. 2007, 248, 58–67. [Google Scholar] [CrossRef]
- Yao, Q.; Doan, L.X.; Zhang, R.; Bharadwaj, U.; Li, M.; Chen, C. Thymosin-α1 modulates dendritic cell differentiation and functional maturation from human peripheral blood CD14+ monocytes. Immunol. Lett. 2007, 110, 110–120. [Google Scholar] [CrossRef]
- Bozza, S.; Gaziano, R.; Bonifazi, P.; Zelante, T.; Pitzurra, L.; Montagnoli, C.; Moretti, S.; Castronari, R.; Sinibaldi, P.; Rasi, G.; et al. Thymosin 1 activates the TLR9/MyD88/IRF7-dependent murine cytomegalovirus sensing for induction of anti-viral responses in vivo. Int. Immunol. 2007, 19, 1261–1270. [Google Scholar] [CrossRef]
- Romani, L.; Bistoni, F.; Montagnoli, C.; Gaziano, R.; Bozza, S.; Bonifazi, P.; Zelante, T.; Moretti, S.; Rasi, G.; Garaci, E.; et al. Thymosin 1: An Endogenous Regulator of Inflammation, Immunity, and Tolerance. Ann. N. Y. Acad. Sci. 2007, 1112, 326–338. [Google Scholar] [CrossRef]
- Serafino, A.; Pica, F.; Andreola, F.; Gaziano, R.; Moroni, N.; Moroni, G.; Zonfrillo, M.; Pierimarchi, P.; Sinibaldi-Vallebona, P.; Garaci, E. Thymosin α1 Activates Complement Receptor-Mediated Phagocytosis in Human Monocyte-Derived Macrophages. J. Innate Immun. 2013, 6, 72–88. [Google Scholar] [CrossRef]
- Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef]
- Wong, C.K.; Lam, C.W.K.; Wu, A.K.L.; Ip, W.K.; Lee, N.L.S.; Chan, I.H.S.; Lit, L.C.W.; Hui, D.S.C.; Chan, M.H.M.; Chung, S.S.C.; et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin. Exp. Immunol. 2004, 136, 95–103. [Google Scholar] [CrossRef]
- Godeau, D.; Petit, A.; Richard, I.; Roquelaure, Y.; Descatha, A. Return-to-work, disabilities and occupational health in the age of COVID-19. Scand. J. Work. Environ. Health 2021, 47, 408–409. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Costantini, C.; van de Veerdonk, F.L.; Romani, L. COVID-19-Associated Pulmonary Aspergillosis: The Other Side of the Coin. Vaccines 2020, 8, 713. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Blish, C.A.; Sallusto, F.; Iwasaki, A. The immunology and immunopathology of COVID-19. Science 2022, 375, 1122–1127. [Google Scholar] [CrossRef]
- Matteucci, C.; Minutolo, A.; Balestrieri, E.; Petrone, V.; Fanelli, M.; Malagnino, V.; Ianetta, M.; Giovinazzo, A.; Barreca, F.; Di Cesare, S.; et al. Thymosin Alpha 1 Mitigates Cytokine Storm in Blood Cells From Coronavirus Disease 2019 Patients. Open Forum Infect. Dis. 2020, 8, ofaa588. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Fei, L.; Xu, W.; Li, W.; Xie, X.; Li, Q.; Chen, L. Efficacy Evaluation of Thymosin Alpha 1 in Non-severe Patients With COVID-19: A Retrospective Cohort Study Based on Propensity Score Matching. Front. Med. 2021, 8, 664776. [Google Scholar] [CrossRef]
- Yu, K.; He, J.; Wu, Y.; Xie, B.; Liu, X.; Wei, B.; Zhou, H.; Lin, B.; Zuo, Z.; Wen, W.; et al. Dysregulated adaptive immune response contributes to severe COVID-19. Cell Res. 2020, 30, 814–816. [Google Scholar] [CrossRef]
- Liu, Y.; Pang, Y.; Hu, Z.; Wu, M.; Wang, C.; Feng, Z.; Mao, C.; Tan, Y.; Liu, Y.; Chen, L.; et al. Thymosin Alpha 1 Reduces the Mortality of Severe Coronavirus Disease 2019 by Restoration of Lymphocytopenia and Reversion of Exhausted T Cells. Clin. Infect. Dis. 2020, 71, 2150–2157. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, J.; Zhu, C.; Liu, L.; Qi, T.; Shen, Y.; Zhang, Y.; Xu, L.; Li, T.; Qian, Z.; et al. Thymosin Alpha-1 Has no Beneficial Effect on Restoring CD4+ and CD8+ T Lymphocyte Counts in COVID-19 Patients. Front. Immunol. 2021, 12, 568789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.; Wang, W.-Y.; Pang, X.-C.; Wang, Z.; Wang, C.-Z.; Zhou, H.; Zheng, B.; Cui, Y.-M. Thymosin-α1 binds with ACE and downregulates the expression of ACE2 in human respiratory epithelia. Front. Biosci. 2022, 27, 48. [Google Scholar] [CrossRef] [PubMed]
- Kharazmi-Khorassani, J.; Asoodeh, A.; Tanzadehpanah, H. Antioxidant and angiotensin-converting enzyme (ACE) inhibitory activity of thymosin alpha-1 (Thα1) peptide. Bioorg. Chem. 2019, 87, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shen, Y.; Wen, Z.; Xu, Q.; Wu, Z.; Feng, H.; Li, Z.; Dong, X.; Huang, S.; Guo, J.; et al. Efficacy of Thymosin Alpha 1 in the Treatment of COVID-19: A Multicenter Cohort Study. Front. Immunol. 2021, 12, 673693. [Google Scholar] [CrossRef]
- Sun, Q.; Xie, J.; Zheng, R.; Li, X.; Chen, H.; Tong, Z.; Du, B.; Qiu, H.; Yang, Y. The effect of thymosin α1 on mortality of critical COVID-19 patients: A multicenter retrospective study. Int. Immunopharmacol. 2020, 90, 107143. [Google Scholar] [CrossRef]
- Wang, Y.; Yan, X.; Huang, C.; Sun, Y.; Yao, C.; Lin, Y.; Xiao, W. Risk factors of mortality and contribution of treatment in patients infected with COVID-19: A retrospective propensity score matched study. Curr. Med. Res. Opin. 2021, 37, 13–19. [Google Scholar] [CrossRef]
- Lin, P.; Chen, W.; Huang, H.; Lin, Y.; Cai, M.; Lin, D.; Cai, H.; Su, Z.; Zhuang, X.; Yu, X. Delayed discharge is associated with higher complement C3 levels and a longer nucleic acid-negative conversion time in patients with COVID-19. Sci. Rep. 2021, 11, 1233. [Google Scholar] [CrossRef]
- Wu, M.; Ji, J.-J.; Zhong, L.; Shao, Z.-Y.; Xie, Q.-F.; Liu, Z.-Y.; Wang, C.-L.; Su, L.; Feng, Y.-W.; Liu, Z.-F.; et al. Thymosin α1 therapy in critically ill patients with COVID-19: A multicenter retrospective cohort study. Int. Immunopharmacol. 2020, 88, 106873. [Google Scholar] [CrossRef]
- Siddiqi, H.K.; Mehra, M.R. COVID-19 illness in native and immunosuppressed states: A clinical–therapeutic staging proposal. J. Heart Lung Transplant. 2020, 39, 405–407. [Google Scholar] [CrossRef]
- Jose, R.J.; Manuel, A. COVID-19 cytokine storm: The interplay between inflammation and coagulation. Lancet Respir. Med. 2020, 8, e46–e47. [Google Scholar] [CrossRef]
- Chisari, F.V.; Ferrari, C. Hepatitis B Virus Immunopathogenesis. Annu. Rev. Immunol. 1995, 13, 29–60. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.-J.; Zhou, Z.-G.; Shen, F.; Li, A.-J.; Chen, M.-S.; Ying, M.-G.; Chen, Z.; Zhang, Y.-X.; Sun, H.-C.; Fan, J. A multicenter, randomized, observation-controlled clinical trial to evaluate the efficacy and safety of thymalfasin adjuvant therapy in patients with HBV-related HCC after curative resection—First announcement of the protocol. Expert Opin. Biol. Ther. 2015, 15 (Suppl. S1), S133–S137. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Chen, S.-R.; Lei, Z.-Y.; Cao, H.-J.; Zhang, S.-Q.; Weng, W.-Z.; Xiong, J.; Lin, D.-N.; Zhang, J.; Zheng, Y.-B.; et al. Safety and efficacy of Thymosin α1 in the treatment of hepatitis B virus-related acute-on-chronic liver failure: A randomized controlled trial. Hepatol. Int. 2022, 16, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Andreone, P.; Cursaro, C.; Gramenzi, A.; Buzzi, A.; Covarelli, M.G.; Giammarino, L.; Miniero, R.; Arienti, V.; Bernardi, M.; Gasbarrini, G. A double-blind, placebo-controlled, pilot trial of thymosin alpha 1 for the treatment of chronic hepatitis C. Liver Int. 2008, 16, 207–210. [Google Scholar] [CrossRef]
- Ciancio, A.; Andreone, P.; Kaiser, S.; Mangia, A.; Milella, M.; Solà, R.; Pol, S.; Tsianos, E.; De Rosa, A.; Camerini, R.; et al. Thymosin alpha-1 with peginterferon alfa-2a/ribavirin for chronic hepatitis C not responsive to IFN/ribavirin: An adjuvant role? J. Viral Hepat. 2012, 19 (Suppl. S1), 52–59. [Google Scholar] [CrossRef]
- McMichael, A.J.; Rowland-Jones, S.L. Cellular immune responses to HIV. Nature 2001, 410, 980–987. [Google Scholar] [CrossRef]
- Garaci, E.; Rocchi, G.; Perroni, L.; D’Agostini, C.; Soscia, F.; Grelli, S.; Mastino, A.; Favalli, C. Combination treatment with zidovudine, thymosin α1 and interferon-α in human immunodeficiency virus infectionand interferon-α in human immunodeficiency virus infection. Int. J. Clin. Lab. Res. 1994, 24, 23–28. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Sifaki, K.; Gumerova, N.I.; Giester, G.; Rompel, A. Synthesis and characterization of the Anderson–Evans tungstoantimonate [Na5(H2O)18{(HOCH2)2CHNH3}2][SbW6O24]. Acta Crystallogr. Sect. C Struct. Chem. 2021, 77, 420–425. [Google Scholar] [CrossRef]
- Christaki, E.; Anyfanti, P.; Opal, S.M. Immunomodulatory therapy for sepsis: An update. Expert Rev. Anti Infect. Ther. 2011, 9, 1013–1033. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, L.; Wang, J.; Gu, T.; Li, S. Clinical effect of Xuebijing combined with thymosinα1 on patients with severe pneumonia complicated with sepsis and its effect on serum inflammatory factors. Cell. Mol. Biol. 2022, 67, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Linye, H.; Zijing, X.; Wei, P.; Chao, H.; Chuan, L.; Tianfu, W. Thymosin alpha-1 therapy improves postoperative survival after curative resection for solitary hepatitis B virus-related hepatocellular carcinoma: A propensity score matching analysis. Medicine 2021, 100, e25749. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.-R.; Guo, Z.; Jiang, J.-H.; Xiang, B.-D.; Li, L.-Q. Thymosin α1 therapy subsequent to radical hepatectomy in patients with hepatitis B virus-associated hepatocellular carcinoma: A retrospective controlled study. Oncol. Lett. 2016, 12, 3513–3518. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Lee, Y.-J.; Kim, W.; Yoon, J.-H.; Jung, E.U.; Park, S.J.; Kim, Y.J.; Lee, H.-S. Efficacy of thymosin α-1 plus peginterferon α-2a combination therapy compared with peginterferon α-2a monotherapy in HBeAg-positive chronic hepatitis B: A prospective, multicenter, randomized, open-label study. Scand. J. Gastroenterol. 2012, 47, 1048–1055. [Google Scholar] [CrossRef]
- Jiang, Y.-F.; Ma, Z.-H.; Zhao, P.-W.; Pan, Y.; Liu, Y.-Y.; Feng, J.-Y.; Niu, J.-Q. Effect of Thymosin-α1 on T-helper 1 Cell and T-helper 2 Cell Cytokine Synthesis in Patients with Hepatitis B Virus e Antigen-positive Chronic Hepatitis B. J. Int. Med. Res. 2010, 38, 2053–2062. [Google Scholar] [CrossRef]
- Lee, H.W.; Lee, J.I.; Um, S.H.; Ahn, S.H.; Chang, H.Y.; Park, Y.K.; Hong, S.P.; Moon, Y.M.; Han, K.-H. Combination therapy of thymosin alpha-1 and lamivudine for HBeAg positive chronic hepatitis B: A prospective randomized, comparative pilot study. J. Gastroenterol. Hepatol. 2008, 23, 729–735. [Google Scholar] [CrossRef]
- Andreone, P.; Gramenzi, A.; Cursaro, C.; Felline, F.; Loggi, E.; D’Errico, A.; Spinosa, M.; Lorenzini, S.; Biselli, M.; Bernardi, M. Thymosin-alpha 1 plus interferon-alpha for naive patients with chronic hepatitis C: Results of a randomized controlled pilot trial. J. Viral Hepat. 2004, 11, 69–73. [Google Scholar] [CrossRef]
- Ramachandran, R.; Katzenstein, D.; Winters, M.A.; Kundu, S.K.; Merigan, T.C. Polyethylene Glycol-Modified Interleukin-2 and Thymosin 1 in Human Immunodeficiency Virus Type 1 Infection. J. Infect. Dis. 1996, 173, 1005–1008. [Google Scholar] [CrossRef]
- Chadwick, D.; Pido-Lopez, J.; Pires, A.; Imami, N.; Gotch, F.; Villacian, J.S.; Ravindran, S.; Paton, N.I. A pilot study of the safety and efficacy of thymosin α1 in augmenting immune reconstitution in HIV-infected patients with low CD4 counts taking highly active antiretroviral therapy. Clin. Exp. Immunol. 2003, 134, 477–481. [Google Scholar] [CrossRef]
- Bagwe, P.V.; Bagwe, P.V.; Ponugoti, S.S.; Joshi, S.V. Peptide-Based Vaccines and Therapeutics for COVID-19. Int. J. Pept. Res. Ther. 2022, 28, 1–16. [Google Scholar] [CrossRef]

| Dosage Regimen | Subjects with Tα1/Subjects | Main Results | Published Year |
|---|---|---|---|
| Tα1 | 269/317 |
| 2022 [76] |
| 306/1026 |
| 2021 [74] | |
| 126/275 |
| 2021 [71] | |
| 232/1388 |
| 2021 [68] | |
| 27/47 |
| 2021 [77] | |
| 327/771 |
| 2021 [75] | |
| 78/127 |
| 2020 [76] | |
| 36/76 |
| 2020 [70] | |
| 102/334 |
| 2020 [78] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tao, N.; Xu, X.; Ying, Y.; Hu, S.; Sun, Q.; Lv, G.; Gao, J. Thymosin α1 and Its Role in Viral Infectious Diseases: The Mechanism and Clinical Application. Molecules 2023, 28, 3539. https://doi.org/10.3390/molecules28083539
Tao N, Xu X, Ying Y, Hu S, Sun Q, Lv G, Gao J. Thymosin α1 and Its Role in Viral Infectious Diseases: The Mechanism and Clinical Application. Molecules. 2023; 28(8):3539. https://doi.org/10.3390/molecules28083539
Chicago/Turabian StyleTao, Nana, Xie Xu, Yuyuan Ying, Shiyu Hu, Qingru Sun, Guiyuan Lv, and Jianli Gao. 2023. "Thymosin α1 and Its Role in Viral Infectious Diseases: The Mechanism and Clinical Application" Molecules 28, no. 8: 3539. https://doi.org/10.3390/molecules28083539
APA StyleTao, N., Xu, X., Ying, Y., Hu, S., Sun, Q., Lv, G., & Gao, J. (2023). Thymosin α1 and Its Role in Viral Infectious Diseases: The Mechanism and Clinical Application. Molecules, 28(8), 3539. https://doi.org/10.3390/molecules28083539








