Golden Chanterelle or a Gold Mine? Metabolites from Aqueous Extracts of Golden Chanterelle (Cantharellus cibarius) and Their Antioxidant and Cytotoxic Activities
Abstract
1. Introduction
2. Results and Discussion
2.1. Phenolic Analysis
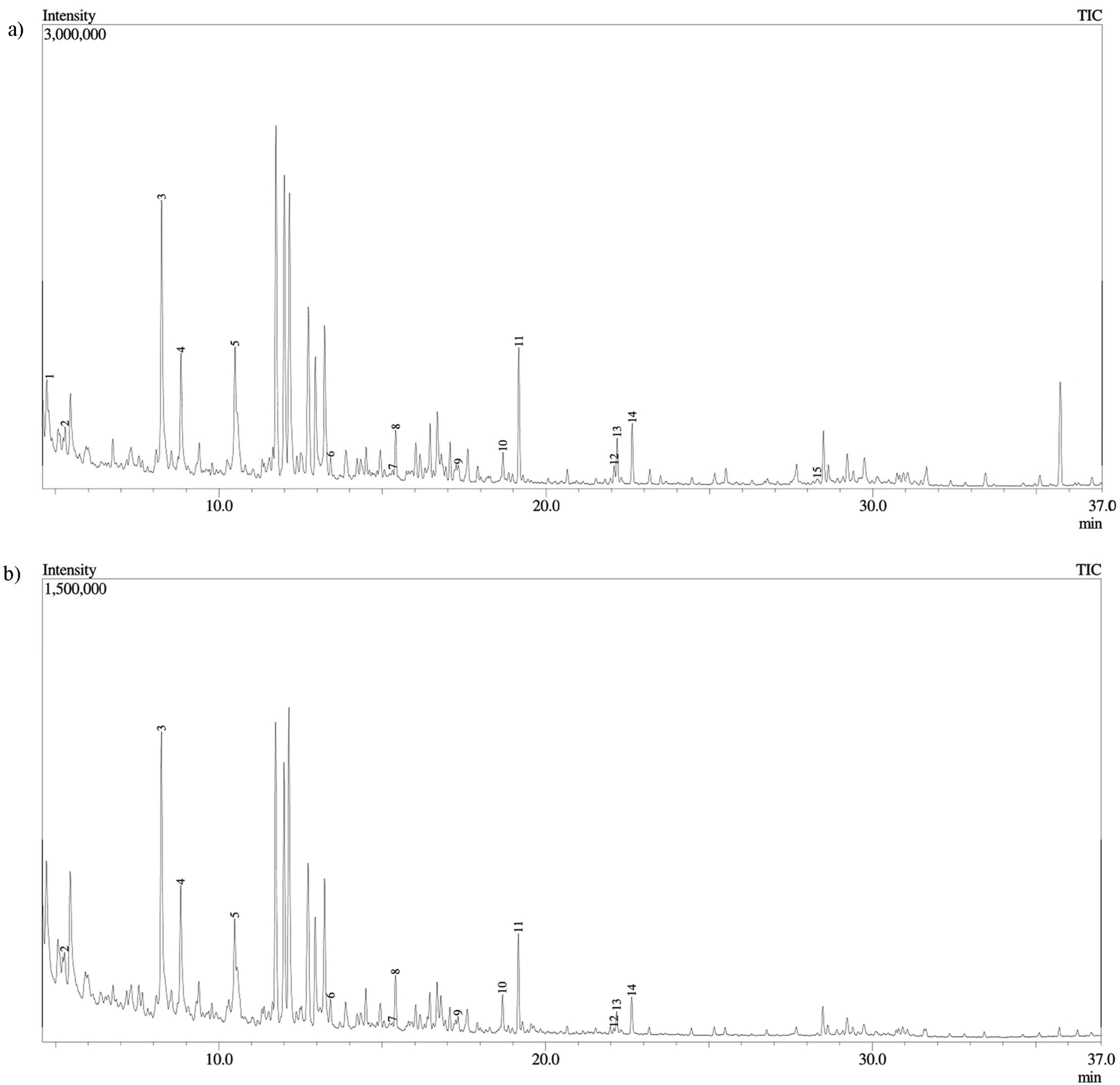
2.2. Derivatized Extract Analysis
2.3. Antioxidant Activity
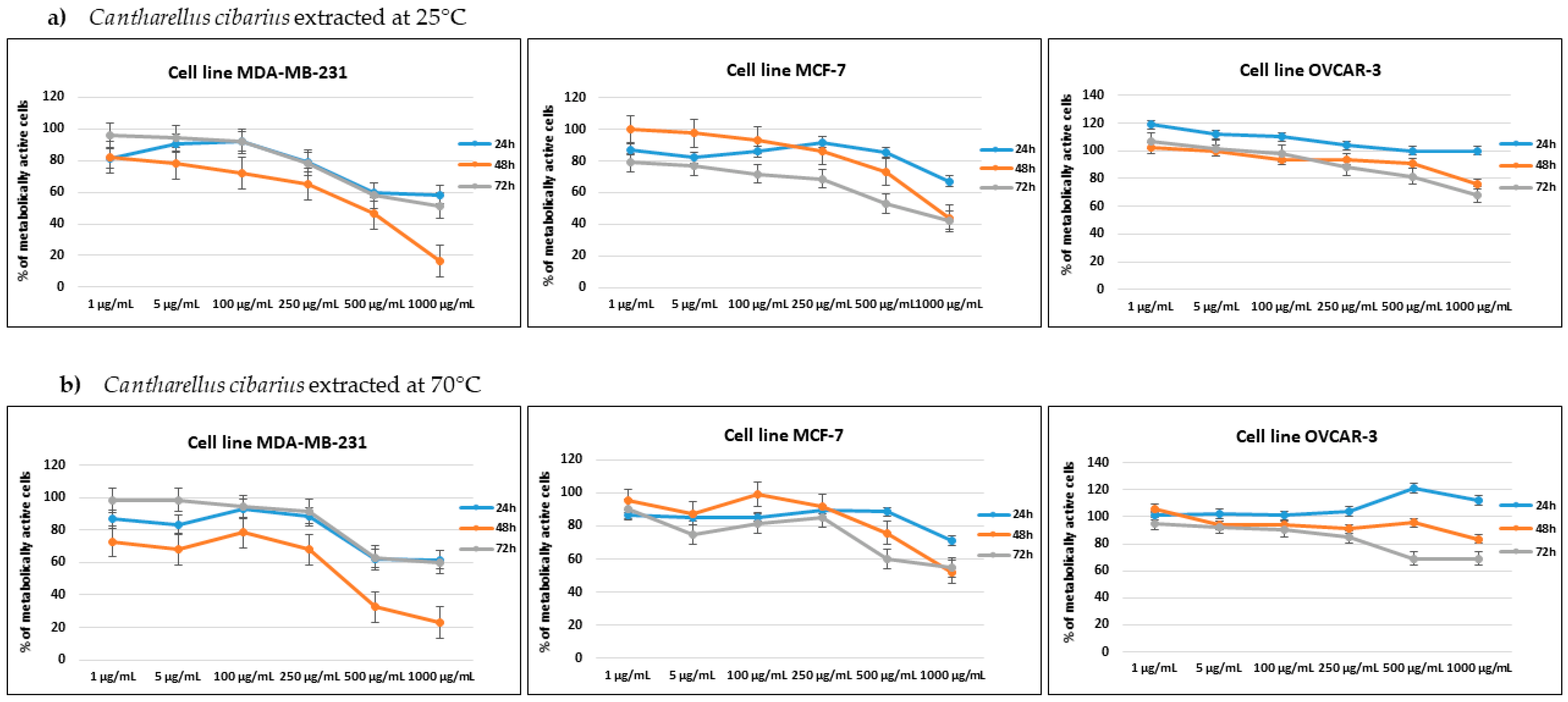
2.4. Cytotoxic Activity
3. Materials and Methods
3.1. Chemicals
3.2. Cantharellus Cibarius Aqueous Extract
3.3. HPLC Conditions for Phenols Determination
3.4. Extract Derivatization
3.5. GC-MS Conditions for Derivatized Extracts
3.6. Antioxidant Activity
3.6.1. Ferric Reducing Antioxidant Power (FRAP) Assay
3.6.2. Oxygen Radical Absorbance Capacity (ORAC) Assay
3.7. Cytotoxic Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Shirur, M.; Barh, A.; Annepu, S.K. Sustainable Production of Edible and Medicinal Mushrooms: Implications on Mushroom Consumption. In Climate Change and Resilient Food Systems: Issues, Challenges, and Way Forward; Hebsale Mallappa, V.K., Shirur, M., Eds.; Springer: Singapore, 2021; pp. 315–346. [Google Scholar]
- Beluhan, S.; Ranogajec, A. Chemical Composition and Non-Volatile Components of Croatian Wild Edible Mushrooms. Food Chem. 2011, 124, 1076–1082. [Google Scholar] [CrossRef]
- Lovrić, M.; Da Re, R.; Vidale, E.; Prokofieva, I.; Wong, J.; Pettenella, D.; Verkerk, P.J.; Mavsar, R. Collection and Consumption of Non-Wood Forest Products in Europe. For. J. For. Res. 2021, 94, 757–770. [Google Scholar] [CrossRef]
- Gałgowska, M.; Pietrzak-Fiećko, R. Evaluation of the Nutritional and Health Values of Selected Polish Mushrooms Considering Fatty Acid Profiles and Lipid Indices. Molecules 2022, 27, 6193. [Google Scholar] [CrossRef] [PubMed]
- Vamanu, E.; Niță, S. Bioactive Compounds, Antioxidant and Anti-Inflammatory Activities of Extracts From Cantharellus Cibarius. Rev. Chim. 2014, 65, 372–379. [Google Scholar]
- Muszyńska, B.; Grzywacz-Kisielewska, A.; Kała, K.; Gdula-Argasińska, J. Anti-Inflammatory Properties of Edible Mushrooms: A Review. Food Chem. 2018, 243, 373–381. [Google Scholar] [CrossRef]
- Muszyńska, B.; Sułkowska-Ziaja, K.; Ekiert, H. Analysis of Indole Compounds in Methanolic Extracts from the Fruiting Bodies of Cantharellus cibarius (the Chanterelle) and from the Mycelium of this Species Cultured in Vitro. J. Food Sci. Tech. 2013, 50, 1233–1237. [Google Scholar] [CrossRef]
- Phillips, K.M.; Ruggio, D.M.; Horst, R.L.; Minor, B.; Simon, R.R.; Feeney, M.J.; Byrdwell, W.C.; Haytowitz, D.B. Vitamin D and Sterol Composition of 10 Types of Mushrooms from Retail Suppliers in the United States. J. Agric. Food Chem. 2011, 59, 7841–7853. [Google Scholar] [CrossRef]
- Palacios, I.; Lozano, M.; Moro, C.; D’Arrigo, M.; Rostagno, M.A.; Martínez, J.A.; García-Lafuente, A.; Guillamón, E.; Villares, A. Antioxidant Properties of Phenolic Compounds Occurring in Edible Mushrooms. Food Chem. 2011, 128, 674–678. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal Mushroom Science: History, Current Status, Future Trends, and Unsolved Problems. Int. J. Med. Mushrooms 2010, 12, 1–16. [Google Scholar] [CrossRef]
- Novakovic, S.; Djekic, I.; Klaus, A.; Vunduk, J.; Djordjevic, V.; Tomović, V.; Šojić, B.; Kocić-Tanackov, S.; Lorenzo, J.M.; Barba, F.J.; et al. The Effect of Cantharellus cibarius Addition on Quality Characteristics of Frankfurter During Refrigerated Storage. Foods 2019, 8, 635. [Google Scholar] [CrossRef]
- Kothari, D.; Patel, S.; Kim, S.K. Anticancer and Other Therapeutic Relevance of Mushroom Polysaccharides: A Holistic Appraisal. Biomed. Pharmacother. 2018, 105, 377–394. [Google Scholar] [CrossRef]
- Moon, B.; Lo, Y.M. Conventional and Novel Applications of Edible Mushrooms in Today’s Food Industry. J. Food Proc. Preserv. 2014, 38, 2146–2153. [Google Scholar] [CrossRef]
- Ferreira, I.C.; Barros, L.; Abreu, R.M. Antioxidants in Wild Mushrooms. Curr. Med. Chem. 2009, 16, 1543–1560. [Google Scholar] [CrossRef]
- Islam, T.; Yu, X.; Xu, B. Phenolic Profiles, Antioxidant Capacities and Metal Chelating Ability of Edible Mushrooms Commonly Consumed in China. LWT—Food Sci. Techn. 2016, 72, 423–431. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Ferreira, I.C.; Baptista, P.; Santos-Buelga, C. Phenolic Acids Determination by HPLC-DAD-ESI/MS in Sixteen Different Portuguese Wild Mushrooms Species. Food Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Muszyńska, B.; Kała, K.; Firlej, A.; Sułkowska-Ziaja, K. Canthaerellus cibarius—Culinary-Medicinal Mushroom Content and Biological Activity. Acta Pol. Pharm. 2016, 73, 589–598. [Google Scholar]
- Muszyńska, B.; Sułkowska-Ziaja, K.; Ekiert, H. Phenolic Acids in Selected Edible Basidiomycota species: Armillaria mellea, Boletus badius, Boletus edulis, Cantharellus cibarius, Lactarius deliciosus and Pleurotus ostreatus. Acta Sci. Polonorum. Hortorum. Cultus. 2013, 12, 107–116. [Google Scholar]
- Dimopoulou, M.; Kolonas, A.; Mourtakos, S.; Androutsos, O.; Gortzi, O. Nutritional Composition and Biological Properties of Sixteen Edible Mushroom Species. Appl. Sci. 2022, 12, 8074. [Google Scholar] [CrossRef]
- Mwangi, R.W.; Macharia, J.M.; Wagara, I.N.; Bence, R.L. The Antioxidant Potential of Different Edible and Medicinal Mushrooms. Biomed. Pharmacoth. 2022, 147, 112621. [Google Scholar] [CrossRef]
- Kozarski, M.; Klaus, A.; Vunduk, J.; Zizak, Z.; Niksic, M.; Jakovljevic, D.; Vrvic, M.M.; Van Griensven, L.J. Nutraceutical Properties of the Methanolic Extract of Edible Mushroom Cantharellus cibarius (fries): Primary Mechanisms. Food Func. 2015, 6, 1875–1886. [Google Scholar] [CrossRef]
- Butkhup, L.; Samappito, W.; Jorjong, S. Evaluation of Bioactivities and Phenolic Contents of Wild Edible Mushrooms from Northeastern Thailand. Food Sci. Biotechnol. 2018, 27, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Xu, L.; Ballantyne, C.M. Dietary and Pharmacological Fatty Acids and Cardiovascular Health. J. Clin. Endoc. Metabol. 2020, 105, 1030–1045. [Google Scholar] [CrossRef] [PubMed]
- Antonyuk, V.; Panchak, L.V.; Antonyuk, L.Y.; Zyn, A.R. Extractive Substances of Fruit Body Golden Chanterelle (Cantharellus cibarius Fr.) and Hedgehog Mushroom (Hydnum repandum fr.). Emir. J. Food Agric. 2020, 32, 826–834. [Google Scholar] [CrossRef]
- Magdziak, Z.; Siwulski, M.; Mleczek, M. Characteristics of Organic Acid Profiles in 16 Species of Wild Growing Edible Mushrooms. J. Environ. Sci. Health Part B 2017, 52, 784–789. [Google Scholar] [CrossRef]
- Valentão, P.; Andrade, P.B.; Rangel, J.; Ribeiro, B.; Silva, B.M.; Baptista, P.; Seabra, R.M. Effect of the Conservation Procedure on the Contents of Phenolic Compounds and Organic Acids in Chanterelle (Cantharellus cibarius) Mushroom. J. Agric. Food Chem. 2005, 53, 4925–4931. [Google Scholar] [CrossRef]
- Nowacka-Jechalke, N.; Nowak, R.; Juda, M.; Malm, A.; Lemieszek, M.; Rzeski, W.; Kaczyński, Z. New Biological Activity of the Polysaccharide Fraction from Cantharellus cibarius and its Structural Characterization. Food Chem. 2018, 268, 355–361. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vannucci, L.; Sima, P.; Richter, J. Beta Glucan: Supplement or drug? From Laboratory to Clinical Trials. Molecules 2019, 24, 1251. [Google Scholar] [CrossRef]
- Kettawan, A.; Chanlekha, K.; Kongkachuichai, R.; Charoensiri, R. Effects of Cooking on Antioxidant Activities and Polyphenol Content of Edible Mushrooms Commonly Consumed in Thailand. Pak. J. Nutr. 2011, 10, 1094–1103. [Google Scholar] [CrossRef]
- Zulueta, A.; Esteve, M.J.; Frígola, A. ORAC and TEAC Assays Comparison to Measure the Antioxidant Capacity of Food Products. Food Chem. 2009, 114, 310–316. [Google Scholar] [CrossRef]
- Shah, P.; Modi, H.A. Comparative study of DPPH, ABTS and FRAP assays for Determination of Antioxidant Activity. Int. J. Res. Appl. Sci. Eng. Technol. 2015, 3, 636–641. [Google Scholar]
- Witkowska, A.M.; Zujko, M.E.; Mirończuk-Chodakowska, I. Comparative Study of Wild Edible Mushrooms as Sources of Antioxidants. Int. J. Med. Mushrooms 2011, 13, 335–341. [Google Scholar] [CrossRef]
- Piljac-Zegarac, J.; Samec, D.; Piljac, A.; Mesić, A.; Tkalcec, Z. Antioxidant Properties of Extracts of Wild Medicinal Mushroom Species from Croatia. Int. J. Med. Mushrooms 2011, 13, 257–263. [Google Scholar] [CrossRef]
- Zhou, X.; Guan, Q.; Wang, Y.; Lin, D.; Du, B. Effect of Different Cooking Methods on Nutrients, Antioxidant Activities and Flavors of Three Varieties of Lentinus edodes. Foods 2022, 11, 2713. [Google Scholar] [CrossRef]
- Lam, Y.S.; Okello, E.J. Determination of Lovastatin, β-Glucan, Total Polyphenols, and Antioxidant Activity in Raw and Processed Oyster Culinary-Medicinal Mushroom, Pleurotus ostreatus (Higher Basidiomycetes). Int. J. Med. Mushrooms 2015, 17, 117–128. [Google Scholar] [CrossRef]
- Flont, M.; Jastrzębska, E.; Brzózka, Z. Synergistic Effect of the Combination Therapy on Ovarian Cancer Cells under Microfluidic Conditions. Anal. Chim. Acta 2020, 1100, 138–148. [Google Scholar] [CrossRef]
- Nowakowski, P.; Markiewicz-Żukowska, R.; Bielecka, J.; Mielcarek, K.; Grabia, M.; Socha, K. Treasures from the Forest: Evaluation of Mushroom Extracts as Anti-Cancer Agents. Biomed. Pharmacother. 2021, 143, 112106. [Google Scholar] [CrossRef]
- Wasser, S. Medicinal Mushrooms as a Source of Antitumor and Immunomodulating Polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar]
- Lemieszek, M.K.; Marques, P.S.; Ribeiro, M.; Ferreira, D.; Marques, G.; Chaves, R.; Pożarowski, P.; Nunes, F.M.; Rzeski, W. Mushroom Small RNAs as Potential Anticancer Agents: A Closer Look at Cantharellus cibarius Proapoptotic and Antiproliferative Effects in Colon Cancer Cells. Food Funct. 2019, 10, 2739–2751. [Google Scholar] [CrossRef]
- Frleta, R.; Popović, M.; Smital, T.; Šimat, V. Comparison of Growth and Chemical Profile of Diatom Skeletonema grevillei in Bioreactor and Incubation-Shaking Cabinet in Two Growth Phases. Mar. Drugs 2022, 20, 697. [Google Scholar] [CrossRef]
- Lisec, J.; Schauer, N.; Kopka, J.; Willmitzer, L.; Fernie, A.R. Gas Chromatography Mass Spectrometry–Based Metabolite Profiling in Plants. Nat. Prot. 2006, 1, 387–396. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Čagalj, M.; Skroza, D.; Razola-Díaz, M.d.C.; Verardo, V.; Bassi, D.; Frleta, R.; Generalić Mekinić, I.; Tabanelli, G.; Šimat, V. Variations in the Composition, Antioxidant and Antimicrobial Activities of Cystoseira compressa during Seasonal Growth. Mar. Drugs 2022, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Popović, M.; Maravić, A.; Čikeš Čulić, V.; Đulović, A.; Burčul, F.; Blažević, I. Biological Effects of Glucosinolate Degradation Products from Horseradish: A Horse that Wins the Race. Biomolecules 2020, 10, 343. [Google Scholar] [CrossRef] [PubMed]

| Phenolic Compound | 25 °C | 70 °C | Concentration Ratio (25 °C/70 °C) | ||
|---|---|---|---|---|---|
| Concentration (mg/L) | Percentage (%) | Concentration (mg/L) | Percentage (%) | ||
| Gallic acid | 0.83 ± 0.00 | 1.01 ±0.02 | 0.86 ± 0.00 | 1.08 ± 0.02 | 0.97 |
| Protocatechuic acid | 0.97 ± 0.00 | 1.05 ± 0.04 | 1.02 ± 0.04 | 1.25 ± 0.02 | 0.95 |
| p-Hydroxybenzoic acid | 1.98 ± 0.02 | 0.85 ± 0.08 | 2.38 ± 0.08 | 1.02 ± 0.02 | 0,83 |
| Caffeic acid | 0.21 ± 0.00 | 0.9 ± 0.03 | 0.23 ±0.01 | 0.89 ± 0.01 | 0.91 |
| Ferulic acid | 0.28 ± 0.00 | 2.27 ± 0.15 | 0.39 ± 0.01 | 2.22 ± 0.1 | 0.72 |
| Quercetin | tr. | - | tr. | - | - |
| Rutin | tr. | - | tr. | - | - |
| Myricetin | 0.47 ± 0.00 | 0.28 ±0.01 | 0.47 ± 0.02 | 0.31 ± 0.00 | 1 |
| Antioxidant Assay | Cantharellus cibarius Aqueous Extract | |
|---|---|---|
| 25 °C | 70 °C | |
| ORAC (μM TE/L) | 19.71 ± 0.64 | 38.72 ± 2.47 |
| FRAP (μM TE/L) | 22.82 ± 0.94 | 41.54 ± 1.54 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Režić Mužinić, N.; Veršić Bratinčević, M.; Grubić, M.; Frleta Matas, R.; Čagalj, M.; Visković, T.; Popović, M. Golden Chanterelle or a Gold Mine? Metabolites from Aqueous Extracts of Golden Chanterelle (Cantharellus cibarius) and Their Antioxidant and Cytotoxic Activities. Molecules 2023, 28, 2110. https://doi.org/10.3390/molecules28052110
Režić Mužinić N, Veršić Bratinčević M, Grubić M, Frleta Matas R, Čagalj M, Visković T, Popović M. Golden Chanterelle or a Gold Mine? Metabolites from Aqueous Extracts of Golden Chanterelle (Cantharellus cibarius) and Their Antioxidant and Cytotoxic Activities. Molecules. 2023; 28(5):2110. https://doi.org/10.3390/molecules28052110
Chicago/Turabian StyleRežić Mužinić, Nikolina, Maja Veršić Bratinčević, Marina Grubić, Roberta Frleta Matas, Martina Čagalj, Tanja Visković, and Marijana Popović. 2023. "Golden Chanterelle or a Gold Mine? Metabolites from Aqueous Extracts of Golden Chanterelle (Cantharellus cibarius) and Their Antioxidant and Cytotoxic Activities" Molecules 28, no. 5: 2110. https://doi.org/10.3390/molecules28052110
APA StyleRežić Mužinić, N., Veršić Bratinčević, M., Grubić, M., Frleta Matas, R., Čagalj, M., Visković, T., & Popović, M. (2023). Golden Chanterelle or a Gold Mine? Metabolites from Aqueous Extracts of Golden Chanterelle (Cantharellus cibarius) and Their Antioxidant and Cytotoxic Activities. Molecules, 28(5), 2110. https://doi.org/10.3390/molecules28052110










