A Comprehensive Review of the Pharmacological Properties and Bioactive Components of Retama monosperma
Abstract
1. Introduction
2. Chemical Composition
2.1. Polyphenols, Flavonoids, and Tannins
2.2. Alkaloids
2.3. Fatty Acids
3. Biological Properties
3.1. Antioxidant and Anti-Aging Activity
3.2. Antimicrobial Activity
3.3. Antiproliferative and Antitumoral Activities
3.4. Anti-Inflammatory
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maghrani, M.; Michel, J.-B.; Eddouks, M. Hypoglycaemic Activity of Retama Raetam in Rats. Phytother. Res. 2005, 19, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Benkhouili, F.Z.; Moutawalli, A.; Benzeid, H.; Doukkali, A.; Zahidi, A. Retama Monosperma (L.) Boiss.: A Review of Its Uses in Traditional Medicine, Chemical Constituents, and Pharmacologic Activities. Phytomed. Plus 2022, 2, 100349. [Google Scholar] [CrossRef]
- Zefzoufi, M.; Fdil, R.; Bouamama, H.; Gadhi, C.; Katakura, Y.; Mouzdahir, A.; Sraidi, K. Effect of Extracts and Isolated Compounds Derived from Retama Monosperma (L.) Boiss. on Anti-Aging Gene Expression in Human Keratinocytes and Antioxidant Activity. J. Ethnopharmacol. 2021, 280, 114451. [Google Scholar] [CrossRef]
- Belayachi, L.; Aceves-Luquero, C.; Merghoub, N.; Bakri, Y.; Fernández de Mattos, S.; Amzazi, S.; Villalonga, P. Retama Monosperma N-Hexane Extract Induces Cell Cycle Arrest and Extrinsic Pathway-Dependent Apoptosis in Jurkat Cells. BMC Complement. Altern. Med. 2014, 14, 38. [Google Scholar] [CrossRef]
- Bouyahya, A.; Abrini, J.; Et-Touys, A.; Bakri, Y.; Dakka, N. Indigenous Knowledge of the Use of Medicinal Plants in the North-West of Morocco and Their Biological Activities. Eur. J. Integr. Med. 2017, 13, 9–25. [Google Scholar] [CrossRef]
- Bourais, I.; Elmarrkechy, S.; Taha, D.; Mourabit, Y.; Bouyahya, A.; el Yadini, M.; Machich, O.; el Hajjaji, S.; el Boury, H.; Dakka, N.; et al. A Review on Medicinal Uses, Nutritional Value, and Antimicrobial, Antioxidant, Anti-Inflammatory, Antidiabetic, and Anticancer Potential Related to Bioactive Compounds of J. Regia. Food Rev. Int. 2022, 1–51. [Google Scholar] [CrossRef]
- Elouafy, Y.; el Idrissi, Z.L.; el Yadini, A.; Harhar, H.; Alshahrani, M.M.; al Awadh, A.A.; Goh, K.W.; Ming, L.C.; Bouyahya, A.; Tabyaoui, M. Variations in Antioxidant Capacity, Oxidative Stability, and Physicochemical Quality Parameters of Walnut (Juglans regia) Oil with Roasting and Accelerated Storage Conditions. Molecules 2022, 27, 7693. [Google Scholar] [CrossRef]
- Elouafy, Y.; el Yadini, A.; el Moudden, H.; Harhar, H.; Alshahrani, M.M.; Awadh, A.A.A.; Goh, K.W.; Ming, L.C.; Bouyahya, A.; Tabyaoui, M. Influence of the Extraction Method on the Quality and Chemical Composition of Walnut (Juglans regia L.) Oil. Molecules 2022, 27, 7681. [Google Scholar] [CrossRef]
- Bouyahya, A.; Belmehdi, O.; Benjouad, A.; Ameziane El Hassani, R.; Amzazi, S.; Dakka, N.; Bakri, Y. Pharmacological Properties and Mechanism Insights of Moroccan Anticancer Medicinal Plants: What Are the next Steps? Ind. Crop. Prod. 2020, 147, 112198. [Google Scholar] [CrossRef]
- Benrahou, K.; Doudach, L.; Mrabti, H.N.; el Guourrami, O.; Zengin, G.; Bouyahya, A.; Cherrah, Y.; Faouzi, M.E.A. Acute Toxicity, Phenol Content, Antioxidant and Postprandial Anti-Diabetic Activity of Echinops Spinosus Extracts. Int. J. Second. Metab. 2022, 9, 91–102. [Google Scholar] [CrossRef]
- Benbacer, L.; Merghoub, N.; el Btaouri, H.; Gmouh, S.; Attaleb, M.; Morjani, H.; Amzazi, S.; el Mzibri, M. Antiproliferative Effect and Induction of Apoptosis by Inula viscosa L. and Retama monosperma L. Extracts in Human Cervical Cancer Cells. In Topics On Cervical Cancer With An Advocacy For Prevention; Rajkumar, R., Ed.; IntechOpen: London, UK, 2012; ISBN 978-953-51-0183-3. [Google Scholar]
- Bouyahya, A.; el Omari, N.; Elmenyiy, N.; Guaouguaou, F.E.; Balahbib, A.; Belmehdi, O.; Salhi, N.; Imtara, H.; Mrabti, H.N.; El-Shazly, M.; et al. Moroccan Antidiabetic Medicinal Plants: Ethnobotanical Studies, Phytochemical Bioactive Compounds, Preclinical Investigations, Toxicological Validations and Clinical Evidences; Challenges, Guidance and Perspectives for Future Management of Diabetes Worldwide. Trends Food Sci. Technol. 2021, 115, 147–254. [Google Scholar] [CrossRef]
- Belmokhtar, Z.; Harche, M.K. In Vitro Antioxidant Activity of Retama monosperma (L.) Boiss. Nat. Prod. Res. 2014, 28, 2324–2329. [Google Scholar] [CrossRef] [PubMed]
- el Hamdani, N.; Zefzoufi, M.; Derhali, S.; el Abbouyi, A.; el Khyari, S.; Mouzdahir, A.; Sraidi, K.; Harjane, T.; Fdil, R. Preliminary Phytochemical Analysis and Antibacterial Potential of Organic Extracts from Aerial Parts of Retama monosperma. J. Mater. Environ. Sci. 2018, 2508, 1889–1898. [Google Scholar]
- Belmokhtar, Z.; Kaid-Harche, M. An Overeview on Phytochemical Composition and Biological Activities of Retama monosperma L Boiss. (Fabaceae). PhytoChem. BioSub. J. 2016, 10, 701–731. [Google Scholar]
- González-Mauraza, H.; Martín-Cordero, C.; Alarcón-De-La-Lastra, C.; Rosillo, M.A.; León-González, A.J.; Sánchez-Hidalgo, M. Anti-Inflammatory Effects of Retama monosperma in Acute Ulcerative Colitis in Rats. J. Physiol. Biochem. 2013, 70, 163–172. [Google Scholar] [CrossRef]
- el Hamdani, N.; Filali-Ansari, N.; Fdil, R.; el Abbouyi, A.; el Khyari, S. Antifungal Activity of the Alkaloids Extracts from Aerial Parts of Retama monosperma. Res. J. Pharm. Biol. Chem. Sci. 2016, 7, 965–971. [Google Scholar]
- Fdil, R.; Tourabi, M.; Derhali, S.; Mouzdahir, A.; Sraidi, K.; Jama, C.; Zarrouk, A.; Bentiss, F. Evaluation of Alkaloids Extract of Retama monosperma (L.) Boiss. Stems as a Green Corrosion Inhibitor for Carbon Steel in Pickling Acidic Medium by Means of Gravimetric, AC Impedance and Surface Studies. J. Mater. Environ. Sci. 2018, 9, 358–369. [Google Scholar] [CrossRef]
- El-Shazly, A.; Ateya, A.M.; Witte, L.; Wink, M. Quinolizidine Alkaloid Profiles of Retama raetam, R. sphaerocarpa and R. monosperma. Zeitschrift für Naturforschung C 1996, 21–30, 301–308. [Google Scholar] [CrossRef]
- Abdelmadjide, S.; Mounir, A.; Atef, C.; Nadia, Z.; Neji, B. Phytochemical Study, Antioxidant and Antimicrobial Activities of Flavonoids and Diethyl Ether Extracts from Leaves and Seeds of Medicinal Plant of Algeria Flora: Retama monosperma (L.) Boiss. PONTE Int. Sci. Res. J. 2020, 76, 42–52. [Google Scholar] [CrossRef]
- León-González, A.J.; Navarro, I.; Acero, N.; Muñoz Mingarro, D.; Martín-Cordero, C. Genus Retama: A Review on Traditional Uses, Phytochemistry, and Pharmacological Activities. Phytochem. Rev. 2018, 17, 701–731. [Google Scholar] [CrossRef]
- Salatino, A.; Gottlieb, O.R. Quinolizidine Alkaloids as Systematic Markers of the Papilionoideae. Biochem. Syst. Ecol. 1980, 8, 133–147. [Google Scholar] [CrossRef]
- el Hamdani, N.; Fdil, R. Evaluation of Fatty Acids Profile and Mineral Content of Retama monosperma (L.) Boiss. of Morocco. J. Mater. Environ. Sci. 2015, 6, 538–545. [Google Scholar]
- Lee, K.W.; Lee, H.J.; Cho, H.Y.; Kim, Y.J. Role of the Conjugated Linoleic Acid in the Prevention of Cancer. Crit. Rev. Food Sci. Nutr. 2005, 45, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Fdil, R.; el Hamdani, N.; el Kihel, A.; Sraidi, K. Distribution of Alkaloids in the Aerial Parts of Retama monosperma (L.) Boiss. in Morocco|Distribution Des Alcaloïdes Dans Les Parties Aériennes de Retama monosperma (L.) Boiss. Du Maroc. Ann. De Toxicol. Anal. 2012, 24, 139–143. [Google Scholar] [CrossRef]
- el Hamdani, N.; Fdil, R.; Tourabi, M.; Jama, C.; Bentiss, F. Alkaloids Extract of Retama Monosperma (L.) Boiss. Seeds Used as Novel Eco-Friendly Inhibitor for Carbon Steel Corrosion in 1 M HCl Solution: Electrochemical and Surface Studies. Appl. Surf. Sci. 2015, 357, 1294–1305. [Google Scholar] [CrossRef]
- el Guiche, R.; Tahrouch, S.; Amri, O.; el Mehrach, K.; Hatimie, A. Antioxidant Activity and Total Phenolic and Flavonoid Contents of 30 Medicinal and Aromatic Plants Located in the South of Morocco. Int. J. New Technol. Res. IJNTR 2015, 1, 263695. [Google Scholar]
- Derhali, S.; el Hamdani, N.; Fdil, R.; Mouzdahir, A.; Sraidi, K. Chemical Composition of Essential Oils of Retama monosperma (L.) Boiss. from Morocco. Res. J. Pharm. Biol. Chem. Sci. 2016, 7, 2102–2106. [Google Scholar]
- Abdelmadjide, S.; Atef, C. Qualitative and Quantitative Identification by GC/MS of the Main Compounds of Extracts Using Solvents of Retama monosperma from the Flora of Algeria. PhytoChem. BioSub. J. 2021, 15, 250. [Google Scholar]
- Merghoub, N.; Benbacer, L.; el Btaouri, H.; Benhassou, H.A.; Terryn, C.; Attaleb, M.; Madoulet, C.; Benjouad, A.; el Mzibri, M.; Morjani, H.; et al. In Vitro Antiproliferative Effect and Induction of Apoptosis by Retama monosperma L. Extract in Human Cervical Cancer Cells. Cell. Mol. Biol. 2011, 57, 1581–1591. [Google Scholar] [CrossRef]
- Bokhari-Taieb Brahimi, H.; Aizi, D.E.; Bouhafsoun, A.; Hachem, K.; Mezemaze, R.; Kaid-Harche, M. Extraction and Analysis of Polysaccharides from Tissues of Retama monosperma Branches Extraction and Analysis of Polysaccharides from Tissues of Retama monosperma Branches. S. Asian J. Exp. Biol. 2019, 9, 214–221. [Google Scholar] [CrossRef]
- Belayachi, L. Screening of North African Medicinal Plant Extracts for Cytotoxic Activity Against Tumor Cell Lines. Eur. J. Med. Plant. 2013, 3, 310–332. [Google Scholar] [CrossRef]
- Yan, Z.; Zhong, Y.; Duan, Y.; Chen, Q.; Li, F. Antioxidant Mechanism of Tea Polyphenols and Its Impact on Health Benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef]
- Gulcin, İ. Antioxidants and Antioxidant Methods: An Updated Overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef] [PubMed]
- Farag, R.S.; Abdel-Latif, M.S.; Abd El Baky, H.H.; Tawfeek, L.S. Phytochemical Screening and Antioxidant Activity of Some Medicinal Plants’ Crude Juices. Biotechnol. Rep. 2020, 28, e00536. [Google Scholar] [CrossRef] [PubMed]
- Elisha, I.L.; Botha, F.S.; McGaw, L.J.; Eloff, J.N. The Antibacterial Activity of Extracts of Nine Plant Species with Good Activity against Escherichia coli against Five Other Bacteria and Cytotoxicity of Extracts. BMC Complement. Altern. Med. 2017, 17, 133. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Last 25 Years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Kavanagh, J.J. Anticancer Therapy Targeting the Apoptotic Pathway. Lancet Oncol. 2003, 4, 721–729. [Google Scholar] [CrossRef]
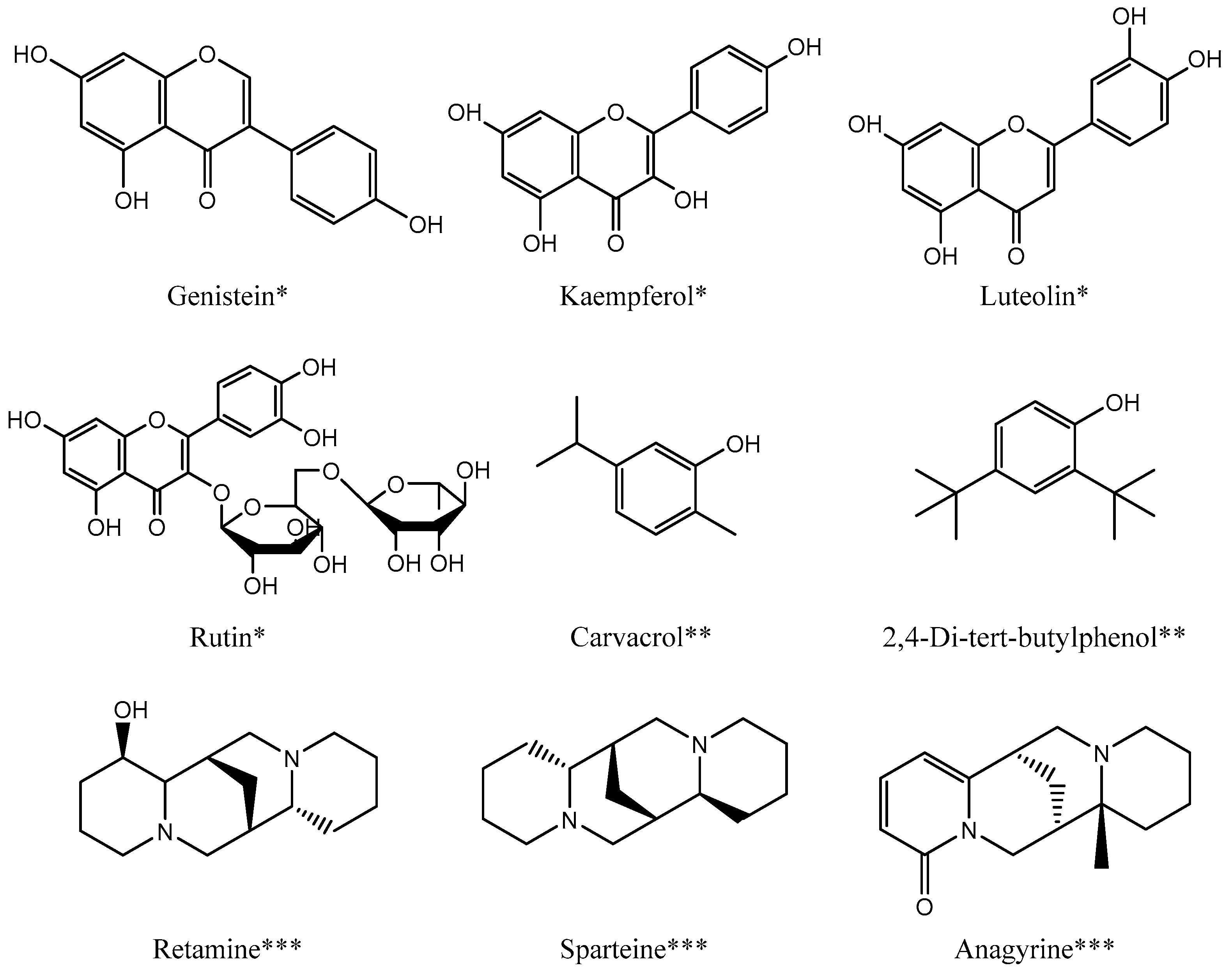
| Part of the Plant | Extract | Major Component | Ref |
|---|---|---|---|
| Stems | Methanol Dichloromethane n-Butanol Ethyl acetate Chloroform | Polyphenols Flavonoids Condensed tannins | [13,14] |
| Methanol | Alkaloid:
| [15,17,18,19,22,25] | |
| Flower | Methanol | Polyphenols Flavonoids Condensed tannins Alkaloids | [13,14] |
| Dichloromethane | Polyphenol Flavonoid Tannins | [14] | |
| Toluene | Polyphenol Flavonoid Condensed tannins | [13] | |
| Diethyl ether | Polyphenol Flavonoid:
| [3] | |
| Seeds | Methanol | Polyphenols Flavonoids Condensed tannins | [13,14] |
Alkaloid:
| [13,15,17,18,19,25,26,27] | ||
| n-Butanol | Polyphenols Flavonoids Condensed tannins | [13] | |
| Hexane | Fatty Acids:
| [2,21,23,28] | |
| Dichloromethane | Polyphenols Flavonoids Tannins | [14] | |
| Ethyl acetate | Polyphenol Flavonoid
| [3,13,14,15] | |
| Mineral: Al, Ba, Cd, Cu, Fe, Mg, Pb, Zn, Mn, Ca, K, Na, P | [21,23] | ||
| Leaves | Methanol | Alkaloid:
| [15,17,18,25] |
| Hexane | α-Linolenic acid Sterols:
| [4] | |
| Ethyl acetate | Ethyl palmitate Phenol: (2,4-Ditertbutylphenol) | [29] | |
| n-Butanol | Methyl palmitate Methyl 7-octadecenoate Alkaloid: (Sparteine) Phenol: (Carvacrol) | [29] | |
| Dichloromethane | Alkaloid:
| [30] | |
| Branches | NaOH 4% | Monosaccharides:
| [31] |
| Whole plant | Aqueous | Flavonoid:
| [16] |
| Plant Part | Extraction Type, Yield, and Studied Dose | Design | Active Compounds | Results | Ref |
|---|---|---|---|---|---|
| Seeds Flowers | -Flowers air-dried for two weeks and extracted three times by maceration at room temperature for three days in 3 L of n-hexane -Maceration with 3 L diethyl ether (at room temperature, three days) -Evaporation of n-hexane under a vacuum using a rotary evaporator gave a diethyl ether extract of flowers. -The same protocol was used on 400 g of seeds air-dried for two weeks -Ground to a fine powder. -The third maceration with ethyl acetate three times (3 L, at room temperature for three days) gave ethyl acetate extract of seeds. (5–100 μg/mL) | in vitro | Genistin Taxifolin Quercetin Genistein 6-Methoxykaemferol Kaempferol Apigenin | Antioxidant activity (EAS IC50 = 15.13 μg/mL compared to BHT IC50 = 30.21 μg/mL) Enhance SIRT1 and SIRT3 genes expression in HaCaT cell anti-aging process in human keratinocytes | [3] |
| Flower Seeds Stems | -Obtain crude extracts of seeds, stems, flowers with methanol 70% -Fractionation by toluene, chloroform, ethyl acetate, butanol (7–10 mg/mL) | in vitro | Total phenolic Flavonoid compounds Condensed Tannins | Antioxidant activity Ethyl acetate extracts from seeds (IC50 = 0.15 ± 0.11 mg/mL) | [13] |
| -The plant samples were air-dried for several weeks -Powdered seeds, stems and flowers -Maceration with methanol three times -Concentrated under reduced pressure -Fractionation with equal volumes of three organic solvents (hexane, dichloromethane, ethyl acetate) -Evaporated Fractions to dryness under vacuum and stored at +4°C -A concentration of 500 mg/mL was prepared by reconstituting the crude extracts in absolute methanol (500 mg/mL) | in vitro | Alkaloids Tannins Flavonoids Saponosides Terpenoids Coumarines | Antibacterial activity against: Bacillus sp B. cereus Listeria ivanovii Staphylococcus aureus C. freundii E. coli Salmonella sp | [14] | |
| Leaves | -Extracted successively using a Soxhlet apparatus with n-hexane and methanol -Obtain hexane and methanolic extract -Evaporated by a Rotavapor to give dried extracts -Extracted with dichloromethane and ethyl acetate to obtain dichloromethane and ethyl acetate fractions (5–80 μg/mL) | in vitro | α-Pinene 1,8-Cineole 9H-pyrrolo [3′,4′:3,4]pyrrolo [2,1-a]phthalazine-9, 11(10H)-dione,10-ethyl-8-phenyl Sparteine Hexadecanoic acid L methyl cytisine 17- oxosparteine 4-(N-(3-trifluoromethylphenyl)-amino)-5,6 dimethyl-7H-pyrro [2.3-d]pyrimidine Lupanine Anagyrine | Antiproliferative effects on human cervical cancer cells | [30] |
| -Extracted by using a Soxhlet apparatus with n-Hexane (1.3 L) and methanol (1.3 L) to obtain hexane and methanolic extract -Evaporated by a Rotavapor to give dried extracts -Extracted with dichloromethane (1.3 L) and ethyl acetate (1.3 L) to obtain dichloromethane fraction and ethyl acetate fraction (1–50 μg/mL) | in vitro | Quinolizidine Alkaloids Sparteine L-methyl cytisine 17-Oxosparteine Lupanine Anagyrine | Antitumoral activity (Rm-DF) was the most active extract, with a significant cytotoxic activity on HeLa and SiHa cells in a dose-dependent manner | [32] | |
| in vitro | Palmitic Acid trimethyl silyl ester Aphylline Phytol Linoleic Acid trimethyl silyl α Linoleic Acid trimethyl silyl ester Octadecanoic Acid, trimethyl silyl ester oleamide/SLEEPAMIDE Eicosanoic Acid, trimethylsilylester Monolupine/anagyrine Hexadecanoic Acid 2,3-bis[(trimethylsilyl)oxy]propyl ester dodecanoic Acid trimethylsilyl ester 1-Tetracosanol Stearic Acid 2,3-bis(trimethylsilyloxy)propyl ester Hexacosanoic Acid Stigmasterol trimethylsilylether β-Sitosteryltrimethylsilylether Campesterol | Anti-leukemic activity Cell cycle arrest and cell death occur through extrinsic apoptosis | [4,32] | ||
| Aerial parts | -Dried aerial parts at room temperature. -Powdered 200 g of plant material -Extracted with water (400 mL) at 70 °C, under agitation using a magnetic stirrer, during 1 h -The water extract was lyophilised to provide a crude water extract with 14 % yield (28.7 g). (18 mg/kg) (9 mg/kg) | in vivo Crohn’s disease model: -Aqueous extract of R. monosperma (9–18 mg/kg p.o.) was suspended in 0.9 % saline solution -Administered by gavage 48, 24 and 1 h prior to the induction of colitis and 24 h later. -Control group received a vehicle in a comparable volume (10 mL/kg animal). -The rats were checked daily for behavior, body weight and stool consistency. -Finally, animals were killed, using an overdose of chloral hydrate 48 h after induction of colitis | Daidzin Rutin Genistin Daidzein Luteolin Apigenin Genistein | Anti-inflammatory effects reduced the inflammation and damage in (TNBS)-induced colonic mucosa | [16] |
| Stems Leaves Flowers Seeds | -Extracted with absolute methanol three times at room temperature -Filtration -Combined methanolic extracts and concentrated to dryness under reduced pressure (31.25–500 µg extract/mL DMSO) | in vitro | Sparteine Dehydrosparteine β-Isosparteine Ammodendrine N-Methylcytisine Dehydrocytisine Cytisine 17-Oxosparteine Isolupanine 5.6-Dehydrolupanine 11.12-Dehydrolupanine Anagyrine Thermopsine | Antifungal activity against: C. albicans C. tropicalis A. niger | [17] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Yadini, A.; Elouafy, Y.; Amiri-Ardekani, E.; Shafiee, M.; Firouzi, A.; Sasani, N.; Khalid, A.; Abdalla, A.N.; Bakrim, S.; Tan, C.S.; et al. A Comprehensive Review of the Pharmacological Properties and Bioactive Components of Retama monosperma. Molecules 2023, 28, 1708. https://doi.org/10.3390/molecules28041708
El Yadini A, Elouafy Y, Amiri-Ardekani E, Shafiee M, Firouzi A, Sasani N, Khalid A, Abdalla AN, Bakrim S, Tan CS, et al. A Comprehensive Review of the Pharmacological Properties and Bioactive Components of Retama monosperma. Molecules. 2023; 28(4):1708. https://doi.org/10.3390/molecules28041708
Chicago/Turabian StyleEl Yadini, Adil, Youssef Elouafy, Ehsan Amiri-Ardekani, Mina Shafiee, Amirhosein Firouzi, Najmeh Sasani, Asaad Khalid, Ashraf N. Abdalla, Saad Bakrim, Ching Siang Tan, and et al. 2023. "A Comprehensive Review of the Pharmacological Properties and Bioactive Components of Retama monosperma" Molecules 28, no. 4: 1708. https://doi.org/10.3390/molecules28041708
APA StyleEl Yadini, A., Elouafy, Y., Amiri-Ardekani, E., Shafiee, M., Firouzi, A., Sasani, N., Khalid, A., Abdalla, A. N., Bakrim, S., Tan, C. S., Goh, K. W., Ming, L. C., & Bouyahya, A. (2023). A Comprehensive Review of the Pharmacological Properties and Bioactive Components of Retama monosperma. Molecules, 28(4), 1708. https://doi.org/10.3390/molecules28041708














