Citrus Waste as Source of Bioactive Compounds: Extraction and Utilization in Health and Food Industry
Abstract
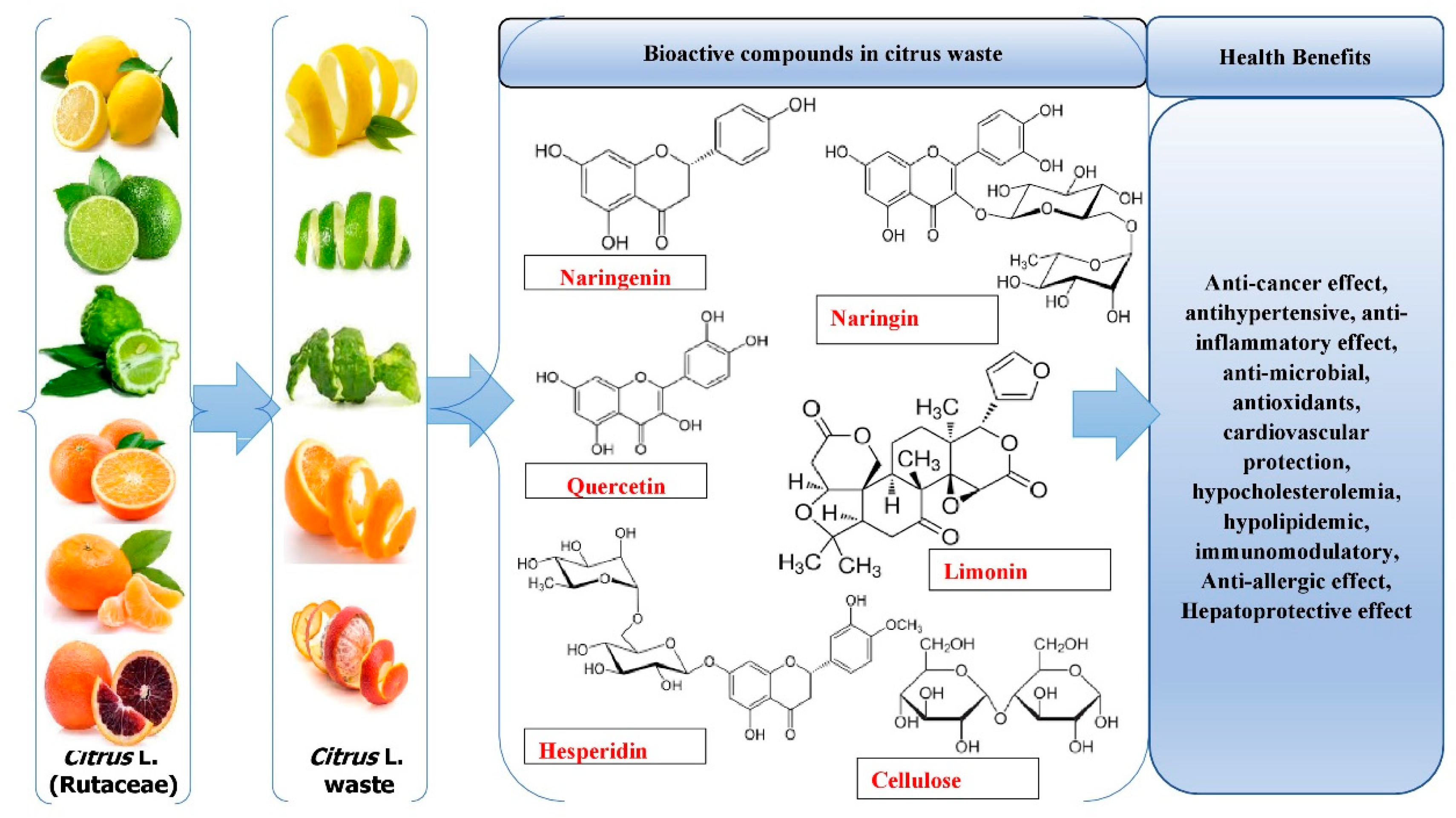
1. Introduction
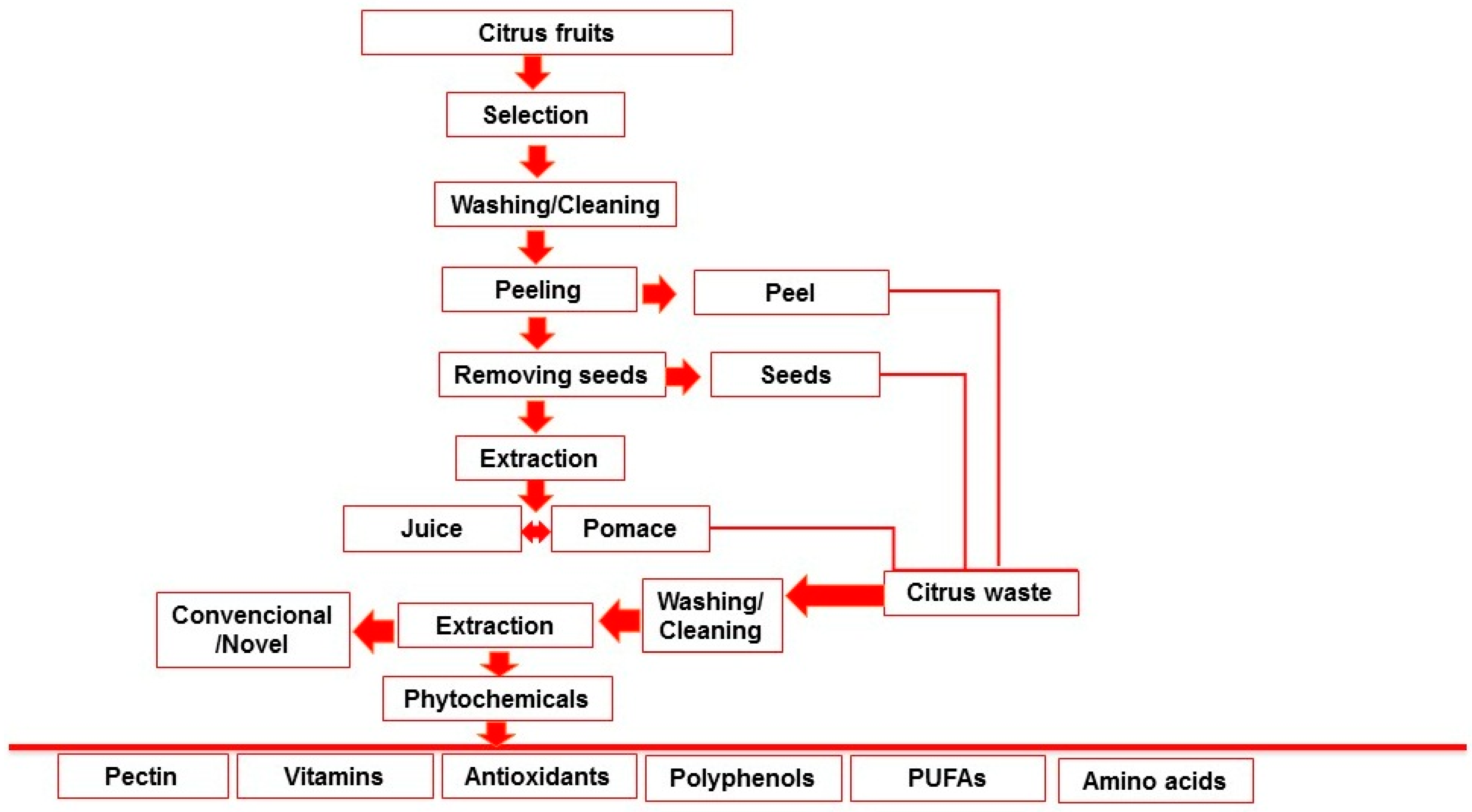
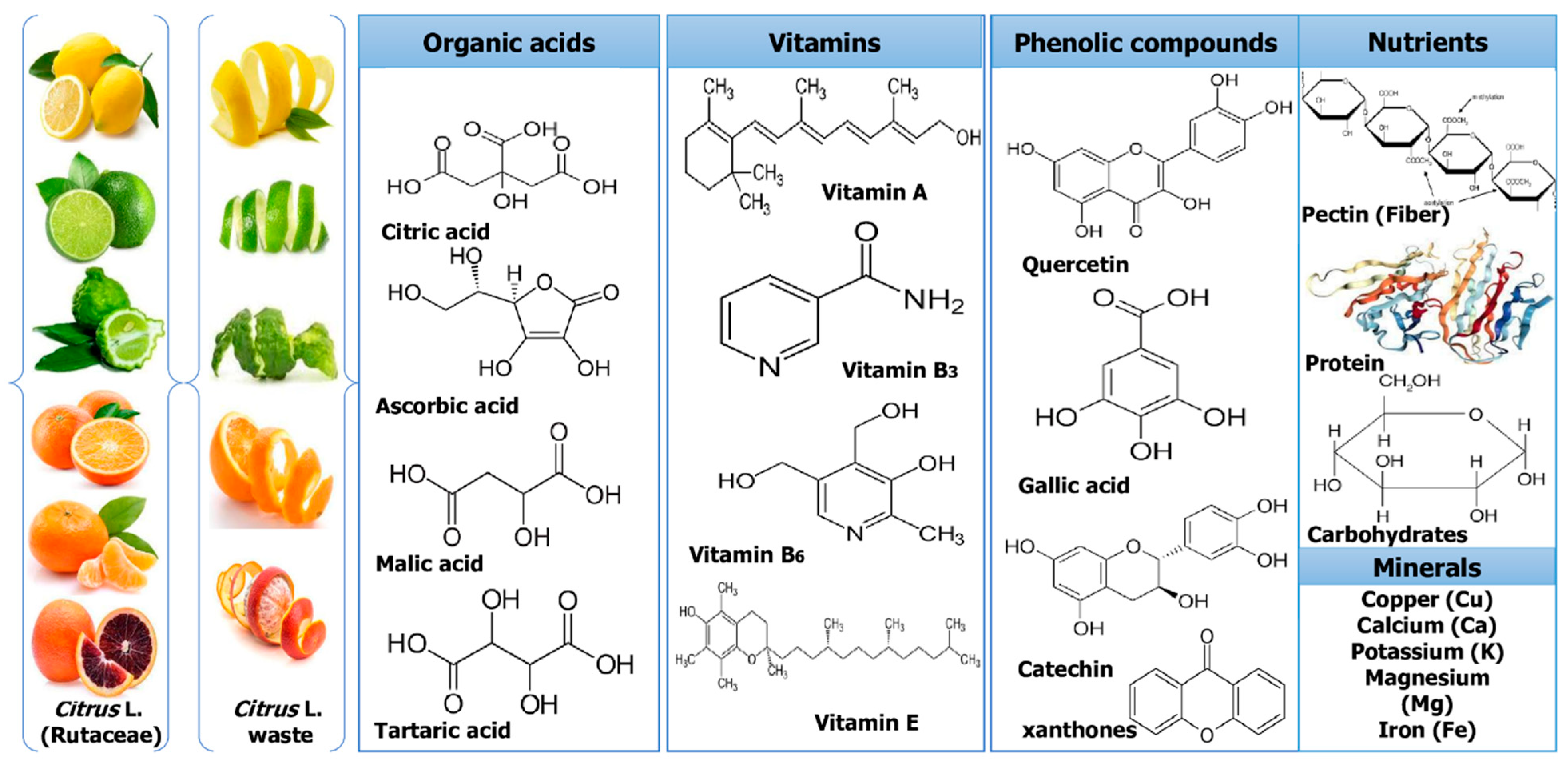
2. Different Sources of Citrus Waste
2.1. Citrus Peel
2.2. Citrus Seeds
2.3. Citrus Pomace
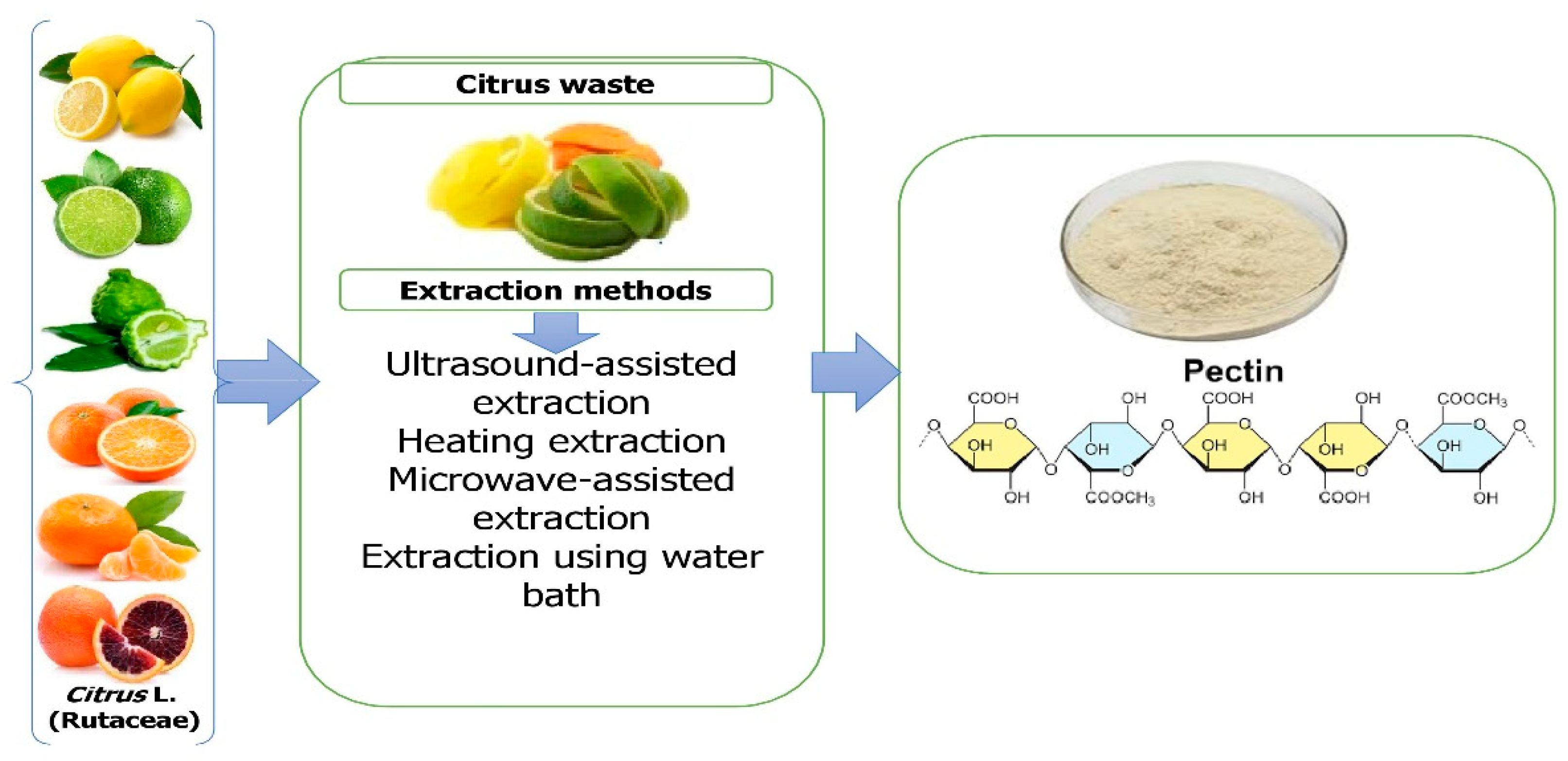
3. Extraction of Bioactive Compounds from Citrus Waste
3.1. Pectin
3.2. Dietary Fiber
3.3. Essential Oils (EOs)
3.4. Carotenoids
3.5. Polyphenols
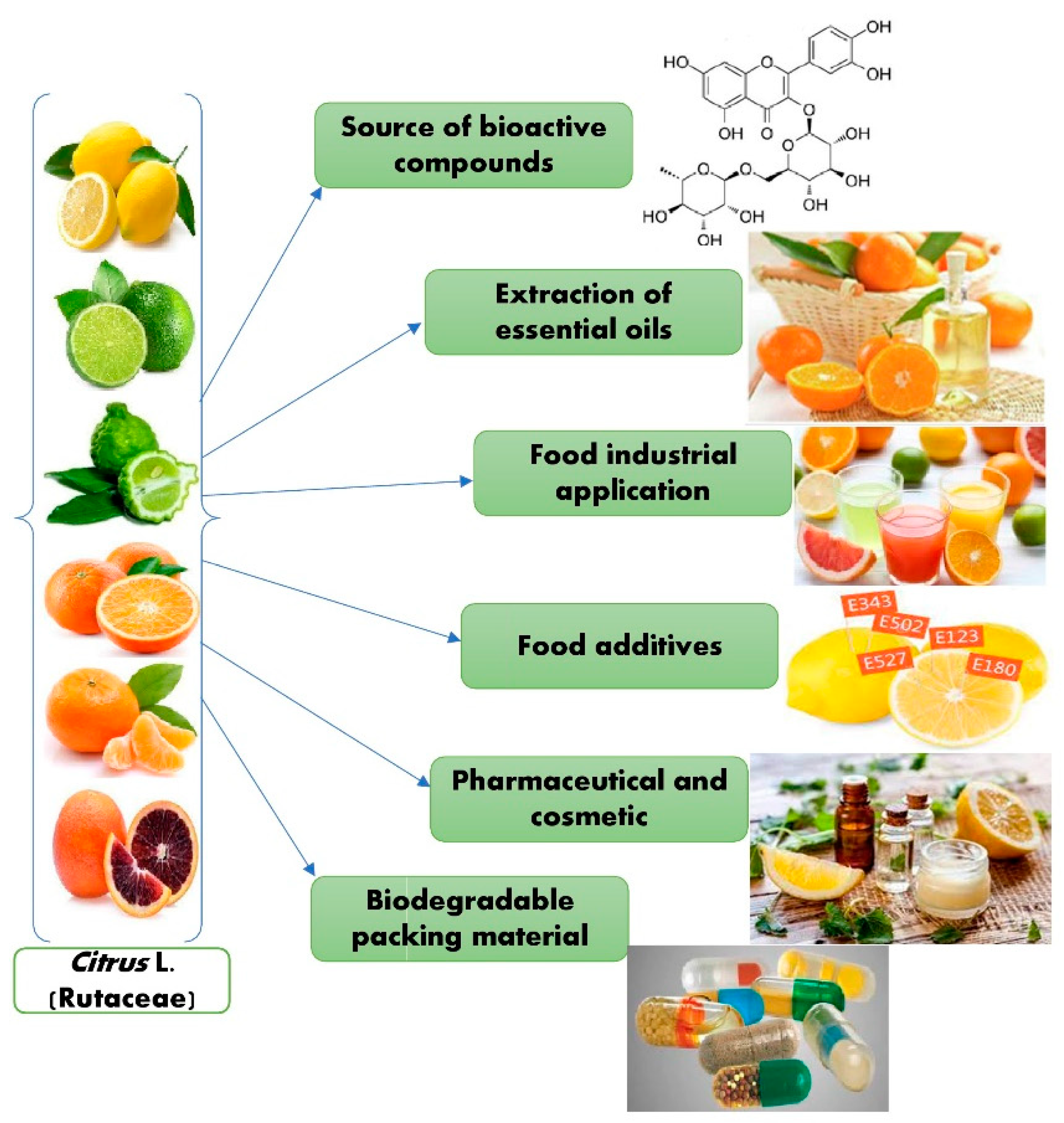
4. Application of Citrus Waste in the Food Industry
4.1. Essential Oil-Containing Polysaccharide-Based Edible Films and Coatings
4.2. Application of Fiber Concentrate in Bakery Products
4.3. Application of Citrus Pectin in Dairy Drinks
4.4. Utilization of Citrus Waste in the Beverage Industry
4.5. Applications of Essential Oils (EOs) for Food Safety
5. Herb–Drug Interaction of Citrus L.
6. Health Benefits of Bioactive Substances Derived from Citrus Waste
6.1. Functional Benefits in the Control of Diabetes
6.2. Anti-Cancer Properties
6.3. Mental Health and Metabolism
6.4. Citrus’ Role in the Prevention of Oxidative Damage and Cardiovascular Disease
6.5. Citrus’ Role in the Prevention of Obesity
6.6. Regulation of Lipoprotein Metabolism
6.7. Phytochemicals as Antiaging Agents
7. Toxicity of Phytochemicals Extracted from Citrus L.
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Zou, Z.; Xi, W.; Hu, Y.; Nie, C.; Zhou, Z. Antioxidant activity of Citrus fruits. Food Chem. 2016, 196, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Heying, E.; Tanumihardjo, S.A. History, global distribution, and nutritional importance of citrus fruits. Com. Rev. Food Sci. Food Saf. 2012, 11, 530–545. [Google Scholar] [CrossRef]
- Rawat, N.; Kiran, S.P.; Du, D.; Gmitter, F.G., Jr.; Deng, Z. Comprehensive meta-analysis, co-expression, and miRNA nested network analysis identifies gene candidates in citrus against Huanglongbing disease. BMC Plant Biol. 2015, 15, 184. [Google Scholar] [CrossRef]
- Anwar, F.; Naseer, R.; Bhanger, M.; Ashraf, S.; Talpur, F.; Aladeduny, F. Physico-chemical characteristics of citrus seeds and seed oils from Pakistan. J. Am. Oil Chem. Soc. 2008, 85, 321–330. [Google Scholar] [CrossRef]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. The medicinal and nutritional role of underutilized citrus fruit Citrus hystrix (Kaffir lime): A review. Drug Invent. 2014, 6, 1–5. [Google Scholar]
- Okwu, D.E. Citrus fruits: A rich source of phytochemicals and their roles in human health. Int. J. Chem. Sci. 2008, 6, 451–471. [Google Scholar]
- Lv, X.; Zhao, S.; Ning, Z.; Zeng, H.; Shu, Y.; Tao, O.; Liu, Y. Citrus fruits as a treasure trove of active natural metabolites that potentially provide benefits for human health. Chem. Cent. J. 2015, 9, 68. [Google Scholar] [CrossRef]
- Saini, R.K.; Ranjit, A.; Sharma, K.; Prasad, P.; Shang, X.; Gowda, K.G.M.; Keum, Y.S. Bioactive Compounds of Citrus Fruits: A Review of Composition and Health Benefits of Carotenoids, Flavonoids, Limonoids, and Terpenes. Antioxidants 2022, 11, 239. [Google Scholar] [CrossRef]
- Ke, Z.; Xu, X.; Nie, C.; Zhou, Z. Citrus flavonoids and human cancers. J. Food Nutr. Res. 2015, 3, 341–351. [Google Scholar] [CrossRef]
- Adenaike, O.; Abakpa, G.O. Antioxidant Compounds and Health Benefits of Citrus Fruits. Eur. J. Nutr. Food Saf. 2021, 13, 65–74. [Google Scholar] [CrossRef]
- Hussain, S.Z.; Naseer, B.; Qadri, T.; Fatima, T.; Bhat, T.A. Citrus fruits—Morphology, taxonomy, composition, and health benefits. In Fruits Grown in Highland Regions of the Himalayas; Springer: Cham, Switzerland, 2021; pp. 229–244. [Google Scholar]
- Bursać Kovačević, D.; Gajdoš Kljusurić, J.; Putnik, P.; Vukušić, T.; Herceg, Z.; Dragović-Uzelac, V. Stability of polyphenols in chokeberry juice treated with gas phase plasma. Food Chem. 2016, 212, 323–331. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Cho, M.H.; Lee, Y.R. Converting citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef]
- Arshad, M.S.; Khalid, W.; Khalid, M.Z.; Maqbool, Z.; Ali, A.; Kousar, S.; Afzal, M.F.; Mukhtar, S.; Rahim, M.A.; Aziz, A.; et al. Sonication Microwave Synergistic Extraction of Bioactive Compounds from Plants, Animals and Others Agro-Industrial Waste Sources; Elsevier Inc.: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Loesecke, H.W.V. Bureau of Agricultural and Industrial Chemistry; Agricultural Research Administration: Washington, DC, USA, 1952; Volume 44, pp. 476–482.
- Vattem, D.; Shetty, K. Solid-state production of phenolic antioxidants from cranberry pomace by Rhizopus oligosporus. Food Biotechnol. 2002, 16, 189–210. [Google Scholar] [CrossRef]
- Sir Elkhatim, K.A.; Elagib, R.A.; Hassan, A.B. Content of phenolic compounds and vitamin C and antioxidant activity in wasted parts of Sudanese citrus fruits. Food Sci. Nutr. 2018, 6, 1214–1219. [Google Scholar] [CrossRef]
- Costanzo, G.; Iesce, M.R.; Naviglio, D.; Ciaravolo, M.; Vitale, E.; Arena, C. Comparative studies on different citrus cultivars: A revaluation of waste mandarin components. Antioxidants 2020, 9, 517. [Google Scholar] [CrossRef]
- Jeong, D.; Park, H.; Jang, B.K.; Ju, Y.; Shin, M.H.; Oh, E.J.; Kim, S.R. Recent advances in the biological valorization of citrus peel waste into fuels and chemicals. Bioresour. Technol. 2021, 323, 124603. [Google Scholar] [CrossRef]
- John, I.; Yaragarla, P.; Muthaiah, P.; Ponnusamy, K.; Appusamy, A. Statistical optimization of acid catalyzed steam pretreatment of citrus peel waste for bioethanol production. Resour.-Effic. Technol. 2017, 3, 429–433. [Google Scholar] [CrossRef]
- Choi, I.S.; Lee, Y.G.; Khanal, S.K.; Park, B.J.; Bae, H.J. A low-energy, cost-effective approach to fruit and citrus peel waste processing for bioethanol production. Appl. Energy 2015, 140, 65–74. [Google Scholar] [CrossRef]
- Zayed, A.; Badawy, M.T.; Farag, M.A. Valorization and extraction optimization of Citrus seeds for food and functional food applications. Food Chem. 2021, 355, 129609. [Google Scholar] [CrossRef]
- Ammerman, C.B.; Arrington, L.R. Re-evaluation of citrus pulp as a feed. In Proceedings of the Florida Nutrition Conference, Gainesville, FL, USA, 2 November 1961. [Google Scholar]
- Rashid, U.; Ibrahimc, M.; Yasin, S.; Yunus, R.; Taufiq-Yap, Y.H.; Gerhard, K. Biodiesel from Citrus reticulata (mandarin orange) seed oil, a potential non-food feedstock. Ind. Crops Prod. 2013, 45, 355–359. [Google Scholar] [CrossRef]
- Wang, L.; Lee, W.W.; Yang, H.W.; Ryu, B.M.; Cui, Y.R.; Lee, S.C.; Lee, T.G.; Jeon, Y.J. Protective Effect of Water Extract of Citrus Pomace against AAPH-Induced Oxidative Stress In Vitro in Vero Cells and In Vivo in Zebrafish. Preventive Nutr. Food Sci. 2018, 23, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.; Schulz, P.; Rizvi, S.S. Valorization of bioactive compounds in fruit pomace from agro-fruit industries: Present Insights and future challenges. Food Biosci. 2021, 44, 101384. [Google Scholar] [CrossRef]
- Papoutsis, K.; Pristijono, P.; Golding, J.B.; Stathopoulos, C.E.; Scarlett, C.J.; Bowyer, M.C.; Vuong, Q.V. Impact of different solvents on the recovery of bioactive compounds and antioxidant properties from lemon (Citrus limon L.) pomace waste. Food Sci. Biotechnol. 2016, 2, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Widmer, W.; Zhou, W.; Grohmann, K. Pretreatment effects on orange processing waste for making ethanol by simultaneous saccharification and fermentation. Bioresour. Technol. 2010, 101, 5242–5249. [Google Scholar] [CrossRef]
- Putnik, P.; Bursać Kovačević, D.; Režek Jambrak, A.; Barba, F.J.; Cravotto, G.; Binello, A.; Shpigelman, A. Innovative “green” and novel strategies for the extraction of bioactive added value compounds from citrus wastes—A review. Molecules 2017, 22, 680. [Google Scholar] [CrossRef]
- Levaj, B.; Dragović-Uzelac, V.; Bursać Kovačević, D.; Krasnići, N. Determination of flavonoids in pulp and peel of mandarin fruits. Agric. Conspec. Sci. 2009, 74, 221–225. [Google Scholar]
- Levaj, B.; Putnik, P.; Linardić, I.; Herceg, Z.; Režek-Jambrak, A.; Kovačević-Bursać, D. Influence of pre-treatment on yield and quality of mandarin juices. In Proceedings of the 6th Central European Congress on Food-CEFood Congress, Novi Sad, Serbia, 2 December 2012. [Google Scholar]
- Sanders, A. Extraction, Encapsulation, and Microfluidization of Naringin from Grapefruit. Doctoral Dissertation, The Ohio State University, Columbus, OH, USA, 2020. [Google Scholar]
- Waghmode, M.S.; Gunjal, A.B.; Nawani, N.N.; Patil, N.N. Management of floral waste by conversion to value-added products and their other applications. Waste Bio. Val. 2018, 9, 33–43. [Google Scholar] [CrossRef]
- Santos, C.M.; Dweck, J.; Viotto, R.S.; Rosa, A.H.; de Morais, L.C. Application of orange peel waste in the production of solid biofuels and biosorbents. Bioresour. Technol. 2015, 196, 469–479. [Google Scholar] [CrossRef]
- Ángel Siles López, J.; Li, Q.; Thompson, I.P. Biorefinery of waste orange peel. Crit. Rev. Biotechnol. 2010, 30, 63–69. [Google Scholar] [CrossRef]
- Chhikara, N.; Kour, R.; Jaglan, S.; Gupta, P.; Gat, Y.; Panghal, A. Citrus medica: Nutritional, phytochemical composition and health benefits—A review. Food Funct. 2018, 9, 1978–1992. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, W.; Xu, Y.; Chen, I.; Cao, J.; Jiang, W. An advance on nutritional profile, phytochemical profile, nutraceutical properties, and potential industrial applications of lemon peels: A comprehensive review. Trends Food Sci. Technol. 2022, 124, 219–236. [Google Scholar] [CrossRef]
- Banerjee, J.; Vijayaraghavan, R.; Arora, A.; Macfarlane, D.R.; Patti, A.F. Lemon juice-based extraction of pectin from mango peels: Waste to wealth by sustainable approaches. ACS Sustain. Chem. Eng. 2016, 4, 5915–5920. [Google Scholar] [CrossRef]
- Shehata, M.G.; Awad, T.S.; Asker, D.; El Shaimy, S.A.; Abd El-Aziz, N.M.; Youssef, M.M. Antioxidant and antimicrobial activities and uplc-esi-ms/ms polyphenolic profile of sweet orange peel extracts. Cur. Res. Food Sci. 2021, 4, 326–335. [Google Scholar] [CrossRef]
- Rezzadori, K.; Benedetti, S.; Amante, E.R. Proposals for the residues recovery: Orange waste as raw material for new products. Food Bioprod. Process. 2012, 90, 606–614. [Google Scholar] [CrossRef]
- Khan, U.M.; Sameen, A.; Aadil, R.M.; Shahid, M.; Sezen, S.; Zarrabi, A.; Butnariu, M. Citrus genus and its waste utilization: A review on health-promoting activities and industrial application. J. Evid. Based Complement. Altern. Med. 2021, 2021, 2488804. [Google Scholar] [CrossRef]
- Ngouémazong, E.D.; Christiaens, S.; Shpigelman, A.; Van Loey, A.; Hendrickx, M. The emulsifying and emulsion-stabilizing properties of pectin: A review. Comp. Rev. Food Sci. Food Saf. 2015, 14, 705–718. [Google Scholar] [CrossRef]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.; De Boeck, G.; Becker, K. Dietary roles of non-starch polysaccharides in human nutrition: A review. Crit. Rev. Food Sci. Nutr. 2012, 52, 899–935. [Google Scholar] [CrossRef]
- May, C.D. Industrial pectins: Sources, production and applications. Carbohydr. Polym. 1990, 12, 79–99. [Google Scholar] [CrossRef]
- Shalini, R.; Gupta, D.K. Utilization of pomace from apple processing industries: A review. J. Food Sci. Technol. 2010, 47, 365–371. [Google Scholar] [CrossRef]
- Koubala, B.B.; Kansci, G.; Mbome, L.I.; Crépeau, M.J.; Thibault, J.F.; Ralet, M.C. Effect of extraction conditions on some physicochemical characteristics of pectins from “Améliorée” and “Mango” mango peels. Food Hydrocoll. 2008, 22, 1345–1351. [Google Scholar] [CrossRef]
- Zhang, L.; Ye, X.; Ding, T.; Sun, X.; Xu, Y.; Liu, D. Ultrasound effects on the degradation kinetics, structure and rheological properties of apple pectin. Ultrason. Sonochem. 2013, 20, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Fishman, M.L.; Cooke, P.H. The structure of high-methoxyl sugar acid gels of citrus pectin is determined by AFM. Carbohydr. Res. 2009, 344, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Ptichkina, N.M.; Markina, O.A.; Rumyantseva, G.N. Pectin extraction from pumpkin with the aid of microbial enzymes. Food Hydrocoll. 2008, 22, 192–195. [Google Scholar] [CrossRef]
- Heitz, M.; Carrasco, F.; Rubio, M.; Chauvette, G.; Chornet, E.; Jaulin, L.; Overend, R.P. Generalized correlations for the aqueous liquefaction of lignocellulosic. Can. J. Chem. Eng. 1986, 64, 647–650. [Google Scholar] [CrossRef]
- Carr, A.G.; Mammucari, R.; Foster, N.R. A review of subcritical water as a solvent and its utilization for the processing of hydrophobic organic compounds. Chem. Eng. J. 2011, 172, 1–17. [Google Scholar] [CrossRef]
- Maxwell, E.G.; Belshaw, N.J.; Waldron, K.W.; Morris, V.J. Pectin—An emerging new bioactive food polysaccharide. Trends Food Sci. Technol. 2012, 24, 64–73. [Google Scholar] [CrossRef]
- Khalid, W.; Arshad, M.S.; Jabeen, A.; Anjum, M.F.; Qaisrani, T.B.; Suleria, H.A.R. Fiber-enriched botanicals: A therapeutic tool against certain metabolic ailments. Food Sci. Nutr. 2022, 10, 3203–3218. [Google Scholar] [CrossRef]
- Sáyago-Ayerdi, S.G.; Brenes, A.; Goñi, I. Effect of grape antioxidant dietary fiber on the lipid oxidation of raw and cooked chicken hamburgers. LWT-Food Sci. Technol. 2009, 42, 971–976. [Google Scholar] [CrossRef]
- de Moraes Crizel, T.; Jablonski, A.; de Oliveira Rios, A.; Rech, R.; Flôres, S.H. Dietary fiber from orange byproducts is a potential fat replacer. LWT-Food Sci. Technol. 2013, 53, 9–14. [Google Scholar] [CrossRef]
- Tanaka, M.; Takamizu, A.; Hoshino, M.; Sasaki, M.; Goto, M. Extraction of dietary fiber from Citrus junos peel with subcritical water. Food Bioprod. Process. 2012, 90, 180–186. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Neng, N.R.; Nogueira, J.M.; Saraiva, J.A.; Nunes, M.L. Chemical composition and antibacterial and antioxidant properties of commercial essential oils. Indus. Crops Prod. 2013, 43, 587–595. [Google Scholar] [CrossRef]
- Lemes, R.S.; Alves, C.C.F.; Estevam, E.B.B.; Santiago, M.B.; Martins, C.H.G.; Dos Santos, T.C.L. Chemical composition and antibacterial activity of essential oils from Citrus Aurantifolia leaves and fruit peel against oral pathogenic Bacteria. An. Acad. Bras. Cienc. 2018, 90, 1285–1292. [Google Scholar] [CrossRef]
- Bayala, B.; Bassole, I.H.; Scifo, R.; Gnoula, C.; Morel, L.; Lobaccaro, J.M.A.; Simpore, J. Anticancer activity of essential oils and their chemical components—A review. Am. J. Cancer Res. 2014, 4, 591. [Google Scholar]
- Javed, S.; Javaid, A.; Nawaz, S.; Saeed, M.K.; Mahmood, Z.; Siddiqui, S.Z.; Ahmad, R. Phytochemistry, GC-MS analysis, antioxidant and antimicrobial potential of essential oil from five citrus species. J. Agricul. Sci. 2014, 6, 201. [Google Scholar] [CrossRef]
- Al-Aamri, M.S.; Al-Abousi, N.M.; Al-Jabri, S.S.; Alam, T.; Khan, S.A. Chemical composition and in-vitro antioxidant and antimicrobial activity of the essential oil of Citrus aurantifolia L. leaves grown in Eastern Oman. J. Taibah Uni. Med. Sci. 2018, 13, 108–112. [Google Scholar] [CrossRef]
- Nidhi, P.; Rolta, R.; Kumar, V.; Dev, K.; Sourirajan, A. Synergistic potential of Citrus aurantium L. essential oil with antibiotics against Candida albicans. J. Ethnophar. 2020, 262, 113135. [Google Scholar] [CrossRef]
- Onyeyirichi, I.; Ogechi, N.; Oche, O.; Jerry, U.; Gero, M. Evaluation of chemical constituent of Citrus medica limonum leaf essential oil. J. Phar. Scientic. Innov. 2014, 3, 306–309. [Google Scholar] [CrossRef]
- Walia, A.; Gupta, A.K.; Sharma, V. Role of bioactive compounds in human health. Acta Sci. Med. 2019, 3, 25–33. [Google Scholar]
- Turner, T.; Burri, B.J. Potential nutritional benefits of current citrus consumption. Agriculture 2013, 3, 170–187. [Google Scholar] [CrossRef]
- Dugo, P.; Škeříková, V.; Kumm, T.; Trozzi, A.; Jandera, P.; Mondello, L. Elucidation of carotenoid patterns in citrus products through comprehensive normal-phase× reversed-phase liquid chromatography. Anal. Chem. 2006, 78, 7743–7750. [Google Scholar] [CrossRef]
- Weissenberg, M.; Schaeffler, I.; Menagem, E.; Barzilai, M.; Levy, A. Isocratic non-aqueous reversed-phase high-performance liquid chromatographic separation of capsanthin and capsorubin in red peppers (Capsicum annuum L.), paprika and oleoresin. J. Chromatogr. A 1997, 757, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Gorinstein, S.; Martín-Belloso, O.; Park, Y.S.; Haruenkit, R.; Lojek, A.; Ĉíž, M.; Trakhtenberg, S. Comparison of some biochemical characteristics of different citrus fruits. Food Chem. 2001, 74, 309–315. [Google Scholar] [CrossRef]
- Tripoli, E.; La Guardia, M.; Giammanco, S.; Di Majo, D.; Giammanco, M. Citrus flavonoids: Molecular structure, biological activity and nutritional properties: A review. Food Chem. 2007, 104, 466–479. [Google Scholar] [CrossRef]
- Kore, P.S.; Virk, A.; Peste, A. Evaluation of in vitro Anthelmintic potential of Quercetin against pheretima posthuma. J. Pharm. Res. 2018, 12, 974–976. [Google Scholar]
- Mahmoud, A.M.; Hernandez Bautista, R.J.; Sandhu, M.A.; Hussein, O.E. Beneficial effects of citrus flavonoids on cardiovascular and metabolic health. Oxid. Med. Cell. Longev. 2019, 2019, 5484138. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Mejía, E.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Citrus peels waste as a source of value-added compounds: Extraction and quantification of bioactive polyphenols. Food Chem. 2019, 295, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Mahato, N.; Sharma, K.; Koteswararao, R.; Sinha, M.; Baral, E.; Cho, M.H. Citrus essential oils: Extraction, authentication and application in food preservation. Crit. Rev. Food Sci. Nutr. 2019, 59, 611–625. [Google Scholar] [CrossRef]
- Bhavaniramya, S.; Vishnupriya, S.; Al-Aboody, M.S.; Vijayakumar, R.; Baskaran, D. Role of essential oils in food safety: Antimicrobial and antioxidant applications. Grain Oil Sci. Technol. 2019, 2, 49–55. [Google Scholar] [CrossRef]
- Anis, A.; Pal, K.; Al-Zahrani, S.M. Essential oil-containing polysaccharide-based edible films and coatings for food security applications. Polymers 2021, 13, 575. [Google Scholar] [CrossRef]
- Perdones, A.; Sánchez-González, L.; Chiralt, A.; Vargas, M. Effect of chitosan–lemon essential oil coatings on storage-keeping quality of strawberry. Postharv. Biol. Technol. 2012, 70, 32–41. [Google Scholar] [CrossRef]
- Moosavy, M.H.; Hassanzadeh, P.; Mohammadzadeh, E.; Mahmoudi, R.; Khatibi, S.A.; Mardani, K. Antioxidant and antimicrobial activities of essential oil of Lemon (Citrus limon) peel in vitro and in a food model. J. Food Qual. Hazards Control. 2017, 4, 42–48. [Google Scholar]
- Ortiz-Flores, M.; Portilla-Martínez, A.; Cabrera-Pérez, F.; Nájera, N.; Meaney, E.; Villarreal, F.; Ceballos, G. PXR is a target of (-)-epicatechin in skeletal muscle. Heliyon 2020, 6, e05357. [Google Scholar] [CrossRef]
- Romero-Lopez, M.R.; Osorio-Diaz, P.; Bello-Perez, L.A.; Tovar, J.; Bernardino-Nicanor, A. Fiber concentrate from orange (Citrus sinensis L.) bagasse: Characterization and application as a bakery product ingredient. Int. J. Mol. Sci. 2011, 12, 2174–2186. [Google Scholar] [CrossRef]
- Naseri, A.T.; Thibault, J.F.; Ralet-Renard, M.C. Citrus pectin: Structure and application in acid dairy drinks. In Tree and Forestry Science and Biotechnology; Global Science Books: Ikenobe, Japan, 2008. [Google Scholar]
- Varmie, E.B.; Mamta Thakur, M. Utilization of citrus processing waste: A review. Pharma. Innov. J. 2021, 10, 682–697. [Google Scholar]
- Okino Delgado, C.H.; Fleuri, L.F. Orange and mango by-products: Agro-industrial waste as a source of bioactive compounds and botanical versus commercial description—A review. Food Rev. Int. 2016, 32, 1–14. [Google Scholar] [CrossRef]
- Khalid, W.; Arshad, M.S.; Aziz, A.; Rahim, M.A.; Qaisrani, T.B.; Afzal, F.; Ali, A.; Ranjha, M.; Khalid, M.Z.; Anjum, F.M. Chia Seeds (Salvia hispanica L.): A Therapeutic Weapon in Metabolic Disorders. Food Sci. Nutr. 2022, 11, 3–16. [Google Scholar] [CrossRef]
- Izzo, A.A. Interactions between herbs and conventional drugs: Overview of the clinical data. Med. Princ. Pract. 2012, 21, 404–428. [Google Scholar] [CrossRef]
- Husain, I.; Bala, K.; Khan, I.A.; Khan, S.I. A review on phytochemicals, pharmacological activities, drug interactions, and associated toxicities of licorice (Glycyrrhiza sp.). Food Front. 2021, 2, 449–485. [Google Scholar] [CrossRef]
- Singh, A.; Zhao, K. Herb–drug interactions of commonly used Chinese medicinal herbs. Int. Rev. Neurob. 2017, 135, 197–232. [Google Scholar]
- Lippert, A.; Renner, B. Herb–Drug Interaction in Inflammatory Diseases: Review of Phytomedicine and Herbal Supplements. J. Clin. Med. 2022, 11, 1567. [Google Scholar] [CrossRef]
- Asher, G.N.; Corbett, A.H.; Hawke, R.L. Common herbal dietary supplement—Drug interactions. Am. Fam. Phy. 2017, 96, 101–107. [Google Scholar]
- Fasinu, P.S.; Bouic, P.J.; Rosenkranz, B. An overview of the evidence and mechanisms of herb–drug interactions. Front. Pharm. 2012, 3, 69. [Google Scholar] [CrossRef] [PubMed]
- Goya-Jorge, E.; Jorge Rodríguez, M.E.; Veitía, M.S.I.; Giner, R.M. Plant occurring flavonoids as modulators of the aryl hydrocarbon receptor. Molecules 2021, 26, 2315. [Google Scholar] [CrossRef]
- Zhou, L.; Cui, M.; Zhao, L.; Wang, D.; Tang, T.; Wang, W.; Qiu, X. Potential metabolic drug–drug interaction of Citrus aurantium L.(Rutaceae) evaluating by its effect on 3 CYP450. Front. Pharm. 2018, 9, 895. [Google Scholar] [CrossRef] [PubMed]
- Lü, J.; Zhang, D.; Zhang, X.; Sa, R.; Wang, X.; Wu, H.; Zhang, B. Network Analysis of the Herb–Drug Interactions of Citrus Herbs Inspired by the “Grapefruit Juice Effect”. ACS Omega 2022, 7, 35911–35923. [Google Scholar] [CrossRef]
- Husain, I.; Dale, O.R.; Martin, K.; Gurley, B.J.; Adams, S.J.; Avula, B.; Khan, S.I. Screening of medicinal plants for possible herb-drug interactions through modulating nuclear receptors, drug-metabolizing enzymes and transporters. J. Ethnopharm. 2023, 301, 115822. [Google Scholar] [CrossRef]
- Makni, M.; Jemai, R.; Kriaa, W.; Chtourou, Y.; Fetoui, H. Citrus limon from Tunisia: Phytochemical and physicochemical properties and biological activities. BioMed Res. Int. 2018, 2018, 6251546. [Google Scholar] [CrossRef]
- USDA. USDA National Nutrient Database for Standard Reference, Release 18. Nutrient Data Laboratory. 2015. Available online: http://www.ars.usda.gov/ba/bhnrc/ndl (accessed on 3 January 2023).
- Nkoana, D.K.; Mashilo, J.; Shimelis, H.; Ngwepe, R.M. Nutritional, phytochemical compositions and natural therapeutic values of citron watermelon (Citrullus lanatus var. citroides): A Review. S. Afr. J. Bot. 2022, 145, 65–77. [Google Scholar] [CrossRef]
- Mäkynen, K.; Jitsaardkul, S.; Tachasamran, P.; Sakai, N.; Puranachoti, S.; Nirojsinlapachai, N.; Adisakwattana, S. Cultivar variations in antioxidant and antihyperlipidemic properties of pomelo pulp. Food Chem. 2013, 139, 735–743. [Google Scholar] [CrossRef]
- Etebu, E.; Nwauzoma, A.B. A review on sweet orange (citrus sinensis l osbeck): Health, diseases and management. Am. J. Res. Commun. 2014, 2, 33–70. [Google Scholar]
- Baghurst, K. The Health Benefits of Citrus Fruits; Horticulture Australia, Ltd.: Sydney, Australia, 2003; pp. 13–14. [Google Scholar]
- Shah, A.; Masoodi, F.A.; Gani, A.; Noor, N. Mosambi (sweet lime). In Antioxidants in Fruits: Properties and Health Benefits; Springer: Singapore, 2020; pp. 125–133. [Google Scholar]
- Zhang, J. Flavonoids in grapefruit and commercial grapefruit juices: Concentration, distribution, and potential health benefits. Proc. Fla. State Hortic. Soc. 2007, 120, 288–294. [Google Scholar]
- Hung, W.L.; Suh, J.H.; Wang, Y. Chemistry and health effects of furanocoumarins in grapefruit. J. Food Drug Anal. 2017, 25, 71–83. [Google Scholar] [CrossRef]
- Li, X.; Meenu, M.; Xu, B. Recent development in bioactive compounds and health benefits of kumquat fruits. Food Rev. Int. 2022, 3, 1–21. [Google Scholar] [CrossRef]
- Lou, S.N.; Ho, C.T. Phenolic compounds and biological activities of small-size citrus: Kumquat and calamondin. J. Food Drug Anal. 2017, 25, 162–175. [Google Scholar] [CrossRef]
- Aruoma, O.I.; Landes, B.; Ramful-Baboolall, D.; Bourdon, E.; Neergheen-Bhujun, V.; Wagner, K.H.; Bahorun, T. Functional benefits of citrus fruits in the management of diabetes. Prev. Med. 2012, 54, S12–S16. [Google Scholar] [CrossRef]
- Hegde, P.S.; Chen, D.S. Top 10 challenges in cancer immunotherapy. Immunity 2020, 52, 17–35. [Google Scholar] [CrossRef]
- Rawson, N.E.; Ho, C.T.; Li, S. Efficacious anti-cancer property of flavonoids from citrus peels. Food Sci. Hum. Wellness 2014, 3, 104–109. [Google Scholar] [CrossRef]
- Ballard, C.R.; Junior, M.R.M. Health benefits of flavonoids. In Bioactive Compounds; Woodhead Publishing: Sawston, UK, 2019; pp. 185–201. [Google Scholar]
- Giammanco, M.; Plescia, F.; Giammanco, M.M.; Leto, G.; Gentile, C. Bioactive effects of citrus flavonoids and role in the prevention of atherosclerosis and cancer. J. Biol. Res. 2022, 50, 340–350. [Google Scholar] [CrossRef]
- Breijyeh, Z.; Karaman, R. Comprehensive review on Alzheimer’s disease: Causes and treatment. Molecules 2020, 25, 5789. [Google Scholar] [CrossRef]
- Freedland, K.E.; Skala, J.A.; Steinmeyer, B.C.; Carney, R.M.; Rich, M.W. Effects of depression on heart failure self-care. J. Card. Fail. 2021, 27, 522–532. [Google Scholar] [CrossRef]
- Kakutani, N.; Yokota, T.; Fukushima, A.; Obata, Y.; Ono, T.; Sota, T.; Kinugawa, S. Impact of citrus fruit intake on the mental health of patients with chronic heart failure. J. Cardiol. 2022, 79, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K.; Nakajima, A.; Guo, Y.; Ohizumi, Y. A Narrative Review of the Effects of Citrus Peels and Extracts on Human Brain Health and Metabolism. Nutrients 2022, 14, 1847. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Azmat, R. Citrus: An ancient fruit of promise for health benefits. In Citrus-Health Benefits and Production Technology; Books on Demand GmbH: Norderstedt, Germany, 2019; pp. 19–30. [Google Scholar]
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Lavie, C.J.; Laddu, D.; Arena, R.; Ortega, F.B.; Alpert, M.A.; Kushner, R.F. Healthy weight and obesity prevention: JACC health promotion series. J. Am. Coll. Cardiol. 2018, 72, 1506–1531. [Google Scholar] [CrossRef]
- Feng, S.; Wang, Y. Citrus phytochemicals and their potential effects on the prevention and treatment of obesity: Review and progress of the past 10 years. J. Food Bioact. 2018, 4, 99–106. [Google Scholar] [CrossRef]
- Tung, Y.C.; Chang, W.T.; Li, S.; Wu, J.C.; Badmeav, V.; Ho, C.T.; Pan, M.H. Citrus peel extracts attenuated obesity and modulated gut microbiota in mice with high-fat diet-induced obesity. Food Funct. 2018, 9, 3363–3373. [Google Scholar] [CrossRef]
- Fardoun, M.M.; Maaliki, D.; Halabi, N.; Iratni, R.; Bitto, A.; Baydoun, E.; Eid, A.H. Flavonoids in adipose tissue inflammation and atherosclerosis: One arrow, two targets. Clin. Sci. 2020, 134, 1403–1432. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Mulvihill, E.E.; Burke, A.C.; Huff, M.W. Citrus flavonoids as regulators of lipoprotein metabolism and atherosclerosis. Annu. Rev. Nutr. 2016, 36, 275–299. [Google Scholar] [CrossRef]
- Stevenie, S.; Girsang, E.; Nasution, A.N.; Lister, I.N.E. Comparison activities of peel and extract of lime (Citrus amblycarpa) as antioxidant and antielastase. Am. Acad. Sci. Res. J. Appl. Sci. Eng. Technol. 2019, 57, 77–84. [Google Scholar]
- Mahato, N.; Sharma, K.; Sinha, M.; Cho, M.H. Citrus waste derived Nutra-/pharmaceuticals for health benefits: Current trends and future perspectives. J. Funct. Foods 2018, 40, 307–316. [Google Scholar] [CrossRef]
- Bailey, R.L.; Gahche, J.J.; Lentino, C.V.; Dwyer, J.T.; Engel, J.S.; Thomas, P.R.; Picciano, M.F. Dietary supplement use in the United States, 2003–2006. J. Nut. 2011, 141, 261–266. [Google Scholar] [CrossRef]
- Yeung, K.S.; Gubili, J.; Cassileth, B. Evidence-based botanical research: Applications and challenges. Hematol./Oncol. Clin. N. Am. 2008, 22, 661–670. [Google Scholar] [CrossRef]
- Shahrokh, L.E.; Lukaszuk, J.M.; Prawitz, A.D. Elderly herbal supplement users less satisfied with medical care than nonusers. J. Am. Diet. Assoc. 2005, 105, 1138–1140. [Google Scholar] [CrossRef]
- Funk, J.; Stoeber, H.; Hauser, E.; Schmidt, H. Molecular analysis of subtilase cytotoxin genes of food-borne Shiga toxin-producing Escherichia coli reveals a new allelic subAB variant. BMC Mic. 2013, 13, 230. [Google Scholar] [CrossRef]
- Lorenz, S.C.; Son, I.; Maounounen-Laasri, A.; Lin, A.; Fischer, M.; Kase, J.A. Prevalence of hemolysin genes and comparison of ehxA subtype patterns in Shiga toxin-producing Escherichia coli (STEC) and non-STEC strains from clinical, food, and animal sources. Appl. Env. Microb. 2013, 79, 6301–6311. [Google Scholar] [CrossRef]
- Rao, M.R.; Palada, M.C.; Becker, B.N. Medicinal and aromatic plants in agroforestry systems. In New Vistas in Agroforestry; Springer: Dordrecht, The Netherlands, 2004; pp. 107–122. [Google Scholar]
- Adewunmi, C.O.; Ojewole, J.A.O. Safety of traditional medicines, complementary and alternative medicines in Africa. Afr. J. Trad. Compl. Alt. Med. 2004, 1, 1–3. [Google Scholar] [CrossRef]
- John, O.R.; Yahaya, A.A.; Emmanuel, A. Aqueous ethanolic extract of Mangifera indica stem bark effect on the biochemical and haematological parameters of albino rats. Arch. Appl. Sci Res. 2012, 4, 1618–1622. [Google Scholar]
- Dybing, E.; Doe, J.; Groten, J.; Kleiner, J.; O’brien, J.; Renwick, A.G.; Younes, M. Hazard characterisation of chemicals in food and diet: Dose response, mechanisms and extrapolation issues. Food Chem. Toxicol. 2002, 40, 237–282. [Google Scholar] [CrossRef]
- Papanastasiou, S.A.; Bali, E.M.D.; Ioannou, C.S.; Papachristos, D.P.; Zarpas, K.D.; Papadopoulos, N.T. Toxic and hormetic-like effects of three components of citrus essential oils on adult Mediterranean fruit flies (Ceratitis capitata). PloS ONE 2017, 12, e0177837. [Google Scholar] [CrossRef]
- Kanes, K.; Tisserat, B.; Berhow, M.; Vandercook, C. Phenolic composition of various tissues of Rutaceae species. Phytochemistry 1993, 32, 967–974. [Google Scholar] [CrossRef]
- Kawaii, S.; Tomono, Y.; Katase, E.; Ogawa, K.; Yano, M. Quantitation of flavonoid constituents in citrus fruits. J. Agric. Food Chem. 1999, 47, 3565–3571. [Google Scholar] [CrossRef] [PubMed]
- Mokbel, M.S.; Suganuma, T. Antioxidant and antimicrobial activities of the methanol extracts from pummelo (Citrus grandis Osbeck) fruit albedo tissues. Eur. Food Res. Technol. 2006, 224, 39–47. [Google Scholar] [CrossRef]
- Do Ngoc, L.; Nguyen, T.T.Q.; Vu, C.P.; Phung, T.H. Effect of pomelo (citrus grandis (l). osbeck) peel extract on lipid-carbohydrate metabolic enzymes and blood lipid, glucose parameters in experimental obese and diabetic mice. VNU J. Sci. Nat. Sci. Tech. 2010, 26, 200–210. Available online: https://js.vnu.edu.vn/NST/article/view/1982 (accessed on 3 January 2023).
- Oyebadejo, S.A.; Solomon, I.P. Acute and sub-acute toxicity study of Citrus limon (L) juice in Sprawgue dawley rats. East Afr. Sch. J. Biotech. Genet. 2019, 1, 2663–7286. [Google Scholar]
- Visakh, N.U.; Pathrose, B.; Narayanankutty, A.; Alfarhan, A.; Ramesh, V. Utilization of Pomelo (Citrus maxima) Peel Waste into Bioactive Essential Oils: Chemical Composition and Insecticidal Properties. Insects 2022, 13, 480. [Google Scholar] [CrossRef]
| Citrus Source | Part | Composition | Food Application | References |
|---|---|---|---|---|
| Citron | Peel, pulp | Antioxidants, vitamin C, phenols, flavanols | Carbonated drinks, alcoholic beverages, jams, syrup | [36] |
| Lemon | Peel | Vitamin C, pectin, phenols, carotenoids | Edible coatings, use as film matrixes | [37,38] |
| Sweet orange | Peel | Narirutin, naringin, quinic acid, sakuranetin | Enhance vegetable oil oxidative stability, candied orange peel, salad dressings, desserts | [39,40] |
| Grapefruit | Peel | Naringin, naringenin, flavonoids, phenols | Jam making, sauces, dessert recipes, flavoring of beverages | [32,41] |
| Citrus Source | Scientific Name | Bioactive/Functional Compounds | Diseases | References |
|---|---|---|---|---|
| Lemon | Citrus limon | Eriocitrin, hesperidin, 6,8-di-c-glu-apigenin, quercetin, hesperetin | Antioxidant action, reduce risk of cardiovascular complications, antimicrobial effects | [95] |
| Citron | Citrus medica | Phenolics, flavonones, vitamin C, pectin | Anti-catarrha, anti-hypertensive, antibacterial, antifungal, anti-cancerous | [96] |
| Pomelo | Citrus maxima | Naringin, naringenin, phenols | Antihyperlipidemic properties | [97] |
| Sweet orange | Citrus sinensis | Hesperidin, sinensitin, liminoids, polyphenols | Prevent arteriosclerosis, reduce risk of kidney stones, reduce cholesterol levels, improve stomach ulcer anti-inflammatory effect | [98] |
| Lime | Citrus lotifolia | Luteolin, tangerine, hesperidin | Improve immunity, prevent kidney stones, promote healthy skin | [99,100] |
| Grapefruit | Citrus paradisi | Naringin, narirutin, neoponcirin, quercetin | Antioxidant, anti-inflammatory, and anti-tumor activity; reduces the risk of atherosclerosis; increases bone cell activity | [101,102] |
| Kumquat | Citrus japonica | Polyphenols, essential oils (EOs), vitamin C | Antioxidant activity, liver protection, anti-cancer activity | [103,104] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maqbool, Z.; Khalid, W.; Atiq, H.T.; Koraqi, H.; Javaid, Z.; Alhag, S.K.; Al-Shuraym, L.A.; Bader, D.M.D.; Almarzuq, M.; Afifi, M.; et al. Citrus Waste as Source of Bioactive Compounds: Extraction and Utilization in Health and Food Industry. Molecules 2023, 28, 1636. https://doi.org/10.3390/molecules28041636
Maqbool Z, Khalid W, Atiq HT, Koraqi H, Javaid Z, Alhag SK, Al-Shuraym LA, Bader DMD, Almarzuq M, Afifi M, et al. Citrus Waste as Source of Bioactive Compounds: Extraction and Utilization in Health and Food Industry. Molecules. 2023; 28(4):1636. https://doi.org/10.3390/molecules28041636
Chicago/Turabian StyleMaqbool, Zahra, Waseem Khalid, Hafiz Taimoor Atiq, Hyrije Koraqi, Zaryab Javaid, Sadeq K. Alhag, Laila A. Al-Shuraym, D. M. D. Bader, Mohammed Almarzuq, Mohamed Afifi, and et al. 2023. "Citrus Waste as Source of Bioactive Compounds: Extraction and Utilization in Health and Food Industry" Molecules 28, no. 4: 1636. https://doi.org/10.3390/molecules28041636
APA StyleMaqbool, Z., Khalid, W., Atiq, H. T., Koraqi, H., Javaid, Z., Alhag, S. K., Al-Shuraym, L. A., Bader, D. M. D., Almarzuq, M., Afifi, M., & AL-Farga, A. (2023). Citrus Waste as Source of Bioactive Compounds: Extraction and Utilization in Health and Food Industry. Molecules, 28(4), 1636. https://doi.org/10.3390/molecules28041636









